Abstract
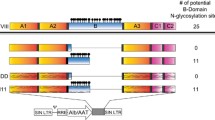
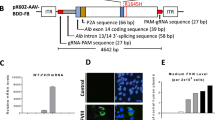
Hemophilia A is caused by a genetic mutation in coagulation factor VIII (FVIII) gene and gene therapy is considered to be a promising strategy for its treatment. We recently demonstrated that co-delivery of two vectors expressing M662C mutated heavy and D1828C mutated light chain genes of B-domain-deleted coagulation factor VIII (BDD-FVIII) leads to inter-chain disulfide cross-linking and improved heavy chain secretion in vitro. In this study, co-injection of both M662C and D1828C mutated BDD-FVIII gene expression vectors into mice resulted in increased heavy chain secretion and coagulation activity in plasma in vivo. Approximately (239±56) ng mL−1 above endogenous levels of transgenic FVIII heavy chain was found in mouse plasma using a chain-specific ELISA. For FVIII coagulation activity, approximately (1.09±0.25) IU mL−1 above endogenous levels were detected in co-injected transgenic mouse plasma using a chromogenic assay. These data demonstrate that inter-chain disulfide bonds likely increase heavy chain secretion and coagulation activity in the plasma of transgenic mice with an improved efficacy of a dual-vector delivery of BDD-FVIII gene. These findings support our ongoing efforts to develop a gene therapy for hemophilia A treatment using dual-AAV vectors.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dooriss K L, Denning G, Gangadharan B, et al. Comparison of factor VIII Transgenes bioengineered for improved expression in gene therapy of hemophilia A. Hum Gene Ther, 2009, 20: 465–478
Jiang H, Lillicrap D, Patarroyo-White S, et al. Multiyear therapeutic benefit of AAV serotypes 2, 6, and 8 delivering factor VIII to hemophilia A mice and dogs. Blood, 2006, 108: 107–115
Burton M, Nakai H, Colosi P, et al. Coexpression of factor VIII heavy and light chain adeno-associated viral vectors produces biologically active protein. Proc Natl Acad Sci USA, 1999, 96: 12725–12730
Sarkar R, Mucci M, Addya S, et al. Long-term efficacy of adenoassociated virus serotypes 8 and 9 in hemophilia a dogs and mice. Hum Gene Ther, 2006, 17: 427–439
Kaufman R J, Wasley L C, Dorner A J. Synthesis, processing, and secretion of recombinant human factor VI11 expressed in mammalian cell. J Biol Chem, 1988, 263: 6352–6362
Zhu F X, Yang S D, Liu Z L, et al. The effect of a secretion-enhanced heavy chain on improving intein-based dual-vector co-delivery of a full-length factor VIII gene. Chin Sci Bull, 2011, 56: 158–163
Gale A J, Radtke K P, Cunningham M A, et al. Intrinsic stability and functional properties of disulfide bond-stabilized coagulation factor VIIIa variants. J Thromb Haemost, 2006, 4: 1315–1322
Zhu F X, Yang S D, Liu Z L, et al. Disulfide cross-linking enhances dual-vector based delivery of split B-domain-deleted coagulation factor VIII gene. Prog Biochem Biophys, 2012, 39: 181–187
Zhu F X, Liu Z L, Chi X Y, et al. Protein trans-splicing based dualvector delivery of the coagulation factor VIII gene. Sci China Life Sci, 2010, 53: 683–689
Chen L X, Zhu F X, Li J, et al. The enhancing effects of the light chain on heavy chain secretion in split delivery of factor VIII gene. Mol Ther, 2007, 15: 1856–1862
Niemeyer G P, Herzog R W, Mount J, et al. Long-term correction of inhibitor-prone hemophilia B dogs treated with liver directed AAV2-mediated factor IX gene therapy. Blood, 2009, 113: 797–806
Arruda V R, Stedman H H, Haurigot V, et al. Peripheral transvenular delivery of adeno-associated viral vectors to skeletal muscle as a novel therapy for hemophilia B. Blood, 2010, 115: 4678–4688
Kaufman R J, Wasley L C, Dorner A J. Synthesis, processing, and secretion of recombinant human factor VI11 expressed in mammalian cell. J Biol Chem, 1988, 263: 6352–6362
Mann K G. Biochemistry and physiology of blood coagulation. Thromb Haemost, 1999, 82: 165–174
Swaroop M, Moussalli M, Pipe S W, et al. Mutagenesis of a potential immunoglobulin-binding protein-binding site enhances secretion of coagulation factor VIII. J Biol Chem, 1997, 272: 24121–24124
Dorner A J, Wasley L C, Kaufman R J. Increased synthesis of secreted proteins induces expression of glucose-regulated proteins in butyrate-treated Chinese hamster ovary cells. J Biol Chem, 1989, 264: 20602–20607
Zhang K, Shen X, Wu J, et al. Endoplasmic reticulum stress activates cleavage of CREBH to induce a systemic inflammatory response. Cell, 2006, 124: 587–599
Chen L X, Lu H, Wang J H, et al. Enhanced factor VIII heavy chain for gene therapy of hemophilia A. Mol Ther, 2009, 17: 417–424
d’Oiron R, Lavergne J M, Lavend’homme R, et al. Deletion of alanine 2201 in the FVIII C2 domain results in mild hemophilia A by impairing FVIII binding to vWF and phospholipids and destroys a major FVIII antigenic determinant involved in inhibitor development. Blood, 2004, 103: 155–157
Fay P J, Haidaris P J, Smudzin T M. Human factor VIIIa subunit structure. Reconstruction of factor VIIIa from the isolated A1/A3-C1-C2 dimer and A2 subunit. J Biol Chem, 1991, 266: 8957–8962
Wise R J, Dorner A J, Krane M, et al. The role of von Willebrand factor multimers and propeptide cleavage in binding and stabilization of factor VIII. J Biol Chem, 1991, 266: 21948–21955
Sabatino D S, Lange A M, Altynova E S, et al. Efficacy and safety of long-term prophylaxis in severe hemophilia A dogs following liver gene therapy using AAV vectors. Mol Ther, 2011, 19: 442–449
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Zhu, F., Liu, Z., Miao, J. et al. Enhanced plasma factor VIII activity in mice via cysteine mutation using dual vectors. Sci. China Life Sci. 55, 521–526 (2012). https://doi.org/10.1007/s11427-012-4333-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4333-8