Abstract
Purpose
The present study presents data on the screening phase (tier 1) of a site-specific ecological risk assessment in a former smelter area heavily contaminated with metals (Santo Amaro, Bahia, Brazil). Joining information from three lines of evidence (LoE), chemical, ecotoxicological, and ecological, integrated risk values were calculated to rank sites within the area and identify those that may need further investigation in tier 2.
Materials and methods
Eleven points were selected up to 1,000 m from the smelter. Three reference points were 3 and 9 km away from the area. Risk values for the chemical LoE were derived from calculating the toxic pressure based on total metal concentrations. Those for the ecotoxicological LoE were based on avoidance (Folsomia candida and Eisenia andrei) and eluate tests (Daphnia magna acute test and Microtox) whereas for the ecological LoE the bait lamina test, soil basal respiration, and vegetation cover were used to derive risk values.
Results and discussion
The chemical LoE showed high risk in those points inside the area where metal loadings exceeded in much the existing soil screening values. Ecotoxicological tools showed a variable response, with tests on soil organisms inducing a higher risk (again at sites inside the smelter and with sandy soils) than tests on eluates. The three parameters composing the ecological LoE revealed a concordant response, despite the lower sensitivity of the vegetation cover. A high risk on this LoE was also observed on those sampling points where a high chemical risk was calculated.
Conclusions
Integrated risk was low outside the smelter area. Inside, a high spatial heterogeneity of risk levels was observed, related to the non homogeneous deposition of smelting residues. Very high risk levels, associated with sandy soils and residue deposits, suggest the need to proceed with remediation actions. However, the uncertainties associated with the contradictory information given by certain LoEs for certain sampling points show the need to confirm potential risks in a tier 2 analysis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The triad approach, originally developed to evaluate sediment quality (Long and Chapman 1985), has been recommended and successfully applied in ecological risk assessment (ERA) of contaminated soils (Wagelmans et al. 2009). Consisting of three lines of evidence (LoE; chemical, ecotoxicological and ecological), the triad approach is usually applied within a tiered system, i.e., information from each LoE is collected at each tier following a step-wise cost-effective process (Jensen and Mesman 2006). While tiers 2 and 3 are performed to reduce uncertainties about the actual risk, tier 1 is essentially a screening phase, aiming to produce a first spatial representation of the risk and to determine whether a site can be excluded from higher tiers of testing (either because it is unlikely to pose a risk to the relevant ecological receptors or because a high risk is detected and there could be a need for immediate mitigation actions), or it needs to be further evaluated (Weeks et al. 2004a; Critto et al. 2007). Thus, the tools used in tier 1 to collect information from each LoE should be not only able to indicate effects, but also rapid, easy to apply and inexpensive, i.e., cost-effective (Jensen and Mesman 2006).
In tier 1, the chemical LoE comprises the comparison of the total concentrations of contaminants at the study sites with soil screening levels. This should be complemented with information derived from ecological surveys (ecological LoE) and ecotoxicological tests (ecotoxicological LoE) (Weeks et al. 2004a; Fernandez et al. 2006). The ecological information at tier 1 is often collected through quick soil fauna or vegetation surveys and by measuring quick microbial parameters (e.g., soil respiration) (Jensen and Mesman 2006). In addition, the bait lamina test developed by Von Törne (1990), being a practical tool to assess soil faunal feeding activity in situ (Larink and Sommer 2002; van Gestel et al. 2003; Hamel et al. 2007), has been proposed as a relevant tool for ecological assessments (Filzek et al. 2004; André et al. 2009; Van Gestel et al. 2009), and has already been successfully tested in tropical soils (Römbke et al. 2006). Regarding the ecotoxicological LoE, short-term cost-effective bioassays evaluating both the habitat and retention functions of the soil are currently used in tier 1. Such bioassays integrate the combined effect of mixtures and that of contaminants not analyzed or for which soil quality levels do not exist (Weeks et al. 2004a; Fernandez et al. 2005; Spurgeon et al. 2005; Jensen and Mesman 2006). Whereas soil extracts (e.g., eluates) are used to perform widely established tests with cladocerans, microalgae, and the luminescent bacteria Vibrio fischeri (van Gestel et al. 2001; Achazi 2002; Loureiro et al. 2005a), soil samples are being increasingly evaluated through avoidance tests with earthworms (Hund-Rinke et al. 2003; Antunes et al. 2008) and collembolans (Natal-da-Luz et al. 2004). Earthworm avoidance tests have been shown to be a useful tool in the screening phase of risk assessment of contaminated soils (Lukkari and Haimi 2005), providing rapid information for future decisions (Schaefer 2003; Loureiro et al. 2005b), while being ecological relevant and of low cost (Yeardley et al. 1996). Although collembola avoidance tests are still under the process of standardization, their use in soil ecotoxicology has been acknowledged (Natal-da-Luz et al. 2004; Aldaya et al. 2006), mainly because the avoidance response of collembola is less influenced by the soil properties than that of earthworms (Natal-da-Luz et al. 2008). However, one of the limitations to use avoidance tests is that some substances are not perceived as repellents by the organisms and consequently are not avoided, leading to an underestimation of the risk (Greenslade and Vaughan 2003). Moreover, high concentrations of some substances (e.g., pesticides acting as AChE inhibitors) may affect mobility of the organisms to such an extent that they are not able to avoid the contaminated soil, creating also biased results (Natal-da Luz, personal communication).
This study aimed to conduct the first step (tier 1) of a site-specific ERA of a metal-contaminated area in Santo Amaro (BA, Brazil), joining information from the three LoE mentioned above. Although a human risk assessment has already been performed in Santo Amaro (FUNASA 2003), human risk-based criteria are unrelated to the ecological parameters that may be important to sustain soil functions (Dawson et al. 2007). Therefore, a site-specific ERA is necessary to evaluate the risks towards key ecological receptors. Besides bringing together chemical and (ecological and biological) effect data, the present study also intended to calculate risk with the purpose of ranking sites within the study area and to identify those that may need to be further investigated. In this way, the present work constitutes an innovative approach in metal-contaminated tropical environments, bringing an important contribution to the resolution of a local problem.
2 Materials and methods
2.1 Study area
This study was carried out in an abandoned lead smelter that was operational between 1960 and 1993, located adjacent to the urban area of Santo Amaro, BA, Brazil (12° 32′ 49″ S, 38° 42′ 43″ W). The area presents a high health risk to animals and humans (Costa 2001; Carvalho et al. 2003), due to high levels of metals in soil and water, as well as by tailings and airborne dust from atmospheric deposition through chimneys emission (up to 3 km from the industrial area), while the smelter was operational (Anjos 2003; Machado et al. 2004). It has been reported that approximately 180,000 m3 of tailings had been deposited around the smelter area from which approximately 55,000 m3 were buried under roads and house's backyards (Machado et al. 2004). In 1995, the Bahia environmental state agency recommended the encapsulation of tailings with the use of organic matter rich soil to mitigate contamination (Anjos 2003). However, because the process was carried out without following the adequate standard procedure, tailings are still exposed and the aerial dispersion of tailings dust is still occurring within and outside the smelter area (Anjos 2003; Machado et al. 2004).
2.2 Soil sampling and selection of reference soils
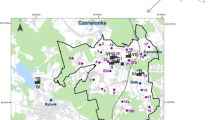
Based on the soil total metal concentrations (Pb, Cd, Cu, Zn) derived from a pre-sampling campaign using six transects on a radial shape (unpublished data), two 1 Km transects (T1 and T3) were defined along the two major gradients of contamination detected. The two transects shared a central point (P0, located next to the smelter facility) and were composed of five points each, located at 20, 50, 150, 400, and 1,000 m from P0 (P20T1-P1000T1 and P20T3-P1000T3; see Fig. 1).
Soils samples at each sampling point consisted of a composite sample made of four subsamples collected at the top 20 cm. Soil was hand mixed on site to homogenize, immediately transported to the laboratory, sieved (≤5 mm), and defaunated by one freeze-thawing cycle. After the physico-chemical characterization of each of the 11 soil samples (see next section), a multivariate factor analysis was run using soil properties data (metals excluded; see next section) aiming to define groups of samples and the main variables defining those groups. Based on this analysis, soil samples were assembled into three groups mainly differing in terms of texture, organic matter content and pH. The obtained results dictated the adoption of a multireference system. Soil from several points in the surrounding of the area were screened, analyzed for metals and soil properties, and three reference soils (the best possible for each identified group of sampling points) were selected at 3 km (Refs. 2 and 3) and 9 km (Ref. 1) from the area (see Fig. 1).
2.3 Soil physico-chemical characterization
Based on the historical use of the site and on a previous study (FUNASA 2003), soils were analyzed for the four main metals responsible for the contamination of the area (Pb, Cd, Cu, and Zn), and also for Cr, Ni, Fe, Co, and Mn. Metals were quantified in the bulk soil by inductively coupled plasma-atomic spectroscopy. Other soil physico-chemical parameters measured were pH (KCl 1M) (ISO 1994a), water holding capacity (ISO 1998), cation exchange capacity (ISO 1994b), organic matter (OM) content (loss on ignition at 500°C for 6 h) and soil texture (LNEC 1970).
2.4 Avoidance tests with Folsomia candida and Eisenia andrei
Avoidance tests with collembolans and earthworms were conducted using dual combinations of each test soil vs. the corresponding reference soil. To validate the test results, dual control tests using OECD (1984a) artificial soil were performed with both test species. Prior to testing, the water content of each soil (including the OECD soil) was adjusted to 50% of its maximum water holding capacity. Both test species originated from laboratory cultures maintained as described by Natal-da-Luz et al. (2009).
Tests with F. candida were carried out based on the ISO draft guideline 17512-2 (ISO 2007a). Cylindrical plastic containers (diameter: 7 cm; height: 6 cm) were divided into two equal sections by a plastic divider introduced vertically. Each container was filled with 30 g fresh weight (FW) of test soil on one side and 30 g FW of the corresponding reference soil on the other side. After removal of the divider, 20 adult collembolans (10 to 12 days old) were placed into the middle line of each test container. After 48 h of incubation, the content of each compartment was emptied into other plastic container to which water and a few drops of blue ink were added. The mixture was gently stirred and the organisms floating on the water surface were counted. Five replicates were prepared for each combination tested, plus one replicate without animals for pH and moisture determination. In the dual control tests with OECD soil the same procedures were adopted but the artificial soil was placed on both sides of the tests container. All tests were performed in a temperature controlled chamber at 25 ± 2°C and with a photoperiod of 16:8 h (light/dark).
Avoidance tests with E. andrei were conducted according to the ISO guideline 17512-1 (ISO 2007b). The test procedures, number of replicates per soil combination and incubation conditions were similar to those used for F. candida. However, rectangular plastic boxes (length, 20 cm; width, 12 cm; height, 5 cm) were used, and 250 g FW of (test, or reference) soil were placed in each section of the test container, and ten adult worms were used per replicate The number of organisms in each section was counted and recorded after a 48 h exposure period. For the dual control tests with OECD soil, the same procedures of the avoidance tests were adopted.
2.5 Daphnia magna lethal test
A 48 h D. magna lethal test (OECD 1984b) was conducted on eluates prepared from all soils. Soil eluates were obtained by shaking on a magnetic stirrer a soil/water mixture (ratio 1:4) for 18 h. The obtained soil suspension was left to settle for 24 h, time after which the supernatant was centrifuged for 7 min at 3370 g, filtered through a Schleicher and Schuell filter paper and stored in plastic bottles at 4°C and in darkness until use (within 10 days). Although filtration may decrease sample toxicity by removing the fraction of the contaminant adsorbed to suspended particles (Weltens et al. 2000), it was a required procedure to eliminate the potential detrimental effects of the suspended particles per se on the biological responses being measured. The pH was not adjusted. The water used to prepare the eluates was reconstituted hard water (ASTM 2002), since it was the media used for organism culturing in the tests as control and dilution media. Organism used for testing were 24-h-old neonates (clone Ircha) from third- to fifth-broods of mothers cultured according to the procedures outlined in Rosa et al. (2010). Four replicates were set up for each treatment with five neonates and 50 mL each. During testing, the incubation conditions were similar to those used for the soil organisms, and no food was provided. After 24 and 48 h exposure periods, the immobility/death of the neonates was checked. Measurements of pH, dissolved oxygen and electrical conductivity were measured at the beginning and at the end of all tests. All soil eluates were first tested at 100%. For eluates where immobility was observed, a dilution series of 100%, 50%, 25%, 12.5%, and 6.25% of the eluate was tested to determine the median lethal dilution (LC50 values).
2.6 V. fischeri luminescent test
The luminescent test with the marine bacterium V. fischeri was carried out on all soil eluates, prepared as described above but using ultra-pure rather than ASTM water, following a previously established protocol (ASTM 2004). All tests were carried out by Cetrel (Camaçari, BA, Brazil). The Microtox toxicity analyzer model 500 (Azur Environmental, Carlsbad, CA, USA) was used to measure the light emission of the bacteria after a 15 min exposure.
2.7 Soil fauna feeding activity using bait lamina
Bait material was prepared in a 1/5/14 ratio of finely ground oat, activated charcoal and cellulose powder (Merck). Five groups (samples) of five bait lamina strips were exposed in each sampling point for 14 days. Baits were inserted vertically into the soil, within an area of 15 × 15 cm at each group. In parallel, soil moisture was determined at each point. After the exposure period, each bait lamina was removed from the soil, conditioned together with the baits from the same group and brought to the laboratory. There, after carefully washing it in water, each bait strip was visually assessed by holding it against a light source and counting the number of pierced (= eaten) holes. No distinction was made between partially or fully pierced holes. The feeding activity per sample (group of five strips) at each sampling point was expressed in percentage, dividing the number of eaten holes by the total number of holes.
2.8 Basal soil respiration
At each sampling point soil samples for the determination of basal respiration were collected using a different procedure than described in Section 2.2. In this case, at each sampling point three parallel transects (5 m apart) were defined. Along each transect, 15 subsamples (10 cm depth) were collected and pooled to form a composite sample. After mixing, the samples were sieved (<5 mm), stored at 4°C and processed within the next 72 h. The methodology to determine the basal soil respiration is described in Alef (1995). Basal soil respiration was measured after 8 days for incubation at 28°C in the dark, with soil moisture adjusted to 60% of water holding capacity. The CO2 evolved from 100 g samples in hermetically sealed containers was trapped in 20 mL of 1 M NaOH. Back-titration with standardized HCl revealed the remaining NaOH and consequently the CO2-C evolved. Results were expressed on an oven-dried soil basis at 105°C for 24 h.
2.9 Vegetation cover
Assessment of percentage of vegetation cover was carried out according to Veiga and Wildner (1993). A plastic grid of 50 × 50 cm, subdivided in small 100 squares of 5 × 5 cm, was randomly released four times (four samples) at each sampling point. The sum of the intersections of small squares over vegetation in each grid represents the percentage of vegetation cover at each sample.
2.10 Data analysis
2.10.1 Ecotoxicological and ecological tests
The avoidance response to each tested soil was calculated according to the ISO guidelines (ISO 2007a, b). Avoidance data for E. andrei at each combination tested was corrected for site-specific properties using a generalized linear model developed by our team based on data gathered with non-contaminated natural soils (unpublished study). Soils properties considered in the model were texture parameters (sand and silt contents) and soil pH. According to this unpublished study, the avoidance response of F. candida showed much less sensitivity to soil properties. Therefore no correction was done for this organism in the current study. The significance of the avoidance responses (p < 0.05) was evaluated using the Fisher exact test (Zar 1996), as described by Natal-da-Luz et al. (2008). For the avoidance tests, a one-tailed test was chosen, and the null hypothesis assumes that half of the individuals are staying in the soil that is being assessed, meaning that there are no avoiders regarding that soil. For the dual control tests, a two-tailed test was chosen, and the null hypothesis assumes an equal distribution of the organisms on both sides of each test container. The 24 and 48 h LC50 values for D. magna and respective 95% confidence limits (CL) were computed using the software PriProbit 1.63, with the probit transformation of the proportion of deaths and the log transformation of the dilution values (Sakuma 1998). For the V. fischeri test, the EC50 values (median effective dilutions) and respective 95% CL were calculated using the Microtox Omni Software 1.18 (Azur Environmental).
For the ecological parameters (bait lamina test, vegetation cover and soil basal respiration) differences between sampling points were evaluated via a one-way analysis of variance followed by the Dunnet's test. In these analyses, an overall reference value was used, based on the values obtained for each reference sampling point. Soil moisture and organic matter contents were used as covariables in the basal respiration ANOVA. Normality and homoscedasticity were checked via the Kolmogorof-Smirnov and Bartlet's tests, respectively. Analyses were done using the Statistica 7.0 software (Stat Soft).
2.10.2 Risk calculations
Risk calculations followed the approach proposed by Jensen and Mesman (2006) where risk values are expressed in a scale ranging from zero (“no risk”) to one (“high risk”). This method assumes that the risk value of the reference soils is zero and that the risk of the tested soils is calculated in relation to the value of the respective reference soil. This implies that results from the various parameters across the three LoE should be made comparable (expressed under the same scale). For each sampling point, the calculation of the risk values was done through three steps: (1) scaling the results of each test/evaluation within each LoE; (2) integrating scaled information and calculating the contribution of each LoE to the overall risk; (3) integrate the information from the three LOEs and calculate the integrated risk.
In the first step, the results of all determinations within each LoE were scaled between zero and one. For the chemical LoE the total content of each metal was used to calculate the specific Toxic Pressure (Potential Affected Fraction (PAF) of species) at each sampling point. This was done based on the mixture model of concentration addition described by De Zwart and Posthuma (2005). The benchmarks (HC50EC50 values) and model parameters used for each metal in these calculations can be found in Rutgers et al. (2008). According to these authors the use of HC50 values derived from species sensitivity distributions based on NOEC values (HC50NOEC) could originate an overestimation of risk values (many values closer to 1). Since no HC50 values based on EC50 values (HC50EC50) are available in literature, they advise to apply the safety factor of 10 to the HC50NOEC and work with those values (HC50EC50 = 10 × HC50NOEC; Rutgers et al. 2008). Prior to calculations, the HC50EC50 values were corrected for sampling site-specific differences (taking into account the organic matter and the clay content of each soil) according to the correction formula described in Boivin et al. (2006). This implies that different HC50EC50 values (HC50cor) were used for each metal at each sampling point. Since HC50 values exist only for some metals, risk derived from the chemical LoE was based only on the concentrations of Pb, Cd, Cu, Zn, Cr, and Ni.
For the ecotoxicological LoE, the scaling of the avoidance data was done based on the percentage of avoidance, where negative values (no avoidance) were set to zero. Since the avoidance response towards a reference soil (when tested against itself) is zero, percentage values (converted between 0 and 1) were used directly as individual risk values. In the D. magna and V. fischeri tests, the LC50 and EC50 values, respectively, expressed as the percentage of dilution of the eluates, were used. For the ecological LoE, the bait lamina (expressed as the percentage of fed holes), the vegetation cover (expressed in percentage values), and the basal soil respiration (expressed as µg CO2-C g–1 day–1) values were scaled using an overall reference value calculated based on the values from the three reference sampling points.
In the second step, the risk derived from each LoE was calculated by integrating the respective scaled information for each parameter. In the chemical LoE this was achieved by estimating the msPAF (multi-substance PAF) by integrating the individual metal PAF's according to a response addition model described by De Zwart and Posthuma (2005). Finally, in step three, the integrated risk (IR) was calculated for each tested soil (sampling point). To evaluate whether the different lines of evidence contributed differently to the total risk, the standard deviation associated to each IR value was also calculated. More details on the calculation involved in each of the three steps (including formulas for each type of data used) can be seen in Jensen and Mesman (2006).
3 Results and discussion
3.1 Soil properties and selection of reference soils
Soils from the study area showed low (<2%) to medium (2 to 6%) organic matter content (according to the USEPA 2004), a cation exchange capacity (CEC) mostly between 30 and 40 meq/100 g, and pH values near neutral, with the exception of soils P1000T1 and Ref.2 with a low pH (Table 1). These characteristics agrees with those reported by Anjos (2003), who identified basic pH high CEC, high clay percentage and high organic matter content as characteristics of soils from the study area.
Results from the multivariate factor analysis indicated that texture (described by coarse sand, silt and clay contents) was the main soil characteristic determining the separation of the soils (along axis 1, explaining 49.5% of the variation), followed by pH (along axis 2, explaining 24.3%) and organic matter content (along axis 3, explaining 12.8% of total variation). Texture variables separated soils of group 2 (highest sand content and lowest silt and clay contents) from all other soils. The latter were then separated into two groups based mainly on their OM content, with group 1 generally presenting lower values than group 3. The reference soil allocated to each of the three soil groups was selected so that its characteristics matched, to the extent possible, these three soil properties, which are known to influence not only the bioavailability of contaminants (Kuperman et al. 2009), but also the avoidance response of the two tested soil-dwelling species (Natal-da-Luz et al. 2008). The heterogeneity of the soil inside the smelter area can be related to the failed attempt to encapsulate the tailings by depositing thousands of cubic meters of soil from regions around (Anjos 2003).
3.2 Soil metal concentrations
Total metal concentrations for each soil are shown in Table 2. For at least one among four metals (Pb, Cd, Cu, and Zn), soils from three sampling points presented levels exceeding the HC50cor values. Among these points, P0 presented a high degree of Zn contamination by exceeding almost three fold the corresponding HC50cor value, whereas points P150T1 and P50T3 presented a critical level of contamination, exceeding by far (between 1.6 and 73.5 times) the screening levels of these four metals.
High levels of metal contamination in the area were also previously reported (Anjos 2003; Machado et al. 2004). Most likely, such contamination levels resulted from the deposition of residues inside the smelting area as well as from the aerial deposition of contaminated particles from the smelter plume while in function, being responsible for the extent of contamination outside the smelter area.
3.3 Avoidance tests with E. andrei and F. candida
Avoidance tests with F. candida fulfilled the validity criteria, since mortality at each combination was less than 20% (ranged between 2% and 18% in avoidance tests, and was 1% in dual control tests), and a homogeneous distribution of individuals in the two compartments was observed in the dual control tests (p > 0.05, Fischer's exact test). Collembolans avoided significantly most of the test soils (Fig. 2). The exception was soil from point P1000T3, where no avoidance was detected. Considering that points P150T1 and P50T3 presented the two most contaminated soils (with the highest concentrations of Cu, Zn, Pb, and Cd), a stronger avoidance response was expected relatively to all other soils. F. candida is known to avoid copper concentrations well below those that impair survival and reproduction (e.g., a 48 h EC50 for avoidance of 61 mg/kg versus a 28 day EC50 for reproduction of 751 mg/kg; Greenslade and Vaughan 2003), and some evidences point that the same can occur with zinc (Natal-da-Luz et al. 2004). However, according to Greenslade and Vaughan (2003), these organisms do not avoid cadmium. Moreover, F. candida seems to be less sensitive to lead than to other metals, as observed by Sandifer and Hopkin (1996) (EC50reproduction=2,970 µg g–1 at pH 6.0), and as reported by Fountain and Hopkin (2001), where F. candida fed on yeast contaminated with Pb up to 49,200 µg g−1 did not exhibited significant change in mortality at all concentrations. In view of these facts, the weak avoidance response observed in these two soils (22% in P150T1 and 11% in P50T3) was most likely due to other factors than due to the total metal loads. Although there are indications that the avoidance response of F. candida is less influenced by soil properties than that of E. andrei (Natal-da-Luz et al. 2008), the fact that soils P150T1 and P50T3 had a higher organic matter content and pH compared to Ref 2, may have influenced the availability of metals to the organisms. Moreover, the very low organic matter content in soils from P0 and P20T1 may have caused the higher avoidance response observed.
Tests with E. andrei also fulfilled the validity criteria since no mortality was found in any soil and the organisms were homogeneously distributed between the two compartments in the dual control tests (p > 0.05 in Fischer's exact test). All soils were significantly avoided by the earthworms (see Fig. 2). An impairment of the habitat function of the soil according to the avoidance criterion proposed by Hund-Rinke and Wiechering (2001), i.e., an avoidance response higher than 60%, was observed for all soils from group 2 and also for soils from points P1000T1 and P50T1. This strong avoidance response may have been induced by the high levels of metals, in some cases exceeding by far the HC50cor values, or by soil related properties known to influence the avoidance behavior of earthworms. Loureiro et al. (2005b) reported avoidance response of E. andrei to copper (EC50 = 181.1 mg/kg Cu) and Lukkari et al. (2005) observed a significant avoidance of Aporrectodea tuberculata to 53 mg/Kg of Cu and 92 mg/Kg of Zn. Moreover, Alvarenga et al. (2008) observed avoidance response of E. fetida to a mine contaminated soil containing 1,250, 362, 264, and 2.6 mg/kg of Pb, Cu, Zn, and Cd, respectively. These findings support our results for soils from group 2, presenting contents of several metals higher than the benchmarks reported by these authors. Besides the high levels of metals, other factors are known to influence the avoidance response of E. andrei. According to Natal-da-Luz et al. (2004, 2008), soils with low pH, low organic matter content and fine texture are avoided by these organisms, which could explain the high avoidance response for soils P1000T1, P20T1, and P50T1 (low pH and low organic matter content, respectively, in comparison with the respective reference soils). In the case of sampling point P1000T1, by being outside the smelter area and in the middle of a pasture, the presence of another type of contamination (e.g., pesticides) eliciting the observed high avoidance response should not be ruled out. The high organic matter content in soil from point P400T1 (5.1%) relatively to the respective reference may explain the lack of a strong avoidance response by E. andrei. Organic matter decreases the bioavailability of metals in soil (Lock et al. 2000; Lock and Janssen 2001) and E. andrei, being a compost worm, is known to prefer soils with high organic matter content.
Overall, earthworms were generally more sensitive to the metal contamination and presented a less variable response than the collembolans. The present results are thus in accordance with the documented sensitivity of earthworms to metals in avoidance tests (Yeardley et al. 1996; Hund-Rinke and Wiechering 2001; Lukkari et al. 2005) and their more consistent response to metal-contaminated soils than collembolans (Natal-da-Luz et al. 2004) confirming their valuable use in ERA (Römbke et al. 2005).
3.4 D. magna lethal test and V. fischeri luminescent test
Lethal effects on D. magna were observed in eluates prepared from soils P150T1 and P1000T1; values of 24 h LC50 of 88% (95% CL; 82–95) and >100% and of 48 h LC50 of 68% (95% CL; 63–74) and 91% (95% CL; 85–97), respectively, were registered. Regarding the V. fischeri test, eluate from soil P50T3 was toxic to the bacteria, with a 15 min EC50 value of 8.6% (95% CL of 1.1–65.3). The present results are in agreement with the fact that metal loads were highest in soils from points P150T1 and P50T3. Although the soil from point P1000T1 was not classified as metal contaminated according to the HC50cor benchmarks used, the response in this soil eluate was most likely related to the low pH (3.7) of the bulk soil, and thus the low pH levels in this soil eluate; pH ranged from 4.84–4.98 to 5.53–6.22 at the start and end of the lethal test, respectively. Actually, lethal effects on cladocerans due to pH alone are likely to occur for values outside the range 6.0–9.0 (USEPA 2002). In agreement, the lack of lethal effects in the eluate prepared from the P0 soil (classified as metal contaminated according to the HC50cor values) was most likely due to the fact that pH level in this eluate was always well above 6.98, i.e., level not only not detrimental for freshwater organisms but at which most metals are not in their dissolved form and thus not bioavailable. The present results suggest that the retention function of soils at most of the sampling points was enough to prevent the mobilization of metals via the water pathway. Bioavailability of metals in soils may depend on several factors, such as pH, organic matter content, cation exchange capacity and clay content (Van Gestel 1992). Due to soil heterogeneity, the sorption potential might vary considerably, resulting in changes of contaminant availability, sometimes even within a small area. With time, sequestration processes become even more pronounced, a phenomenon generally referred to as “ageing”. In general, most of the soils sampled have low organic matter content, however, all soils (except P1000T1 and Ref 2) have an alkaline pH, typical of soils of this region (Anjos 2003), which favors soil adsorption and restricts metal bioavailability. Moreover, soil “ageing” may be occurring in the area between the closure of the smelter in 1993 and the last attempt to rehabilitate the residue piles in 2001 (Anjos 2003).
3.5 Bait lamina test
The average feeding activity observed at the sampling points was in general significantly lower than the overall reference value (Table 3). Lower feeding activity was registered at sampling points within the smelter area, mainly at those associated to a high degree of contamination (P0, P150T1, and P50T3) or to a low organic matter content (P20T1 and P50T1). Unexpectedly, point P400T3 also presented a low feeding activity, but other soil or habitat parameters must explain these results. With the exception of sampling point P1000T1, bait lamina data was, in general, in good agreement with the E. andrei avoidance responses, i.e., high avoidance was usually associated to a low feeding activity. This is in agreement with van Gestel et al. (2003) that found a strong association between soil fauna feeding activity measured by bait lamina and earthworm densities. The observed decline in feeding activity at contaminated sampling points may suggest an impact of metals on the soil fauna, especially earthworms, and eventually on invertebrate abundance and diversity (Weeks et al. 2004b). Similar results were also reported by Filzek et al. (2004) and André et al. (2009) on metal-contaminated areas in the UK and Portugal, respectively.
3.6 Basal soil respiration
Basal soil respiration was significantly lower in sampling points inside the smelter area, either those with high metal contents (P0, P150T1, P50T3, and P150T3) or those with very low organic matter content (P20T1 and P50T1), despite the correction for this parameter (see Table 3). These results showed impairment in microbial activity, which indicates a probable negative effects on nutrients cycle in points inside the smelter area. Respiration is a functional parameter widely used to indicate the microbial activity related to nutrient cycling (Araújo and Monteiro 2007). Results obtained by Zimakowska-Gnoinska et al (2000) confirmed that soil respiration can be used for estimations and comparisons of soil ecological conditions and biological activity of soils. These authors observed a significantly lower oxygen consumption in soil samples from contaminated sites in comparison to uncontaminated sites. Similar results were obtained by Gülser and Erdogan (2008) that presented a negative correlation between soil respiration and metal contents in roadside fields of intensive traffic areas.
3.7 Vegetation cover
Vegetation cover ranged between 20–100% (see Table 3). Like for the other two ecological parameters, and despite the large variability in the data, a significant reduction of the vegetation cover in comparison to the overall reference situation was observed in most sampling points within the smelter area (P0, P20T1, P150T1, P20T3, and P50T3). These points, together with P50T1 and P150T3 (where a reduction in vegetation cover was also observed), correspond to sites where tailings were deposed and where a non-successful re-vegetating action took place. At these sites vegetation was dominated by one herbaceous species. In some of these points, evidences of erosion were observed, which could have originated the delay of the natural regeneration process.
Our results are in agreement with Salemaa et al (2001). When studying plant diversity and cover along a metal pollution gradient in a smelter area in SW Finland, these authors found that these parameters decreased in soils with metals and sulphur, and increased with distance from the smelter. Similarly to our findings, they also found that a few tolerant species dominated the ground vegetation on the most polluted sites. They also found that the understory vegetation near the smelter was more damaged than trees, which confirms the importance of including understory vegetation in monitoring programmes.
According to Godínez-Alvarez et al. (2009) vegetation cover is an important indicator of soil quality. However when used alone (without information on other vegetation parameters like biomass or species richness) data obtained should be interpreted carefully, since a higher cover does not necessarily indicate a good soil quality. In this study, vegetation cover showed to be a less sensitive parameter than bait lamina or microbial basal respiration, being able to detect differences only on those heavy degraded soils. However, due to the extreme simplicity in terms of sampling, it can be considered a good parameter for the screening phase when used together with other ecological parameters.
3.8 Lines of evidence and integrated risk
Table 4 shows the individual contribution and the combined calculated risk values for each LoE. Sampling points presenting very high risk values (above 0.75) derived from the chemical LoE were, as expected, those where the metal concentrations exceed the HC50cor values (P0, P150T1, and P50T3) or were near that threshold (P150T3; see also Table 2). Regarding the ecotoxicological LoE, the differences in sensitivity of the screening tests were clearly visible in their contribution for the calculated combined risk, with avoidance tests indicating a higher risk than aquatic tests. The highest risk values (0.60 to 0.82) for this LoE were found in the more sandy soils (sampling points from group 2), with clay and silt soils presenting lower risk values (P1000T1 was the exception to this trend due to the high contribution of the earthworm avoidance test). Despite the differences in sensitivity, the contributions of the three parameters from the ecological LoE were concordant with the other LoE's in indicating greater risk values (above 0.60) for sampling sites from group 2 (sandy soils with high metal content), and for P150T3 (soil with high metal loadings).
The combination of the three lines of evidence into an integrated ecological risk value (IR) for tier 1, showed the spatial heterogeneity of the risk along the study area. However, high levels of risk were found at sampling points within the smelter area, particularly in soils with a coarse texture (soils from group 2; Fig. 3). Very high integrated risk values (IR > 0.75) were calculated for sampling points P0, P150T1, and P50T3 which, according to the Dutch limit acceptable values according to land use (Jensen and Mesman 2006) restricts their use to industrial activities and requires sealed soils. Among the latter soils, the weight of evidence was strong, since a high level of risk was indicated by each of the three lines of evidence (as illustrated in Fig. 3 by the low levels of standard deviation and balanced triangular graphs).
Integrated ecological risk values (+ standard deviation) (Min, 0; Max, 1) for each sampling point, combining information from the chemical, ecotoxicological, and ecological lines of evidence. Points with grey bars are located inside of the smelter area. Different bands indicate limits of accepted risk values for different soil uses (A agriculture, R residential, I industrial; asterisks indicate necessity of sealed soils) according to Jensen and Mesman (2006). Triangles on top of each bar represent the contribution of each LoE for the integrated risk value being an indicator of the weight of evidence (on the top right the example the length of each axis of the triangle represent maximum risk (1) from each LoE)
Sampling points P20T1 and P150T3 showed a moderate risk (0.51 ≤ IR ≤ 0.75) (see Fig. 3). However, the weight of evidence was not as strong as for the three soils described earlier, as seen by the slightly higher standard deviation associated to the IR values. At sampling point P20T1 the low risk indicated by the chemical LoE did not agree with the high risk values for both the ecotoxicological and the ecological LoE's. The observed low metal loadings on this soil, and the consequent low chemical risk values obtained, were unexpected due to the spatial location of the sampling point. However this is another clear sign of the spatial heterogeneity of the contamination within the smelter area and the degree of uncertainty associated to this spatial variation. This type of discrepancy, i.e., high effects on the biological parameters but low risk shown by the chemical parameters, may lead to the discussion about the weighting of the different LoE's. In principle each LoE is equally weighted, but under special circumstances a differential weighting can be attributed. Rutgers and Jensen (2010) mention, as examples, that the lack of proper reference sites or a deficient chemical characterization are situations where a lower weight should be attributed to the ecological and to the chemical LoE's, respectively. In this study, the lower and unexpected risk value obtained for the chemical LoE could justify the attribution of a lower weight to this LoE. However, the option of attributing the same weight to each LoE was followed, especially because the existing experiences in attributing different weights to different LoE's are scarce for the terrestrial environment. Moreover this discrepancy should be confirmed on a tier 2 assessment. In sampling point P150T3, the low weight of evidence was originated by the low risk values of the ecotoxicological LoE (0.3, see Table 4). The reason behind this low risk value is the very low sensitivity of the aquatic tests performed. Although these findings should be further investigated in a tier 2 evaluation, they trigger the discussion of differential weighting, this time within each LoE.
A low risk (IR ≤ 0.50) was associated to sampling points P50T1, P400T1, P1000T1, P20T3, P400T3, and P1000T3, all clay based soils. On points P50T1, P20T3, P400T3, and P1000T3 all the lines of evidence pointed to the same direction (a general low risk), despite the high impact on soil fauna feeding activity found in P400T3 (a trend not followed by the other two ecological parameters measured). However for the other two sampling points some uncertainties still persisted (IR value with a high standard deviation). The risk at sampling point P1000T1 was just indicated by the ecotoxicological LoE, as the chemical and ecological LoE indicated no risk. The high toxicity of this soil, mainly indicated by the earthworm avoidance, can either be related to the low pH value of the soil or to the presence of contaminants not analyzed in this study, namely pesticides. Nevertheless the tendency of earthworm avoidance tests to produce high risk values (near 1) when the percentage avoidance is used directly as individual risk value in the scaling process (see methods section) should not be neglected. One could argue that another type of scaling process could be used instead with this type of data. However, this problem can be minimized since avoidance tests with earthworms should be only one of the ecotoxicological tools applied at this tier. For sampling point P400T1 the high standard deviation associated to the IR value was due to the indication of low or no risk by the ecotoxicological and ecological LoE’s, respectively, and to the low to moderate risk signaled by the chemical LoE, related to the levels of Pb and Zn. This result may indicate that the metal levels at this point had a low bioavailability, thus, that this degree of uncertainty needs to be further confirmed in a second tier of risk analysis.
These results confirm the added value of deriving risk values in site-specific risk assessment not only by adopting the triad approach, but also by obtaining ecotoxicological information using different test organisms, covering different sensitivities and exposure routes. As shown in this study, biological testing is of direct relevance to the principle of significant harm, because the organisms will respond to the bioavailable fraction of the contaminant (Spurgeon et al. 2005). Furthermore, ecological data derived also from different parameters give information about the structure and function of soils, linked directly to the aim of diversity protection, complementing and improving the ecological relevance of the risk assessment process. Either a positive response in an ecotoxicological screening test, an exceedance of soil screening levels or evidences of damage to the ecological structure and functioning of soils, is sufficient to warrant progression to the next tier in the process, where a reduction of uncertainties is done through sublethal bioassays, determination of the available fraction of contaminants and inclusion of additional ecological data.
4 Conclusions
In general, integrated risk was low outside the smelter area, although some uncertainties were observed that need further investigation on the next tier. Inside the smelter area a high spatial heterogeneity of risk levels was observed, probably related to the non homogeneous deposition of smelting residues. Very high levels of risk were observed mainly in sampling points having sandy soils, and possibly associated to residue deposits. This high risk (above 0.75) may indicate the need to proceed with some remediation action. However, due to several uncertainties associated to the contradictory information given by the lines of evidence in some sampling points, there is a need to confirm the potential risk in a tier 2 analysis. With this aim, further data from the three lines of evidence (including evaluation of metal extractable concentrations, sublethal bioassays and additional ecological surveys) is being collected.
References
Achazi R (2002) Invertebrates in risk assessment: development of a test battery and of short term biotests for ecological risk assessment of soil. J Soils Sediments 2:174–178
Aldaya MM, Lors C, Salmon S, Ponge JF (2006) Avoidance bio-assays may help of soil to test the ecological significance pollution. Environ Pollut 140:173–180
Alef K (1995) Soil respiration. In: Alef K, Nannipieri P (eds) Methods in applied soil microbiology and biochemistry. Academic Press, London, pp 214–219
Alvarenga P, Palma P, Gonçalves A, Fernandes R, Varennes A, Vallini G, Duarte E, Cunha-Queda A (2008) Evaluation of tests to assess the quality of mine-contaminated soils. Environ Geochem Health 30:95–99
André A, Antunes SC, Goncalves F, Pereira R (2009) Bait-lamina assay as a tool to assess the effects of metal contamination in the feeding activity of soil invertebrates within a uranium mine area. Environ Pollut 157:2368–2377
Anjos JSA (2003) Avaliação da eficiência de uma zona alagadiça (wetland) no controle da poluição por metais pesados: o caso da Plumbum em Santo Amaro da Purificação/BA. Thesis, University of São Paulo, Brazil
Antunes SC, Castro BB, Pereira R, Goncalves F (2008) Contribution for tier 1 of the ecological risk assessment of Cunha Baixa uranium mine (Central Portugal): II. Soil ecotoxicological screening. Sci Total Environ 390:387–395
Araújo ASF, Monteiro RTR (2007) Indicadores biológicos de qualidade do solo. Biosci J 23:66–75
ASTM (2002) Standard guide for conducting acute toxicity tests on test materials with fishes, macroinvertebrates, and amphibians. E 729-96. Annual Book of ASTM Standards 11.05
ASTM (2004) Test Method D5660-96, Standard test method for assessing the microbial detoxification of chemically luminescent marine bacterium. D5660-96
Boivin MEY, Greve GD, Kools SAE, van der Wurff AWG, Leeflang P, Smit E, Breure AM, Rutgers M, van Straalen NM (2006) Discriminating between effects of metals and natural variables in terrestrial bacterial communities. Appl Soil Ecol 34:103–113
Carvalho FM, Neto AMS, Tavares TM, Costa ACA, Chaves CR, Nascimento LD, Reis MA (2003) Chumbo no sangue de crianças e passivo ambiental de uma fundição de chumbo no Brasil. Pan Am J Public Health 13:19–23
Costa ACA (2001) Avaliação de alguns efeitos do passivo ambiental de uma metalurgia de chumbo em Santo Amaro da Purificação, Bahia. Dissertation, Federal University of Bahia
Critto A, Torresan S, Semenzin E, Giove S, Mesman M, Schouten AJ, Rutgers M, Marcomini A (2007) Development of a site-specific ecological risk assessment for contaminated sites: part I. A multi-criteria based system for the selection of ecotoxicological tests and ecological observations. Sci Total Environ 379:16–33
Dawson JJC, Godsiffe EJ, Thompson IP, Ralebitso-Senior TK, Killham KS, Paton GI (2007) Application of biological indicators to assess recovery of hydrocarbon impacted soils. Soil Biol Biochem 39:164
De Zwart D, Posthuma L (2005) Complex mixture toxicity for single and multiple species: proposed methodologies. Environ Toxicol Chem 24:2665–2676
Fernandez MD, Cagigal E, Vega MM, Urzelai A, Babin M, Pro J, Tarazona JV (2005) Ecological risk assessment of contaminated soils through direct toxicity assessment. Ecotoxicol Environ Saf 62:174–184
Fernandez MD, Vega MM, Tarazona JV (2006) Risk-based ecological soil quality criteria for the characterization of contaminated soils. Combination of chemical and biological tools. Sci Total Environ 366:466–484
Filzek PDB, Spurgeon DJ, Broll G, Svendsen C, Hankard PK, Parekh N, Stubberud HE, Weeks JM (2004) Metal effects on soil invertebrate feeding: measurements using the bait lamina method. Ecotoxicology 13:807–816
Fountain MT, Hopkin SP (2001) Continuous monitoring of Folsomia candida (Insecta: Collembola) in a metal exposure test. Ecotox Environ Saf 48:275–286
FUNASA (Fundação Nacional de Saúde) (2003) Avaliação de risco à saúde humana por metais em Santo Amaro da Purificação, Bahia. http://portal.saude.gov.br/portal/svs/visualizar_texto.cfm?idtxt=24117
Godínez-Alvarez H, Herrick JE, Mattocks M, Toledo D, Van Zee J (2009) Comparison of three vegetation monitoring methods: their relative utility for ecological assessment and monitoring. Ecological Indicators 9:1001–1008
Greenslade P, Vaughan GT (2003) A comparison of Collembola species for toxicity testing of Australian soils. Pedobiologia 47:171–179
Gülser F, Erdogan E (2008) The effects of heavy metal pollution on enzyme activities and basal soil respiration of roadside soils. Environ Monit Assess 145:127–133
Hamel C, Schellenberg MP, Hanson K, Wang H (2007) Evaluation of the “bait-lamina test” to assess soil microfauna feeding activity in mixed grassland. Appl Soil Ecol 36:199
Hund-Rinke K, Wiechering H (2001) Earthworm avoidance test for soil assessments. An alternative for acute and reproduction tests. J Soils Sediments 1:15–20
Hund-Rinke K, Achazi R, Römbke J, Warnecke D (2003) Avoidance test with Eisenia fetida as indicator for the habitat function of soils: results of a laboratory comparison test. J Soils Sediments 3:7–12
ISO (1994a) Soil quality – Determination of pH. ISO 10390. International Organization for Standardization, Geneva
ISO (1994b) Soil quality—determination of effective cation exchange capacity and base saturation level using barium chloride solution. ISO 11260. International Organization for Standardization, Geneva
ISO (1998) Soil quality—determination of the water-retention characteristic—laboratory methods. ISO 11274. International Organization for Standardization, Geneva
ISO (2007a) Soil quality—avoidance test for testing the quality of soils and effects of chemicals—part 2: test with collembolans (Folsomia candida) ISO Draft 17512-2. International Organization for Standardization, Geneva
ISO (2007b) Soil quality—avoidance test for testing the quality of soils and the toxicity of chemicals—test with earthworms (Eisenia fetida). ISO 17512-1. International Organization for Standardization, Geneva
Jensen J, Mesman M (eds) (2006) Ecological risk assessment of contaminated land—decision support for site specific investigations. RIVM report 711701047. p 136
Kuperman RG, Checkai TR, Garcia M, Römbke J, Stephenson GL, Sousa JP (2009) State of the science and the way forward for the ecotoxicological assessment of contaminated land. Pesq Agropec Bras 44:811–824
Larink O, Sommer R (2002) Influence of coated seeds on soil organisms tested with bait lamina. Eur J Soil Biol 38:287–290
LNEC (1970) Solos—análise granulométrica por peneiração húmida. LNEC-E 239. Laboratório Nacional de Engenharia Civil, Lisboa, Portugal
Lock K, Janssen CR, Coen WM (2000) Multivariate test designs to asses the influence of zinc and cadmium bioavailability in soils on the toxicity to Enchytraeus albidus. Environ Toxicol Chem 19:2666–2671
Lock K, Janssen CR (2001) Effect of clay and organic matter type on the ecotoxicity of zinc and cadmium to the potworm Enchytraeus albidus. Chemosphere 44:1669–1672
Long ER, Chapman PM (1985) A sediment quality triad: measures of sediment contamination, toxicity and infaunal community composition in Puget sound. Mar Pollut Bull 16:405–415
Loureiro S, Ferreira ALG, Soares AMM, Nogueira AJA (2005a) Evaluation of the toxicity of two soils from Jales Mine (Portugal) using aquatic bioassays. Chemosphere 61:168–177
Loureiro S, Soares AMVM, Nogueira AJA (2005b) Terrestrial avoidance behaviour tests as screening tool to assess soil contamination. Environ Pollut 138:121–131
Lukkari T, Haimi J (2005) Avoidance of Cu- and Zn-contaminated soil by three ecologically different earthworm species. Ecotoxicol Environ Saf 62:35–41
Lukkari T, Aatsinki M, Vaisanen A, Haimi J (2005) Toxicity of copper and zinc assessed with three different earthworm tests. Appl Soil Ecol 30:133–146
Machado SL, Ribeiro LD, Kiperstok A, Botelho MAB, Carvalho MF (2004) Diagnóstico da contaminação por metais pesados em Santo Amaro—BA. Eng Sanit Ambient 9:140–155
Natal-da-Luz T, Ribeiro R, Sousa JP (2004) Avoidance tests with Collembola and earthworms as early screening tools for site-specific assessment of polluted soils. Environ Toxicol Chem 23:2188–2193
Natal-da-Luz T, Römbke J, Sousa JP (2008) Avoidance tests in site-specific risk assessment: influence of soil properties on the avoidance response of collembola and earthworms. Environ Toxicol Chem 27:1112–1117
Natal-da-Luz T, Tidona S, Jesus B, Morais P, Sousa J (2009) The use of sewage sludge as soil amendment. The need for an ecotoxicological evaluation. J Soils Sediments 9:246–260
OECD (1984a) Earthworm, acute toxicity tests. Guidelines for testing of chemicals, 207. Organization for Economic Co-operation and Development, Paris
OECD (1984b) Daphnia sp., acute immobilization test and reproduction test. OECD 202. OECD – Organization for Economic Cooperation and Development, Paris
Römbke J, Jansch S, Didden W (2005) The use of earthworms in ecological soil classification and assessment concepts. Ecotoxicol Environ Saf 62:249–265
Römbke J, Hofer H, Garcia MVB, Martius C (2006) Feeding activities of soil organisms at four different forest sites in Central Amazonia using the bait lamina method. J Trop Ecol 22:313–320
Rosa R, Moreira-Santos M, Lopes I, Silva L, Rebola J, Mendonça E, Picado A, Ribeiro R (2010) Comparison of a test battery for assessing the toxicity of a bleached-kraft pulp mill effluent before and after secondary treatment implementation. Environ Monit Assess 161:439–451
Rutgers M, Jensen J (2010) Site-specific ecological risk assessment. In: Swartjes FA (ed) Dealing with Contaminated Sites: From Theory towards Practical Application. Springer
Rutgers M, Tuinstra J, Spijker J, Mesman M, Wintersen A, Posthuma L (2008) Ecological risks of soil contamination in the second step of the remediation criterion. Report 711701072, RIVM, Bilthoven, The Netherlands (in Dutch with English summary)
Sakuma M (1998) Probit analysis of preference data. Appl Entomol Zool 33:339–347
Salemaa M, Vahan-Majamaa I, Deromeu J (2001) Understory vegetation along a heavy-metal pollution gradient in SW Finland. Environ Pollut 112:339–350
Sandifer RD, Hopkin SP (1996) Effects of temperature on the relative toxicities of Cd, Cu, Pb and Zn to Folsomia candida (Collembola). Ecotoxicol Environ Saf 37:125–130
Schaefer M (2003) Behavioural Endpoints in earthworm ecotoxicology: evaluation of different test systems in soil toxicity assessment. J Soils Sediments 3:79–84
Spurgeon DJ, Svendensen C, Hankard PK (2005) Biological methods for assessing potentially contaminated soils. In: Thompson KC, Wadhia K, Loibner AP (eds) Environmental toxicity testing. Blackwell Publishing CRC Press, Oxford, pp 163–205
USEPA (2002) Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms. EPA-821-R-02-012. United States Environmental Protection Agency, Washington DC
USEPA (2004) Framework for inorganic metals risk assessment. Draft EPA/630/P-04/068B, United States Environmental Protection Agency, Washington DC
Van Gestel CAM (1992) The influence of soil characteristics on the toxicity of chemicals for earthworms: a review. In: Greig-Smith PW, Becker H, Edwards PW, Heimbach F (eds) Ecotoxicology of earthworms. Intercept Ltd., Andover, pp 44–54
Van Gestel CAM, van der Waarde JJ, Derksen JGM, van der Hoek EE, Veul M, Bouwens S, Rusch B, Kronenburg R, Stokman GNM (2001) The use of acute and chronic bioassays to determine the ecological risk and bioremediation efficiency of oil-polluted soils. Environ Toxicol Chem 20:1438–1449
Van Gestel CAM, Kruidenier M, Berg MP (2003) Suitability of wheat straw decomposition, cotton strip degradation and bait-lamina feeding tests to determine soil invertebrate activity. Biol Fert Soils 37:386–386
Van Gestel CAM, Koolhaas JE, Hamers T, van Hoppe M, van Roovert M, Korsman C, Reinecke S (2009) Effects of metal pollution on earthworm communities in a contaminated floodplain area: Linking biomarker, community and functional responses. Environ Pollut 157:895–903
Veiga MD, Wildner LDP (1993) Manual para la instalación y conducción de experimentos de perdida de suelos. EPAGRI/FAO, Santiago
Von Törne E (1990) Assessing feeding activities of soil living. I. Bait-lamina test animals. Pedobiologia 34:89–101
Wagelmans M, Derksen JGM, Kools SAE, Faber J, van der Pol J, Mesman M, Lud D (2009) Evaluation of the implementation of Triad in assessing ecological risks. SKB (The Centre for Soil Quality Management and Knowledge Transfer) project report. The Netherlands. p 46
Weeks JM, Sorokin N, Johnson I, Whitehouse P, Ashton D, Spurgeon D, Hankard P, Svendsen C, Hart A (2004a) Biological Test Methods for Assessing Contaminated Land, Stage 2: a demonstration of the use of a framework for the ecological risk assessment of land contamination. Environment Agency of England and Wales, Science Report P5-069/TR1
Weeks JM, Spurgeon DJ, Svendsen C, Hankard PK, Kammenga JE, Dallinger R, Kohler HR, Simonsen V, Scott-Fordsmand J (2004b) Critical analysis of soil invertebrate biomarkers: a field case study in Avonmouth, UK. Ecotoxicology 13:817–822
Weltens R, Goossens R, Van Puymbroeck S (2000) Ecotoxicity of contaminated suspended solids for filter feeders (Daphnia magna). Bull Environ Contam Toxicol 39:315–323
Yeardley RB, Lazorchak JM, Gast LC (1996) The potential of an earthworm avoidance test for evaluation of hazardous waste sites. Environ Toxicol Chem 15:1532–1537
Zar J (1996) Biostatistical analysis. Hall International, London
Zimakowska-Gnoinska D, Bech J, Tobias FJ (2000) Assessment of the heavy metal pollution effects on the soil respiration in the Baix LLobregat (Catalonia, NE Spain). Environ Monit Assess 61:301–313
Acknowledgments
The authors would like to thank Cetrel S/A—Empresa de Proteção Ambiental (Camaçari, BA, Brazil) for the Microtox analysis. A special thanks to Alice Andrade, Salomão Pinho and Bruno Abdon for their help in the field work, and to Tiago Natal-da-Luz and Sónia Chelinho for their help in the laboratory. The authors would also like to thank Dr. Michiel Rutgers for his fundamental contribution to improve the manuscript, and also to the two anonymous reviewers for their comments and suggestions. The study was supported through two Ph.D. grants to Júlia C. Niemeyer, from Program Alban (E05D058118BR, from Fev/06 to Fev/07) and Fundação para a Ciência e a Tecnologia (FCT, Portugal; SFRH/BD/28796/2006).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Jörg Römbke
Rights and permissions
About this article
Cite this article
Niemeyer, J.C., Moreira-Santos, M., Nogueira, M.A. et al. Environmental risk assessment of a metal-contaminated area in the Tropics. Tier I: screening phase. J Soils Sediments 10, 1557–1571 (2010). https://doi.org/10.1007/s11368-010-0255-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-010-0255-x