Abstract
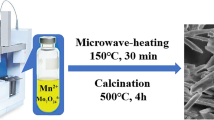
Manganese oxides with different crystalline phases and morphologies were prepared by calcining MnCO3 precursors. The MnCO3 precursors with different morphologies were obtained through a green route under hydrothermal conditions with orange pericarp extracting solution as the reducing agent. By calcining the precursor under different temperatures and atmospheres, MnO x with different stoichiometric ratios (i.e., MnO, MnO2, Mn2O3, and Mn3O4) can be obtained. Electrochemical studies reveal that among these manganese oxides, MnO or MnO2 are more suitable as supercapacitor working electrodes than Mn2O3 or Mn3O4. They exhibit high specific capacitance up to 296.6 F/g and also possess good cycling stability, which make them potential electrode materials for supercapacitors.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Yang ZG, Zhang JL, Kintner-Meyer MCW, Lu XC, Choi D, Lemmon JP, Liu J. Electrochemical energy storage for green grid. Chem Rev, 2011, 111: 3577–3613
Wagner FT, Lakshmanan B, Mathias MF. Electrochemistry and the future of the automobile. J Phys Chem Lett, 2010, 1: 2204–2219
Miller JR, Simon P. Electrochemical capacitors for energy management. Science, 2008, 321: 651–652
Chen LF, Huang ZH, Liang HW, Gao HL, Yu SH. Three-dimensional heteroatom-doped carbon nanofiber networks derived from bacterial cellulose for supercapacitors. Adv Funct Mater, 2014, 24: 5104–5111
Chen LF, Zhang XD, Liang HW, Kong MG, Guan QF, Chen P, Wu ZY, Yu SH. Synthesis of nitrogen-doped porous carbon nanofibers as an efficient electrode material for supercapacitors. ACS Nano, 2012, 6: 7092–7102
Chen LF, Huang ZH, Liang HW, Yao WT, Yu ZY, Yu SH. Flexible all-solid-state high-power supercapacitor fabricated with nitrogen-doped carbon nanofiber electrode material derived from bacterial cellulose. Energy Environ Sci, 2013, 6: 3331–3338
Kaempgen M, Chan CK, Ma J, Cui Y, Gruner G. Printable thin film supercapacitors using single-walled carbon nanotubes. Nano Lett, 2009, 9: 1872–1876
Zhang LL, Zhou R, Zhao XS. Graphene-based materials as supercapacitor electrodes. J Mater Chem, 2010, 20: 5983–5992
Wang YG, Li HQ, Xia YY. Ordered whiskerlike polyaniline grown on the surface of mesoporous carbon and its electrochemical capacitance performance. Adv Mater, 2006, 18: 2619–2623
Sivakkumar SR, Ko JM, Kim DY, Kim BC, Wallace GG. Performance evaluation of CNT/polypyrrole/MnO2 composite electrodes for electrochemical capacitors. Electrochim Acta, 2007, 52: 7377–7385
Luo JY, Cheng L, Xia YY. LiMn2O4 hollow nanosphere electrode material with excellent cycling reversibility and rate capability. Electrochem Commun, 2007, 9: 1404–1409
Lee SW, Kim J, Chen S, Hammond PT, Yang SH. Carbon nanotube/manganese oxide ultrathin film electrodes for electrochemical capacitors. ACS Nano, 2010, 4: 3889–3896
Chen PC, Shen GZ, Shi Y, Chen HT, Zhou CW. Preparation and characterization of flexible asymmetric supercapacitors based on transition-metal-oxide nanowire/single-walled carbon nanotube hybrid thin-film electrodes. ACS Nano, 2010, 4: 4403–4411
Mao L, Zhang K, Chan HSO, Wu JS. Nanostructured MnO2/graphene composites for supercapacitor electrodes: the effect of morphology, crystallinity and composition. J Mater Chem, 2012, 22: 1845–1851
Yan J, Fan ZJ, Wei T, Qian WZ, Zhang ML, Wei F. Fast and reversible surface redox reaction of graphene-MnO2 composites as supercapacitor electrodes. Carbon, 2010, 48: 3825–3833
Lei ZB, Shi FH, Lu L. Incorporation of MnO2-coated carbon nanotubes between graphene sheets as supercapacitor electrode. ACS Appl Mater Interfaces, 2012, 4: 1058–1064
Zhang X, Ji LY, Zhang SC, Yang WS. Synthesis of a novel polyaniline-intercalated layered manganese oxide nanocomposite as electrode material for electrochemical capacitor. J Power Sources, 2007, 173: 1017–1023
Liu R, Lee SB. MnO2/poly(3,4-ethylenedioxythiophene) coaxial nanowires by one-step coelectrodeposition for electrochemical energy storage. J Am Chem Soc, 2008, 130: 2942–2943
Liu R, Duay J, Lee SB. Redox exchange induced MnO2 nanoparticle enrichment in poly(3,4-ethylenedioxythiophene) nanowires for electrochemical energy storage. ACS Nano, 2010, 4: 4299–4307
Yang DF. Pulsed laser deposition of cobalt-doped manganese oxide thin films for supercapacitor applications. J Power Sources, 2012, 198: 416–422
Yang DF. Pulsed laser deposition of vanadium-doped manganese oxide thin films for supercapacitor applications. J Power Sources, 2013, 228: 89–96
Reddy ALM, Shaijumon MM, Gowda SR, Ajayan PM. Multi-segmented Au-MnO2/carbon nanotube hybrid coaxial arrays for high-power supercapacitor applications. J Phys Chem C, 2010, 114: 658–663
Hou Y, Cheng YW, Hobson T, Liu J. Design and synthesis of hierarchical MnO2 nanospheres/carbon nanotubes/conducting polymer ternary composite for high performance electrochemical electrodes. Nano Lett, 2010, 10: 2727–2733
Bao LH, Zang JF, Li XD. Flexible Zn2SnO4/MnO2 core/shell nanocable-carbon microfiber hybrid composites for high-performance super-capacitor electrodes. Nano Lett, 2011, 11: 1215–1220
Fischer AE, Pettigrew KA, Rolison DR, Stroud RM, Long JW. Incorporation of homogeneous, nanoscale MnO2 within ultraporous carbon structures via self-limiting electroless deposition: implications for electrochemical capacitors. Nano Lett, 2007, 7: 281–286
Jiang J, Li YY, Liu JP, Huang XT, Yuan CZ, Lou XW. Recent advances in metal oxide-based electrode architecture design for electro-chemical energy storage. Adv Mater, 2012, 24: 5166–5180
Fei JB, Cui Y, Yan XH, Qi W, Yang Y, Wang KW, He Q, Li JB. Controlled preparation of MnO2 hierarchical hollow nanostructures and their application in water treatment. Adv Mater, 2008, 20: 452–456
Li ZQ, Ding Y, Xiong YJ, Yang Q, Xie Y. One-step solution-based catalytic route to fabricate novel α-MnO2 hierarchical structures on a large scale. Chem Commun, 2005: 918–920
Yu P, Zhang X, Wang DL, Wang L, Ma YW. Shape-controlled synthesis of 3D hierarchical MnO2 nanostructures for electrochemical supercapacitors. Cryst Growth Des, 2009, 9: 528–533
Duay J, Sherrill SA, Gui Z, Gillette E, Lee SB. Self-limiting electrodeposition of hierarchical MnO2 and M(OH)2/MnO2 nanofibril/nanowires: mechanism and supercapacitor properties. ACS Nano, 2013, 7: 1200–1214
Komaba S, Tsuchikawa T, Ogata A, Yabuuchi N, Nakagawa D, Tomita M. Nano-structured birnessite prepared by electrochemical activation of manganese(III)-based oxides for aqueous supercapacitors. Electrochim Acta, 2012, 59: 455–463
Tian N, Zhou ZY, Sun SG, Ding Y, Wang ZL. Synthesis of tetrahexahedral platinum nanocrystals with high-index facets and high rlectro-oxidation vctivity. Science, 2007, 316: 732–735
Yan J, Fan Z, Wei T, Cheng J, Shao B, Wang K, Song L, Zhang M. Carbon nanotube/MnO2 composites synthesized by microwave-assisted method for supercapacitors with high power and energy densities. J Power Sources, 2009, 194: 1202–1207
Subramanian V, Zhu HW, Wei BQ. Synthesis and electrochemical characterizations of amorphous manganese oxide and single walled carbon nanotube composites as supercapacitor electrode materials. Electrochem Commun, 2006, 8: 827–832
Hu CC, Chang KH, Hsu TY. The synergistic influences of OH− concentration and electrolyte conductivity on the redox behavior of Ni(OH)2/NiOOH. J Electrochem Soc, 2008, 155: F196–F200
Jiang H, Zhao T, Ma J, Yan CY, Li CZ. Ultrafine manganese dioxide nanowire network for high-performance super capacitors. Chem Commun, 2011, 47: 1264–1266
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Du, W., Xu, X., Zhang, D. et al. Green synthesis of MnO x nanostructures and studies of their supercapacitor performance. Sci. China Chem. 58, 627–633 (2015). https://doi.org/10.1007/s11426-014-5242-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-014-5242-4