Abstract
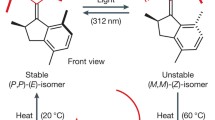
Molecular switches that can undergo reversible switching between two or more different states in response to external stimuli have been used in the fabrication of various optoelectronic devices and smart materials for many decades, and also found many applications in sensing, molecular self-assembly and photo-controlled biological systems. Recently, mechanically interlocked molecules, such as rotaxanes and catenanes, and molecular rotary motors based on overcrowded alkenes have emerged as two new kinds of molecular switches. Some novel applications of above-mentioned molecular switches have been discovered. In this mini review, we mainly highlight noticeable achievements over the past decade in this field, and summarize the applications of new types of molecular switches, for instance, controlling the chiral space to regulate catalytic reaction as organocatalysts, controlling molecular motions, synthesizing a peptide in a sequence-specific manner and modulating the wettability of the self-assembled monolayers.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Tian H, Wang S. Photochromic bisthienylethene as multi-function switches. Chem Commun, 2007: 781–792
Zhang JJ, Zou Q, Tian H. Photochromic materials: more than meets the eye. Adv Mater, 2013, 25: 378–399
Tian H, Yang SJ. Recent progresses on diarylethene based photochromic switches. Chem Soc Rev, 2004, 33: 85–97
Dong HL, Zhu HF, Meng Q, Gong X, Hu WP. Organic photoresponse materials and devices. Chem Soc Rev, 2012, 41: 1754–1808
Cui XN, Zhao JZ, Zhou YH, Ma J, Zhao YL. Reversible photoswitching of triplet-triplet annihilation upconversion using dithienylethene photochromic switches. J Am Chem Soc, 2014, 136: 9256–9259
Kudernac T, Kobayashi T, Uyama A, Uchida K, Nakamura S, Feringa BL. Tuning the temperature dependence for switching in dithienylethene photochromic switches. J Phys Chem A, 2013, 117: 8222–8229
Li B, Wang JY, Wen HM, Shi LX, Chen ZN. Redox-modulated stepwise photochromism in a ruthenium complex with dual dithienylethene-acetylides. J Am Chem Soc, 2012, 134: 16059–16067
Tian H, Feng Y. Next step of photochromic switches? J Mater Chem, 2008, 18: 1617–1622
Mengel AKC, He B, Wenger OS. A triarylamine-triarylborane dyad with a photochromic dithienylethene bridge. J Org Chem, 2012, 77: 6545–6552
Riskin M, Gutkin V, Felner I, Willner I. Photochemical and electrochemical encoding of erasable magnetic patterns. Angew Chem Int Ed, 2008, 47: 4416–4420
Flling J, Polyakova S, Belov V, van Blaaderen A, Bossi ML, Hell SW. Synthesis and characterization of photoswitchable fluorescent silica nanoparticles. Small, 2008, 4: 134–142
Zhang J, Riskin M, Tel-Vered R, Tian H, Willner I. Photochemical switching of the phase-transition temperatures of p-NIPAM-Pt nanoparticles thermosensitive polymer composites associated with electrodes: functional electrodes for switchable electrocatalysis. Chem Eur J, 2011, 17: 11237–11242
Niazov T, Shlyahovsky B, Willner I. Photoswitchable electrocatalysis and catalyzed chemiluminescence using photoisomerizable monolayer-functionalized surfaces and Pt nanoparticles. J Am Chem Soc, 2007, 129: 6374–6375
Wang Y, Urbas A, Li Q. Reversible visible-light tuning of self-organized helical superstructures enabled by unprecedented light-driven axially chiral molecular switches. J Am Chem Soc, 2012, 134: 3342–3345
Crivillers N, Orgiu E, Reinders F, Mayor M, Samorì P. Optical modulation of the charge injection in an organic field-effect transistor based on photochromic self-assembled-monolayer-functionalized electrodes. Adv Mater, 2011, 23: 1447–1452
Angelos S, Yang YW, Khashab NM, Stoddart JF, Zink JI. Dual-controlled nanoparticles exhibiting AND logic. J Am Chem Soc, 2009, 131: 11344–11346
Izquierdo-Serra M, Gascón-Moya M, Hirtz JJ, Pittolo S, Poskanzer, KE, Ferrer È, Alibés R, Busqué F, Yuste R, Hernando R, Gorostiza P. Two-photon neuronal and astrocytic stimulation with azobenzene-based photoswitches. J Am Chem Soc, 2014, 136: 8693–8701
Shao N, Jin JY, Wang H, Zheng J, Yang RH, Chan WH, Abliz Z. Design of bis-spiropyran ligands as dipolar molecule receptors and application to in vivo glutathione fluorescent probes. J Am Chem Soc, 2010, 132: 725–736
Shiraishi Y, Sumiya S, Hirai T. Highly sensitive cyanide anion detection with a coumarin-spiropyran conjugate as a fluorescent receptor. Chem Commun, 2011, 47: 4953–4955
Dube H, Ajami D, Rebek Jr J. Photochemical control of reversible encapsulation. Angew Chem Int Ed, 2010, 49: 3192–3195
Zhang H, Kou XX, Zhang Q, Qu DH, Tian H. Altering intercomponent interactions in a photochromic multi-state [2]rotaxane. Org Biomol Chem, 2011, 9: 4051–4056
Singer M, Jäschke A. Reversibly photoswitchable nucleosides: synthesis and photochromic properties of diarylethene-functionalized 7-deazaadenosine derivatives. J Am Chem Soc, 2010, 132: 8372–8377
McCullagh M, Franco I, Ratner MA, Schatz GC. DNA-based optomechanical molecular motor. J Am Chem Soc, 2011, 133: 3452–3459
Andersson J, Li S, Lincoln P, Andréasson J. Photoswitched DNA-binding of a photochromic spiropyran. J Am Chem Soc, 2008, 130: 11836–11837
Guo XF, Huang LM, O’Brien S, Kim P, Nuckolls C. Directing and sensing changes in molecular conformation on individual carbon nanotube field effect transistors. J Am Chem Soc, 2005, 127: 15045–15047
Shen Q, Wang LJ, Liu S, Cao Y, Gan L, Guo XF, Steigerwald ML, Shuai ZG, Liu ZF, Nuckolls C. Photoactive gate dielectrics. Adv Mater, 2010, 22: 3282–3287
Orgiu E, Samorì P. 25th Anniversary article: organic electronics marries photochromism: generation of multifunctional interfaces, materials, and devices. Adv Mater, 2014, 26: 1827–1845
Zhang HT, Guo XF, Hui JS, Hu SX, Xu W, Zhu DB. Interface engineering of semiconductor/dielectric heterojunctions toward functional organic thin-film transistors. Nano Lett, 2011, 11: 4939–4946
Ma X, Tian H. Bright functional rotaxanes. Chem Soc Rev, 2010, 39: 70–80
Qu DH, Tian H. Novel and efficient templates for assembly of rotaxanes and catenanes. Chem Sci, 2011, 2: 1011–1015
Zhou WD, Xu JL, Zheng HY, Yin XD, Zuo ZH, Liu HB, Li YL. Distinct nanostructures from a molecular shuttle: effects of shuttling movement on nanostructural morphologies. Adv Funct Mater, 2009, 19: 141–149
Li H, Li X, Cao ZQ, Qu DH, Ågren H, Tian H. A switchable bis-branched [1]rotaxane featuring dual-mode molecular motions and tunable molecular aggregation. ACS Appl Mater Interfaces, 2014, 6: 18921–18929
Carroll GT, Pollard MM, Deldena R, Feringa BL. Controlled rotary motion of light-driven molecular motors assembled on a gold film. Chem Sci, 2010, 1: 97–101
Cnossen A, Hou LL, Pollard MM, Wesenhagen PV, Browne WR, Feringa BL. Driving unidirectional molecular rotary motors with visible light by intra-and intermolecular energy transfer from palladium porphyrin. J Am Chem Soc, 2012, 134: 17613–17619
Filatov M, Olivucci M. Designing conical intersections for light-driven single molecule rotary motors: from precessional to axial motion. J Org Chem, 2014, 79: 3587–3600
de Jong JJ, Lucas LN, Kellogg RM, van Esch JH, Feringa BL. Reversible optical transcription of supramolecular chirality into molecular chirality. Science, 2004, 304: 278–281
Wang JB, Feringa BL. Dynamic control of chiral space in a catalytic asymmetric reaction using a molecular motor. Science, 2011, 331: 1429–1432
Blanco V, Carlone A, Hänni KD, Leigh DA, Lewandowski B. A rotaxane-based switchable organocatalyst. Angew Chem Int Ed, 2012, 51: 5166–5169
Blanco V, Leigh DA, Marcos V, Morales-Serna JA, Nussbaumer AL. A switchable [2]rotaxane asymmetric organocatalyst that utilizes an acyclic chiral secondary amine. J Am Chem Soc, 2014, 136: 4905–4908
Qu DH, Feringa BL. Controlling molecular rotary motion with a self-complexing lock. Angew Chem Int Ed, 2010, 49: 1107–1110
Coskun A, Friedman DC, Li H, Patel K, Khatib HA, Stoddart JF. A light-gated STOP-GO molecular shuttle. J Am Chem Soc, 2009, 131: 2493–2495
Lewandowski B, De Bo G, Ward JW, Papmeyer M, Kuschel S, Aldegunde MJ, Gramlich PME, Heckmann D, Goldup SM, D’Souza DM, Fernandes AE, Leigh DA. Sequence-specific peptide synthesis by an artificial small-molecule machine. Science, 2013, 339: 189–193
De Bo G, Kuschel S, Leigh DA, Lewandowski B, Papmeyer M, Ward JW. Efficient assembly of threaded molecular machines for sequence-specific synthesis. J Am Chem Soc, 2014, 136: 5811–5814
Qu DH, Tian H. Synthetic small-molecule walkers at work. Chem Sci, 2013, 4: 3031–3035
Chen KY, Ivashenko O, Carroll GT, Robertus J, Kistemaker JCM, London G, Browne WR, Rudolf P, Feringa BL. Control of surface wettability using tripodal light-activated molecular motors. J Am Chem Soc, 2014, 136: 3219–3224
Traut T. Allosteric Regulatory Enzymes. New York: Springer, 2008
Mortezaei S, Catarineu NR, Canary JW. A redox-reconfigurable, ambidextrous asymmetric catalyst. J Am Chem Soc, 2012, 134: 8054–8057
Blanco V, Carlone A, Hänni KD, Leigh DA, Lewandowski B. A rotaxane-based switchable organocatalyst. Angew Chem Int Ed, 2012, 51: 5166–5169
Neilson BM, Bielawski CW. Photoswitchable NHC-promoted ring-opening polymerizations. Chem Commun, 2013, 49: 5453–5455
Dawson PE, Kent SBH. Synthesis of native proteins by chemical ligation. Annu Rev Biochem, 2000, 69: 923–960
Thomas CR, Ferris DP, Lee JH, Choi E, Cho MH, Kim ES, Stoddart JF, Shin JS, Cheon J, Zink JI. Noninvasive remote-controlled release of drug molecules in vitro using magnetic actuation of mechanized nanoparticles. J Am Chem Soc, 2010, 132: 10623–10625
Cao ZQ, Miao Q, Zhang Q, Li H, Qu DH, Tian H. A fluorescent bistable [2]rotaxane molecular switch on SiO2 nanoparticles. Chem Commun, 2015, 51: 4973–4976
Yang H, Yuan B, Zhang X. Supramolecular chemistry at interfaces: host-guest interactions for fabricating multifunctional biointerfaces. Acc Chem Res, 2014, 47: 2106–2115
Yang YW, Sun YL, Song N. Switchable host-guest systems on surfaces. Acc Chem Res, 2014, 47: 1950–1960
Yang H, Ma Z, Yuan B, Wang ZQ, Zhang X. Supramolecular polymerization at the interface: layer-by-layer assembly driven by host-enhanced p-p interaction. Chem Commun, 2014, 50: 11173–11176
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, H., Qu, DH. Recent advances in new-type molecular switches. Sci. China Chem. 58, 916–921 (2015). https://doi.org/10.1007/s11426-015-5417-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-015-5417-7