Abstract
Purpose
Nitrous oxide (N2O) measured simultaneously with di-nitrogen (N2) emissions from soils are greatly uncertain due to large temporal and spatial variations. This study aims to report N2O, N2, N2/N2O, 15N-N2O, wheat-maize annual grain yields, and yield-scaled N2O and N2O plus N2 emissions on the responses to different nitrogen (N) fertilizer rates in a winter wheat-summer maize cropping system. Furthermore, this study also seeks to determine controlling factors for N2O, N2, and N2/N2O emissions and significantly investigate the relationship between the soil-climate measured factors and 15N-N2O.
Materials and method
Three N inputs and control treatments, 0, CK; 200, LN; 400, MN; and 600, HN kg N ha−1 year−1 were set since 1998. Direct measurement method has been used to quantify N2O and N2 emissions.
Results
Our results indicated that the effects of long-term N fertilization significantly increased N2O and N2 and also reduced N2/N2O emission ratios as described by exponential functions. Using structural equation modeling (SEM), NH4+, WFPS, NO3-, and DOC were revealed to be main controlling factors for N2O, while N2 by DOC, NO3-, WFPS, and temperature finally N2/N2O was positively related to temperature. Furthermore, the 15N-N2O was positively related to N2/N2O ratios, indicating that denitrification is the dominant process at the study site. The yield-scaled N2O emissions followed the order HM>MN>LN>CK, and they were 1.56, 1.47, and 1.07 times greater than CK, respectively. Total yield-scaled N2O plus N2 were in the order of CK>HN>MN>LN.
Conclusion
N fertilization has shown strong impact not only on N2O, N2, and N2/N2O emissions but also on yield-scaled N2O and N2O plus N2 emissions. High agronomic nitrogen use efficiency (NUE), low yield-scaled N2O emissions, and low cumulative N2O plus N2 emissions were observed at 200-LN treatment, suggesting this rate to be an optimum and sustainable agricultural management practice with no significant crop yield reduction as compared to the current farmers’ practice of 400 kg N ha−1 year−1.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Globally, a half of nitrogen (N) fertilizer applied into agricultural soils is uptaken by crops, while another half either remains in the soils or lost as reactive N (Nr) to the atmosphere (de Vries et al. 2011; Lassaletta et al. 2014). Since N plays a vital role in plant growth, the increase in usage of N fertilizer has enhanced crop yields that facilitated to supply food to ever-growing world population, estimated to more than 7.2 billion inhabitants today (Erisman et al. 2008; Grafton et al. 2015). Since, N is one of the macronutrient limiting factors for crop growth (Wang et al. 2010), the increase of N fertilizer application rates will also increase N2O and N2 losses from agricultural soil (Wang et al. 2020).
Nitrous oxide (N2O) is a potent and long-lived major greenhouse gas that contributes to ozone depletion and global warming (Forster et al. 2007; Saggar et al. 2007). Since pre-industrial (1750) time, atmospheric concentration of N2O has been increasing at a rate of 0.8 ppb year−1 (Prinn et al. 2000; IPCC 2007) that makes it to increase from 270 to 330 ppb by 2019 (NoAA 2019). Fertilized agricultural soils are estimated to emit 4.1 Tg N2O annually (IPCC 2013) that makes it a significant source of N2O among other anthropogenic sources. Fan et al. (2017) reported its contribution as more than 60% of worldwide anthropogenic N2O emissions. However, N2O measured simultaneous with N2 have not been well documented in agricultural soils.
Nitrification and denitrification are two microbial processes considered to be main soil N2O formation (Saggar et al. 2013; Timilsina et al. 2020a), while denitrification process reduces N2O formed into N2 under conducive conditions (e.g. Dannenmann et al. 2008; Saggar et al. 2013). Exponential relationships between N fertilizer rates and direct N2O emissions have been well documented (Kim et al. 2013; Cui et al. 2014; Shcherbak et al. 2014; Song et al. 2018), while some other studies also reported linear relationships between N fertilizer rates and direct N2O emissions (Liu et al. 2012; Huang et al. 2017). However, the relationships between N application rates and direct N2O and N2 in agricultural soils remain unclear.
N2O emitted through nitrification are more depleted in 15N isotopic composition than denitrification (Baggs 2008; Park et al. 2011). Studying the 15N isotopic composition of soil-emitted N2O with soil variables and soil N2O and N2 losses would further enhance our understanding of N2O formation and reduction processes in the current study. Moreover, soil-emitted 15N-N2O from fertilized and unfertilized agricultural soil will help to insight each contribution in depletion of 15N isotopic composition of atmospheric N2O.
North China Plain (NCP) is the key agricultural area mainly yielding cereals, and if compared to other regions in China, the previous study reported that 26% and 76% of total maize and wheat were produced there, respectively (Tan et al. 2017), but the N fertilizer input is up to 550–600 kg N ha−1 year−1, and the amount is quite high as compared to the required N by crops ranging from 200 to 300 kg N ha−1 year−1 (Meng et al. 2012), and also Wang et al. (2010) proposed the N fertilizer rate of 360 kg N ha−1 year−1 for effective use of N fertilizer. In addition, Pan et al. (2012) reported the average annual N deposition of 61 kg per hectare per year in NCP. Hence, there is a surplus of N in the soil. Moreover, in vegetables, excessive N fertilizer application had been also observed in different climatic regions of China; Zhang et al. (2020) proposed that 81–90% of N2O emissions could be reduced when N fertilizer input had been optimized as well.
Many studies reported N2O and N2 emissions from soils using one of the three measurement approaches: acetylene (C2H2) inhibition technique (e.g., Yoshinari and Knowles 1976; Miller et al. 2008; Qin et al. 2012; Qin et al. 2013; Guo et al. 2018) and 15N isotope labeling method (e.g., Hauck and Melsted 1956; Cai et al. 2001; Mathieu et al. 2006; Ruser et al. 2006) and helium-oxygen incubation approach known as direct method (e.g., Butterbach-Bahl et al. 2002; Qin et al. 2017). The lack of a robust direct measurement methods to measure N2 emissions, as final denitrification estimations (IPCC 2013), has caused a considerable underestimation or overestimation of N2. For instance, C2H2 inhibition technique is cheap and easy to conduct but underestimates denitrification, hence underestimation of N2 emissions as well (Butterbach-Bahl et al. 2013; Qin et al. 2012), while the 15N isotope labeling method has been at first limited by more laboratory procedures and expensive equipment as compared to C2H2 inhibition (Hauck and Melsted 1956). Later Ruser et al. (2006) reported high detection limit that identified overestimation of N2. For example, Qin et al. (2017), Friedl et al. (2020), and Wang et al. (2020) have strongly recommended the use of helium-oxygen direct method to estimate N2O and N2 emissions to elucidate uncertainty for N budgets, thus attaining realistic and unbiased measurements of N2O and N2. It is well known that N2O measurements have been intensively improved at both spatial and temporal scales, but N2 measurements against high atmospheric N2 background remain a challenge either in situ or in laboratory.
In the current study, we used a robust direct method, helium-oxygen incubation for N2O and N2 measurement (Qin et al. 2017); it is harmless although it needs high gas tightness to avoid atmospheric N2 leakage (Groffman et al. 2006). This will elucidate dynamics of N2O and N2 emissions from long-term N inputs by mimicking field conditions in laboratory in a way to comprehend the N cycle. Therefore, a field study was handled to evaluate the responses of N2O and N2, 15N isotopic composition of N2O, annual wheat and maize grain yields, yield-scaled N2O and N2 emissions, and agronomic nitrogen use efficiency (NUE) to a long-term N fertilized agricultural soil, and to the best of our knowledge less work has been done. The objectives of the current study are (i) to explore the effects of long-term N fertilizer application rates on N2O and N2, annual grain yields (wheat plus maize), and agronomic NUE and to quantify the yield-scaled N2O and N2O plus N2; (ii) to relate the soil-climate controlling factors to the emissions of N2O, N2, and their ratios N2/N2O; and (iii) to understand the relationship between 15N in soil-emitted N2O and the measured factors.
2 Materials and methods
2.1 Field site description
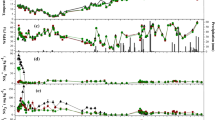
The field site is a long-term N fertilization experiment located in the Northern region of NCP known as Luancheng Agro-ecosystem Experimental Station (37°53′N, 114°41′E, 50 m) of the Chinese Academy of Sciences (CAS) in Hebei province, China. The soil of this region is categorized as a silt loam Haplic Cambisol (Mueller et al. 2013), and this experimental site has a typical monsoon climate classified as temperate semi-arid. The annual precipitation and annual temperature were observed 540 mm and 12.7°, respectively. The trend of daily precipitation, soil temperature (5-cm depth), and average air temperature during the study period are obtained from the meteorological station records; details are shown in (Fig. 1). The NCP is a distinctive representative of the high-yield agricultural region in China; about 90% of the cultivated land is winter wheat (Triticum aestivum L.) and summer maize (Zea mays L.) (Tan et al. 2017). Maximum annual soil (5-cm depth) temperature was 31.88 °C, and minimum annual soil temperature was − 3.8 °C, and the average is 15.1 °C. More rainfall was noticed in summer season, while there was dry cold soil in winter in the NCP. The annum cumulative rainfall is 349.2 mm (Fig. 1).
2.2 Experimental design and soil sample collection
Each experimental plot has an area of 70 m2 (7 m × 10 m) in a long-term experimental field of winter wheat-summer maize rotation system established in 1998. The experiment has three N inputs and control treatments; each has three replicates in our study design: 0 (control, CK), 200 (low nitrogen, LN), 400 (medium nitrogen, MN), and 600 (high nitrogen, HN) kg N ha−1 year−1. For winter wheat, a half of total annual N fertilizer rates were applied, respectively, to treatments in a split dosage at planting in October and at stem elongation in April, while for summer maize, the other a half of total annual N fertilizer rates were applied once, respectively, to treatments during flowering stage in August. As an additional mineral macronutrient, all plots received double superphosphate (65 kg ha−1 year−1) and potassium chloride (75 kg ha−1 year−1); these macronutrients were applied once a year as basal fertilizers during winter wheat planting in October. The field was plowed once after maize harvest in October, and the straw was mechanically chopped into smaller parts and integrated into the topsoil by rotary tillage. The details of the fertilization practices and other crop management activities are shown in Table S1.
2.3 Measurements of N2O and N2 fluxes and 15N isotopic composition of N2O
A robust direct measurement technique was used to incubate soil cores using two cylinders in one (inward cylinder was placed into outward one) setup, and afterward, they were sealed well to avoid atmospheric N2 contamination, and the soil core atmosphere was substituted by helium-oxygen. One to three soil samples were collected per month, depending on the N fertilizer application, and irrigation throughout the observational period and 21 sampling campaigns was reported. A stainless steel cylinder opened at both ends, a hammer, and a flat timber were used at the field to acquire soil cores vertically without much soil disturbance. The depth of soil core was 10 cm, and diameter was 19 cm. The diameters of inward and outward cylinders were 19 cm and 26 cm and the heights were 15 cm and 18 cm, respectively. After soil sample collection, the headspaces of both inward and outward cylinders were evacuated and filled with an artificial helium, 79%, and oxygen, 21%, for times varying from 4 to 5 in the laboratory.
Double cylinders were used to directly quantify N2O and N2 headspace concentrations after soil cores were evacuated as described in our previous study (Qin et al. 2017). Concentrations of N2O were analyzed by an electron capture detector while N2 by a thermal conductivity detector of the gas chromatography for 24 h after soil core sampling. During concentration measurement of N2O and N2, field environmental conditions were simulated in laboratory. Furthermore, this was enhanced by placing cylinders outdoor in the field. After mimicking the field environmental conditions, the cylinders were brought back in the laboratory again, and the same procedures were repeated. The N2O and N2 fluxes were calculated based on our previous study (Qin et al. 2017).
Gas samples were also taken for 15N isotope analysis of N2O after the cylinders’ atmosphere exchange by gas flow helium and oxygen. 120-ml gas sample was taken from each inward cylinder headspace and another 120 ml of gas sample was taken after incubation period using glass bottles. Then, the 훿15N of the soil-emitted N2O were quantified using an isotope ratio mass spectrometer (IRMS) (Delta V Plus. Thermo Fisher, Germany). As the N2O in the sample represented the 15N isotopic composition of both atmospheric and soil-emitted N2O, the equation from Snider et al. (2017) was used to calculate the 훿15N of soil-emitted N2O.
2.4 Annual grain yields (wheat plus maize) measurement and yield-scaled N2O and N2 emissions
Annual grain yields (wheat and maize) expressed in million grams per hectare (Mg ha−1) were calculated separately during harvesting seasons from an area of 12 m2 (3 × 4 m) in middle of each plot. Weights of ready maize cobs were determined and recorded, while wheat ears were even counted and then dried at 60 °C during 48 h to determine and record their dry weights. Yield-scaled N2O and N2 emissions (kg N Mg−1 grain) were calculated by dividing annual cumulative N2O and N2 emissions by annual wheat plus maize grain yields, respectively. Agronomic nitrogen use efficiency (NUE) was calculated by dividing annual wheat plus maize grain yields by annual N fertilizer (kg kg−1) rates in all N fertilized treatments except from CK.
2.5 Soil physicochemical analyses
Auxiliary soil analyses were conducted on 10-cm depth. During each sampling time, 5 soil cores were collected in plastic bags using auger from each plot in the respect to replicates. The collected soils of each plot were mixed thoroughly and stored in laboratory at 4 °C before further analyses of the soil parameters. NH4+ and NO3- were measured by Smartchem 140 analyzer and dual wavelength ultraviolet spectrophotometer, respectively. For soil NH4+ and NO3- extracts, 10-g fresh soil was extracted by shaking for 1 h using 50 ml of a 1 M potassium chloride solution. DOC was extracted and analyzed as described by (Mulvaney et al. 1997). Soil characteristics at field observational site, bulk density, soil pH, total organic matter, available phosphorus, and total nitrogen, were analyzed as described by Wang et al. (2014). Table S2 summarizes soil properties at field observational site. The measured gravimetric soil water content (%) obtained by drying the soil at 105 ± 0.05 °C for 24 h was converted to soil water filled pore space WFPS (%) based on (Franzluebbers 1999).
2.6 Data analyses
Structural equation modeling (SEM) analyses were carried out using the Lavaan R package. SEM was used to identify the relationships between WFPS, NO3- , DOC, NH4+, and soil temperature (5-cm depth) and N2O, N2 fluxes, and N2/N2O ratios. The quality of the SEM was assessed by using the Chi-square (X2) goodness of fit statistics (P > 0.05, this indicates statistical significant model fit), the root mean square error of approximation value (RMSEA, the smaller the better), and Akaike information criteria (AIC) (Kline 2011). Pathways relationships at *P < 0.05; **P < 0.01, and ***P<0.001 were considered significant. Analysis of variances (ANOVA) with a least significant difference test (LSD, P < 0.05) was used to test for differences in N2O and N2 annual cumulative emissions, N2/N2O ratios, 훿15N of soil-emitted N2O, annual wheat plus maize grain yields, agronomic NUE, and yield-scaled N2O and N2 between the treatments. All other data analyses were executed using OriginPro8 (Origin Lab Ltd., Guangzhou, China).
3 Results
3.1 Soil physicochemical properties
Soil DOC results showed a remarkable trend where highly fertilized soils have larger amount of DOC content compared to control (Fig. 2c), and average values were 40.54 ± 0.7, 47.84 ± 0.7, 50.93 ± 0.8, and 52.76 ± 0.9 from CK, LN, MN, and HN treatments, respectively. The trend of DOC was as follows: CK<LN<MN<HN, and statistical difference was obtained between all fertilized soil and CK, while HN was also statistically significant with LN. Soil WFPS (Fig. 2d) showed its highest peak in April after irrigation, and there was no statistical difference among treatments, and average values were 54.32 ± 1.7, 55.12 ± 2.1, 59.13 ± 1.7, and 58.96 ± 1.6 from CK, LN, MN, and HN treatments, respectively. Soil NH4+-N (Fig. 2a) was low, and some peaks were observed after N fertilizer application under sufficient soil moisture. Long-term N fertilization increased the NO3--N concentrations in soil, and the average values were 5.83 ± 0.4, 35.73 ± 2.1, 55.35 ± 3.1, and 63.61 ± 3.4 in the CK, LN, MN, and HN treatments, respectively, and there was a statistical significance between all treatments except HN and MN. The trend for both NO3--N and NH4+-N concentrations in the soil was CK<LN<MN<HN at our study site (Fig. 3a, b). NH4+-N average values were 0.86 ± 0.06, 1.2 ± 0.08, 1.35 ± 0.11, and 1.55 ± 0.13 from CK, LN, MN, and HN treatments, respectively, and the statistical significance was only observed between HN and CK and MN and CK.
(a) NH4+-N (mg kg−1), (b) NO3--N (mg kg−1), (c) DOC (mg kg−1) for dried soil, and (d) WFPS (w/w, %) at our observational site in the all treatments. Solid arrows on top signify N fertilizer application and dashed arrows indicate irrigation. Vertical bars denote standard errors for each mean treatment (n = 3)
3.2 Temporal patterns of N2O and N2 emissions and N2/N2O emission ratios
N fertilizer and irrigation practices greatly regulated the emissions of N2O. Comparing fertilized treatments among themselves and furthermore, to unfertilized treatments, the N fertilization rates greatly enhanced N2O emissions (Fig. 3a). N2O emissions were highest from the HN and lowest from CK treatment, and the average values were 1.18 ± 0.1, 6.07 ± 0.6, 9.81 ± 1.2, and 10.88 ± 1.1 g N ha−1 day−1 from CK, LN, MN, and HN treatments, respectively. Thus, HN>MN>LN>CK was the observed trend. Throughout the annual measurement period, there were three main peaks in the fertilized treatments (Fig. 3a); the first peak occured in October 2018, just after the basal N fertilizer application and flood irrigation. The second peak which was the highest among all occurred in April 2019, immediately after the top dressing of N fertilizer application and irrigation. The third happened in August 2019; immediately after N fertilizer application, there was no irrigation because it was a rainy period and the soil had enough moisture.
N2 flux peaks were detected a few days after N fertilizer application and flood irrigation (Fig. 3b). The emissions from three fertilized treatments were much larger than the CK treatment, and the average values were 21.91 ± 1.2, 52.17 ± 2.2, 60.92 ± 2.3, and 67.71 ± 2.9 g N ha−1 day−1 from CK, LN, MN, and HN treatments, respectively. This clearly demonstrates how long-term N fertilization induced the N2 fluxes. Moreover, within fertilized treatments, irrigation induced peak in N2 fluxes. From early spring throughout summer to autumn, in our observational site, there are enough soil moisture (WFPS) (Fig. 2d) and conducive temperature (Fig. 1) that favored N2O production and reduction to form high N2 fluxes.
In the current study, the N2/N2O ratios of emissions ranged from 1.7 to 34.6 (Fig. 3c). Overall mean N2/N2O emission ratios were 20.9 ± 0.8, 13.2 ± 0.8, 11.4 ± 0.8, and 10.3 ± 0.7 from CK, LN, MN, and HN treatments, respectively. The general trend of N2/N2O emission ratios was observed highest in summer and lowest towards the end of winter.
3.3 The annual cumulated N2O and N2 emissions and the overall mean of N2/N2O ratios to N fertilizer application rates
The annual cumulative emissions of N2O are significantly different among all treatments, and average values obtained were 0.40 ± 0.02, 1.91 ± 0.1, 2.83 ± 0.3, and 3.30 ± 0.3 kg N ha−1 year−1 from CK, LN, MN, and HN treatments, respectively (Fig. 4). Therefore, annual cumulative N2 emissions and average values were 7.89 ± 0.4, 18.02 ± 0.7, 21.46 ± 0.8, and 23.73 ± 1.1 kg N ha−1 year−1 from CK, LN, MN, and HN treatments, respectively. Among treatments, CK and LN are significantly different with MN and HN (Fig. 4). The overall N2/N2O ratios were in the trend of CK > LN > MN > HN treatments. The cumulative N2O and N2 emissions increased exponentially, while the N2/N2O ratios decreased exponentially with the increase of N fertilizer application rates. The differences of N2/N2O ratios between the LN, MN, and HN treatments were small and insignificant (Fig. 4). The total annual cumulative N2O and N2 emissions were exponentially related to N fertilizer application rates at P = 0.006 and P = 0.015, respectively (Fig. 4), throughout the year, while N2/N2O was negatively related to the N fertilizer application rate at P < 0.016.
3.4 Relationships between N2O, N2 emissions, and N2/N2O emission ratios with soil parameters
Structural equation model analyses were applied to evaluate effects of the selected soil-environmental factors, i.e., dissolved organic carbon (DOC), water-filled pore space (WFPS), nitrate (NO3--N), ammonium (NH4+-N), and soil temperature at 5 cm depth (ST). These soil factors are considered to critically induce N2O and N2 fluxes and N2/N2O ratios. N2O fluxes were positively related to NH4+-N, WFPS, NO3--N, and DOC while negatively related to ST (Fig. 5). N2 fluxes were positively related to DOC, WFPS, NO3—N, and ST (Fig. 5). N2/N2O ratios were positively related to ST and negatively related to NH4+-N and NO3--N content (Fig. 5).
The results of structural equation modeling (SEM) for the effects of temperature (Temp), WFPS, DOC, NH4+, and NO3- on N2O, N2, and N2:N2O during whole observational duration. The model is a good fit to the data: Chi-square (X2) = 1.522; P = 0.467, df = 2; RSMEA = 0.252; AIC = 51.22 for N2O; Chi-square (X2) = 0.471; P = 0.492, df = 1; RSMEA = 0.297; AIC = 52.471 for N2 and Chi-square (X2) = 0.979; P = 0.323, df = 1; RSMEA = 0.223; AIC = 40.97 for N2:N2O. Green arrows denote positive relationships, red arrows indicate negative relationships and the relationships are denoted on the arrows at *P < 0.05; **P < 0.01; and ***P < 0.001. None significant coefficients are not shown. Numbers next to the arrows denote the (positive or negative) path coefficients
3.5 15N isotopic signatures in soil-emitted N2O and its relationship with the N2/N2O ratios
The N fertilized soils emitted more depleted δ15N of N2O, while unfertilized soil shows less depleted values (Fig. 6a). The average values of δ15N of N2O soil-emitted were − 7.9‰ ± 0.9, − 13.6‰ ± 1.2, − 16.1‰ ± 1.1, and − 18.5‰ ± 1.1 from CK, LN, MN, and HN treatments, respectively. There was statistical difference between CK and other fertilized treatments, while within fertilized treatments, only LN and HN were statically different. Our study results showed a positive correlation between the N2/N2O emission ratios and the δ15N of soil-emitted N2O at P < 0.0366 (Fig. 6b), demonstrating that the increase of the emission ratios of N2/N2O is associated with less depleted δ15N of soil-emitted N2O.
3.6 Annual grain yields (wheat plus maize) and NUE as responses to N fertilizer rates
The results of annual grain yields (wheat plus maize) in the current study were averagely 3086, 13,635, 14,729, and 16,216 Mg ha−1 year−1 in CK, LN, MN, and HN, respectively. There were 4.41, 4.77, and 5.25 times the annual grain yields greater in LN, MN, and HN treatments, respectively, as compared to CK treatment. The annual grain yields were statistically different between CK and fertilized treatments. Moreover, there was a significant difference in annual grain yields between HN and LN, but no significant difference was observed between LN and MN and HN and MN. The responses of N fertilizer rates to annual grain yields were well described by exponential relationship (Fig. 7A). A linear model described well the responses of agronomic NUE and how it was inversely related to N fertilizer rates. There is a clear difference (P < 0.001) between all fertilized treatments for agronomic NUE (Fig. 7B). Therefore, the results from annual grain yields and agronomic NUE (Fig. 7A, B) demonstrate that the overdose N fertilizer rates could lead to no more annual grain yields.
Annual grain yields (wheat plus maize) (A). Agronomic nitrogen use efficiency (NUE) (B). Yield-scaled N2O and N2O + N2 (kg N Mg−1 grain) (C). Total cumulative N2O plus N2 in response to N fertilizer rates (D). Data point refers to mean ± stand error (n = 3) and lowercase letters denote significant differences between treatments
3.7 Yield-scaled N2O and N2O plus N2 emissions
Yield-scaled N2O and N2O plus N2 (kg N Mg−1 grain) have been a good indicator to show how annual grain yields (wheat plus maize) are related to annual cumulative N2O and N2 emissions. The average results of total yield-scaled N2O plus N2 were 2.69, 1.46, 1.64, and 1.67 kg N Mg−1 grain in CK, LN, MN, and HN, respectively (Fig. 7C). The lowest yield-scaled N2O plus N2 were observed in LN, and interestingly no statistical difference was observed among N fertilized treatments, but there was a statistical difference between N fertilized treatments and CK. However, yield-scaled N2O emissions ranged from 0.13 to 0.20 kg N Mg−1 grain. The yield-scaled N2O emissions followed the order hereafter: HM>MN>LN>CK, and they were 1.56, 1.47, and 1.07 times greater than CK, respectively. Statistical difference was observed between CK and N fertilized treatment MN and HN, while no statistical difference was observed between CK and LN treatments (Fig. 7C).
4 Discussion
4.1 Fluxes of N2O and N2, their ratios N2/N2O, and controlling factors from agricultural soil
In the current study, we aimed to relate soil-climate controlling factors to N2O and N2 emissions and even their ratios N2/N2O. The results indicated that N2O emissions responded to N fertilizer rates among fertilized treatments and were far large compared to unfertilized soil (CK). Thus, annual cumulative N2O emissions increased with increasing N input rates as given in details hereafter, 0.40, 1.91, 2.83, and 3.30 kg N ha−1 year−1 from CK, LN, MN, and HN treatments respectively (Fig. 4). Our findings were supported by many previous studies reporting the responses of N2O emissions to N fertilizer rates (e.g., Huang et al. 2017; Song et al. 2018). N2O emissions on the daily basis range from 0.4 to 36.8 g N ha−1 day−1 (Fig. 3a); our results are in agreement with the previous studies (Song et al. 2018; Chen et al. 2019; Wang et al. 2020) in agricultural soils. The N fertilization in combination with flooding irrigation results in remarkable peaks of N2O emissions, indicating that high soil WFPS accelerated the denitrification process (Fig. 2d) in combination with N inputs.
Overall, N2O fluxes are positively related to NH4+-N, WFPS, NO3--N, and DOC (Fig. 5). NH4+-N and NO3--N are the main substrate for nitrification and denitrification, respectively (Butterbach-Bahl et al. 2013). Moreover, positive relationships between soil N2O emissions and NH4+-N and NO3--N denote that both the nitrification and denitrification occurred at our study site, and our results were supported by various studies (Baggs 2008; Park et al. 2011; Baily et al. 2012; Li et al. 2020). The positive relationship between WFPS and N2O emissions confirms that high WFPS can accelerate denitrification process by creating anaerobic condition (Butterbach-Bahl et al. 2013; Hu et al. 2013). The positive relationship between N2O emissions and soil DOC in our study explained that readily available carbon accelerates denitrification. Low temperature at 5-cm depth and rainfall was observed during winter, and similarly N2O emissions were also low. The reason for this may be due to the fact that nitrifying and denitrifying bacteria were in unfavorable conditions. The previous studies reported that when soil temperature was below 10 °C, bacteria responsible for nitrification and denitrification also became dormant (Smith et al. 2010; Hu et al. 2013). Soil moisture (WFPS) is mainly attributed to flood irrigation and precipitation during our study period, and N2O emission peaks are observed after N fertilizer application and flood irrigation (Fig. 3a).
Annual cumulative N2 fluxes are larger than N2O fluxes in our current study. This has evidently shown the potentiality of N2 emissions from direct measurement method as obtained hereafter: 7.89, 18.02, 21.46, and 23.73 kg N ha−1 year−1 from CK, LN, MN, and HN treatments respectively (Fig. 4). Daily N2 emissions range from 13.5 to 109.8 g N per hectare per day (Fig. 3b). Based on structural equation modeling applied in this study, N2 fluxes are positively related to DOC, NO3--N, WFPS, and soil temperature at 5-cm depth (Fig. 5). The current study is consistent with findings reported by Yuan et al. (2019) emphasizing that DOC strongly induces denitrification rate. Many previous studies also found relationships between N2 emissions and temperature (e.g., Butterbach-Bahl et al. 2002; Hu et al. 2013). Wang et al. (2020) by using a modified gas-flow-soil-core method reported 7.2 and 2.4 kg N ha−1 cumulative N2O and N2, respectively, in summer maize season by applying 260 kg N ha−1, and these results confirmed that N2 emissions were dominant N losses in agricultural soil. Moreover, similar results have been observed in our current study that N fertilizer application rates enhanced more N2 fluxes than N2O (Fig. 4). So, we conclude that high cumulative N2 emissions from long-term N fertilized treatments might be due to higher available DOC along with N inputs that induced NO3- in our study site (Fig. 2b, c). This enhances the evidence that the presence of high DOC, NO3- , WFPS, and soil temperature is a conducive condition for high N2 fluxes (Fig. 5).
The current study has revealed that the mean N2/N2O emission ratios at the end of this study are 20.9, 13.2, 11.4, and 10.3 from CK, LN, MN, and HN treatments, respectively (Fig. 4). The daily N2/N2O emission ratio ranges from 1.7 to 34.6 (Fig. 3c). Our findings are in agreement with previous studies that have documented that N2 emissions are the main N losses from denitrification, hence higher N2/N2O emission ratios (Werner et al. 2014; Chen et al. 2019; Wang et al. 2020). Overall total treatments, N2/N2O emission ratios are negatively related to NH4+ and NO3- and positively to soil temperature at 5-cm depth (Fig. 5). As both NH4+ and NO3- increased with the increase of N inputs compared to control treatment (Fig. 2a, b), while N2/N2O ratio decreased with the increase of N inputs compared to control treatment, this might have led to negative relationship between the N2/N2O ratio and NH4+ and NO3-.
The relatively high overall mean N2/N2O emission ratios throughout the year (Fig. 4) gave more insights on the N losses in a form of N2 since within global N cycle, N2 fluxes from soils were poorly understood due to the difficulties to measure them (Galloway et al. 2008; Groffman 2012). Qin et al. (2012) reported that low N2 emissions from agricultural soil were reported basically using the C2H2 inhibition technique, which possibly underestimate N2 emissions, hence leading to low N2/N2O emission ratios. We report that overall mean N2/N2O emission ratios are negative exponentially correlated to N fertilizer rates (Fig.4). So, it has been recommended that more N2 production is stimulated in higher temperature (Saggar et al. 2013; Wang et al. 2020), leading to positive relationship between soil temperature and N2/N2O ratio (Fig. 5).
4.2 Annual cumulative N2O and N2 emissions and the overall mean N2/N2O ratios in response to N fertilizer application rates
The cumulative N2O and N2 emissions increased exponentially at P < 0.006 and P < 0.015, respectively; however, the N2/N2O ratios decrease exponentially at P < 0.016 with the increase of N fertilizer application rates (Fig. 4). The exponential relationships between N fertilizer application rates and N2O, N2 and N2/N2O ratios might be due to exceeding the optimum N fertilizer rates (Song et al. 2018).
Interestingly, N fertilizer application rates greatly increased N2O emissions by 4.7, 7.0, and 8. 2 times higher in LN, MN, and HN than the CK soil. However, the fertilization application rates only increase N2 emission by 2.2, 2.7, and 3.0 times higher in LN, MN, and HN than CK soil. This shows that fertilization greatly increases N2O but not N2 to that extent. This might be the reason for the negative exponential relationship between N fertilizer application rate and N2/N2O ratios. The total measured N2O plus N2 fluxes in our study are 8.3, 20.9, 24.3, and 27.0 kg N ha−1 year−1 from CK, LN, MN, and HN treatments, respectively (Fig. 7D). As there is a chance for denitrification under lower soil profiles (Wang et al. 2013; Yuan et al. 2019), there is a possibility that we missed some gaseous N2O and N2 emissions underground since we only measured upper 10 cm of the soil profile. Moreover, recent studies further suggest that plants may also have NO3--NO2--NO-N2O pathway of N2O production (Timilsina et al. 2020a; Timilsina et al. 2020b) which possibly indicates the underestimation of denitrification pathway in our study.
4.3 The δ15N of soil-emitted N2O and its relationship with the measured parameters
It is evidently shown that highly fertilized soils have more depleted isotopic signatures, while unfertilized soils are less depleted (Fig. 6a). The highly depleted and less depleted values of 15N isotope of soil-emitted N2O are − 29.83‰ and − 3.66‰ (HN); − 31.06‰ and − 3.96‰ (MN); − 28.72‰ and − 1.92‰ (LN); and − 25.66‰ and 1.62‰ (CK), respectively (Fig. 6a). The mean values of 15N in soil-emitted N2O were − 7.89 ± 0.88, − 13.61 ± 1.21, − 16.13 ± 1.12, and − 18.54 ± 1.11 from CK, LN, MN, and HN treatments, respectively. There were significant differences (P < 0.001) of 15N in soil-emitted N2O between unfertilized (CK) and fertilized (LN, MN, and HN) treatments, while within fertilized treatments, i.e., LN and HN have significant difference (P <0.01). The values of 15N in soil-emitted N2O from the current study were consistently observed within range from various previous studies (Pérez et al. 2001; Xiong et al. 2009; Park et al. 2011; Timilsina et al., 2020c). Previously 15N in soil-emitted N2O were used to predicate the N2O formation processes in the soil; however, using only 15N in soil-emitted N2O may mislead interpretation (Xiong et al. 2009; Timilsina et al., 2020c) because there was no any relationship between 15N in soil-emitted N2O and other soil parameters (like WFPS, NO3-, NH4+, DOC) in our study. Therefore, it may mislead the conclusion as well. Moreover, more powerful tool like site preference of N2O was not used in our study to describe the pathways of N2O formation (Park et al. 2011; Timilsina et al., 2020c). Nevertheless, 15N composition of soil-emitted N2O shows relationship only with N2/N2O ratio (Fig. 6b). To our knowledge, this is the first study to evaluate the relationship between gaseous N2O and N2 measured simultaneously using direct soil core measurement method and 15N isotopic compositions of N2O emitted. The 15N isotopic signatures of soil-emitted N2O from unfertilized and fertilized soils are greatly different (Pérez et al. 2001; Park et al. 2011; Timilsina et al., 2020c). The current study is consistent with the previous studies where it demonstrates the clear difference between fertilized and unfertilized soil. N2O produced during nitrification is more depleted as a fractionation factor by the nitrification process is generally higher than the denitrification process (Yoshida 1988; Baggs 2008). The results of our study show a positive significant correlation between the N2/N2O emission ratios and the δ15N-N2O at P < 0.0366 (Fig. 6b), indicating that N2O emissions were mainly delivered from denitrification process, while nitrification may probably contributed less.
4.4 Responses of wheat-maize annual grain yields; annual cumulative N2O and N2 emissions; yield-scaled N2O and N2 emissions to N fertilizer rates
Considering agronomic NUE, yield-scaled N2O and N2O plus N2 emissions, the annual grain yields, and total N2O plus N2, we proposed that sustainable agricultural practices can be achieved at LN (200 kg N ha−1 year−1), indicating that adopting this N fertilizer input will decrease 50% as compared to current farmers’ practice of 400 kg N ha−1 year−1 in this agricultural region with no grain yields decrease. This is enhanced by exponential relationship between N fertilizer rates and the annual grain yields with no statistical difference between LN and MN treatments (Fig. 7A). Therefore, a linear relationship is observed between agronomic NUE and N fertilized treatments (Fig. 7B). Interestingly, the optimal N input proposed in the current study will curb as much as 66.7% of N inputs from conventional N fertilizer rate of 600 kg N ha−1 year−1. The current findings are in agreement with Huang et al., (2017) that compared optimal N input (294 kg N ha−1 year−1) against conventional N input (560 kg N ha−1 year−1), and the results proved that 48% reduction of N fertilizer rate had occurred due to that shift with no significant decrease in grain yields. Hence, their practices also reduced cumulative N2O and yield-scaled N2O emissions by 18% and 38%, respectively. In our site, cumulative N2O emissions in LN were reduced by 32.5 % and 42% as compared to MN and HN, respectively. Evidently, Ju et al. (2009) reported that up to 50% of N losses to the environment can be cut off with no statistical crop yield decrease, while N input would be reduced up to 30–60%. Yield-scaled N2O in LN decreased by 27.0% and 31.3% as compared to MN and HN, respectively. While yield-scaled N2O plus N2 in LN reduced by 11.2% and 12.3% as compared to MN and HN, respectively. Our study is first to report yield-scaled N2O and N2; there are no existing data to compare with our findings.
In the current study, yield-scaled N2O emissions are ranging from 0.13 to 0.20 kg N Mg−1 grain (Fig. 7C), and they are similar to those found by Huang et al. (2017) (0.142 to 0.304 kg N Mg−1 grain) for maize-wheat double cropping system in NCP, for maize-soybean rotation and single maize (Adviento-Borbe et al. 2007) (0.079 to 0.3 kg N Mg−1 grain), (0.099 to 0.281 kg N Mg−1 grain), respectively, and for continuous maize by Mosier et al. (2006) (0.064 to 0.31 kg N Mg−1 grain). However, in the current study, the reported yield-scaled N2O plus N2 ranging from 1.46 to 2.69 kg N Mg−1 grain (Fig. 7C) are new findings and have never been reported before. As N is major macronutrient for crops in agricultural soils, both N2O and N2 emissions are forms of N losses that may be a matter of concern for optimum crop production and sustainable dosage of N input, even though inert N2 does not harm the atmosphere. Thus, enhancement of environmental sustainability and optimum cereal yields when excessive N fertilizer is cut off while N2O emissions reduced was previously suggested (e.g., Sun and Huang 2012; Tian et al. 2012).
5 Conclusion
Long-term N fertilizer inputs have resulted in higher DOC, NO3-, and NH4+ in LN, MN, and HN compared to CK treatment. Moreover, this led to higher rates of N2O and N2. Both positive and negative relationships between soil-environmental parameters and N2O, N2, and N2/N2O ratio indicate their heterogeneous roles in N2O formation and reduction processes. Decreasing the ratio of N2/N2O with the increase of N fertilizer application rates indicates that N2O and N2 depend on the N fertilizer application rates. The positive relation between 15N isotope of N2O and N2/N2O ratio indicates that denitrification might be the main process of N2O formation in the wheat and maize agricultural soil. The exponential relationships between emissions of N2O, N2, N2/N2O, and N fertilizer application rates indicate the excessive N input compared to the crop need at the current study site. For the environmental risk protection, there is a need to apply optimal N in agro-ecosystem in a way to increase yield along with alleviating detrimental environmental consequences caused by overdose from long-term N application. So, treatment that received 200 kg N ha−1 year−1 was observed as optimum and future sustainable agricultural practice because it has high agronomic NUE, optimal wheat-maize annual grain yields, low yield-scaled N2O emissions, and low N2O plus N2 emissions as compared to MN and HN treatments in North China Plain. Future research should focus not only on NO3- leaching and N2O emissions but also emissions of N2 along with crop productivity in the intensive agricultural soils using different crops.
References
Adviento-Borbe MAA, Haddix ML, Binder DL, Walters DT, Dobermann A (2007) Soil greenhouse gas fluxes and global warming potential in four high - yielding maize systems. Global Change Biol 13(9):1972–1988. https://doi.org/10.1111/j.1365-2486.2007.01421.x
Mosier AR, Halvorson AD, Reule CA, Liu XJ (2006) Net global warming potential and greenhouse gas intensity in irrigated cropping systems in northeastern colorado. Journal of Environmental Quality 35(4):1584–1598
Baggs EM (2008) A review of stable isotope techniques for N2O source partitioning in soils: recent progress, remaining challenges and future considerations. Rapid Commun Mass Sp 22(11):1664–1672. https://doi.org/10.1002/rcm.3456
Baily A, Watson CJ, Laughlin R, Matthews D, McGeough K, Jordan P (2012) Use of the 15N gas flux method to measure the source and level of N2O and N2 emissions from grazed grassland. Nutr Cycl Agroecosys 94(2-3):287–298. https://doi.org/10.1007/s10705-012-9541-x
Butterbach-Bahl K, Baggs EM, Dannenmann M, Kiese R, Zechmeister-Boltenstern S (2013) Nitrous oxide emissions from soils: how well do we understand the processes and their controls? Philos T Roy Soc B 368(1621):20130122. https://doi.org/10.1098/rstb.2013.0122
Butterbach-Bahl K, Willibald G, Papen H (2002) Soil core method for direct simultaneous determination of N2 and N2O emissions from forest soils. Plant Soil 240(1):105–116. https://doi.org/10.1023/A:1015870518723
Cai Z, Laughlin RJ, Stevens RJ (2001) Nitrous oxide and dinitrogen emissions from soil under different water regimes and straw amendment. Chemosphere 42(2):113–121. https://doi.org/10.1016/S0045-6535(00)00116-8
Chen T, Oenema O, Li J, Misselbrook T, Dong W, Qin S, Hu C (2019) Seasonal variations in N2 and N2O emissions from a wheat–maize cropping system. Biol Fert Soil 1-13:539–551. https://doi.org/10.1007/s00374-019-01373-8
Cui Z, Wang G, Yue S, Wu L, Zhang W, Zhang F, Chen X (2014) Closing the N-use efficiency gap to achieve food and environmental security. Environ Sci Technol 48(10):5780–5787. https://doi.org/10.1021/es5007127
Dannenmann M, Butterbach-Bahl K, Gasche R, Willibald G, Papen H (2008) Dinitrogen emissions and the N2:N2O emission ratio of a Rendzic Leptosol as influenced by pH and forest thinning. Soil Biol Biochem 40(9):2317–2323. https://doi.org/10.1016/j.soilbio.2008.05.009
de Vries W, Leip A, Reinds GJ, Kros J, Lesschen JP, Bouwman AF (2011) Comparison of land nitrogen budgets for European agriculture by various modeling approaches. Environ Pollut 159(11):3254–3268. https://doi.org/10.1016/j.envpol.2011.03.038
Erisman JW, Sutton MA, Galloway J, Klimont Z, Winiwarter W (2008) How a century of ammonia synthesis changed the world. Nat Geosci 1(10):636–639. https://doi.org/10.1038/ngeo325
Fan C, Chen H, Li B, Xiong Z (2017) Biochar reduces yield-scaled emissions of reactive nitrogen gases from vegetable soils across China. Biogeosciences. 14(11):2851–2863. https://doi.org/10.5194/bg-14-2851-2017
Forster, P., Ramaswamy, V., Artaxo, P., Berntsen, T., Betts, R., Fahey, D. W. and Nganga, J. (2007). Changes in atmospheric constituents and in radiative forcing. Chapter2. In Climate Change 2007. The Physical Science Basis.
Franzluebbers AJ (1999) Microbial activity in response to water-filled pore space of variably eroded southern Piedmont soils. Appl Soil Ecol 11(1):91–101. https://doi.org/10.1016/S0929-1393(98)00128-0
Friedl J, Cardenas LM, Clough TJ, Dannenmann M, Hu C, Scheer C (2020) Measuring denitrification and the N2O/(N2O+ N2) emission ratio from terrestrial soils. Curr Opin Env Sust 47:61–71. https://doi.org/10.1016/j.cosust.2020.08.006
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320(5878):889–892. https://doi.org/10.1126/science.1136674
Grafton RQ, Williams J, Jiang Q (2015) Food and water gaps to 2050: preliminary results from the global food and water system (GFWS) platform. Food Secur 7(2):209–220. https://doi.org/10.1007/s12571-015-0439-8
Groffman PM (2012) Terrestrial denitrification: challenges and opportunities. Ecological Processes 1(1):1–11. https://doi.org/10.1186/2192-1709-1-11
Groffman PM, Altabet MA, Böhlke JK, Butterbach-Bahl K, David MB, Firestone MK, Voytek MA (2006) Methods for measuring denitrification: diverse approaches to a difficult problem. Ecol Appl 16(6):2091–2122. https://doi.org/10.1890/1051-0761(2006)016[2091:MFMDDA]2.0.CO;2
Guo L, Wang X, Diao T, Ju X, Niu X, Zheng L, Han X (2018) N2O emission contributions by different pathways and associated microbial community dynamics in a typical calcareous vegetable soil. Environ Pollut 242:2005–2013. https://doi.org/10.1016/j.envpol.2018.07.028
Hauck RD, Melsted SW (1956) Some aspects of the problem of evaluating denitrification in soils. Soil Sci Soc Am J 20(3):361–364. https://doi.org/10.2136/sssaj1956.03615995002000030017x
Hu XK, Su F, Ju XT, Gao B, Oenema O, Christie P, Zhang FS (2013) Greenhouse gas emissions from a wheat–maize double cropping system with different nitrogen fertilization regimes. Environ. Pollut 176:198–207. https://doi.org/10.1016/j.envpol.2013.01.040
Huang T, Yang H, Huang C, Ju X (2017) Effect of fertilizer N rates and straw management on yield-scaled nitrous oxide emissions in a maize-wheat double cropping system. Field Crops Research 204:1–11. https://doi.org/10.1016/j.fcr.2017.01.004
IPCC. 2007. Climate Change 2007: The Physical Science Basis. Working group I contribution to the fourth assessment report of the Intergovernmental Panel on Climate Change (Vol. 4). Cambridge university press.
IPCC. 2013. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, United Kingdom and New York, NY, 662 USA.
Ju XT, Xing GX, Chen XP, Zhang SL, Zhang LJ, Liu XJ, Zhang FS (2009) Reducing environmental risk by improving N management in intensive Chinese agricultural systems. P Natl Acad Sci 106(9):3041–3046. https://doi.org/10.1073/pnas.0813417106
Kim DG, Hernandez-Ramirez G, Giltrap D (2013) Linear and nonlinear dependency of direct nitrous oxide emissions on fertilizer nitrogen input: a meta-analysis. Agr Ecosyst Environ 168:53–65. https://doi.org/10.1016/j.agee.2012.02.021
Kline RB (2011) Convergence of structural equation modeling and multilevel modeling. SEGA publication. https://doi.org/10.4135/9781446268261.n31
Lassaletta L, Billen G, Grizzetti B, Anglade J, Garnier J (2014) 50 year trends in nitrogen use efficiency of world cropping systems: the relationship between yield and nitrogen input to cropland. Environ Res Lett 9(10):105011. https://doi.org/10.1088/1748-9326/9/10/105011
Li Z, Xia S, Zhang R, Zhang R, Chen F, Liu Y (2020) N2O emissions and product ratios of nitrification and denitrification are altered by K fertilizer in acidic agricultural soils. Environ Pollut 115065:115065. https://doi.org/10.1016/j.envpol.2020.115065
Liu C, Wang K, Zheng X (2012) Responses of N2O and CH4 fluxes to fertilizer nitrogen addition rates in an irrigated wheat-maize cropping system in northern China. Biogeosciences 9(2):839–850. https://doi.org/10.5194/bg-9-839-2012
Mathieu O, Hénault C, Lévêque J, Baujard E, Milloux MJ, Andreux F (2006) Quantifying the contribution of nitrification and denitrification to the nitrous oxide flux using 15 N tracers. Environ Pollut 144(3):933–940. https://doi.org/10.1016/j.envpol.2006.02.005
Meng Q, Sun Q, Chen X, Cui Z, Yue S, Zhang F, Römheld V (2012) Alternative cropping systems for sustainable water and nitrogen use in the North China Plain. Agr Ecosyst Environ 146(1):93–102. https://doi.org/10.1016/j.agee.2011.10.015
Miller MN, Zebarth B, Dandie CE, Burton DL, Goyer C, Trevors JT (2008) Crop residue influence on denitrification, N2O emissions and denitrifier community abundance in soil. Soil Biol Biochem 40(10):2553–2562. https://doi.org/10.1016/j.soilbio.2008.06.024
Mueller L, Shepherd G, Schindler U, Ball BC, Munkholm LJ, Hennings V, Hu C (2013) Evaluation of soil structure in the framework of an overall soil quality rating. Soil Till Res 127:74–84. https://doi.org/10.1016/j.still.2012.03.002
Mulvaney RL, Khan SA, Stevens WB, Mulvaney CS (1997) Improved diffusion methods for determination of inorganic nitrogen in soil extracts and water. Biol Fert Soil 24(4):413–420. https://doi.org/10.1007/s003740050266
NoAA, ESLR. https://www.esrl.noaa.gov/gmd/hats/insitu/cats/conc.php?site=brw&gas=n2o Accessed on 01 June 2019.
Pan YP, Wang YS, Tang GQ, Wu D (2012) Wet and dry deposition of atmospheric nitrogen at ten sites in Northern China. Atmos Chem Phys 12(14):6515–6535. https://doi.org/10.5194/acp-12-6515-2012
Park S, Pérez T, Boering KA, Trumbore SE, Gil J, Marquina S, Tyler SC (2011) Can N2O stable isotopes and isotopomers be useful tools to characterize sources and microbial pathways of N2O production and consumption in tropical soils? Global Biogeochem. Cy. 25(1). https://doi.org/10.1029/2009GB003615
Pérez T, Trumbore SE, Tyler SC, Matson PA, Ortiz-Monasterio I, Rahn T, Griffith DWT (2001) Identifying the agricultural imprint on the global N2O budget using stable isotopes. J Geophysi Res Atmos 106(D9):9869–9878. https://doi.org/10.1029/2000JD900809
Prinn RG, Weiss RF, Fraser PJ, Simmonds PG, Cunnold DM, Alyea FN, O'Doherty S, Salameh P, Miller BR, Huang J, Wang RHJ, Hartley DE, Harth C, Steele LP, Sturrock G, Midgley PM, McCulloch A (2000) A history of chemically and radiatively important gases in air deduced from ALE/GAGE/AGAGE. Journal of Geophysical Research: Atmospheres 105(D14):17751–17792
Qin S, Clough T, Luo J, Wrage-Mönnig N, Oenema O, Zhang Y, Hu C (2017) Perturbation-free measurement of in situ dinitrogen emissions from denitrification in nitrate-rich aquatic ecosystems. Water Research 109:94–101
Qin S, Hu C, Oenema O (2012) Quantifying the underestimation of soil denitrification potential as determined by the acetylene inhibition method. Soil Biol Biochem 47:14–17. https://doi.org/10.1016/j.soilbio.2011.12.019
Qin S, Yuan H, Dong W, Hu C, Oenema O, Zhang Y (2013) Relationship between soil properties and the bias of N2O reduction by acetylene inhibition technique for analyzing soil denitrification potential. Soil Biol. Biochem. 66:182–187. https://doi.org/10.1016/j.soilbio.2013.07.016
Ruser R, Flessa H, Russow R, Schmidt G, Buegger F, Munch JC (2006) Emission of N2O, N2 and CO2 from soil fertilized with nitrate: effect of compaction, soil moisture and rewetting. Soil Biol Biochem 38(2):263–274. https://doi.org/10.1016/j.soilbio.2005.05.005
Saggar S, Giltrap DL, Li C, Tate KR (2007) Modelling nitrous oxide emissions from grazed grasslands in New Zealand. Agr Ecosyst Environ 119(1-2):205–216. https://doi.org/10.1016/j.agee.2006.07.010
Saggar S, Jha N, Deslippe J, Bolan NS, Luo J, Giltrap DL, Tillman RW (2013) Denitrification and N2O: N2 production in temperate grasslands: Processes, measurements, modelling and mitigating negative impacts. Sci Total Environ 465:173–195. https://doi.org/10.1016/j.scitotenv.2012.11.050
Shcherbak I, Millar N, Robertson GP (2014) Global metaanalysis of the nonlinear response of soil nitrous oxide (N2O) emissions to fertilizer nitrogen. P Natl Acad Sci U S A 111(25):9199–9204. https://doi.org/10.1073/pnas.1322434111
Smith J, Wagner-Riddle C, Dunfield K (2010) Season and management related changes in the diversity of nitrifying and denitrifying bacteria over winter and spring. Appl Soil Ecol 44(2):138–146. https://doi.org/10.1016/j.apsoil.2009.11.004
Snider DM, Wagner-Riddle C, Spoelstra J (2017) Stable isotopes reveal rapid cycling of soil nitrogen after manure application. J Environ Qual 46:261–271. https://doi.org/10.2134/jeq2016.07.0253
Song X, Liu M, Ju X, Gao B, Su F, Chen X, Rees RM (2018) Nitrous oxide emissions increase exponentially when optimum nitrogen fertilizer rates are exceeded in the North China Plain. Environ Sci Techol 52(21):12504–12513. https://doi.org/10.1021/acs.est.8b03931
Sun W, Huang Y (2012) Synthetic fertilizer management for China’s cereal crops has reduced N2O emissions since the early 2000s. Environ Pollut 160:24–27. https://doi.org/10.1016/j.envpol.2011.09.006
Tan Y, Xu C, Liu D, Wu W, Lal R, Meng F (2017) Effects of optimized N fertilization on greenhouse gas emission and crop production in the North China Plain. Field Crop Res 205:135–146. https://doi.org/10.1016/j.fcr.2017.01.003
Tian H, Lu C, Melillo J, Ren W, Huang Y, Xu X, Liu J (2012) Food benefit and climate warming potential of nitrogen fertilizer uses in China. Environ Res Lett 7(4):044020. https://doi.org/10.1088/1748-9326/7/4/044020
Timilsina A, Bizimana F, Pandey B, Yadav RKP, Dong W, Hu C (2020a) Nitrous oxide emissions from paddies: understanding the role of rice plants. Plants-Basel 9(2):180. https://doi.org/10.3390/plants9020180
Timilsina A, Dong W, Luo J, Lindsey S, Wang Y, Hu C (2020c) Nitrogen isotopic signatures and fluxes of N2O in response to land-use change on naturally occurring saline–alkaline soil. Sci Rep 10(1):1–13. https://doi.org/10.1038/s41598-020-78149-w
Timilsina A, Zhang C, Pandey B, Bizimana F, Dong W, Hu C (2020b) Potential pathway of nitrous oxide formation in plants. Front Plant Sci 11:1177. https://doi.org/10.3389/fpls.2020.01177
Wang Y, Hu C, Ming H, Oenema O, Schaefer DA, Dong W, Li X (2014) Methane, carbon dioxide and nitrous oxide fluxes in soil profile under a winter wheat-summer maize rotation in the North China Plain. PloS One 9(6):e98445. https://doi.org/10.1371/journal.pone.0098445
Wang YY, Hu CS, Ming H, Zhang YM, Li XX, Dong WX, Oenema O (2013) Concentration profiles of CH4, CO2 and N2O in soils of a wheat–maize rotation ecosystem in North China Plain, measured weekly over a whole year. Agr Ecosyst Environ 164:260–272. https://doi.org/10.1016/j.agee.2012.10.004
Wang R, Pan Z, Zheng X, Ju X, Yao Z, Butterbach-Bahl K, Huang B (2020) Using field-measured soil N2O fluxes and laboratory scale parameterization of N2O/(N2O+ N2) ratios to quantify field-scale soil N2 emissions. Soil Biol Biochem 148:107904. https://doi.org/10.1016/j.soilbio.2020.107904
Wang Y, Wang E, Wang D, Huang S, Ma Y, Smith CJ, Wang L (2010) Crop productivity and nutrient use efficiency as affected by long-term fertilisation in North China Plain. Nutr Cycl Agroecosys 86(1):105–119. https://doi.org/10.1007/s10705-009-9276-5
Werner C, Reiser K, Dannenmann M, Hutley LB, Jacobeit J, Butterbach-Bahl K (2014) N2O, NO, N2 and CO2 emissions from tropical savanna and grassland of northern Australia: an incubation experiment with intact soil cores. Biogeosciences 11(21):6047–6065. https://doi.org/10.5194/bg-11-6047-2014
Xiong ZQ, Khalil MAK, Xing G, Shearer MJ, Butenhoff C (2009) Isotopic signatures and concentration profiles of nitrous oxide in a rice-based ecosystem during the drained crop-growing season. J Geophys Res Biogeosci 114(G2). https://doi.org/10.1029/2008JG000827
Yoshida N (1988) 15N-depleted N2O as a product of nitrification. Nature 335(6190):528–529
Yoshinari T, Knowles R (1976) Acetylene inhibition of nitrous oxide reduction by denitrifying bacteria. Biochem Biophys Res Common 69(3):705–710. https://doi.org/10.1016/0006-291X(76)90932-3
Yuan H, Qin S, Dong W, Hu C, Manevski K, Li X (2019) Denitrification rate and controlling factors for accumulated nitrate in the deep subsoil of intensive farmlands: a case study in the North China Plain. Pedosphere 29(4):516–526. https://doi.org/10.1016/S1002-0160(17)60472-7
Zhang J, Li H, Deng J, Wang L (2020) Assessing impacts of nitrogen management on nitrous oxide emissions and nitrate leaching from greenhouse vegetable systems using a biogeochemical model. Geoderma 382:114701. https://doi.org/10.1016/j.geoderma.2020.114701
Acknowledgments
Authors are thankful to editor and reviewers for their constructive comments and suggestions. We would also like to thank Mr Junqi Yang for his tireless assistance in the fieldwork.
Funding
This work was funded by the National Natural Science Foundation of China (No. 41530859) and the National Key Research and Development Program of China (2016YFD0800100, 2018YFC0213301) and (2016YFD0800102-4, 2018YFC0213300).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Authors declare no conflicts of interest.
Additional information
Responsible editor: Zucong Cai
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Bizimana, F., Timilsina, A., Dong, W. et al. Effects of long-term nitrogen fertilization on N2O, N2 and their yield-scaled emissions in a temperate semi-arid agro-ecosystem. J Soils Sediments 21, 1659–1671 (2021). https://doi.org/10.1007/s11368-021-02903-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-021-02903-4