Abstract
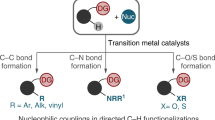
In the past decade, transition-metal-catalyzed C-H functionalization by weak coordination has emerged as a practical and powerful tool to access many valuable chemicals. Two classes of weakly coordinating directing groups, commonly occurring functional groups, and easily removable auxiliaries, have been found to be efficient and practical for C-H activation reactions. This mini-review contains examples of recent research advances on transition-metal-catalyzed SP2 C-H functionalization via weak coordination, using Ru, Rh, and Pd. A number of weakly coordinating functional groups (e.g., ketone, ester, carbamate, tertiary amide, ether, thioether, alcohol, and some others) are covered. As the field of transition-metal-catalyzed C-H functionalization continues to develop and more synthetically useful chemo-, regio-, and enantioselective reactions catalyzed by transition metal via weak coordination are discovered, this promising and attractive strategy will play a more important role in modern organic synthesis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kakiuchi F, Chatani N. Catalytic Methods for C-H bond functionalization: application in organic synthesis. Adv Synth Catal, 2003, 345: 1077–1101
Dick AR, Sanford MS. Transition metal catalyzed oxidative functionalization of carbon-hydrogen bonds. Tetrahedron, 2006, 62: 2439–2463
Godula K, Sames D. C-H bond functionalization in complex organic synthesis. Science, 2006, 312: 67–72
Yu JQ, Giri R, Chen X. σ-Chelation-directed C-H functionalizations using Pd(II) and Cu(II) catalysts: regioselectivity, stereoselectivity and catalytic turnover. Org Biomol Chem, 2006, 4: 4041–4047
Alberico D, Scott ME, Lautens M. Aryl-aryl bond formation by transition-metal-catalyzed direct arylation. Chem Rev, 2007, 107: 174–238
Ackermann L, Vicente R, Kapdi AR. Transition-metal-catalyzed direct arylation of (hetero)arenes by C-H bond cleavage. Angew Chem Int Ed, 2009, 48: 9792–9826
Daugulis O, Do HQ, Shabashov D. Palladium- and copper-catalyzed arylation of carbon-hydrogen bonds. Acc Chem Res, 2009, 42: 1074–1086
Colby DA, Bergman RG, Ellman JA. Rhodium-catalyzed C-C bond formation via heteroatom-directed C-H bond activation. Chem Rev, 2010, 110: 624–655
Lyons TW, Sanford MS. Palladium-catalyzed ligand-directed C-H functionalization reactions. Chem Rev, 2010, 110: 1147–1169
Xu LM, Li BJ, Yang Z, Shi ZJ. Organopalladium(IV) chemistry. Chem Soc Rev, 2010, 39: 712–733
Ackermann L, Vicente R, Kapdi AR. Carboxylate-assisted transition-metalcatalyzed C-H bond functionalizations: mechanism and scope. Chem Rev, 2011, 111: 1315–1345
Colby DA, Tsai AS, Bergman RG, Ellman JA. Rhodium catalyzed chelation-assisted C-H bond functionalization reactions. Acc Chem Res, 2012, 45: 814–825
Neufeldt SR, Sanford MS. Controlling site selectivity in palladium-catalyzed C-H bond functionalization. Acc Chem Res, 2012, 45: 936–946
Yu DG, Li BJ, Shi ZJ. Challenges in C-C bond formation through direct transformations of sp2 C-H bonds. Tetrahedron, 2012, 68: 5130–5136
Yan GB, Wu XM, Yang MH. Transition-metal-catalyzed additions of C-H bonds to C-X (X = N, O) multiple bonds via C-H bond activation. Org Biomol Chem, 2013, 11: 5558–5578
Zheng QZ, Jiao N. Transition-metal-catalyzed ketonedirected ortho-C-H functionalization reactions. Tetrahedron Lett, 2014, 55: 1121–1126
Engle KM, Mei TS, Wasa M, Yu JQ. Weak coordination as a powerful means for developing broadly useful C-H functionalization reactions. Acc Chem Res, 2012, 45: 788–802
Murai S, Kakiuchi F, Sekine S, Tanaka Y, Kamatani A, Sonoda M, Chatani N. Efficient catalytic addition of aromatic carbon-hydrogen bonds to olefins. Nature, 1993, 366: 529–531
Kakiuchi F, Yamamoto Y, Chatani N, Murai S. Catalytic addition of aromatic C-H bonds to acetylenes. Chem Lett, 1995, 8: 681–682
Kakiuchi F, Kan S, Igi K, Chatani N, Murai S. A ruthenium-catalyzed reaction of aromatic ketones with arylboronates: a new method for the arylation of aromatic compounds via C-H bond cleavage. J Am Chem Soc, 2003, 125: 1698–1699
Martinez R, Chevalier R, Darses S, Genet JP. A versatile ruthenium catalyst for C-C bond Formation by C-H bond activation. Angew Chem Int Ed, 2006, 45: 8232–8235
Martinez R, Simon MO, Chevalier R, Pautigny C, Genet JP, Darses S. C-C bond formation via C-H bond activation using an in situ-generated ruthenium catalyst. J Am Chem Soc, 2009, 131: 7887–7895
Thirunavukkarasu VS, Ackermann L. Ruthenium-catalyzed C-H bond oxygenations with weakly coordinating ketones. Org Lett, 2012, 14, 6206–209
Shan G, Han XS, You SY, Lin Y, Rao Y. Broadening the catalyst and reaction scope of regio- and chemoselective C-H oxygenation: a convenient and scalable approach to 2-acylphenols by intriguing Rh(II) and Ru(II) catalysis. Org Biomol Chem, 2013, 1: 2318–2322
Kim J, Kim J, Chang S. Ruthenium-catalyzed direct C-H amidation of arenes including weakly coordinating aromatic ketones. Chem Eur J, 2013, 19: 7328–7333
Zheng QZ, Liang YF, Qin C, Jiao N. Ru(II)-catalyzed intermolecular C-H amidation of weakly coordinating ketones. Chem Commun, 2013, 49: 5654–5656
Bhanuchandra M, Yadav MR, Rit RK, Kuram MR, Sahoo A. Ru(II)-catalyzed intermolecular ortho-C-H amidation of aromatic ketones with sulfonyl azides. Chem Commun, 2013, 49: 5225–5257
Sonoda M, Kakiuchi F, Kamatani A, Chatani N, Murai S. Ruthenium-catalyzed addition of aromatic esters at the ortho C-H Bonds to olefins. Chem Lett, 1996, 109–110
Kakiuchi F, Ohtaki H, Sonoda M, Chatani N, Murai S. Mechanistic study of the Ru(H)2(CO)(PPh3)3-catalyzed addition of C-H bonds in aromatic esters to olefin. Chem Lett, 2001: 918–919
Nishimura T, Washitake Y, Uemura S. Ruthenium/halide catalytic system for C-C bond forming reaction between alkynes and unsaturated carbonyl compounds. Adv Synth Catal, 2007, 349: 2563–2571
Neisius NM, Plietker B. The ruthenium-catalyzed hydrovinylation of internal alkynes by acrylates: an atom economic approach to highly substituted 1,3-dienes. Angew Chem Int Ed, 2009, 48:5752–5755
Graczyk K, Ma W, Ackermann L. Oxidative alkenylation of aromatic esters by ruthenium-catalyzed twofold C-H bond cleavages. Org Lett, 2012, 14: 4110–4113
Yang YQ, Lin Y, Rao Y. Ruthenium(II)-catalyzed synthesis of hydroxylated arenes with ester as an effective directing group. Org Lett, 2012, 14: 2874–2877
Li B, Ma JF, Liang Y, Wang NC, Xu SS, Song H, Wang BQ. Regio- and stereoselective olefination of phenol carbamates through C-H bond functionalization. Eur J Org Chem, 2013: 1950–1962
Reddy MC, Jeganmohan M. Ruthenium-catalyzed selective aerobic oxidative ortho-alkenylation of substituted phenols with alkenes through C-H bond activation. Eur J Org Chem, 2013: 1150–1157
Liu W, Ackermann L. Ortho- and para-selective ruthenium-catalyzed C(sp2)-H oxygenations of phenol derivatives. Org Lett, 2013, 15: 3484–3486
Thirunavukkarasu VS, Hubrich J, Ackermann L. Ruthenium-catalyzed oxidative C(sp2)-H bond hydroxylation: site-selective C-O bond formation on benzamides. Org Lett, 2012, 14: 4210–4213
Yang F, Ackermann L. Ruthenium-catalyzed C-H oxygenation on aryl weinreb amides. Org Lett, 2013, 15: 718–720
Yang XL, Shan G, Rao Y. Synthesis of 2-aminophenols and heterocycles by Ru-catalyzed C-H mono- and dihydroxylation. Org Lett, 2013, 15: 2334–2337
Muralirajan K, Parthasarathy K, Cheng CH. Regioselective synthesis of indenols by rhodium-catalyzed C-H activation and carbocyclization of aryl ketones and alkynes. Angew Chem Int Ed, 2011, 50, 4169–4172
Patureau FW, Besset T, Kuhl N, Glorius F. Diverse strategies toward indenol and fulvene derivatives: Rh-catalyzed C-H activation of aryl ketones followed by coupling with internal alkynes. J Am Chem Soc, 2011, 133: 2154–2156
Wencel-Delord J, Nimphius C, Wang H, Glorius F. Rhodium(III) and hexabromobenzene: a catalyst system for the cross-dehydrogenative coupling of simple arenes and heterocycles with arenes bearing directing groups. Angew Chem Int Ed, 2012, 51: 13001–13005
Shi XY, Li CJ. Synthesis of indene frameworks via rhodium-catalyzed cascade cyclization of aromatic ketone and unsaturated carbonyl compounds. Org Lett, 2013, 15: 1476–1479
Shin K, Baek Y, Chang S. Direct C-H amination of arenes with alkyl azides under rhodium catalysis. Angew Chem Int Ed. 2013, 52: 8031–8036
Schroder N, Wencel-Delord J, Glorius F. High-yielding, versatile, and practical [Rh(III)Cp*]-catalyzed ortho bromination and iodination of arenes. J Am Chem Soc, 2012, 134: 8298–8301
Park SH, Kim JY, Chang S. Rhodium-catalyzed selective olefination of arene esters via C-H bond activation. Org Lett, 2011, 13: 2372–2375
Gong TJ, Xiao B, Liu ZJ, Wan, Xu J, Luo DF, Fu Y, Liu L. Rhodium-catalyzed selective C-H activation/olefination of phenol carbamates. Org Lett, 2011, 13: 3235–3237
Feng C, Loh TP. Rhodium-catalyzed direct ortho C-H olefination of phenol derivatives. Chem Commun 2011, 47: 10458–10460
Zhang Q, Yu HZ, Li YT, Liu L, Huang Y, Fu Y. Computational study on mechanism of Rh(III)-catalyzed oxidative Heck coupling of phenol carbamates with alkenes. Dalton Trans, 2013, 42: 4175–4184
Yu M, Xie YJ, Xie CS, Zhang YH. Palladium-catalyzed C-H alkenylation of arenes using thioethers as directing groups. Org Lett, 2012, 14: 2164–2167
Yao JZ, Yu M, Zhang YH. Thioethers as directing group for the palladium-catalyzed direct arylation of arenes. Adv Synth Catal, 2012, 354, 3205–3210
Samanta r, Antonchick AP. Palladium-catalyzed double C-H activation directed by sulfoxides in the synthesis of dibenzothiophenes. Angew Chem Int Ed, 2011, 50: 5217–5220
Zhang XS, Zhu QL, Zhang YF, Li YB, Shi ZJ. Controllable mono-/dialkenylation of benzyl thioethers through Rh-catalyzed aryl C-H activation. Chem Eur J, 2013, 19: 11898–111903
Satoh T, Kametani Y, Terao Y, Miura M, Namura M. Palladium-catalyzed multiple arylation of phenyl ketones with aryl bromides. Tetrahedron Lett, 1999, 40: 5345–5348
Gandeepan P, Parthasarathy K, Cheng CH. Synthesis of phenanthrone derivatives from sec-alkyl aryl ketones and aryl halides via a palladium-catalyzed dual C-H bond activation and enolate cyclization. J Am Chem Soc, 2010, 132: 8569–8571
Xiao B, Gong TJ, Xu J, Liu ZJ, Liu L. Palladium-catalyzed intermolecular directed C-H amidation of aromatic ketones. J Am Chem Soc, 2011, 133: 1466–1474
Gandeepan P, Hung CH, Cheng CH. Pd-catalyzed double C-H bond activation of diaryl ketones for the synthesis of fluorenones. Chem Commun, 2012, 48: 9379–9381
Li H, Zhu RY, Shi WJ, He KH, Shi ZJ. Synthesis of fluorenone derivatives through Pd-catalyzed dehydrogenative cyclization. Org Lett, 2012, 14: 4850–4853
Shan G, Yang XL, Ma LL, Rao Y. Pd-catalyzed C-H oxygenation with TFA/TFAA: expedient access to oxygen-containing heterocycles and late-stage drug modification. Angew Chem Int Ed, 2012, 51: 13070–13074
Mo FY, Trzepkowski LJ, Dong GB. Synthesis of ortho-acylphenols through the palladium-catalyzed ketone-directed hydroxylation of arenes. Angew Chem Int Ed, 2012, 51: 13075–13079
Choy PY, Kwong FY. Palladium-catalyzed ortho-CH-bond oxygenation of aromatic ketones. Org Lett, 2013, 15: 270–273
Sun XY, Shan G, Sun YH, Rao Y. Regio- and chemoselective C-H chlorination/bromination of electron-deficient arenes by weak coordination and study of relative directing-group abilities. Angew Chem Int Ed, 2013, 52: 4440–4444
Xiao B, Fu Y, Xu J, Gong TJ, Dai JJ, Yi J, Liu L. Pd(II)-catalyzed C-H activation/aryl-aryl coupling of phenol esters. J Am Chem Soc, 2010, 132: 468–469
Bedford RB, Webstera RL, Mitchellb CJ. Palladium-catalysed ortho-arylation of carbamate-protected phenols. Org Biomol Chem, 2009, 7: 4853–4857
Zhao XD, Yeung CS, Dong VM. Palladium-catalyzed ortho-arylation of O-phenylcarbamates with simple arenes and sodium persulfate. J Am Chem Soc, 2010, 132: 5837
John A, Nicholas KM. Palladium catalyzed C-H functionalization of O-arylcarbamates: selective ortho-bromination using NBS. J Org Chem, 2012, 77: 5600–5605
Sun XY, Sun YH, Zhang C, Rao Y. Room-temperature Pd-catalyzed C-H chlorination by weak coordination: one-pot synthesis of 2-chlorophenols with excellent regioselectivity. Chem Commun, 2014, 50: 1262–1264
Lu Y, Wang DH, Engle KM, Yu JQ. Pd(II)-catalyzed hydroxyl-directed C-H olefination enabled by monoprotected amino acid ligands. J Am Chem Soc 2010, 132: 5916–5921
Wang X, Lu Y, Dai HX, Yu JQ. Pd(II)-catalyzed hydroxyl-directed C-H activation/C-O cyclization: expedient construction of dihydrobenzofurans. J Am Chem Soc, 2010, 132: 12203–12205
Wang H, Li G, Engle KM, Yu JQ, H. Davies ML. Sequential C-H functionalization reactions for the enantioselective synthesis of highly functionalized 2,3-dihydrobenzofurans. J Am Chem Soc, 2013, 135: 6774–6777
Chidipudi SR, Wieczysty MD, Khan I, Lam HW. Synthesis of benzopyrans by Pd(II)- or Ru(II)-catalyzed C-H alkenylation of 2-aryl-3-hydroxy-2-cyclohexenones. Org Lett, 2013, 15: 570–573
Xiao B, Gong TJ, Liu ZJ, Liu JH, Luo DF, Xu J, Liu L. Synthesis of dibenzofurans via palladium-catalyzed phenol-directed C-H activation/C-O cyclization. J Am Chem Soc, 2011, 133: 9250–9253
Lu Y, Leow D, Wang X, Engle KM, Yu JQ. Hydroxyl-directed C-H carbonylation enabled by mono-N-protected amino acid ligands: an expedient route to 1-isochromanones. Chem Sci, 2011, 2: 967–971
Huang A, Ghavtadze N, Chattopadhyay B, Gevorgyan V. Synthesis of catechols from phenols via pd-catalyzed silanol-directed C-H oxygenation. J Am Chem Soc, 2011, 133: 17630–1733
Li G, Leow D, Wan L, Yu JQ, Ether-directed ortho-C-H olefination with a palladium(II)/monoprotected amino acid catalyst. Angew Chem Int Ed, 2013, 52: 1245–1247
Gandeepan P, Cheng CH. Allylic carbon-carbon double bond directed Pd-catalyzed oxidative ortho-olefination of arenes. J Am Chem Soc, 2012, 134: 5738–5741
Minami Y, Shiraishi Y, Yamada K, Hiyama T. Palladium-catalyzed cycloaddition of alkynyl aryl ethers with internal alkynes via selective ortho C-H activation. J Am Chem Soc, 2012, 134: 6124–6127
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, Y., Shan, G. & Yang, X. Some recent advances in transition-metal-catalyzed ortho SP2 C-H functionalization using Ru, Rh, and Pd. Sci. China Chem. 57, 930–944 (2014). https://doi.org/10.1007/s11426-014-5130-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-014-5130-y