Abstract
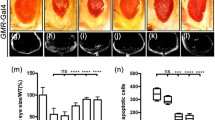
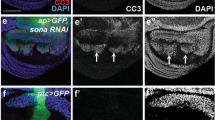
Ribosomal protein L8 is a component of the 60S subunit of the ribosome and is involved in protein synthesis but its role in Drosophila development is not well understood. We depleted L8 through RNA interference (RNAi) to examine its effects on fly development both in vivo and in vitro. The results demonstrated that L8 RNAi caused embryonic or first-larval lethality, delay of larval development, defects in eye and wing morphology, and dramatically reduced the number of S2 cells. This indicated that L8 plays a crucial role in Drosophila development. Acridine orange staining of the wing discs showed that apoptosis occurred when L8 was depleted, indicating that depletion of L8 is tightly connected to apoptosis. RT-PCR analyses of the transcription level of genes that are known to be key factors in apoptosis (p53, hid, reaper, dark, Dcp-1) and cell cycle regulation (cdc45, MCM3, cyclin B, incenp) in L8-deficient S2 cells, were consistent with their role in apoptosis induction and cell cycle arrest. These results indicate that depletion of L8 strongly impairs Drosophila development, and that this depletion is associated with cell proliferation arrest and apoptosis, in which p53 may play a central role.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chen F W, Ioannou Y A. Ribosomal proteins in cell proliferation and apoptosis. Int Rev Immunol, 1999, 18: 429–448 1:CAS:528:DC%2BD3cXjvFCqtg%3D%3D, 10672495
Volarevic S, Stewart M J, Ledermann B, et al. Proliferation, but not growth, blocked by conditional deletion of 40S ribosomal protein S6. Science, 2000, 288: 2045–2047 1:CAS:528:DC%2BD3cXkt1Kjs74%3D, 10.1126/science.288.5473.2045, 10856218
Volarevic S, Thomas G. Role of S6 phosphorylation and S6 kinase in cell growth. Prog Nucleic Acid Res Mol Biol, 2001, 65: 101–127 1:CAS:528:DC%2BD3MXnt1Wgs7g%3D, 10.1016/S0079-6603(00)65003-1, 11008486
Lohrum M A, Ludwig R L, Kubbutat M H, et al. Regulation of HDM2 activity by the ribosomal protein L11. Cancer Cell, 2003, 3: 577–587 1:CAS:528:DC%2BD3sXltlSmsL0%3D, 10.1016/S1535-6108(03)00134-X, 12842086
Wool I G. Extraribosomal functions of ribosomal proteins. Trends Biochem Sci, 1996, 21: 164–165 1:CAS:528:DyaK28XivFentbk%3D, 8871397
Soulet F, Al Saati T, Roga S, et al. Fibroblast growth factor-2 interacts with free ribosomal protein S19. Biochem Biophys Res Commun, 2001, 289: 591–596 1:CAS:528:DC%2BD3MXosFGit70%3D, 10.1006/bbrc.2001.5960, 11716516
Draptchinskaia N, Gustavsson P, Andersson B, et al. The gene encoding ribosomal protein S19 is mutated in Diamond-Blackfan anaemia. Nat Genet, 1999, 21: 169–175 1:CAS:528:DyaK1MXpsVCluw%3D%3D, 10.1038/5951, 9988267
Fisher E M, Beer-Romero P, Brown L G, et al. Homologous ribosomal protein genes on the human X and Y chromosomes: escape from X inactivation and possible implications for Turner syndrome. Cell, 1990, 63: 1205–1218 1:CAS:528:DyaK3MXhsl2ns7w%3D, 10.1016/0092-8674(90)90416-C, 2124517
O’Brien T W, O’Brien B J, Norman R A. Nuclear MRP genes and mitochondrial disease. Gene, 2005, 354: 147–151 10.1016/j.gene.2005.03.026, 15908146
Ruggero D, Pandolfi P P. Does the ribosome translate cancer? Nat Rev Cancer, 2003, 3: 179–192 1:CAS:528:DC%2BD3sXhsFGhtro%3D, 10.1038/nrc1015, 12612653
Lindsley D L, Sandler L, Baker B S, et al. Segmental aneuploidy and the genetic gross structure of the Drosophila genome. Genetics, 1972, 71: 157–184 1:STN:280:DyaE383htlejuw%3D%3D, 4624779
Schultz J. The Minute reaction in the development of Drosophila melanogaster. Genetics, 1929, 14: 366–419 1:STN:280:DC%2BD2s%2FmsVGmtQ%3D%3D, 17246581
Lambertsson A. The minute genes in Drosophila and their molecular functions. Adv Genet, 1998, 38: 69–134 1:CAS:528:DyaK1MXmtlOr, 10.1016/S0065-2660(08)60142-X, 9677706
Enerly E, Larsson J, Lambertsson A. Silencing the Drosophila ribosomal protein L14 gene using targeted RNA interference causes distinct somatic anomalies. Gene, 2003, 320: 41–48 1:CAS:528:DC%2BD3sXosFyjtb0%3D, 10.1016/S0378-1119(03)00827-8, 14597387
Ruhf M L, Meister M. The Drosophila homologue of ribosomal protein L8. Insect Biochem Mol Biol, 1999, 29: 349–353 1:CAS:528:DyaK1MXjtFSktb0%3D, 10.1016/S0965-1748(99)00009-0, 10333574
Xu R, Deng K, Zhu Y, et al. A large-scale functional approach to uncover human genes and pathways in Drosophila. Cell Res, 2008, 18: 1114–1127 1:CAS:528:DC%2BD1cXhtlWrsbfM, 10.1038/cr.2008.295, 18957936
Ni J Q, Liu L P, Hess D, et al. Drosophila ribosomal proteins are associated with linker histone H1 and suppress gene transcription. Genes Dev, 2006, 20: 1959–1973 1:CAS:528:DC%2BD28XnsVyjt7k%3D, 10.1101/gad.390106, 16816001
Hauck B, Gehring W J, Walldorf U. Functional analysis of an eye specific enhancer of the eyeless gene in Drosophila. Proc Natl Acad Sci USA, 1999, 96: 564–569 1:CAS:528:DyaK1MXmtlClsg%3D%3D, 10.1073/pnas.96.2.564, 9892673
Halder G, Callaerts P, Flister S, et al. Eyeless initiates the expression of both sine oculis and eyes absent during Drosophila compound eye development. Development, 1998, 125: 2181–2191 1:CAS:528:DyaK1cXksFanu74%3D, 9584118
Huang H, Potter C J, Tao W, et al. PTEN affects cell size, cell proliferation and apoptosis during Drosophila eye development. Development, 1999, 126: 5365–5372 1:CAS:528:DC%2BD3cXmtFOltg%3D%3D, 10556061
Schuler M, Bossy-Wetzel E, Goldstein J C, et al. p53 induces apop tosis by caspase activation through mitochondrial cytochrome c release. J Biol Chem, 2000, 275: 7337–7342 1:CAS:528:DC%2BD3cXhvVGrs74%3D, 10.1074/jbc.275.10.7337, 10702305
Chooi W Y, James S M, Burns D K. Decrease in ribosomal proteins 1, 2/3, L4, and L7 in Drosophila melanogaster in the absence of X rDNA. Mol Gen Genet, 1982, 187: 364–369 1:CAS:528:DyaL38XmtFeit7w%3D, 10.1007/BF00332613, 6817032
Grether M E, Abrams J M, Agapite J, et al. The head involution defective gene of Drosophila melanogaster functions in programmed cell death. Genes Dev, 1995, 9: 1694–1708 1:CAS:528:DyaK2MXnt1SksL4%3D, 10.1101/gad.9.14.1694, 7622034
White K, Grether M E, Abrams J M, et al. Genetic control of programmed cell death in Drosophila. Science, 1994, 264: 677–683 1:CAS:528:DyaK2cXksFChtbo%3D, 10.1126/science.8171319, 8171319
Brodsky M H, Weinert B T, Tsang G, et al. Drosophila melanogaster MNK/Chk2 and p53 regulate multiple DNA repair and apoptotic pathways following DNA damage. Mol Cell Biol, 2004, 24: 1219–1231 1:CAS:528:DC%2BD2cXmsFWntQ%3D%3D, 10.1128/MCB.24.3.1219-1231.2004, 14729967
White K, Tahaoglu E, Steller H. Cell killing by the Drosophila gene reaper. Science, 1996, 271: 805–807 1:CAS:528:DyaK28Xptlyisg%3D%3D, 10.1126/science.271.5250.805, 8628996
Zhou L, Schnitzler A, Agapite J, et al. Cooperative functions of the reaper and head involution defective genes in the programmed cell death of Drosophila central nervous system midline cells. Proc Natl Acad Sci USA, 1997, 94: 5131–5136 1:CAS:528:DyaK2sXjtlenu7k%3D, 10.1073/pnas.94.10.5131, 9144202
Song Z, McCall K, Steller H. DCP-1, a Drosophila cell death protease essential for development. Science, 1997, 275: 536–540 1:CAS:528:DyaK2sXnvVChuw%3D%3D, 10.1126/science.275.5299.536, 8999799
Quinn L M, Dorstyn L, Mills K, et al. An essential role for the caspase dronc in developmentally programmed cell death in Drosophila. J Biol Chem, 2000, 275: 40416–40424 1:CAS:528:DC%2BD3MXhsFersA%3D%3D, 10.1074/jbc.M002935200, 10984473
Li X, Scuderi A, Letsou A, et al. B56-associated protein phosphatase 2A is required for survival and protects from apoptosis in Drosophila melanogaster. Mol Cell Biol, 2002, 22: 3674–3684 1:CAS:528:DC%2BD38XjslWlsbc%3D, 10.1128/MCB.22.11.3674-3684.2002, 11997504
Alenzi F Q. Links between apoptosis, proliferation and the cell cycle. Br J Biomed Sci, 2004, 61: 99–102 1:CAS:528:DC%2BD2cXmvVWquro%3D, 15250676
Pollok S, Bauerschmidt C, Sanger J, et al. Human Cdc45 is a proliferation-associated antigen. Febs J, 2007, 274: 3669–3684 1:CAS:528:DC%2BD2sXosVOitLc%3D, 10.1111/j.1742-4658.2007.05900.x, 17608804
Schwab B L, Leist M, Knippers R, et al. Selective proteolysis of the nuclear replication factor MCM3 in apoptosis. Exp Cell Res, 1998, 238: 415–421 1:CAS:528:DyaK1cXhtVGitL4%3D, 10.1006/excr.1997.3850, 9473350
Krause K, Wasner M, Reinhard W, et al. The tumour suppressor protein p53 can repress transcription of cyclin B. Nucleic Acids Res, 2000, 28: 4410–4418 1:CAS:528:DC%2BD3cXosVemtL8%3D, 10.1093/nar/28.22.4410, 11071927
Sugihara Y, Honda H, Iida T, et al. Proteomic analysis of rodent ribosomes revealed heterogeneity including ribosomal proteins L10-like, L22-like 1, and L39-like. J Proteome Res, 2010, 9: 1351–1366 1:CAS:528:DC%2BC3cXhsVGitrk%3D, 10.1021/pr9008964, 20063902
Cheeseman I M, Anderson S, Jwa M, et al. Phospho-regulation of kinetochore-microtubule attachments by the Aurora kinase Ipl1p. Cell, 2002, 111: 163–172 1:CAS:528:DC%2BD38Xotlaiu7c%3D, 10.1016/S0092-8674(02)00973-X, 12408861
Bolton M A, Lan W, Powers S E, et al. Aurora B kinase exists in a complex with survivin and INCENP and its kinase activity is stimulated by survivin binding and phosphorylation. Mol Biol Cell, 2002, 13: 3064–3077 1:CAS:528:DC%2BD38XntlGis78%3D, 10.1091/mbc.E02-02-0092, 12221116
Anderson S J, Lauritsen J P, Hartman M G, et al. Ablation of ribosomal protein L22 selectively impairs alphabeta T cell development by activation of a p53-dependent checkpoint. Immunity, 2007, 26: 759–772 1:CAS:528:DC%2BD2sXntlKnurc%3D, 10.1016/j.immuni.2007.04.012, 17555992
Sulic S, Panic L, Barkic M, et al. Inactivation of S6 ribosomal protein gene in T lymphocytes activates a p53-dependent checkpoint response. Genes Dev, 2005, 19: 3070–3082 1:CAS:528:DC%2BD28Xmt1w%3D, 10.1101/gad.359305, 16357222
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, H., Pan, L. & Gou, K. Depletion of ribosomal protein L8 impairs Drosophila development and is associated with apoptosis. Sci. China Life Sci. 53, 1092–1097 (2010). https://doi.org/10.1007/s11427-010-4059-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-010-4059-4