Abstract
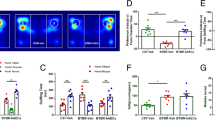
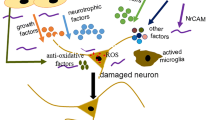
Human amniotic epithelial cells (HAECs), which exhibit characteristics of embryonic and pluripotent stem cells, could be utilized for cell therapy without legal or ethical problems. Double-transgenic (TG) mice (n=20) and wild-type (WT) mice (n=20) were randomly assigned to two groups, respectively. The transplantation group was treated with HAECs and the control group with PBS. A six-radial arm water maze was used to assess spatial memory. Immunofluorescence was utilized to track HAEC survival. Immunohistochemistry was used to determine octamer-binding protein 4 (oct-4) and nanog expression in the HAECs. High-performance liquid chromatography (HPLC) was used to measure acetylcholine levels in the hippocampus. The density of cholinergic neurons in the basal forebrain and nerve fibers in the hippocampus was measured following acetylcholinesterase staining. Results showed that transplanted HAECs survived for at least eight weeks and migrated to the third ventricle without immune rejection. Graft HAECs also expressed the specific stem cell markers oct-4 and nanog. Compared with the control group, HAEC transplantation significantly ameliorated spatial memory deficits in TG mice, as well as increased acetylcholine levels and the number of hippocampal cholinergic neurites. Intracerebroventricular HAEC transplantation improved spatial memory in double-TG mice, and results suggested that increased acetylcholine levels in the hippocampus, released by surviving cholinergic neurites, were responsible for this improvement.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Breteler M M. Mapping out biomarkers for Alzheimer’s disease. JAMA, 2011, 305: 304–305
Bartus R T, Dean III R L, Beer B, et al. The cholinergic hypothesis of geriatric memory dysfunction. Science, 1982, 217: 408–417
Selkoe D J. Toward a comprehensive theory for Alzheimer’s disease. Hypothesis: Alzheimer’s disease is caused by the cerebral accumulation and cytotoxicity of amyloid beta-protein. Ann N Y Acad Sci 2000, 924: 17–25
Savonenko A, Xu G M, Melnikova T, et al. Episodic-like memory deficits in the APPswe/PS1 dE9 mouse model of Alzheimer’s disease: relationships to beta-amyloid deposition and neurotransmitter abnormalities. Neurobiol Dis, 2005, 18: 602–617
German D C, Yazdani U, Speciale S G, et al. Cholinergic neuropathology in a mouse model of Alzheimer’s disease. J Comp Neurol, 2003, 462: 371–381
Ibach B, Haen E. Acetylcholinesterase inhibition in Alzheimer’s disease. Curr Pharm Des, 2004, 10: 231–251
Schifilliti D, Santamaria L B, Rosa G, et al. Cholinergic central system, Alzheimer’s disease, and anesthetics liaison: a vicious circle? J Alzheimers Dis, 2010, 22: 35–41
Marlatt M W, Lucassen P J. Neurogenesis and Alzheimer’s disease: Biology and pathophysiology in mice and men. Curr Alzheimer Res, 2010, 7: 113–125
Adinolfi M, Akle C A, McColl I, et al. Expression of HLA antigens, beta 2-microglobulin and enzymes by human amniotic epithelial cells. Nature, 1982, 295: 325–327
Uchida S, Inanaga Y, Kobayashi M, et al. Neurotrophie function of conditioned medium from human amniotic epithelial cells. Neurosci Res, 2000, 62: 585–590
Tamagawa T, Oi S, Ishiwata I, et al. Differentiation of mesenchymal cells derived from human amniotic membranes into hepatocyte-like cells in vitro. Hum Cell, 2007, 20: 77–84
Enosawa S, Sakuragawa N, Suzuki S. Possible use of amniotic cells for regenerative medicine. Nippon Rinsho, 2003, 61: 396–400
Sakuragawa N, Thangavel R, Mizuguchi M, et al. Expression of markers for both neuronal and glial cells in human amniotic epithelial cells. Neurosci Lett, 1996, 209: 9–12
Xue S R, Li Q, Khalil Z. Alterations in vascular reactivity in single-and double-transgenic mice coexpressing human APP-C100 and mutant SOD1 genes. Chin Med J (Engl), 2002, 115: 696–701
Yang X X, Xue S R, Dong W L, et al. Therapeutic effect of human amniotic epithelial cell transplantation into the lateral ventricle of hemiparkinsonian rats. Chin Med J (Engl), 2009, 122: 2449–2254
Liu T, Wu J, Huang Q, et al. Human amniotic epithelial cells ameliorate behavioral dysfunction and reduce infarct size in the rat middle cerebral artery occlusion model. Shock, 2008, 29: 603–611
Barinaga M. Fetal neuron grafts pave the way for stem cell therapies. Science, 2000, 287: 1421–1422
Le Belle J E, Svendsen C N. Stem cells for neurodegenerative disorders: where we can go from here? BioDrugs, 2002, 16: 389–401
Cho M S, Hwang D Y, Kim D W. Efficient derivation of functional dopaminergic neurons from human embryonic stem cells on a large scale. Nat Protoc, 2008, 3: 1888–1894
Bilic G, Zeisberger S M, Mallik A S, et al. Comparative characterization of cultured human term amnion epithelial and mesenchymal stromal cells for application in cell therapy. Cell Transplant, 2008, 17: 955–968
Uchida S, Suzuki Y, Araie M, et al. Factors secreted by human amniotic epithelial cells promote the survival of rat retinal ganglion cells. Neurosci Lett, 2003, 341: 1–4
Arnhold S, Post C, Glüer S, et al. Neuronal characteristics of amniotic fluid derived cells after adenoviral transformation. Cell Biol Int, 2008, 32: 1559–1566
Nilbratt M, Porras O, Marutle A, et al. Neurotrophic factors promote cholinergic differentiation in human embryonic stem cell-derived neurons. J Cell Mol Med, 2010, 14: 1476–1484
Straten G, Eschweiler G W, Maetzler W, et al. Glial cell-line derived neurotrophic factor (GDNF) concentrations in cerebrospinal fluid and serum of patients with early Alzheimer’s disease and normal controls. J Alzheimers Dis, 2009, 18: 331–337
Miki T, Lehmann T, Cai H, et al. Stem cell characteristics of amniotic epithelial cells. Stem Cells, 2005, 23: 1549–1559
Kakishita K, Elwan M A, Nakao N, et al. Human amniotic epithelial cells produce dopamine and survive after implantation into the striatum of a rat model of Parkinson’s disease: a potential source of donor for transplantation therapy. Exp Neurol, 2000, 165: 27–34
Hagan J J, Salamone J D, Simpson J. Place navigation in rats is impaired by lesions of medial septum and diagonal band but not nucleus basalis magnocellularis. Brain Res, 1988, 27: 9–16
Winkler J, Thal L J, Gage F H, et al. Cholinergic strategies for Alzheimer’s disease. J Mol Med, 1998, 76: 555–567
Machova E, Rudajev V, Smyckova H, et al. Function cholinergic damage develops with amyloid accumulation in young adult APPswe/PS1 dE9 transgenic mice. Neurobiol Dis, 2010, 38: 27–35
Reisere R S, Harrison E E, Syverud D C, et al. Impaired spatial learning in the APPswe+PSEN1DeltaE9 bigenic mouse model of Alzheimer’s disease. Genes Brain Behav, 2007, 6: 54–65
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Xue, S., Chen, C., Dong, W. et al. Therapeutic effects of human amniotic epithelial cell transplantation on double-transgenic mice co-expressing APPswe and PS1ΔE9-deleted genes. Sci. China Life Sci. 55, 132–140 (2012). https://doi.org/10.1007/s11427-012-4283-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4283-1