Abstract
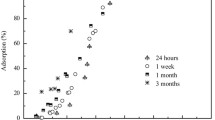
The sorption behavior and microscopic sequestration mechanisms of radionuclide 63Ni(II) on mordenite as a function of aging time, ionic strength, initial 63Ni(II) concentrations, solid content and coexistent electrolyte ions were investigated by the combination of batch and EXAFS techniques. Macroscopic experiment results show that the sorption of 63Ni(II) is dependent on ionic strength at pH<7, and independent of ionic strength at pH>7. The sorption percentage of 63Ni(II) on mordenite increases with increasing solid content, while the sorption capacity decreases as solid content increases. The presence of different electrolyte ions can enhance or inhibit the sorption of Ni(II) on mordenite in various degrees. EXAFS analysis results of the samples under three different ionic strengths suggest that the retained 63Ni(II) in these samples exists in an octahedral environment with six water ligands. In the initial period of rapid uptake, the sorption of 63Ni(II) is dominated by the formation of inner-sphere surface complexes. As aging time increases, 63Ni(II) sequestration behavior tends to be mainly controlled by the formation of Ni phyllosilicate co-precipitates and/or Ni(OH)2(s) precipitates. Results for the second shell fit of the sample prepared at an initial 63Ni(II) concentration of 100 mg/L indicate the possible formation of Ni polynuclear surface complexes. Both the macroscopic sorption data and the molecular level evidence of 63Ni(II) surface speciation at the mordenite/water interfaces should be factored into better predictions of the mobility and bioavailability of 63Ni(II) in environment mediums.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Faur-Brasquet C, Kadirvelu K, Le Cloirec P. Removal of metal ions from aqueous solution by adsorption onto activated carbon cloths: Adsorption competition with organic matter. Carbon, 2002, 40: 2387–2392
Gonzalez-Serrano E, Cordero T, Rodriguez-Mirasol J, Cotoruelo L, Rodriguez JJ. Removal of water pollutants with activated carbons prepared from H3PO4 activation of lignin from kraft black liquors. Water Res, 2004, 38: 3043–3050
Hale SE, Tomaszewski JE, Luthy RG, Werner D. Sorption of dichlorodiphenyltrichloroethane (DDT) and its metabolites by activated carbon in clean water and sediment slurries. Water Res, 2009, 43: 4336–4346
Osmanlioglu AE. Treatment of radioactive liquid waste by sorption on natural zeolite in Turkey. J Hazard Mater, 2006, 137: 332–335
Rao GPC, Satyaveni S, Ramesh A, Seshaiah K, Murthy KSN, Choudary NV. Sorption of cadmium and zinc from aqueous solutions by zeolite 4A, zeolite 13X and bentonite. J Environ Manage, 2006, 81: 265–272
Ochoa-Herrera V, Sierra-Alvarez R. Removal of perfluorinated surfactants by sorption onto granular activated carbon, zeolite and sludge. Chemosphere, 2008, 72: 1588–1593
Yang ST, Sheng GD, Tan XL, Hu J, Du JZ, Montavon G, Wang XK. Determination of Ni(II) uptake mechanisms on mordenite surfaces: a combined macroscopic and microscopic approach. Geochim Cosmochim Acta, 2011, 75: 6520–6534
Hu J, Xie Z, He B, Sheng G, Chen C, Li J, Chen Y, Wang X. Sorption of Eu(III) on GMZ bentonite in the absence/presence of humic acid studied by batch and XAFS techniques. Sci China Chem, 2010, 53: 1420–1428
Schmidt GT, Vlasova N, Zuzaan D, Kersten M, Daus B. Adsorption mechanism of arsenate by zirconyl-functionalized activated carbon. J Colloid Interface Sci, 2008, 317: 228–234
Ankudinov AL, Rehr JJ. Relativistic calculations of spin-dependent X-ray absorption spectra. Phys Rev B, 1997, 56: 1712–1715
Strawn, DG, Scheidegger, AM, Sparks, DL. Kinetics and mechanisms of Pb(II) sorption and desorption at the aluminum oxide-water interface. Environ Sci Technol, 1998, 32: 2596–2601
Sheng GD, Yang ST, Sheng J, Hu J, Tan XL, Wang XK. Macroscopic and microscopic investigation of Ni(II) sequestration on diatomite by batch, XPS and EXAFS techniques. Environ Sci Technol, 2011, 45: 7718–7726
Scheidegger, AM, Strawn, DG, Lamble, GM, Sparks, DL. The kinetics of mixed Ni-Al hydroxide formation on clay and aluminum oxide minerals: A time-resolved XAFS study. Geochim Cosmochim Acta, 1998, 62: 2233–2245
Thompson, HA, Parks, GA, Brown, GE Jr. Dynamic interactions of dissolution, surface adsorption, and precipitation in an aging cobalt(II)-clay-water system. Geochim Cosmochim Acta, 1999, 63: 1767–1779
Shao D, Xu D, Wang S, Fan Q, Wu W, Dong Y, Wang X. Modeling of radionickel sorption on MX-80 bentonite as a function of pH and ionic strength. Sci China Ser B-Chem, 2009, 52: 362–371
Yang ST, Li JX, Shao DD, Hu J, Wang XK. Adsorption of Ni(II) on oxidized multi-walled carbon nanotubes: effect of contact time, pH, foreign ions and PAA. J Hazard Mater, 2009, 166: 109–116
Wuhan University. Analytical Chemistry (5th ed). Beijing: Higher Education Press, 2006, 385–403
Reddad Z, Gerente C, Andres Y, Le Cloirec P. Adsorption of several metal ions onto a low-cost biosorbent: kinetic and equilibrium studies. Environ Sci Technol, 2002, 36: 2067–2073
Chen CL, Wang XK. Adsorption of Ni(II) from aqueous solution using oxidized multiwall carbon nanotubes. Ind Eng Chem Res, 2006, 45: 9144–9149
Mercer KL, Tobiason JE. Removal of arsenic from high ionic strength solutions: Effects of ionic strength, pH, and preformed versus in situ formed HFO. Environ Sci Technol, 2008, 42: 3797–3802
Wang XS, Huang J, Hu HQ, Wang J, Qin Y. Determination of kinetic and equilibrium parameters of the batch adsorption of Ni(II) from aqueous solutions by Na-mordenite. J Hazard Mater, 2007, 142: 468–476
Huang JH, Liu YF, Wang XG. Selective adsorption of tannin from flavonoids by organically modified attapulgite clay. J Hazard Mater, 2008, 160: 382–387
Bhattacharyya KG, Gupta SS. Adsorption of Fe(III), Co(II) and Ni(II) on ZrO-kaolinite and ZrO-montmorillonite surfaces in aqueous medium. Colloids Surf A, 2008, 317: 71–79
Sheng GD, Yang ST, Zhao DL, Sheng J, Wang XK. Adsorption of Eu(III) on titanate nanotubes studied by a combination of batch and EXAFS technique. Sci China Chem, 2011, 54: 1–13
Collins CR, Vala RK, Sherman DM. Effect of inorganic and organic ligands on the mechanism of cadmium sorption to goethite. Geochim Cosmochim Acta, 1999, 63: 2989–3002
Buerge-Weirich B, Hari R, Xue H. Adsorption of Cu, Cd, and Ni on goethite in the presence of natural groundwater ligands. Environ Sci Technol, 2002, 36: 328–336
Boily JF, Sjoberg S, Persson P, Structures and stabilities of Cd(II) and Cd (II)-phthalate complexes at the goethite/water interface. Geochim Cosmochim Acta, 2005, 69: 3219–3235
Esmadi F, Simm J. Sorption of cobalt(II) by amorphous ferric hydroxide. Colloids Surf A, 1995, 104: 265–270
Tan XL, Wang XK, Chen CL, Sun AH. Effect of soil humic and fulvic acids, pH and ionic strength on Th(IV) sorption to TiO2 nanoparticles. Appl Radiat Isot, 2007, 65: 375–381
Harter RD, Naidu R. An assessment of environmental and solution parameter impact on trace-metal sorption by soil. Soil Sci Soc Am J, 2001, 65: 597–612
Yang ST, Li JX, Lu Y, Chen YX, Wang XK. Sorption of Ni(II) on GMZ bentonite: effects of pH, ionic strength, foreign ions, humic acid and temperature. Appl Radiat Isot, 2009, 67: 1600–1608
Xu D, Tan XL, Chen CL, Wang XK. Removal of Pb(II) from aqueous solution by oxidized multiwalled carbon nanotubes. J Hazard Mater, 2008, 154: 407–416
Fan QH, Shao DD, Lu Y, Wu WS, Wang XK. Effect of pH, ionic strength, temperature and humic substances on the sorption of Ni(II) to Na-attapulgite. Chem Eng J, 2009, 150: 188–195
Shukla A, Zhang YH, Dubey P, Margrave JL, Shukla SS. The role of sawdust in the removal of unwanted materials from water. J Hazard Mater, 2002, 95: 137–152
Tan XL, Hu J, Montavon G, Wang XK. Sorption speciation of nickel(II) onto Ca-montmorillonite: batch, EXAFS techniques and modeling. Dalton Trans, 2011, 40: 10953–10960
Bargar JR, Brown GE Jr, Parks GA. Surface complexation of Pb(II) at oxide-water interfaces: I. XAFS and bond-valence determination of mononuclear and polynuclear Pb(II) sorption products on aluminum oxides. Geochim Cosmochim Acta, 1997, 61: 2617–2637
Cheah SF, Brown, GE Jr, Parks GA. XAFS spectroscopy study of Cu(II) sorption on amorphous SiO2 and γ-Al2O3: Effect of substrate and time on sorption complexes. J Colloid Interface Sci, 1998, 208: 110–128
Dähn R, Scheidegger AM, Manceau A, Schlegel ML, Baeyens B, Bradbury MH, Morales M. Neoformation of Ni phyllosilicate upon Ni uptake on montmorillonite: a kinetics study by powder and polarized extended X-ray absorption fine structure spectroscopy. Geochim Cosmochim Acta, 2002, 66: 2335–2347
Dähn R, Scheidegger AM, Manceau A, Schlegel ML, Baeyens B, Bradbury MH, Chateigner D. Structural evidence for the sorption of Ni(II) atoms on the edges of montmorillonite clay minerals: a polarized X-ray absorption fine structure study. Geochim Cosmochim Acta, 2003, 67: 1–15
Lee S, Anderson PR, Bunker GB, Karanfil C. EXAFS study of Zn sorption mechanisms on montmorillonite. Environ Sci Technol, 2004, 38: 5426–5432
Scheidegger AM, Strawn DG, Lamble GM, Sparks DL. The kinetics of mixed Ni-Al hydroxide formation on clay and aluminum oxide minerals: a time-resolved XAFS study. Geochim Cosmochim Acta, 1998, 62: 2233–2245
Mandaliev P, Dähn R, Tits J, Wehrli B, Wieland E. EXAFS study of Nd(III) uptake by amorphous calcium silicate hydrates (C-S-H). J Colloid Interface Sci, 2010, 342: 1–7
Charlet L, Manceau A. Evidence for the neoformation of clays upon sorption of Co(II) and Ni(II) on silicates. Geochim Cosmochim Acta, 1994, 58: 2577–2582
Spadini L, Manceau A, Schindler PW, Charlet L. Structure and stability of Cd2+ surface complexes on ferric oxides. I. results from EXAFS spectroscopy. J Colloid Interface Sci, 1994, 168: 73–86
Roberts DR, Scheidegger AM, Sparks DL. Kinetics of mixed Ni-Al precipitate formation on a soil clay fraction. Environ Sci Technol, 1999, 33: 3749–3754
Ford RG, Scheckel KG, Sparks DL, Scheinost AC. The link between clay mineral weathering and the stabilization of Ni surface precipitates. Environ Sci Technol, 1999, 33: 3140–3144
Elzinga EJ, Sparks DL. Reaction condition effects on nickel sorption mechanisms in illite-water suspensions. Soil Sci Soc Am J, 2001, 65: 94–101
Strathmann TJ, Myneni SB. Effect of soil fulvic acid on nickel(II) sorption and bonding at the aqueous-boehmite (γ-AlOOH) interface. Environ Sci Technol, 2005, 39: 4027–4034
Papelis C, Brown GE Jr, Parks GA, Leckie JO. X-ray-absorption spectroscopic studies of cadmium and selenite adsorption on aluminum-oxides. Langmuir, 1995, 11: 2041–2048
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, S., Sheng, G., Guo, Z. et al. Investigation of radionuclide 63Ni(II) sequestration mechanisms on mordenite by batch and EXAFS spectroscopy study. Sci. China Chem. 55, 632–642 (2012). https://doi.org/10.1007/s11426-011-4482-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-011-4482-9