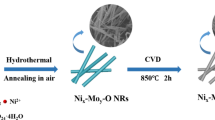
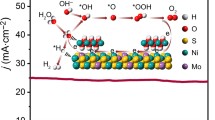
Abstract
Hydrogen is one of the most promising energy carriers to replace fossil fuels and electrolyzing water to produce hydrogen is a very effective method. However, designing highly active and stable non-precious metal hydrogen evolution electrocatalysts that can be used in universal pH is a huge challenge. Here, we have reported a simple strategy to develop a highly active and durable non-precious MoO2-Ni electrocatalyst for hydrogen evolution reaction (HER) in a wide pH range. The MoO2-Ni catalyst exhibits a superior electrocatalytic performance with low overpotentials of 46, 69, and 84 mV to reach -10 mA cm-2 in 1.0 M KOH, 0.5 M H2SO4, and 1.0 M PBS electrolytes, respectively. At the same time, the catalyst also shows outstanding stability over a wide pH range. It is particularly noted that the catalytic performance of MoO2-Ni in alkaline solution is comparable to the highest performing catalysts reported. The outstanding HER performance is mainly attributed to the collective effect of the rational morphological design, electronic structure engineering, and strong interfacial coupling between MoO2 and Ni in heterojunctions. This work provides a viable method for the synthesis of inexpensive and efficient HER electrocatalysts for the use in wide pH ranges.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lai J, Huang B, Chao Y, Chen X, Guo S. Adv Mater, 2019, 31: 1805541
Shi Y, Zhou Y, Yang DR, Xu WX, Wang C, Wang FB, Xu JJ, Xia XH, Chen HY. J Am Chem Soc, 2017, 139: 15479–15485
Song F, Li W, Yang J, Han G, Liao P, Sun Y. Nat Commun, 2018, 9: 4531
Zhou P, He J, Zou Y, Wang Y, Xie C, Chen R, Zang S, Wang S. Sci China Chem, 2019, 62: 1365–1370
Elbert K, Hu J, Ma Z, Zhang Y, Chen G, An W, Liu P, Isaacs HS, Adzic RR, Wang JX. ACS Catal, 2015, 5: 6764–6772
Chen Z, Song Y, Cai J, Zheng X, Han D, Wu Y, Zang Y, Niu S, Liu Y, Zhu J, Liu X, Wang G. Angew Chem Int Ed, 2018, 57: 5076–5080
Zhou M, Weng Q, Popov ZI, Yang Y, Antipina LY, Sorokin PB, Wang X, Bando Y, Golberg D. ACS Nano, 2018, 12: 4148–4155
Li J, Chen J, Wang Q, Cai WB, Chen S. Chem Mater, 2017, 29: 10060–10067
Chen J, Liu J, Xie JQ, Ye H, Fu XZ, Sun R, Wong CP. Nano Energy, 2019, 56: 225–233
Liu B, Huo L, Gao Z, Zhi G, Zhang G, Zhang J. Small, 2017, 13: 1700092
Liu X, Li W, Zou S. J Mater Chem A, 2018, 6: 17067–17074
Saat G, Balci FM, Alsaç EP, Karadas F, Dag Ö. Small, 2018, 14: 1701913
Yang ZD, Chang ZW, Xu JJ, Yang XY, Zhang XB. Sci China Chem, 2017, 60: 1540–1545
Zhang C, Hou M, Cai X, Lin J, Liu X, Wang R, Zhou L, Gao J, Li B, Lai L. J Mater Chem A, 2018, 6: 15630–15639
Wang M, Li Z, Wang C, Zhao R, Li C, Guo D, Zhang L, Yin L. Adv Funct Mater, 2017, 27: 1701014
Zhou M, Liu Z, Song Q, Li X, Chen B, Liu Z. Appl Catal B-Environ, 2019, 244: 188–196
Chen L, Zhang J, Ren X, Ge R, Teng W, Sun X, Li X. Nanoscale, 2017, 9: 16632–16637
Zhang ZS, Fu XP, Wang WW, Jin Z, Song QS, Jia CJ. Sci China Chem, 2018, 61: 1389–1398
Qi J, Zhang W, Cao R. Chem Commun, 2017, 53: 9277–9280
Ji D, Peng L, Shen J, Deng M, Mao Z, Tan L, Wang M, Xiang R, Wang J, Shah SSA. Chem Commun, 2019, 55: 3290–3293
Kauppinen MM, Melander MM, Bazhenov AS, Honkala K. ACS Catal, 2018, 8: 11633–11647
Yan H, Qin XT, Yin Y, Teng YF, Jin Z, Jia CJ. Appl Catal B-Environ, 2018, 226: 182–193
Gong M, Zhou W, Tsai MC, Zhou J, Guan M, Lin MC, Zhang B, Hu Y, Wang DY, Yang J, Pennycook SJ, Hwang BJ, Dai H. Nat Commun, 2014, 5: 4695
Liu X, Ni K, Niu C, Guo R, Xi W, Wang Z, Meng J, Li J, Zhu Y, Wu P, Li Q, Luo J, Wu X, Mai L. ACS Catal, 2019, 9: 2275–2285
Zhang R, Wang X, Yu S, Wen T, Zhu X, Yang F, Sun X, Wang X, Hu W. Adv Mater, 2017, 29: 1605502
Adam A, Suliman MH, Siddiqui MN, Yamani ZH, Merzougui B, Qamar M. ACS Appl Mater Interfaces, 2018, 10: 29407–29416
Lu XF, Yu L, Lou XWD. Sci Adv, 2019, 5: eaav6009
Mahmood J, Li F, Jung SM, Okyay MS, Ahmad I, Kim SJ, Park N, Jeong HY, Baek JB. Nat Nanotech, 2017, 12: 441–446
Yao RQ, Zhou YT, Shi H, Zhang QH, Gu L, Wen Z, Lang XY, Jiang Q. ACS Energy Lett, 2019, 4: 1379–1386
Zheng J, Sheng W, Zhuang Z, Xu B, Yan Y. Sci Adv, 2016, 2: e1501602
Sheng W, Zhuang Z, Gao M, Zheng J, Chen JG, Yan Y. Nat Commun, 2015, 6: 5848
Li Q, Zou X, Ai X, Chen H, Sun L, Zou X. Adv Energy Mater, 2018, 1803369
Mishra IK, Zhou H, Sun J, Qin F, Dahal K, Bao J, Chen S, Ren Z. Energy Environ Sci, 2018, 11: 2246–2252
Yao Q, Huang B, Zhang N, Sun M, Shao Q, Huang X. Angew Chem Int Ed, 2019, 58: 13983–13988
Kumar P, Singh M, Reddy GB. Mater Res Express, 2017, 4: 036405
Ku JH, Jung YS, Lee KT, Kim CH, Oh SM. J Electrochem Soc, 2009, 156: A688
Li Z, Yu C, Wen Y, Gao Y, Xing X, Wei Z, Sun H, Zhang YW, Song W. ACS Catal, 2019, 9: 5084–5095
Yang L, Yu J, Wei Z, Li G, Cao L, Zhou W, Chen S. Nano Energy, 2017, 41: 772–779
Guo J, Wu C, Zhang J, Yan P, Tian J, Shen X, Isimjan TT, Yang X. J Mater Chem A, 2019, 7: 8865–8872
Guo J, Wang B, Yang D, Wan Z, Yan P, Tian J, Isimjan TT, Yang X. Appl Catal B-Environ, 2020, 265: 118584
Jin H, Liu X, Jiao Y, Vasileff A, Zheng Y, Qiao SZ. Nano Energy, 2018, 53: 690–697
Wan J, Wu J, Gao X, Li T, Hu Z, Yu H, Huang L. Adv Funct Mater, 2017, 27: 1703933
Zhou Y, Huang W, Zhang X, Wang M, Zhang L, Shi J. Chem Eur J, 2017, 23: 17029–17036
Ito Y, Ohto T, Hojo D, Wakisaka M, Nagata Y, Chen L, Hu K, Izumi M, Fujita J, Adschiri T. ACS Catal, 2018, 8: 3579–3586
Chi JQ, Chai YM, Shang X, Dong B, Liu CG, Zhang W, Jin Z. J Mater Chem A, 2018, 6: 24783–24792
Boppella R, Tan J, Yang W, Moon J. Adv Funct Mater, 2019, 29: 1807976
Yang L, Zeng L, Liu H, Deng Y, Zhou Z, Yu J, Liu H, Zhou W. Appl Catal B-Environ, 2019, 249: 98–105
Xiao P, Ge X, Wang H, Liu Z, Fisher A, Wang X. Adv Funct Mater, 2015, 25: 1520–1526
Youn DH, Han S, Kim JY, Kim JY, Park H, Choi SH, Lee JS. ACS Nano, 2014, 8: 5164–5173
Yang X, Lu AY, Zhu Y, Hedhili MN, Min S, Huang KW, Han Y, Li LJ. Nano Energy, 2015, 15: 634–641
Qi Y, Zhang L, Sun L, Chen G, Luo Q, Xin H, Peng J, Li Y, Ma F. Nanoscale, 2020, 12: 1985–1993
Chen YY, Zhang Y, Zhang X, Tang T, Luo H, Niu S, Dai ZH, Wan LJ, Hu JS. Adv Mater, 2017, 29: 1703311
Yang X, Lu AY, Zhu Y, Min S, Hedhili MN, Han Y, Huang KW, Li LJ. Nanoscale, 2015, 7: 10974–10981
Li Y, Wang H, Xie L, Liang Y, Hong G, Dai H. J Am Chem Soc, 2011, 133: 7296–7299
Acknowledgments
This work has been supported by the National Natural Science Foundation of China (21965005), Natural Science Foundation of Guangxi Province (2018GXNSFAA294077 and 2018GXNSFAA281220), Project of High-Level Talents of Guangxi (F-KA18015 and 2018ZD004).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflict of interest
The authors declare that they have no conflict of interest.
Supporting information
The supporting information is available online at http://chem.scichina.com and http://springerlink.bibliotecabuap.elogim.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, B., Huang, H., Huang, M. et al. Electron-transfer enhanced MoO2-Ni heterostructures as a highly efficient pH-universal catalyst for hydrogen evolution. Sci. China Chem. 63, 841–849 (2020). https://doi.org/10.1007/s11426-019-9721-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-019-9721-0