Abstract
Moutan Cortex and its major compounds have been shown to possess various biological activities, including anti-inflammatory properties. However, the effects of Moutan Cortex aqueous fraction (MCA) and its molecular mechanisms have yet to be elucidated. In this study, we attempted to evaluate the effects of MCA on mast cell-mediated allergy inflammation in vitro and in vivo compared with major Moutan Cortex compounds. Thus, we examined the anti-inflammatory effects of a water extract of Moutan Cortex by comparing the inhibition of β-hexosaminadase and tumor necrosis factor-α (TNF-α) release in an aqueous fraction with other major compounds of Moutan Cortex. The inhibitory mechanism of MCA was investigated by western blotting in IgE-mediated DNP-BSA-stimulated RBL-2H3 cells. We confirmed the pharmacological effects of MCA on compound 48/80-induced allergic reactions in a mouse model by assessing scratching behavior and passive cutaneous anaphylaxis (PCA)-like reaction. Consequently, MCA inhibited IgE-mediated DNP-BSA-induced β-hexosaminadase and TNF-α release via inactivation of p38, ERK, Akt, and NF-κB in RBL-2H3 cells. MCA reduced compound 48/80-induced PCA reaction and scratching behavior in mice. This inhibitory effect of MCA is more potent than major compounds of Moutan Cortex. In conclusion, our results suggest that MCA has more potential in the treatment of allergic inflammatory diseases compared to other major compounds of Moutan Cortex.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Allergic inflammation is a hypersensitivity disorder of the immune system and occurs in several important medical conditions, such as allergic eczema, atopic dermatitis, allergic rhinitis, and other allergy-related diseases. It represents an important public health issue due to its impact on quality of life and its socioeconomic burden [1]. Allergy is induced by the sudden systemic release of inflammatory mediators such as histamine, serotonin, and various pro-inflammatory cytokines from mast cells.
Mast cells are the primary effector cells involved in the allergic inflammatory reaction [2, 3]. The degranulation process starts by the interaction of antigens with immunoglobulin (Ig)E molecules already bound to high affinity Fc receptors (FcεRI) on the surface of mast cells [4]. In addition, mast cells secrete a variety of bioactive substances such as histamine and proinflammatory cytokines such as interleukin (IL)-6 and TNF against various stimuli [5]. These mediators contribute to inflammation process through the activation and migration of immune cells [6]. Therefore, the inhibition of these mediators can be a useful therapeutic strategy for allergic inflammatory diseases.
In traditional oriental medicines, Moutan Cortex, the root of Paeonia suffruticosa, functions as a heat-clearing and blood-cooling agent, promoting blood flow and dissipating blood stasis; for these reasons, it is used to treat inflammatory diseases [7]. In recent studies, ethanol or methanol extract of Moutan Cortex and its major compounds, such as paeonol and paeoniflorin, have been described to possess anti-inflammatory and anti-allergic activity that reduces the production of NO and prostaglandin E2 (PGE2) and the expression of inducible NO synthase (iNOS), cyclooxygenase-2 (COX-2), tumor necrosis factor-α (TNF-α), IL-1β and IL-6, phosphorylated inhibitor of IκBα, and the activation of nuclear factor κB (NF-κB) [8–12]. Although water extract of Moutan Cortex has been used in traditional formulas, its effect on allergic inflammatory responses is unknown.
In this study, we prepared an aqueous fraction from water extract and studied the inhibitory effect and mechanisms of a Moutan Cortex aqueous fraction (MCA) on IgE-sensitized and DNP-BSA-stimulated allergic reactions in mast cells. In addition, compound 48/80-mediated acute allergic reactions, such as systemic anaphylaxis-like reaction and scratching behavior, were also inhibited by MCA compared with other compounds in a mouse model. Collectively, MCA may be useful for the treatment of allergic inflammatory diseases.
Materials and methods
Antibodies and reagents
Anti-phospho Akt (Ser-473), ERK (Thr-202/Tyr-204), p38 (Thr-180/Tyr-182), and JNK (Thr-183/Tyr-185) antibodies were purchased from Cell Signaling Technology (Danvers, MA). Antibodies against Akt (C-20), p38 (C-20-G), JNK (FL), ERK1 (C-16), and β-actin (C-11) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Anti-dinitrophenyl (DNP) IgE monoclonal antibody and compound 48/80 were obtained from Sigma (St. Louis, MO). Dinitrophenyl-bovine serum albumin (DNP-BSA) was obtained from Calbiochem (San Diego, CA). Paeonol and paeoniflorin were obtained from Santa Cruz Biotechnology. Paeonolide was obtained from the Tokyo Chemical Industry (Tokyo, Japan). Methyl gallate and Evans blue were purchased from Wako Pure Chemical Industries (Tokyo, Japan).
Cell culture
Rat basophilic leukemia mast cell line RBL-2H3 was purchased from ATCC (Manassas, VA). This cell line was maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10 % heat-inactivated fetal calf serum, 100 units/mL penicillin, and 100 μg/mL streptomycin at 37 °C in a humidified incubator under 5 % CO2.
Mice
ICR 6-week-old female mice were purchased from Japan SLC (Hamamatsu, Japan). They were housed four per cage in a laminar airflow room and maintained at 22 ± 1 °C with a relative humidity of 55 ± 10 % throughout the study. The study was conducted in accordance with the standards established in the Guidelines for the Care and Use of Laboratory Animals of Toyama University.
Preparation of MCA
Moutan Cortex prepared from the root bark of P. suffruticosa Andrews produced in China was purchased from TOCHIMOTO TENKAIDO Pharmacy Co. (Osaka, Japan). Moutan cortex (45 g) was extracted at 95–98 °C for 1 h in 1,700 mL distilled water (DW) and then freeze dried. A 100 mg freeze-dried extract was dissolved in 2 mL DW and adsorbed onto solid phase in a Sep-Pak C18 cartridge (Waters, Milford, MA). Components were eluted with a 10 mL solution of 8 mL DW and 20 % methanol (2 mL), and the solvent was removed by a rotary evaporator. The residual substance was named as MCA.
Assays of cell viability
Cell viability was quantified using the cell proliferation reagent 2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2, 4-disulfophenyl)-2H-tetrazolium, monosodium salt (WST-8; Dojindo, Kumamoto, Japan). Cells were plated 2 × 103 cells/well in 96-well microplates and then incubated for 1 h. Medium containing MCA was then added to the wells. After 24 h incubation, the medium was changed and 10 μL of the WST-8 solution was added. Absorbance was measured at 450 nm.
β-hexosaminidase assay
The release of mast cell mediators by exocytosis was monitored by the β-hexosaminidase assay. RBL-2H3 cells (1 × 105 cells) in 48-well plates were sensitized with anti-DNP IgE (100 ng/mL) for 6 h. The cells were then washed with Siraganian buffer (pH 7.2, 119 mM NaCl, 5 mM KCl, 0.4 mM MgCl2, 25 mM PIPES), incubated in Siraganian buffer containing 5.6 mM CaCl2 and 0.1 % bovine serum albumin (BSA) for 10 min, and then incubated in pretreated MCA (1 and 10 μg/mL) for 30 min. Thereafter, the cells were stimulated for 1 h with DNP-BSA (1 μg/mL), which activates RBL-2H3 cells into producing allergic reactions. The supernatant (50 μL) was mixed with an equal volume (20 μL) of substrate solution (2 mM p-nitrophenyl-N-acetyl-β-d-glucosaminide in 0.1 M sodium citrate buffer, pH 1.5), and the mixture was incubated for 1 h at 37 °C. The reaction was terminated by adding 200 μL stopping buffer (0.1 M Na2CO3/NaHCO3, pH 10.0). Absorbance at 405 nm was measured with a microplate reader. MCA-mediated inhibition of β-hexosaminidase release was expressed as the inhibition percentage and was calculated using the following equation:
where A control is the absorbance of the control and A sample is the absorbance of the samples.
Real-time RT-PCR
RBL cells (2 × 105 cells) were seeded in six-well plates and sensitized with 100 ng/mL anti-DNP IgE for 1 h at 37 °C, and then phosphate-buffered saline (PBS) buffer, incubated with sample or medium for 1 h at 37 °C. For degranulation, cells were stimulated with 1 μg/mL DNP-BSA for 30 min at 37 °C. Total RNAs were prepared using the RNeasy Mini kit (Qiagen, Valencia, CA). First-strand cDNAs were synthesized by SuperScript II reverse transcriptase (Invitrogen, Carlsbad, CA). The cDNAs were amplified using FastStart Essential DNA Green Master (Roche, Pleasanton, CA). Forward/reverse RT-PCR primer pairs for mouse cDNAs were as follows: TNF-α (5′-TCTTCTCATTCCTGCTTGTGG-3′/5′-GGTCTGGGCCATAGAACTGA-3′); β-actin (5′-CTAAGGCCAACCGTGAAAAG-3′/5′-ACCAGAGGCATACAGGGACA-3′). Real-time RT-PCR was performed using a Lightcycler nano system (Roche). All data were normalized to β-actin mRNA levels.
Measurement of TNF-α production
RBL-2H3 cells (2 × 105 cells) were incubated with anti-DNP IgE (100 μg/mL) for 6 h and treated with MCA (0.1, 1, and 10 μg/mL) for 30 min. The cells were washed with Siraganian buffer and incubated in Siraganian buffer containing 5.6 mM CaCl2 and 0.1 % BSA. The cells were stimulated with DNP-BSA (1 μg/mL) for 2 h. The supernatants (100 μL per well) of these cells were determined using a mouse TNF-α ELISA kit (eBioscience, San Diego, CA) according to the manufacturer’s instructions.
Western blotting analysis
Cells were harvested and plated in a 6-cm dish (1 × 106 cells) for 1 h. Cells were then sensitized with DNP-IgE (100 ng/mL) for 1 h and stimulated with DNP-BSA (1 μg/mL) for 30 min. The whole-cell lysates were prepared with lysis buffer [25 mM HEPES (pH 7.7), 0.3 M NaCl, 1.5 mM MgCl2, 0.2 mM EDTA, 10 % Triton X-100, 20 mM β-glycerophosphate, 1 mM sodium orthovanadate, 1 mM dithiothreitol (DTT), 10 μg/mL aprotinin, and 10 μg/mL leupeptin]. Cell lysates were collected from the supernatant after centrifugation at 14,000 rpm for 10 min. Nuclear extracts were prepared by suspending cells in 420 μL buffer A [10 mM HEPES (pH 7.9), 10 mM KCL, 0.1 mM EDTA, 0.1 mM EGTA, 1 mM DTT, 1 mM PMSF, 20 mM β-glycerophosphate, 0.1 mM sodium orthovanadate, 10 μg/mL aprotinin, and 10 μg/mL leupeptin] and chilled on ice for 15 min. Next, 25 μL 10 % Nonidet P-40 was added, and the suspension was vortexed vigorously for 10 s. The nuclear pellets were suspended in 50 μL buffer B [20 mM HEPES (pH 7.9), 0.4 M NaCl, 1 mM EDTA, 1 mM EGTA, 1 mM DTT, 1 mM PMSF, 20 mM β-glycerophosphate, 1 mM sodium orthovanadate, 10 μg/ml aprotinin, and 10 μg/ml leupeptin]. The mixture was kept on ice for 15 min with frequent agitation. Nuclear extracts were prepared by centrifugation at 15,000 rpm for 5 min. Cell lysate was resolved by SDS-PAGE and transferred to an immobilon-P-nylon membrane (Millipore, Bedford, MA). The membrane was blocked with Block Ace (Dainippon Pharmaceutical Co., Suita, Japan) and probed with primary antibodies. The antibodies were detected using horseradish peroxidase-conjugated anti-rabbit and anti-mouse immunoglobulin G (Dako, Carpinteria, CA), and blots were detected using the enhanced chemiluminescence system (Amersham Biosciences, Piscataway, NJ).
Scratching behavior
Azelastine, MCA and Moutan Cortex major compounds were suspended in physiological saline and administrated with 100 μL to the dorsal skin 1 h before the start of behavioral observations. Prior to the observations, the animals were placed individually in an acrylic cage containing four wells (13 × 9 × 35 cm) for at least 1 h to allow them to acclimate. Compound 48/80 (10 μg/50 μL) was injected intradermally into the central part of the mouse dorsal skin. Immediately after injection, the mice were placed back into the same wells, and their behavior was videotaped for 1 h. Playback of the video allowed for determination of hind-paw scratching.
Compound 48/80-induced a passive cutaneous anaphylaxis (PCA)-like reaction
The day before the experiments, hair was removed with hair clippers from the rostral part of the back or cheek. Ethanol (100 μL) was applied to the mice for 10 min, and MCA (10, 100 μg/100 μL) was administered intravenously 30 min before the challenge. Each mouse received an injection of compound 48/80 (10 μg/50 μL) in saline containing 1 % Evans blue via tail vein 1 h later. The mice were killed using cervical dislocation after the challenge, and the dorsal skin was collected in order to measure the pigment area and incubated with formamide for overnight. The absorbance at 620 nm was measured using a microplate reader.
Liquid chromatography–mass spectrometry analysis
The liquid chromatography–mass spectrometry (LC-MS) analysis of MCA was performed using a Bruker esquire 3000 quadrupole ion trap mass spectrometer, coupled to an Agilent 1100 Series HPLC system, operated in the positive electrospray ionization (ESI) mode scanning from m/z 200 to 2000 [source voltage, +4.5 kV; capillary temperature, 360 °C; Nebulizer gas, 7 l/min; column, Waters Atrantis T3 (2.1 × 150 mm); column temperature, 40 °C]. The mobile phase was a binary eluent of (A) 0.1 % formic acid solution and (B) CH3CN under the following gradient conditions: 0–40 min linear gradient from 5 % to 25 % B, 40–70 min 25 % to 100 % B, 70–80 min isocratic at 100 % B. The flow rate was 0.2 mL/min. The water extract of Moutan Cortex and MCA were dissolved in HPLC grade H2O (10 mg/mL) and filtered using an Advantec DISMIC-13HP filter (0.45 μm, Toyo Roshi Kaisha, Japan), and aliquots of the filtrate (5 μL) were injected for analysis. The standards, paeoniflorin and paeonol, were dissolved in HPLC grade methanol (1 mg/mL) and 5 μL of both solutions were injected for analysis.
Statistical analysis
The results are presented as a summary of the data from three experiments. Data were analyzed by the Student’s t test and presented as the mean ± standard deviation (SD). Results with P < 0.05 were considered significant.
Results
Effect of MCA on cytotoxicity and β-hexodaminidase release
The WST assay was used to determine the viability of RBL-2H3 cells exposed to MCA. As shown in Fig. 1a, up to 100 μg/mL MCA did not cause any cytotoxicity after 24 h incubation compared with the control, whereas 1,000 μg/mL MCA showed cytotoxicity. RBL-2H3 cells were used to assess in vitro degranulation-inhibitory activity induced by the IgE–antigen complex through a β-hexodaminidase assay. The amount of β-hexodaminidase was determined by fluorometric and ELISA methods. MCA (10 and 100 μg/mL) inhibited release of β-hexodaminidase from RBL-2H3 cells, but not in a dose-dependent manner (Fig. 1b; Supplementary Material 1). Therefore, we used a maximum concentration of MCA of 10 μg/mL for in vitro experiments.
Cytotoxic effects on RBL-2H3 cells and inhibitory effects on β-hexosaminidase release by Moutan Cortex aqueous fraction (MCA). a Cell viability of MCA on RBL-2H3 cells. RBL-2H3 cells (2 × 104 cells) were cultured with 100 ng/mL anti-DNP IgE for 12 h. The cytotoxicity of MCA on RBL-2H3 cells was determined by WST-8 assay after treatment with MCA for 24 h. b Inhibitory effect of MCA on β-hexosaminidase release from RBL-2H3 cells. RBL-2H3 cells were sensitized with DNP-IgE (100 ng/mL) for 12 h and pretreated with MCA (1–100 μg/mL). After 1 h, cells were stimulated with DNP-BSA for 30 min, and β-hexosaminidase release was measured by enzymatic assay. Data are the mean ± SD of three independent experiments. * P < 0.05 vs control
Effect of MCA on mRNA expression and protein secretion of TNF-α
Among the proinflammatory cytokines, TNF-α is released from mast cells by degranulation and consequently aggravate allergic inflammation [2]. Therefore, we investigated the effect of MCA on IgE-sensitized and DNP-BSA-induced production of TNF-α from RBL-2H3 cells. Treatment with MCA significantly suppressed expression of TNF-α more than other major compounds of Moutan Cortex (Fig. 2a). Additionally, MCA treatment resulted in a significant suppression of TNF-α release from RBL-2H3 cells compared to paeonol (Fig. 2b).
Inhibitory effect of MCA on TNF-α expression and secretion. a mRNA levels of TNF-α. RBL-2H3 cells (1 × 106 cells) were sensitized with 100 ng/mL anti-DNP IgE for 12 h and pretreated with various doses of MCA (0.1, 1, and 10 μg/mL) or paeonol (100 μM), paeoniflorin (50 μM), paeonolide (50 μM), or methyl gallate (10 μg/mL) for 1 h. Cells were stimulated with 10 μg/mL DNP-BSA for 30 min. b Secretion of TNF-α. RBL-2H3 cells (5 × 105 cells) were sensitized with 100 ng/mL anti-DNP IgE for 12 h and pretreated with various doses of MCA (0.1, 1, and 10 μg/mL) or paeonol (100 μM) for 1 h. Cells were stimulated with 10 μg/mL DNP-BSA for 1 h, and the supernatant was collected for ELISA. Results are expressed as mean ± SD (n = 5). B, Blank; C, control; P, paeonol; PF, paeoniflorin; PN, paeonolide; MG, methyl gallate. # P < 0.05 vs blank, ** P < 0.01, *** P < 0.001 vs control
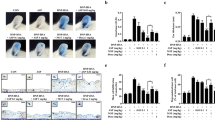
Effect of MCA on mitogen-activated protein kinase phosphorylation and degradation of IκBα and NF-κB activation in RBL-2H3 cells
The production of pro-inflammatory cytokines is induced by allergen stimulation via activation of mitogen-activated protein kinases (MAPKs) and the NF-κB pathway. For this reason, the NF-κB signaling pathway is an important component in the regulation of inflammatory responses [15, 16]. To determine which molecules are regulated by MCA in an allergic reaction, western blotting analysis was conducted. Phosphorylation of p38, ERK, and Akt were increased markedly 30 min after treatment with DNP-BSA in RBL-2H3 cells. As shown in Fig. 3a, the phosphorylation of p38, ERK, and Akt was dose-dependently inhibited by MCA treatment, whereas phosphorylation of JNK did not change. Because suppression of NF-κB activation has been linked with anti-inflammation, we theorized that the NF-κB activation pathway might be regulated by MCA; therefore, we evaluated cytosol IκBα and nuclear NF-κB levels via Western blot analysis. As shown in Fig. 3b, we determined that DNP-BSA treatment effectively induced the degradation of IκBα and NF-κB translocation. However, 10 μg/mL MCA inhibited IκBα degradation and nuclear levels of NF-κB in RBL-2H3 cells.
Effect of MCA on IgE-sensitized and DNP-BSA-induced mitogen-activated protein kinases (MAPKs) and the NF-κB signaling pathway. a Phosphorylation of MAPKs in DNP-BSA-stimulated RBL-2H3 cells. The cells were pretreated with MCA (0.1, 1, and 10 μg/mL) and paeonol (100 μM) for 1 h and then stimulated with DNP-BSA for 30 min. b Degradation of IκBα and NF-κB activation. The cytosolic extracts were prepared and evaluated for IκBα. Nuclear extracts were prepared and evaluated for p65 via Western blot analysis. B, Blank; C, control; P, paeonol
Effect of MCA on compound 48/80-induced scratching behavior
We examined the effect of MCA on compound 48/80-induced scratching behavior. MCA at doses of 100 and 300 μg potently inhibited scratching behavior by 49 % and 38 %, respectively; paeoniflorin and methyl gallate have been shown to inhibit scratching behavior by 17 % and 15 %, respectively (Fig. 4).
Effect of MCA on compound 48/80-induced scratching behavior. Azelastine (1 %), MCA (100 and 300 μg), and compounds of Moutan cortex paeonol (100 μM), paeoniflorin (50 μM), paeonolide (50 μM), and methyl gallate (10 μg) were administrated on dorsal skin 1 h before administering compound 48/80 (10 ng/spot). Azelastine, a representative anti-allergy drug [20], is a H1-receptor antagonist and decreases the mediator release from mast cells and was used as a positive control. P, Paeonol; PF, paeoniflorin; PN, paeonolide; MG, methyl gallate. Each value represents the mean ± SD (n = 4). *P < 0.05, **P < 0.01, ***P < 0.001 vs control group
Effect of MCA on compound 48/80-induced PCA-like reaction
PCA induced by injection of DNP-IgE and compound 48/80 is one of the most important in vivo models for a local allergic reaction [17, 18]. The PCA reaction was dose-dependently inhibited by MCA at the doses of 10 and 100 μg when mice were injected intradermally with 10 ng/mL compound 48/80 (Fig. 5).
Effect of MCA on the PCA-like reaction induced by compound 48/80. Compound 48/80 (10 ng/spot) was injected intradermally into the dorsal skin of mice. After 1 h, the mice were intravenously challenged with 1 % Evans blue dye as described in the text. The indicated doses of MCA were treated on the dorsal skin before antigen stimulation. Each value represents the mean ± SD (n = 5). *P < 0.05 vs control group
LC–MS and LC UV analysis
Total ion chromatograms (TIC) of MCA, water extract of Moutan Cortex and the standards paeoniflorin and paeonol were obtained from LC–MS analysis under the same conditions (Fig. 6). Components observed in TIC of MCA (Fig. 6a) were detected before the retention time of 34 min, compared to TIC of water extract of Moutan Cortex (Fig. 6b). Paeoniflorin (Fig. 6c)—a major compound from Moutan Cortex—was detected in MCA, while paeonol (Fig. 6d) was not found in MCA.
Discussion
In this study, we investigated the effect of MCA on mast cell-mediated allergic inflammation. Major compounds of MCA have been shown to have an anti-inflammatory effect [11, 12]. We assumed that not only lipophilic extracts and compounds but also water extract has an anti-allergic function because formulas containing Moutan Cortex are boiled in water in traditional medicine. We therefore compared the effect of MCA and major compounds on mast cell degranulation and pro-inflammatory cytokine production.
The rat basophilic leukemia cell line RBL-2H3, displaying characteristics of mucosal-type mast cells, is considered to be a good model for studying comprehensive events on mast cell activation [19]. TNF-α—a representative pro-inflammatory cytokine—is presented in the cytoplasmic granules of mast cells and is released through mast cell degranulation immediately after antigenic stimulation [20]. In addition, cross-linking of FcεRI can lead to an increase in TNF-α mRNA expression. TNF-α exacerbates allergic symptoms by inducing chemotaxis of neutrophils and production of other inflammatory cytokines and plays a critical role in allergic inflammation [2]. We found that MCA significantly inhibited TNF-α production in RBL-2H3 cells (Fig. 3). It is also known that paeonol functions as a regulator of inflammation through inhibition of TNF-α [11]. Taken together, the main cause of the anti-inflammatory effect of Moutan Cortex is the inhibition of TNF-α production.
The anti-allergic and anti-inflammatory activities of an 80 % ethanol extract of Moutan Cortex via regulating phosphorylation of p38, ERK, Akt and degranulation of IκBα and NF-κB have been elucidated [9]. Therefore, we investigated expression of MAPKs. Our results showed an inhibitory effect of MCA on mast cells through the suppression of MAPK phosphorylation (Fig. 3). MCA significantly inhibited the activation of p38, ERK, and Akt (Fig. 3a) and MCA blocked both phosphorylation of IκBα and nuclear translocation of NF-κB (Fig. 3b). MCA showed higher inhibitory activity because it controlled phosphorylation of p38, ERK, and Akt, whereas lipophilic fractions inhibited the phosphorylation of p38 and ERK in the MAPK pathway [12]. In addition, the effect of MCA in allergic inflammation was compared with paeonol, which is the main compound of Moutan Cortex. Paeonol has been reported to significantly inhibit the release of histamine, IL-4 and TNF-α [11]. However, we found no evidence that paeonol had an inhibitory effect on the activation of the MAPK pathway in RBL-2H3 cells (Fig. 3). Thus, further study is needed to investigate the pathway of the anti-allergic effect of paeonol in mast cells.
In this study we selected skin application to investigate the effect of MCA on immediate hypersensitivity in vivo model. However, skin absorption of the aqueous fraction is very low; therefore, ethanol was applied to increase skin penetration before we spread the MCA on the dorsal skin. Additionally, we increased the MCA concentration to 100 μg and 300 μg. MCA reduced allergic responses at concentrations of 100 μg and 300 μg (Figs. 4, 5) in the scratching behavior and PCA reaction experiments. Further studies are needed that focus on the permeability of the aqueous fraction for external application.
In addition, azelastine was used as a positive control in the in vivo model. Azelastine is a potent histamine antagonist and has been formulated both as eye drops and nasal spray [21]. It suppresses degranulation of mast cells by inhibition of TNF-α secretion in RBL-2H3 cells [22]. We found that MCA also significantly decreased scratching behavior while azelastine was most effective in experimental groups (Fig. 4). This result suggests that MCA has potential use as an anti-allergenic herbal medicine and could be used as an external preparation.
Moutan Cortex and its major compounds paeonol and paeoniflorin showed anti-allergic and anti-inflammatory effects [9–12]. Ethanol extract of Moutan Cortex decreased the release of histamine (10–100 μg/mL), TNF-α and IL-6 (50–200 μg/mL) in mast cells. In in vivo experiments, scratching behavior and PCA reaction were inhibited by 10 mg/kg and 200 mg/kg Moutan Cortex ethanol extract, respectively [10, 11]. Among the Moutan Cortex major compounds, paeonol and paeoniflorin at a dose of 50 mg/kg inhibited the PCA reaction and scratching behavior. Moreover, paeonol decreased β-hexosaminidase, IL-4 and TNF-α release (100 μM) and histamine release (0.01–1 mg/mL) in mast cells [11, 12]. In recent study, the antiallergic effect of Moutan radicis cortex water extract (MW) and its lipophilic extracts were reported. Interestingly, MW significantly suppressed allergic responses such as systemic anaphylactic shock, PCA reaction and histamine release, whereas the ethylacetate layer did not inhibit the production of antigen-specific IgG1 and IgE antibody in mice [23]. Similarly, MCA inhibited β-hexosaminidase and TNF-α release at a dose of 10 μg/mL (Figs. 1, 2; Supplementary Material 1), whereas paeonol showed an inhibitory effect at a dose of 100 μM [11, 12]. These results suggested that Moutan Cortex water extract is more effective in countering allergic responses compared with lipophilic extract. It has been reported that Paeonia suffruticosa water extract contains paeonol, paeoniflorin, derivatives of paeoniflorin (oxypaeoniflorin, benzoylpaeoniflorin), benzoic acid and 1,2,3,4,6-Penta-O-galloyl-β-d-glucopyranose (PGG) [24]. Of these constituents, PGG shows anti-allergic effects through reduction of IL-4-induced IgE production via inhibiting the JAK3/STAT6 signaling pathway and down-regulation of mast cell surface FcεRI expression in vitro and in vivo [25, 26]. Therefore, further studies are required to identify the constituents, including PGG, of Paeonia suffruticosa water extract that have anti-allergic effects.
Conclusion
We have shown for the first time that the inhibitory effect of MCA is more potent than that of other compounds. This study provides evidence for the traditional and pharmacological use of Moutan Cortex water extract in the treatment of allergic inflammation. Further studies are needed to clarify the medicinal properties of MCA and examine application methods.
References
Pawankar R, Canonica GW, Holgate, ST, Lockey RF (2012) WAO White book on allergy 2011–2012: executive summary. http://www.worldallergy.org/publications/wao_white_book.pdf
Bischoff SC (2007) Role of mast cells in allergic and non-allergic immune responses: comparison of human and murine data. Nat Rev Immunol 7:93–104
Theoharides TC, Kempuraj D, Tagen M, Conti P, Kalogeromitros D (2007) Differential release of mast cell mediators and the pathogenesis of inflammation. Immunol Rev 217:65–78
Yamasaki S, Saito T (2005) Regulation of mast cell activation through FcepsilonRI. Chem Immunol Allergy 87:22–31
Galli SJ, Kalesnikoff J, Grimbaldeston MA, Piliponsky AM, Williams CM, Tsai M (2005) Mast cells as “tunable” effector and immunoregulatory cells: recent advances. Annu Rev Immunol 23:749–786
Galli SJ, Tsai M (2010) Mast cells in allergy and infection: versatile effector and regulatory cells in innate and adaptive immunity. Eur J Immunol 40:1843–1851
Huang KC (1999) The pharmacology of Chinese herbs. CRC, New York, pp 403–404
Chun SC, Jee SY, Lee SG, Park SJ, Lee JR, Kim SC (2007) Anti-inflammatory activity of the methanol extract of moutan cortex in LPS-activated Raw264.7 cells. Evid Based Complement Alternat Med 4:327–333
Hong MH, Kim JH, Na SH, Bae H, Shin YC, Kim SH, Ko SG (2010) Inhibitory effects of Paeonia suffruticosa on allergic reactions by inhibiting the NF-kappaB/IkappaB-alpha signaling pathway and phosphorylation of ERK in an animal model and human mast cells. Biosci Biotechnol Biochem 74:1152–1156
Jiang S, Nakano Y, Yatsuzuka R, Ono R, Kamei C (2007) Inhibitory effects of Moutan cortex on immediate allergic reactions. Biol Pharm Bull 30:1707–1710
Kim SH, Kim SA, Park MK, Kim SH, Park YD, Na HJ, Kim HM, Shin MK, Ahn KS (2004) Paeonol inhibits anaphylactic reaction by regulating histamine and TNF-alpha. Int Immunopharmacol 4:279–287
Lee B, Shin YW, Bae EA, Han SJ, Kim JS, Kang SS, Kim DH (2008) Antiallergic effect of the root of Paeonia lactiflora and its constituents paeoniflorin and paeonol. Arch Pharm Res 31:445–450
Alfonso A, Cabado AG, Vieytes MR, Botana LM (2000) Functional compartments in rat mast cells for cAMP and calcium on histamine release. Cell Signal 12:343–350
Kaliner M, Austen KF (1974) Cyclic AMP, ATP, and reversed anaphylactic histamine release from rat mast cells. J Immunol 112:664–674
Shakoory B, Fitzgerald SM, Lee SA, Chi DS, Krishnaswamy G (2004) The role of human mast cell-derived cytokines in eosinophil biology. J Interferon Cytokine Res 24:271–281
Wong CK, Wang CB, Ip WK, Tian YP, Lam CW (2005) Role of p38 MAPK and NF-kB for chemokine release in coculture of human eosinophils and bronchial epithelial cells. Clin Exp Immunol 139:90–100
Wershil BK, Mekori YA, Murakami T, Galli SJ (1987) 125I-fibrin deposition in IgE-dependent immediate hypersensitivity reactions in mouse skin. Demonstration of the role of mast cells using genetically mast cell-deficient mice locally reconstituted with cultured mast cells. J Immunol 139:2605–2614
Choi YH, Chai OH, Han EH, Choi SY, Kim HT, Song CH (2010) Lipoic acid suppresses compound 48/80-induced anaphylaxis-like reaction. Anat Cell Biol 43:317–324
Passante E, Frankish N (2009) The RBL-2H3 cell line: its provenance and suitability as a model for the mast cell. Inflamm Res 58:737–745
Walsh LJ, Trinchieri G, Waldorf HA, Whitaker D, Murphy GF (1991) Human dermal mast cells contain and release tumor necrosis factor alpha, which induces endothelial leukocyte adhesion molecule 1. Proc Natl Acad Sci USA 88:4220–4224
Chand N, Pillar J, Diamantis W, Sofia RD (1985) Inhibition of allergic histamine release by azelastine and selected antiallergic drugs from rabbit leukocytes. Int Arch Allergy Appl Immunol 77:451–455
Hide I, Toriu N, Nuibe T, Inoue A, Hide M, Yamamoto S, Nakata Y (1997) Suppression of TNF-alpha secretion by 10% azelastine in a rat mast (RBL-2H3) cell line: evidence for differential regulation of TNF-alpha release, transcription, and degranulation. J Immunol 159:2932–2940
Lee SW, Kim DH, Seol IC, Kim SH (2001) Antiallergic effect of water and EA extract of Moutan radicis cortex. Korean J Orient Physiol Pathol 15:631–635
Fujiwara H, Tabuchi M, Yamaguchi T, Iwasaki K, Furukawa K, Sekiguchi K, Ikarashi Y, Kudo Y, Higuchi M, Saido TC, Maeda S, Takashima A, Hara M, Yaegashi N, Kase Y, Arai H (2009) A traditional medicinal herb Paeonia suffruticosa and its active constituent 1,2,3,4,6-penta-O-galloyl-beta-d-glucopyranose have potent anti-aggregation effects on Alzheimer’s amyloid beta proteins in vitro and in vivo. J Neurochem 109:1648–1657
Kim YH, Yoshimoto M, Nakayama K, Tanino S, Fujimura Y, Yamada K, Tachibana H (2013) Tannic acid, a higher galloylated pentagalloylglucose, suppresses antigen-specific IgE production by inhibiting ɛ germline transcription induced by STAT6 activation. FEBS Open Bio 5:341–345
Kageyama-Yahara N, Suehiro Y, Maeda F, Kageyama S, Fukuoka J, Katagiri T, Yamamoto T, Kadowaki M (2009) Pentagalloylglucose down-regulates mast cell surface FcepsilonRI expression in vitro and in vivo. FEBS Lett 584:111–118
Acknowledgments
This study was supported by a Grant-in-Aid for Health Labour Sciences Research Grant and a Grant-in-Aid for the Cooperative Research Project from Institute of Natural Medicine, University of Toyama.
Conflict of interest
We have no financial relationships to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kee, JY., Inujima, A., Andoh, T. et al. Inhibitory effect of Moutan Cortex aqueous fraction on mast cell-mediated allergic inflammation. J Nat Med 69, 209–217 (2015). https://doi.org/10.1007/s11418-014-0880-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-014-0880-6