Abstract
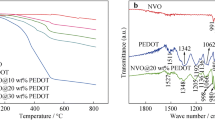
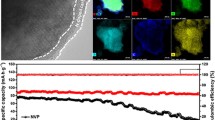
The key to the development of sodium ion battery is materials with a high rate capacity and cycle stability. Conducting coating is an efficient approach to improve electrochemical performance. As a case study, the Na3V2(PO4)3@PEDOT composite was prepared through an in-situ self-decorated conducting polymer route without further calcination. The Na3V2(PO4)3 electrode with a 7% poly(3,4-ethylenedioxythiophene) (PEDOT) coating can deliver an initial reversible capacity of 100 mA h g−1 at 1 cycle, and 82% capacity retention over 200 cycles. The results also show that the Na3V2(PO4)3 electrode without and with a thick PEDOT coating exhibits poor electrochemical performance, indicating that an appropriate coating layer is important for improving electronic conductivity and regulating Na-ion insertion. Therefore, this work offers possibility to promote the electrochemical performance of poor-conducting materials in sodium-ion batteries using an in-situ self-decorated conducting polymer.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Dunn B, Kamath H, Tarascon JM. Science, 2011, 334: 928–935
Li H, Wang Z, Chen L, Huang X. Adv Mater, 2009, 21: 4593–4607
Goodenough JB, Kim Y. Chem Mater, 2010, 22: 587–603
Palomares V, Casas-Cabanas M, Castillo-Martínez E, Han MH, Rojo T. Energy Environ Sci, 2013, 6: 2312
Ellis BL, Nazar LF. Curr Opin Solid State Mater Sci, 2012, 16: 168–177
Guan W, Pan B, Zhou P, Mi J, Zhang D, Xu J, Jiang Y. ACS Appl Mater Interfaces, 2017, 9: 22369–22377
Li S, Guo J, Ye Z, Zhao X, Wu S, Mi JX, Wang CZ, Gong Z, McDonald MJ, Zhu Z, Ho KM, Yang Y. ACS Appl Mater Interfaces, 2016, 8: 17233–17238
Chen CY, Matsumoto K, Nohira T, Hagiwara R. Electrochem Commun, 2014, 45: 63–66
Treacher JC, Wood SM, Islam MS, Kendrick E. Phys Chem Chem Phys, 2016, 18: 32744–32752
Wei S, Mortemard de Boisse B, Oyama G, Nishimura S, Yamada A. ChemElectroChem, 2016, 3: 209–213
Barpanda P, Oyama G, Nishimura SI, Chung SC, Yamada A. Nat Commun, 2014, 5: 4358–4366
Singh P, Shiva K, Celio H, Goodenough JB. Energy Environ Sci, 2015, 8: 3000–3005
Rajagopalan R, Chen B, Zhang Z, Wu XL, Du Y, Huang Y, Li B, Zong Y, Wang J, Nam GH, Sindoro M, Dou SX, Liu HK, Zhang H. Adv Mater, 2017, 29: 1605694
Zhu Q, Chang X, Sun N, Liu H, Chen R, Wu F, Xu B. J Mater Chem A, 2017, 5: 9982–9990
Barpanda P, Ye T, Avdeev M, Chung SC, Yamada A. J Mater Chem A, 2013, 1: 4194–4197
Kim H, Shakoor RA, Park C, Lim SY, Kim JS, Jo YN, Cho W, Miyasaka K, Kahraman R, Jung Y, Choi JW. Adv Funct Mater, 2013, 23: 1147–1155
Barpanda P, Liu G, Mohamed Z, Ling CD, Yamada A. Solid State Ion, 2014, 268: 305–311
Jian Z, Zhao L, Pan H, Hu YS, Li H, Chen W, Chen L. Electrochem Commun, 2012, 14: 86–89
Jung YH, Lim CH, Kim DK. J Mater Chem A, 2013, 1: 11350
Saravanan K, Mason CW, Rudola A, Wong KH, Balaya P. Adv Energy Mater, 2013, 3: 444–450
Zhu C, Song K, van Aken PA, Maier J, Yu Y. Nano Lett, 2014, 14: 2175–2180
Uebou Y, Kiyabu T, Okada S, Yamaki J-i. The Reports of Institute of Advanced Material Study. Fukuoka: Kyushu University, 2002. 1–5
Lim SY, Kim H, Shakoor RA, Jung Y, Choi JW. J Electrochem Soc, 2012, 159: A1393–A1397
Zhang W, Liu Y, Chen C, Li Z, Huang Y, Hu X. Small, 2015, 11: 3822–3829
Zhang J, Fang Y, Xiao L, Qian J, Cao Y, Ai X, Yang H. ACS Appl Mater Interfaces, 2017, 9: 7177–7184
Guo JZ, Wu XL, Wan F, Wang J, Zhang XH, Wang RS. Chem Eur J, 2015, 21: 17371–17378
Jiang Y, Yang Z, Li W, Zeng L, Pan F, Wang M, Wei X, Hu G, Gu L, Yu Y. Adv Energy Mater, 2015, 5: 1402104
Li S, Dong Y, Xu L, Xu X, He L, Mai L. Adv Mater, 2014, 26: 3545–3553
Li H, Yu X, Bai Y, Wu F, Wu C, Liu LY, Yang XQ. J Mater Chem A, 2015, 3: 9578–9586
Chekannikov A, Kapaev R, Novikova S, Tabachkova N, Kulova T, Skundin A, Yaroslavtsev A. J Solid State Electrochem, 2017, 21: 1615–1624
Fang Y, Xiao L, Ai X, Cao Y, Yang H. Adv Mater, 2015, 27: 5895–5900
Rui X, Sun W, Wu C, Yu Y, Yan Q. Adv Mater, 2015, 27: 6670–6676
Vadivel Murugan A, Muraliganth T, Manthiram A. Electrochem Commun, 2008, 10: 903–906
Park KS, Schougaard SB, Goodenough JB. Adv Mater, 2007, 19: 848–851
Kim J, Yoo JK, Jung YS, Kang K. Adv Energy Mater, 2013, 3: 1004–1007
Lepage D, Michot C, Liang G, Gauthier M, Schougaard SB. Angew Chem Int Ed, 2011, 50: 6884–6887
Nie P, Zhu Y, Shen L, Pang G, Xu G, Dong S, Dou H, Zhang X. J Mater Chem A, 2014, 2: 18606–18612
Chiu WW, Travaš-Sejdić J, Cooney RP, Bowmaker GA. J Raman Spectrosc, 2006, 37: 1354–1361
Selvaganesh SV, Mathiyarasu J, Phani KLN, Yegnaraman V. Nanoscale Res Lett, 2007, 2: 546–549
Garreau S, Louarn G, Buisson JP, Froyer G, Lefrant S. Macromolecules, 1999, 32: 6807–6812
Wu CZ, Xie Y, Lei LY, Hu SQ, OuYang CZ. Adv Mater, 2006, 18: 1727–1732
Chen Q, Wang J, Tang Z, He W, Shao H, Zhang J. Electrochim Acta, 2007, 52: 5251–5257
Silversmit G, Depla D, Poelman H, Marin GB, De Gryse R. J Electron Spectrosc Relat Phenom, 2004, 135: 167–175
Conte M, Budroni G, Bartley JK, Taylor SH, Carley AF, Schmidt A, Murphy DM, Girgsdies F, Ressler T, Schlögl R, Hutchings GJ. Science, 2006, 313: 1270–1273
Acknowledgments
This work was supported by the National Key Research Program of China (2016YFB0100400), and the National Natural Science Foundation of China (21373155, 21333007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J., Yuan, T., Wan, H. et al. Surface-engineering enhanced sodium storage performance of Na3V2(PO4)3 cathode via in-situ self-decorated conducting polymer route. Sci. China Chem. 60, 1546–1553 (2017). https://doi.org/10.1007/s11426-017-9125-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-017-9125-y