Abstract
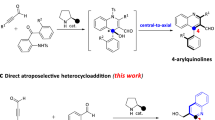
The catalytic atroposelective synthesis of axially chiral isochromenone-indoles has been established by the strategy of designing homophthalic anhydride-based indole derivatives as a new type of indole-based platform molecules for dynamic kinetic resolution. By this strategy, a wide range of axially chiral isochromenone-indoles were synthesized in high yields with excellent enantioselectivities (up to 98% yield, 97% ee) via the catalytic asymmetric sulfonylation reaction of homophthalic anhydride-based indole derivatives with aryl sulfonyl chlorides under the catalysis of chiral quaternary ammonium salt as a phase-transfer catalyst

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bonne D, Rodriguez J. Chem Commun, 2017, 53: 12385–12393
Bonne D, Rodriguez J. Eur J Org Chem, 2018, 2018(20–21): 2417–2431
Wang YB, Tan B. Acc Chem Res, 2018, 51: 534–547
Zhang S, Liao G, Shi B. Chin J Org Chem, 2019, 39: 1522–1528
Liao G, Zhou T, Yao QJ, Shi BF. Chem Commun, 2019, 55: 8514–8523
Da BC, Xiang SH, Li S, Tan B. Chin J Chem, 2021, 39: 1787–1796
Carmona JA, Rodríguez-Franco C, Fernández R, Hornillos V, Lassaletta JM. Chem Soc Rev, 2021, 50: 2968–2983
Cheng JK, Xiang SH, Li S, Ye L, Tan B. Chem Rev, 2021, 121: 4805–4902
Liu CX, Zhang WW, Yin SY, Gu Q, You SL. J Am Chem Soc, 2021, 143: 14025–14040
He XL, Wang C, Wen YW, Wang Z, Qian S. ChemCatChem, 2021, 13: 3547–3564
Wu YJ, Liao G, Shi BF. Green Synthesis Catal, 2022, 3: 117–136
Rodríguez-Salamanca P, Fernández R, Hornillos V, Lassaletta JM. Chem Eur J, 2022, 28: e202104442
Norton RS, Wells RJ. J Am Chem Soc, 1982, 104: 3628–3635
Ito C, Thoyama Y, Omura M, Kajiura I, Furukawa H. Chem Pharm Bull, 1993, 41: 2096–2100
Zhang Q, Mándi A, Li S, Chen Y, Zhang W, Tian X, Zhang H, Li H, Zhang W, Zhang S, Ju J, Kurtán T, Zhang C. Eur Org Chem, 2012, 2012(27): 5256–5262
Jiang F, Chen KW, Wu P, Zhang YC, Jiao Y, Shi F. Angew Chem Int Ed, 2019, 58: 15104–15110
Wang CS, Li TZ, Liu SJ, Zhang YC, Deng S, Jiao Y, Shi F. Chin Chem, 2020, 38: 543–552
Chen KW, Chen ZH, Yang S, Wu SF, Zhang YC, Shi F. Angew Chem Int Ed, 2022, 61: e202116829
Xia W, An QJ, Xiang SH, Li S, Wang YB, Tan B. Angew Chem Int Ed, 2020, 59: 6775–6779
Baumann T, Brückner R. Angew Chem Int Ed, 2019, 58: 4714–4719
He T, Peng L, Li S, Hu F, Xie C, Huang S, Jia S, Qin W, Yan H. Org Lett, 2020, 22: 6966–6971
Ma C, Sheng FT, Wang HQ, Deng S, Zhang YC, Jiao Y, Tan W, Shi F. Am Chem Soc, 2020, 142: 15686–15696
Liu SJ, Chen ZH, Chen JY, Ni SF, Zhang YC, Shi F. Angew Chem Int Ed, 2022, 61: e202112226
Li TZ, Liu SJ, Tan W, Shi F. Chem Eur J, 2020, 26: 15779–15792
Ototake N, Morimoto Y, Mokuya A, Fukaya H, Shida Y, Kitagawa O. Chem Eur J, 2010, 16: 6752–6755
Wang L, Zhong J, Lin X. Angew Chem Int Ed, 2019, 58: 15824–15828
Zhang J, Xu Q, Wu J, Fan J, Xie M. Org Lett, 2019, 21: 6361–6365
Sun L, Chen H, Liu B, Chang J, Kong L, Wang F, Lan Y, Li X. Angew Chem Int Ed, 2021, 60: 8391–8395
Kim A, Kim A, Park S, Kim S, Jo H, Ok KM, Lee SK, Song J, Kwon Y. Angew Chem Int Ed, 2021, 60: 12279–12283
Ren Q, Cao T, He C, Yang M, Liu H, Wang L. ACS Catal, 2021, 11: 6135–6140
Wang F, Jing J, Zhao Y, Zhu X, Zhang XP, Zhao L, Hu P, Deng WQ, Li X. Angew Chem Int Ed, 2021, 60: 16628–16633
Mi R, Chen H, Zhou X, Li N, Ji D, Wang F, Lan Y, Li X. Angew Chem Int Ed, 2022, 61: e202111860
Wang ZS, Zhu LJ, Li CT, Liu BY, Hong X, Ye LW. Angew Chem Int Ed, 2022, 61: e202201436
Zhang HH, Wang CS, Li C, Mei GJ, Li Y, Shi F. Angew Chem Int Ed, 2017, 56: 116–121
Qi LW, Mao JH, Zhang J, Tan B. Nat Chem, 2018, 10: 58–64
Lu DL, Chen YH, Xiang SH, Yu P, Tan B, Li S. Org Lett, 2019, 21: 6000–6004
Bisag GD, Pecorari D, Mazzanti A, Bernardi L, Fochi M, Bencivenni G, Bertuzzi G, Corti V. Chem Eur J, 2019, 25: 15694–15701
Ding WY, Yu P, An QJ, Bay KL, Xiang SH, Li S, Chen Y, Houk KN, Tan B. Chem, 2020, 6: 2046–2059
Mao JH, Wang YB, Yang L, Xiang SH, Wu QH, Cui Y, Lu Q, Lv J, Li S, Tan B. Nat Chem, 2021, 13: 982–991
Xu WL, Zhao WM, Zhang RX, Chen J, Zhou L. Chem Sci, 2021, 12: 14920–14926
Yang H, Sun HR, He RQ, Yu L, Hu W, Chen J, Yang S, Zhang GG, Zhou L. Nat Commun, 2022, 13: 632
Wu P, Yu L, Gao CH, Cheng Q, Deng S, Jiao Y, Tan W, Shi F. Fund Res, 2022, doi: doi: doi: https://doi.org/10.1016/j.fmre.2022.01.002
For metal-catalysis: He C, Hou M, Zhu Z, Gu Z. ACS Catal, 2017, 7: 5316–5320
He XL, Zhao HR, Song X, Jiang B, Du W, Chen YC. ACS Catal, 2019, 9: 4374–4381
Xi CC, Zhao XJ, Tian JM, Chen ZM, Zhang K, Zhang FM, Tu YQ, Dong JW. Org Lett, 2020, 22: 4995–5000
Shaaban S, Li H, Otte F, Strohmann C, Antonchick AP, Waldmann H. Org Lett, 2020, 22: 9199–9202
Li X, Zhao L, Qi Z, Li X. Org Lett, 2021, 23: 5901–5905
Wang CS, Wei L, Fu C, Wang XH, Wang CJ. Org Lett, 2021, 23: 7401–7406
Hu YL, Wang Z, Yang H, Chen J, Wu ZB, Lei Y, Zhou L. Chem Sci, 2019, 10: 6777–6784
Peng L, Li K, Xie C, Li S, Xu D, Qin W, Yan H. Angew Chem Int Ed, 2019, 58: 17199–17204
He YP, Wu H, Wang Q, Zhu J. Angew Chem Int Ed, 2020, 59: 2105–2109
Xu D, Huang S, Hu F, Peng L, Jia S, Mao H, Gong X, Li F, Qin W, Yan H. CCS Chem, 2022, 4: 2686–2697
Zou Y, Wang P, Kong L, Li X. Org Lett, 2022, 24: 3189–3193
Ma C, Jiang F, Sheng FT, Jiao Y, Mei GJ, Shi F. Angew Chem Int Ed, 2019, 58: 3014–3020
Sheng FT, Li ZM, Zhang YZ, Sun LX, Zhang YC, Tan W, Shi F. Chin J Chem, 2020, 38: 583–589
Sheng FT, Yang S, Wu SF, Zhang YC, Shi F. Chin J Chem, 2022, 40: 2151–2160
Tian M, Bai D, Zheng G, Chang J, Li X. J Am Chem Soc, 2019, 141: 9527–9532
Wang JY, Sun M, Yu XY, Zhang YC, Tan W, Shi F. Chin J Chem, 2021, 39: 2163–2171
Zhu S, Chen YH, Wang YB, Yu P, Li SY, Xiang SH, Wang JQ, Xiao J, Tan B. Nat Commun, 2019, 10: 4268
Chen YH, Li HH, Zhang X, Xiang SH, Li S, Tan B. Angew Chem Int Ed, 2020, 59: 11374–11378
Zhang YC, Jiang F, Shi F. Acc Chem Res, 2020, 53: 425–446
Hang QQ, Liu SJ, Yu L, Sun TT, Zhang YC, Mei GJ, Shi F. Chin J Chem, 2020, 38: 1612–1618
Li TZ, Liu SJ, Sun YW, Deng S, Tan W, Jiao Y, Zhang YC, Shi F. Angew Chem Int Ed, 2021, 60: 2355–2363
Wang JY, Zhang S, Yu XY, Wang YH, Wan HL, Zhang S, Tan W, Shi F. Tetrahedron Chem, 2022, 1: 100007
Manoni F, Connon SJ. Angew Chem Int Ed, 2014, 53: 2628–2632
Wu JL, Du BX, Zhang YC, He YY, Wang JY, Wu P, Shi F. Adv Synth Catal, 2016, 358: 2777–2790
Jarvis CL, Hirschi JS, Vetticatt MJ, Seidel D. Angew Chem Int Ed, 2017, 56: 2670–2674
Jolliffe JD, Armstrong RJ, Smith MD. Nat Chem, 2017, 9: 558–562
Kumarasamy E, Raghunathan R, Sibi MP, Sivaguru J. Chem Rev, 2015, 115: 11239–11300
CCDC 2173719 (3aa) contains the Supporting Information
Acknowledgements
This work was supported by the National Science Foundation of China (22125104, 21831007, 22101103) and the Natural Science Foundation of Jiangsu Province (BK20201018). We are grateful to Prof. Shu Zhang and Prof. Yinchun Jiao for their kind help in biological evaluation and theoretical calculation on the rotational barrier.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at http://chem.scichina.com and http://springerlink.bibliotecabuap.elogim.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
Rights and permissions
About this article
Cite this article
Hang, QQ., Wu, SF., Yang, S. et al. Design and catalytic atroposelective synthesis of axially chiral isochromenone-indoles. Sci. China Chem. 65, 1929–1937 (2022). https://doi.org/10.1007/s11426-022-1363-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1363-y