Abstract
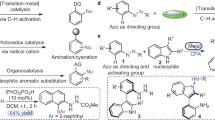
Asymmetric synthesis of enantioenriched azo compounds bearing tetrasubstituted stereocenter was achieved through chiral N,N′-dioxide/metal Lewis acid promoted interrupted Japp-Klingemann reaction of aryldiazonium tetrafluoroborate salts with nucleophiles under mild conditions. This protocol features wide substrate scope and good functional group compatibility. Azaarene-containing chiral azo compounds were stable enough in Japp-Klingemann reaction condition. The key to success of the reaction was the employment of metal salt/N,N′-dioxide ligand and the dual-task roles of the base. Moreover, the X-ray crystal structure of Ni(II)/N,N′-dioxide/substrate complex confirmed that the substrate was activated by bidentate coordination, which shed light on the origin of chiral control of the reaction.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Li JJ. In Name Reactions: A Collection of Detailed Reaction Mechanisms. Berlin: Springer, 2009. 302
Japp FR, Klingemann F. Ber, 1887, 20: 2942
Japp FR, Klingemann F. Ber, 1887, 20: 3284
Japp FR, Klingemann F. Ber, 1887, 20: 3398
Humphrey GR, Kuethe JT. Chem Rev, 2006, 106: 2875–2911
Mo F, Qiu D, Zhang Y, Wang J. Acc Chem Res, 2018, 51: 496–506
Mo F, Qiu D, Zhang L, Wang J. Chem Rev, 2021, 121: 5741–5829
Li L, Chen JJ, Li YJ, Bu XB, Liu Q, Zhao YL. Angew Chem Int Ed, 2015, 54: 12107–12111
Arkhipov AV, Arkhipov VV, Cossy J, Kovtunenko VO, Mykhailiuk PK. Org Lett, 2016, 18: 3406–3409
Wu D, Wang Y, Zhou J, Sun Q, Zhao Y, Xu X. Org Lett, 2019, 21: 8722–8725
Li W, Liu X, Hao X, Hu X, Chu Y, Cao W, Qin S, Hu C, Lin L, Feng X. J Am Chem Soc, 2011, 133: 15268–15271
Nelson HM, Patel JS, Shunatona HP, Toste FD. Chem Sci, 2015, 6: 170–173
Merino E. Chem Soc Rev, 2011, 40: 3835
Bafana A, Devi SS, Chakrabarti T. Environ Rev, 2011, 19: 350–371
Breul AM, Hager MD, Schubert US. Chem Soc Rev, 2013, 42: 5366
Dong M, Babalhavaeji A, Samanta S, Beharry AA, Woolley GA. Acc Chem Res, 2015, 48: 2662–2670
Cao B, Yang M, Mao C. Acc Chem Res, 2016, 49: 1111–1120
Garg RP, Qian XL, Alemany LB, Moran S, Parry RJ. Proc Natl Acad Sci USA, 2008, 105: 6543–6547
Cassano R, Trombino S, Cilea A, Ferrarelli T, Muzzalupo R, Picci N. Chem Pharm Bull, 2010, 58: 103–105
Kennedy DA, Vembu N, Fronczek FR, Devocelle M. J Org Chem, 2011, 76: 9641–9647
Dietz FR, Prechter A, Gröger H, Heinrich MR. Tetrahedron Lett, 2011, 52: 655–657
Movassaghi M, Ahmad OK, Lathrop SP. J Am Chem Soc, 2011, 133: 13002–13005
Blair LM, Sperry J. J Nat Prod, 2013, 76: 794–812
Jain A, Gupta Y, Jain SK. Crit Rev Ther Drug Carrier Syst, 2006, 23: 349–400
Dana S, Sahoo H, Bhattacharyya A, Mandal A, Prasad E, Baidya M. ChemistrySelect, 2017, 2: 2029–2033
Sultan HA, Dhumad AM, Hassan QMA, Fahad T, Emshary CA, Raheem NA. Spectrochim Acta Part A-Mol Biomol Spectr, 2021, 251: 119487
Crespo-Peña A, Monge D, Martín-Zamora E, Alvarez E, Fernández R, Lassaletta JM. J Am Chem Soc, 2012, 134: 12912–12915
Fernández M, Uria U, Vicario JL, Reyes E, Carrillo L. J Am Chem Soc, 2012, 134: 11872–11875
Nelson HM, Reisberg SH, Shunatona HP, Patel JS, Toste FD. Angew Chem Int Ed, 2014, 53: 5600–5603
Liu XH, Lin LL, Feng XM. Acc Chem Res, 2011, 44: 574–587
Liu X, Zheng H, Xia Y, Lin L, Feng X. Acc Chem Res, 2017, 50: 2621–2631
Zheng K, Liu X, Feng X. Chem Rev, 2018, 118: 7586–7656
For selected examples of azaarene-containing compounds, see: (a) Vitaku E, Smith DT, Njardarson JT. J Med Chem, 2014, 57: 10257–10274
Keri RS, Patil MR, Patil SA, Budagumpi S. Eur J Med Chem, 2015, 89: 207–251
Yin Y, Li Y, Gonçalves TP, Zhan Q, Wang G, Zhao X, Qiao B, Huang KW, Jiang Z. J Am Chem Soc, 2020, 142: 19451–19456
Wang SQ, Liu ZC, Yue WJ, Yin L. Angew Chem Int Ed, 2021, 60: 4604–4608
Chang X, Yang Y, Shen C, Xue KS, Wang ZF, Cong H, Tao HY, Chung LW, Wang CJ. J Am Chem Soc, 2021, 143: 3519–3535
de Angelis L, Crawford AM, Su YL, Wherritt D, Arman H, Doyle MP. Org Lett, 2021, 23: 925–929
Bertuzzi G, Thøgersen MK, Giardinetti M, Vidal-Albalat A, Simon A, Houk KN, Jørgensen KA. J Am Chem Soc, 2019, 141: 3288–3297
Zhao P, Li Z, He J, Liu X, Feng X. Sci China Chem, 2021, 64: 1355–1360
Pan GH, He CL, Chen M, Xiong Q, Cao WD, Feng XM. CCS Chem, 2021, 3: 2012
Cahard D, Xu X, Couve-Bonnaire S, Pannecoucke X. Chem Soc Rev, 2010, 39: 558–568
Lectard S, Hamashima Y, Sodeoka M. Adv Synth Catal, 2010, 352: 2708–2732
Zhu Y, Han J, Wang J, Shibata N, Sodeoka M, Soloshonok VA, Coelho JAS, Toste FD. Chem Rev, 2018, 118: 3887–3964
He ZT, Jiang X, Hartwig JF. J Am Chem Soc, 2019, 141: 13066–13073
Yang SQ, Wang YF, Zhao WC, Lin GQ, He ZT. J Am Chem Soc, 2021, 143: 7285–7291
CCDC 2038543 (3a) contains the Supporting Information
CCDC 2097126 (6c) contains the Supporting Information
Berrisford DJ, Bolm C, Sharpless KB. Angew Chem Int Ed, 1995, 34: 1059–1070
Abramovitch RA, Saha JG. Tetrahedron, 1965, 21: 3297–3303
Shaaban S, Jolit A, Petkova D, Maulide N. Chem Commun, 2015, 51: 13902–13905
Felipe-Blanco D, Gonzalez-Gomez JC. Eur J Org Chem, 2019, 2019(47): 7735–7744
CCDC 2091454 (Ni(II)/L-PiEt2/1a) contains the Supporting Information
He X, Zhang Q, Wang W, Lin L, Liu X, Feng X. Org Lett, 2011, 13: 804–807
Zhou L, Liu X, Ji J, Zhang Y, Hu X, Lin L, Feng X. J Am Chem Soc, 2012, 134: 17023–17026
Liu Y, Liu X, Hu H, Guo J, Xia Y, Lin L, Feng X. Angew Chem Int Ed, 2016, 55: 4054–4058
Hu H, Liu Y, Lin L, Zhang Y, Liu X, Feng X. Angew Chem Int Ed, 2016, 55: 10098–10101
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21890723, 21921002).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflict of interest
The authors declare no conflict of interest.
Supporting information
The supporting information is available online at http://chem.scichina.com and http://springerlink.bibliotecabuap.elogim.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Electronic Supplementary Material
11426_2021_1149_MOESM1_ESM.pdf
Catalytic asymmetric synthesis of chiral azo compounds via interrupted Japp-Klingemann reaction with aryldiazonium salts
Rights and permissions
About this article
Cite this article
Wang, Y., Yihuo, A., Wang, L. et al. Catalytic asymmetric synthesis of chiral azo compounds via interrupted Japp-Klingemann reaction with aryldiazonium salts. Sci. China Chem. 65, 546–553 (2022). https://doi.org/10.1007/s11426-021-1149-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-021-1149-1