Abstract
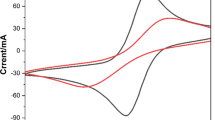
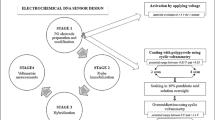
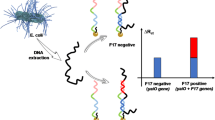
An electrochemical DNA biosensor for specific-sequences detection of Vibrio parahaemolyticus (VP) was fabricated. A single-stranded 20-mer oligonucleotide (ssDNA) and 6-mercapto-1-hexanol (MCH) were immobilized via a thiol linker on gold disk electrodes by self-assembling. The ssDNA underwent hybridization in a hybridization solution containing complementary or non-complementary or single base pair mismatched DNA sequences of VP. Examination of changes in response to these three target DNAs showed that the developed biosensor had a high selectivity and sensitivity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Janda JM, Powers C, Bryant RG, Abbott SL. Current perspectives on the epidemiology and pathogenesis of clinically significant Vibrio spp. Clin Microbiol Rev, 1988, 1(3): 245–267
Kaysner CA. Compendium of Methods for the Microbiological Examination of Foods. 3rd ed. Washington: American Public Health Association, 1992. 451–473
Sarkar BL, Nair GB, Sircar BK, Pal SC. Incidence and level of vibrio parahaemolyticus associated with freshwater plankton. Appl Environ Microbiol, 1983, 46(1): 288–290
DePaola A, Hopkins LH, Peeler JT, Wentz B, McPhearson RM. Incidence of Vibrio parahaemolyticus in US coastal waters and oysters. Appl Environ Microbiol, 1990, 56(8): 2299–2302
Kim YB, Okuda J, Matsumoto C, Takahashi N, Hashimoto S, Nishibuchi M. Identification of Vibrio parahaemolyticus strains at the species level by PCR targeted to the toxR gene. J Clin Microbiol, 1999, 37(4): 1173–1177
Lee CY, Pan SF, Chen CH. Sequence of a cloned pR72H fragment and its use for detection of Vibrio parahaemolyticus in shellfish with the PCR. Appl Environ Microbiol, 1995, 61(4): 1311–1317
Honda T, Miwatani T, Yabushita Y, Koike N, Okada K. A novel method to chemically immobilize antibody on nylon and its application to the rapid and differential detection of two Vibrio parahaemolyticus toxins in a modified enzyme-linked immunosorbent assay. Clinical and Vaccine Immunology, 1995, 2(2): 177–181
Grieshaber D, MacKenzie R, Voros J, Reimhult E. Electrochemical biosensors-sensor principles and architectures. Sensors-Basel, 2008, 8(3): 1400–1458
Palecek E, Fojta M, Jelen F. New approaches in the development of DNA sensors: hybridization and electrochemical detection of DNA and RNA at two different surfaces. Bioelectrochemistry, 2002, 56(1–2): 85–90
Ricci F, Plaxco KW. E-DNA sensors for convenient, label-free electrochemical detection of hybridization. Microchim Acta, 2008, 163(3–4): 149–155
Loaiza OA, Campuzano S, Pedrero M, Pingaron JM. Designs of enterobacteriaceae lac Z gene DNA gold screen printed biosensors. Electroanal, 2008, 20(13): 1397–1405
Susmel S, Guilbault GG, O’sullivan CK. Demonstration of labeless detection of food pathogens using electrochemical redox probe and screen printed gold electrodes. Biosens Bioelectron, 2003, 18(7): 881–889
Lucarelli F, Marrazza G, Turner APF, Mascini M. Carbon and gold electrodes as electrochemical transducers for DNA hybridisation sensors. Biosens Bioelectron, 2004, 19(6): 515–530
Nuzzo RG, Allara DL. Adsorption of bifunctional organic disulfides on gold surfaces. J Am Chem Soc, 1983, 105(13): 4481–4483
Ulman A. Formation and structure of self-assembled monolayers. Chem Rev, 1996, 96(4): 1533–1554
Del Giallo ML, Lucarelli F, Cosulich E, Pistarino E, Santamaria B, Marrazza G, Mascini M. Steric factors controlling the surface hybridization of PCR amplified sequences. Anal Chem, 2005, 77(19): 6324–6330
Fan CH, Li GX, Gu QR, Zhu JQ, Zhu DX. Electrochemical detection of Cecropin CM4 gene by single stranded probe and cysteine modified gold electrode. Anal Lett, 2000, 33(8): 1479–1490
Kafka J, Panke O, Abendroth B, Lisdat F. A label-free DNA sensor based on impedance spectroscopy. Electrochim Acta, 2008, 53(25): 7467–7474
Herne TM, Tarlov MJ. Characterization of DNA probes immobilized on gold surfaces. J Am Chem Soc, 1997, 119(38): 8916–8920
Levicky R, Herne TM, Tarlov MJ, Satijas SK. Using self-assembly to control the structure of DNA monolayers on gold: a neutron reflectivity study. J Am Chem Soc, 1998, 120(38): 9787–9792
Kerman K, Ozkan D, Kara P, Meric B, Gooding JJ, Ozsoz M. Voltammetric determination of DNA hybridization using methylene blue and self-assembled alkanethiol monolayer on gold electrodes. Anal Chim Acta, 2002, 462(1): 39–47
Steichen M, Buess-Herman C. Electrochemical detection of the immobilization and hybridization of unlabeled linear and hairpin DNA on gold. Electrochem Commun, 2005, 7(4): 416–420
Tani A, Thomson AJ, Butt JN. Methylene blue as an electrochemical discriminator of single- and double-stranded oligonucleotides immobilised on gold substrates. Analyst, 2001, 126(10): 1756–1759
Yau HCM, Chan HL, Sui SF, Yang MS. Integrity and redox properties of homogeneous and heterogeneous DNA films on gold surface probed by cyclic voltammetry. Thin Solid Films, 2002, 413(1–2): 218–223
Lee TMH, Hsing IM. Sequence-specific electrochemical detection of asymmetric PCR amplicons of traditional Chinese medicinal plant DNA. Anal Chem, 2002, 74(19): 5057–5062
Pang DW, Qi YP, Wang ZL, Cheng JK, Wang JW. Electrochemical oxidation of DNA at a gold microelectrode. Electroanal, 1995, 7(8): 774–777
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lan, M., Zhou, Q., Zhao, Y. et al. Electrochemical detection of a Vibrio parahaemolyticus sequence-specific gene based on a gold electrode modified with a single stranded probe. Sci. China Chem. 53, 1366–1370 (2010). https://doi.org/10.1007/s11426-010-3091-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-010-3091-3