Abstract
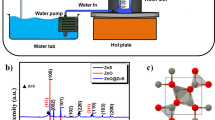
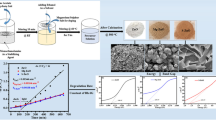
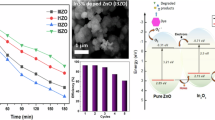
Low-cost and scalable preparation, high photocatalytic activity, and convenient recycle of ZnO nanopowders (NPs) would determine their practical application in purifying wastewater. In this contribution, ZnO NPs were scalably synthesized via the simple reaction of Zn powder with H2O vapor in autoclave. The structural, morphological and optical properties of the samples were systematically characterized by X-ray diffraction, scanning electron microscopy, Fourier transform infrared spectra, transmission electron microscopy, Micro-Raman, photoluminescence, and ultraviolet-visible spectroscopy. The as-prepared ZnO NPs are composed of nanoparticles with 100–150 nm in diameter, and have a small Brunauer-Emmett-Teller surface area of 6.85 m2/g. The formation of ZnO nanoparticles is relative to the peeling of H2 release. Furthermore, the product has big strain-stress leading to the red-shift in the band gap of product, and shows a strong green emission centered at 515 nm revealing enough atomic defects in ZnO NPs. As a comparison with P25, the obtained dust gray ZnO NPs have a strong absorbance in the region of 200–700 nm, suggesting the wide wave-band utilization in sunlight. Based on the traits above, the ZnO NPs show excellent photocatalytic activity on the degradation of rhodamine B (Rh-B) under solar light irradiation, close to that under UV irradiation. Importantly, the ZnO NPs could be well recycled in water due to the quick sedimentation in themselves in solution. The low-cost and scalable preparation, high photocatalytic activity, and convenient recycle of ZnO NPs endow themselves with promising application in purifying wastewater.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Khin MM, Nair AS, Babu V, Murugan R, Ramakrishna S. Energy Environ Sci, 2012, 5: 8075–8109
Chan SHS, Wu TY, Juan JC, Teh CY. J Chem Tech Biotech, 2011, 86: 1130–1158
Ullah I, Ali S, Hanif MA, Shahid SA. Intern J Chem Biochem Sci, 2012, 2: 60–77
Zhang DQ, Li GS, Yu JC. Advanced photocatalytic nanomaterials for degrading pollutants and generating fuels by sunlight. In: Zhang L, Ed. Energy Efficiency and Renewable Energy Through Nanotechnology. London: Springer London, 2011. 679–716
Chen X, Mao SS. Chem Rev, 2007, 107: 2891–2959
Li Y, Zhou X, Hu X, Zhao X, Fang P. J Phys Chem C, 2009, 113: 16188–16192
Behnajady MA, Modirshahla N, Hamzavi R. J Hazard Mater, 2006, 133: 226–232
Xu LP, Hu YL, Pelligra C, Chen CH, Jin L, Huang H, Sithambaram S, Aindow M, Joesten R, Sui SL. Chem Mater, 2009, 21: 2875–2885
Meng SG, Li DZ, Zheng XZ, Wang JX, Chen J, Fang JL, Shao Y, Fu XZ. J Mater Chem A, 2013, 1: 2744–2747
Dijken AV, Janssen AH, Smitsmans HP, Vanmaekelbergh D, Meijerink A. Chem Mater, 1998, 10: 3513–3522
Pudukudy M, Hetieqa A, Yaakob Z. Appl Surf Sci, 2014, 319: 221–229
Meulenkamp EA. J Phys Chem B, 1998, 102: 7764–7769
Velmurugan R, Selvam K, Krishnakumar B, Swaminathan M. Sep Purif Technol, 2011, 80: 119–124
Subash B, Krishnakumar B, Swaminathan M, Shanthi M. Langmuir, 2013, 29: 939–949
Tan TV, Río Ld, Valdés-Solís T, Marbán G. Appl Catal B: Environ, 2013, 140–141: 189–198
Lv YY, Yu LS, Huang HY, Feng YY, Chen DZ, Xie X. Nanotechnology, 2012, 23: 065402–065409
Perales-percz Q, Parra-palomino A, Singhal R, Voyles Y, Zhu Y, Jia W, Tomar MS. Nanotechnology, 2007, 18: 315606–315611
Liu C, Hu Z, Wu Q, Wang XZ, Chen Y, Sang H, Zhu JM, Deng SZ, Xu NS. J Am Chem Soc, 2005, 127: 1318–1322
Chen X, Ma J, Hu Z, Wu Q, Chen Y. J Am Chem Soc, 2005, 127: 7982–7983
Mclaren A, Valdes-Solis T, Li GQ, Tsang SC. J Am Chem Soc, 2009, 131: 12540–12541
Pan ZW, Dai ZR, Wang ZL. Science, 2001, 291: 1947–1949
Kruk M, Jaroniec M. Chem Mater, 2001, 13: 3169–3183
Decremps F, Porres JP, Saitta AM, Chervin JC, Polian A. Phys Rev B, 2002, 65: 092101–092104
Zhang Y, Jia HB, Wang RM, Chen CP, Luo XP, Yu DP, Lee C. Appl Phys Lett, 2003, 83: 4631–4633
Teng XM, Fan HT, Pan SS, Ye C, Li GH. J Phys D: Appl Phys, 2006, 39: 471–476
Greene LE, Law M, Goldberger J, Kim F, Johnson JC, Zhang YFR, Saykally J, Yang PD. Angew Chem Int Ed, 2003, 42: 3031–3034
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Lv, Y., Yu, L., Li, C. et al. ZnO nanopowders and their excellent solar light/UV photocatalytic activity on degradation of dye in wastewater. Sci. China Chem. 59, 142–149 (2016). https://doi.org/10.1007/s11426-015-5438-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-015-5438-2