Abstract
Purpose
The aim of this study was to assess phosphorus-retention (Pret) capacities via P adsorption (Pads) and desorption (Pdes) by sediments collected from six different sources associated with various origins, physical, and chemical characteristics.
Materials and methods
Sediment samples were collected in the State of Florida, the USA, from estuary, marine, wetland, canal, river, and lake, respectively. Phosphorus adsorption and desorption for each sediment were evaluated in three types of ambient water, i.e., marine, wetland, and canal, with different rates of phosphate added and then desorbed by chloride (Cl−). Capacities in adsorption and desorption of P by various sediments were evaluated with different isotherm models to compare their potentials and stabilities in P retention.
Results and discussion
Sediments from the canal and lake had the greatest native adsorbed P and the highest zero equilibrium P concentration. Sediment from the estuary had the highest P adsorption and followed by those from wetland and marine origins, respectively. Phosphorus desorption from sediments by replacing exchangeable P with Cl− (20 mmol L−1 KCl) was obtained from an excellent fit of the data by an exponential growth model of desorption kinetics. The fractions of retained P (Pret = Pads − Pdes) were as high as 85–98% in the studied sediments, which displayed strong P retention capacities by all these sediments.
Conclusions
High capacities to retain P by sediments from estuary and wetland may play a critical role in buffering some chemical and ecological changes and benefit aquatic eco-environments by preventing P rapid release to the overlying water column.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Phosphorus (P) concentrations and bioactivities in aquatic environments are major concerns due to the potential of P to cause eutrophication. Sediment is a dominant P reservoir in many aquatic ecosystems (Lebo 1991; Zwolsman 1994; Conley et al. 1995; Paludan and Morris 1999) because suspended particulates in water columns that eventually become bottom sediments (Gibbs 1981) possess strong affinities for dissolved P (Meybeck 1982). Phosphorus adsorption and precipitation have been identified as important mechanisms for P removal from aquatic ecosystems by the solid phase, which may govern dissolved phosphate concentrations in the overlying water (Mortimer 1941; Stumm and Leckie 1970; Froelich et al. 1982). However, sediment accretion is a major sink for P in most aquatic environments, including wetlands.
Sediment accretion rates that depend on water velocity and P loading, which improves the aquatic biomass production and the detritus deposition, vary from a few millimeter to >10 mm year−1 in the Florida Everglades and other wetlands (Craft and Richardson 1993; Reddy et al. 1993). For example, up to 385 Mg year−1 of P loading may be delivered through 2,500 km of canals and levees to the three water conservation areas (WCAs) of the Everglades (SFWMD 1992; Reddy et al. 1998; Diaz et al. 2006). Therefore, sediments can function either as a P sink or a source, depending on sediment geochemical or physicochemical characteristics (Richardson 1985; Reddy et al. 1995). For example, P exchange across the sediment–water interface, via adsorption and desorption, plays a crucial role in governing the availability of water-soluble P (Froelich 1988).
Phosphorus binding forms in sediments vary greatly with their origins and geochemical compositions. In most Florida aquatic environments, especially in the south, sediments contain an abundance of calcium carbonate mainly in the canal and marine sediments, which precipitates substantial amounts of P to form hardly water-soluble Ca-bound P, such as hydroxylapatite [Ca10(PO4)6(OH)2], fluoroapatite [Ca10(PO4)6(F, OH)2], and chloroapatite [Ca10(PO4)6Cl2] (Morel and Hering 1993; De Kanel and Morse 1978). Under some environmental conditions, such as sediments from the wetland of Florida Everglades and the Lake Okeechobee, besides organic P, Fe- and Al-bound P as metal oxides might be a major fraction (Reddy et al. 1995, 1998). Furthermore, geophysical properties such as particle size distribution associated with the total surface area might be dominant factors in determining P retention. Indeed, the amount of P adsorbed increases exponentially with decreasing particle size (Walter and Morse 1984; Zhang and Huang 2007). In some marine sediments, the proportion of fine particles is much lower than that of coarse particles. Nevertheless, a small amount of fine particles may play a very important role in P adsorption because of the large cumulative reactive surface area. For example, in sediments in which fine particles accounted for only about 16% of the total weight, they accounted for 76% of the total surface area (Walter and Morse 1984). In addition, fine particles are readily resuspended by disturbances, such as a wave action, and reside longer than coarse particles in the water column; thus fine particles have more opportunities to interact with dissolved P than coarse particles.
In order to assess the impacts of sediments on P retention and their potential to buffer and conserve aquatic ecosystems, it is important to understand the P sedimentation process in different environments especially the effects of specific sediment characteristics, such as pH value, metal oxides, mineral particle size, and organic matter contents, etc., which may greatly affect P adsorption and desorption behaviors.
Commonly, the evaluation of P desorption behavior under conditions judged to simulate the natural environment has been conducted using a certain concentration of salt, such as 0.02 mol L−1 KCl, in distilled or deionized water. However, such an artificial solution differs greatly from the ambient water in which the corresponding sediment is located. To overcome this flaw in analytical methodology, three types of naturally occurring ambient surface water were collected, i.e., from wetland, canal, and Atlantic Ocean (marine) sites. In the current experiment involving the addition of a series of P standards, these natural water samples were used to measure P adsorption, while the same types of water samples with 20 mmol L−1 KCl as a neutral salt were used to simulate P desorption reactions for exchangeable P from mineral particle surfaces.
The main objectives of the current study are (a) to determine if the above sediments could adsorb additional P from the ambient water, (b) to evaluate P desorption behavior in order to assess P retention potential for each of the various sediments, and (c) to predict adsorption and desorption kinetics by fitting different isotherms.
2 Materials and methods
2.1 Description of sampling sites
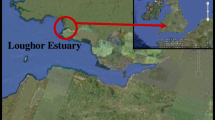
Six types of sediment samples were collected from the following sources in the State of Florida (USA): St. John’s River (river), St. Lucie Estuary (estuary), Lake Okeechobee (lake), canals (canal), the Everglades wetlands (wetland), and Florida Bay (marine) adjacent to the southern tip of the peninsula, respectively (Fig. 1). The St. John’s River, which is the longest river in Florida, runs 500 km northward from its headwaters in Indian River County to discharge into the Atlantic Ocean at Jacksonville in Duval County. The small difference in the elevation (about 10 m) between the headwaters and the mouth causes both the water velocity and volume to be extremely low. Because the very low velocity strongly impedes pollutant flushing, the vulnerability of the river ecosystem is a serious concern. The major pollutant sources include discharges from wastewater treatment plants and runoff from urban and agricultural areas (SJRWMD 2008).
The St. Lucie Estuary is a major component of the southern portion of the Indian River Lagoon which is located on the southeast coast of Florida and discharges into the Atlantic Ocean through the St. Lucie Inlet (Woodward-Clyde 1994). Due to extensive urban and agricultural drainage projects in the St. Lucie Estuary watershed, this historic drainage basin area has been greatly expanded to almost 2,006 km2 (775 m2), and the Estuary has been linked to Lake Okeechobee through the C-44 canal (Haunert and Steward 1994).
Lake Okeechobee, the second largest freshwater lake located wholly within the continental US with a drainage basin of more than 11,913 km2, covers 1,890 km2 with an average depth of only 2.7 m. Lake Okeechobee which has a capacity of 3.8 billon m3 of water is a major water source for south Florida communities and the Everglades.
Everglades Water Conservation Area 3 (WCA-3) which is considered to be the least impacted of the three WCAs being the most distant from the Everglades Agricultural Area (Reddy et al. 1998), occupies 232,600 ha dominated by sawgrass (Cladium spp.), marsh, tree islands, wet prairies, and aquatic sloughs. The L-28 canal and its interceptor canal from which the canal sediment and water samples were collected, lie along the western boundary of this WCA and together with the levees deliver water to the Everglades wetland.
Florida Bay, which is shallow covering an area of 2,100 to 2,600 km2 (Phirman 2000), lies between the southern end of the Florida mainland (the Everglades) and the Florida Keys. This estuarine area is fed mostly by runoff from the Everglades and sloughs, such as Taylor Slough, through sheet flow and from creeks.
2.2 Sample collection
A helicopter and motor boat were used to collect samples from wetlands and other sites, respectively. Intact sediment cores were collected using a polycarbonate tube (7 cm i.d.) with a sharp end for sampling wetlands in shallow water and a piston corer sampler (Fisher et al. 1992) attached to the polycarbonate tube for sampling sites with deep water. The core samples were transported to the laboratory, sliced into a series of 5-cm thick subsamples from top, air dried, ground, and sieved passing through a 1-mm sieve prior to chemical analysis and P sorption/desorption experiments. Due to a large variation in sampling depths of various sources (Table 1), only the top layer (0–5 cm) of each core column with four to six samples from each type was used in this experiment. Natural waters corresponding to the sediments from canal, wetland, and Atlantic Ocean (marine) were collected for the P adsorption/desorption experiments on their own and other sediments from similar environments.
2.3 Sample analysis
Sediment physical and chemical properties were assayed using the following methods. The pH and electrical conductivity (EC) were measured in the supernatant of a 1:2.5 (sediment: water) suspension. Sediment organic carbon and calcium carbonate (CaCO3) were determined by weight loss on ignition at 550°C for 12 h and pressure calcimeter methods (Wagner et al. 1998), respectively. Particle size distribution was obtained from the micropipette method (Miller and Miller 1987). Extractable Fe and Al were analyzed by inductively coupled plasma-optical emission spectroscopy (ICP-OES, Ultima 2C, Horiba, Jobin Yvon, Inc., Edison, NJ, USA) after extraction with 1 mol L−1 NH4-oxalate (Olson and Ellis 1982). Selected sediment characteristics are presented in Table 1.
The pH and EC values of surface water samples collected from wetland, canal, and marine were measured as for the sediment samples. Water total P was determined by a colorimetric method using an auto-analyzer (AA-3), which was specifically for very low level of P with the detection limit of 0.002 mg L−1 after digestion with HNO3 and HClO4 in accordance with EPA Method 365.4. Water ortho-P was measured using the same approach as used for total P after filtration through a 0.45-µm Magna nylon neutral membrane (Fisher Scientific). Ca, Mg, and Fe were quantified by ICP-OES after the samples had been filtered in the same way as for ortho-P. NO3-N, SO4 2−, and Cl− in the filtrate were determined by ion chromatography (CD25, Dionex, Sunnyvale, CA). Phosphorus desorbed by KCl was analyzed by AQ2+ auto-analyzer (AQ-2 Discrete Automated Analyzer, Seal-Analytical, Mequon, WI, USA) with an appropriate range of P concentrations. All these analyses were accomplished in our laboratory (Soil and Water Research) certified by National Environmental Laboratory Accreditation Conference.
2.4 Phosphorus adsorption experiment
Ambient waters collected from the L-28 canal in the Everglades or from the wetland of WCA-3 were used for the P adsorption studies of canal and lake sediments or both the wetland and river sediments, respectively. Marine water from the Atlantic Ocean of Florida Bay was used for the P adsorption studies on both marine and estuary sediments. Chemical properties of the various waters are presented in Table 2.
Two sets laboratory P adsorption experiments were conducted: (a) in low (0.01–0.1 mmol L−1) and (b) high P concentration (0.1–1.0 mmol L−1) ranges in order to cover the whole range of the prevailing P concentrations in the natural waters from various sources. For instance, the ortho-P concentration from marine water was as low as 0.06 µmol L−1 while that from the canal was as high as 0.7 µmol L−1 (see Table 2). The standard (reagent grade P) was added to each of the above waters to make two series of standard P solutions, i.e., (a) low range: 0, 0.01, 0.02, 0.03, 0.04, 0.05, 0.06, 0.07, 0.08, 0.09, and 0.1 mmol L−1 and (b) high range: 0, 0.1, 0.2, 0.4, 0.6, 0.8, and 1.0 mmol L−1. Next, 30 mL of each of the above P solutions was added to 1 g of each of the sediment samples, shaken at 100–120 rpm for 24 h (Gomez et al. 1999; Yoo et al. 2006) and centrifuged at 1,800 g for 15 min. The supernatant was filtered through a 0.45-µm Magna nylon neutral membrane and analyzed with a AQ2+ auto-analyzer for P as described above, and the residues were retained for the desorption experiment.
Linear, Freundlich, and Langmuir equations were used to plot the data obtained with the sediments. For this purpose, the difference between the initial (Pi) and equilibrium (Pe) concentration is the quantity of P adsorbed (Pads) by the respective sediment:
When Pi > Pe, net adsorption occurs; when Pi < Pe, negative adsorption (desorption) occurs; and when Pe = Pi, Pads = 0, neither adsorption nor desorption occurs and the P concentration at this point is called the zero equilibrium P concentration (EPC0) and represents the maximum potential for adsorbing P from the aqueous phase.
The linear equation was used to obtain the native adsorbed P (NAP) and EPC0 with the low range of P concentrations:
where Pads is the adsorbed P, K (slope) is a constant, Pe is the equilibrium P concentration, and NAP (intercept) is the amount of P that had been adsorbed on the sediment prior to the experiment. The EPC0 is given by the Pe = NAP/K when Pads = 0, i.e., the equilibrium P concentration at the point where neither adsorption nor desorption occurs.
The Freundlich equation is as follows:
where K f and n are empirical constants.
The Langmuir equation is as follows:
where Pe is the equilibrium P concentration, Pads is the adsorbed P concentration, Kl is a constant related to the energy that bonds P to the sediment, and Pmax is the P maximum adsorption capacity of the sediment.
The Langmuir equation for two-surface sorption is as follows:
where b1 and b2 are the high affinity and low affinity P sorption maxima and k1 and k2 are related to high affinity and low affinity bonding energies of P sorption, respectively. Based on the method described by previous researchers (Holford et al. 1974; Shuman 1975; Posner and Bowden 1980; Sposito 1982), a plot of Pads/Pe vs. Pads yields 2 straight lines if the data fit the isotherm equation well.
2.5 Phosphorus desorption experiment
After decanting the supernatant from the adsorption experiment, 20 mL of the natural water containing 20 mmol L−1 KCl was added to the corresponding sediment residues. The samples were shaken at 100–120 rpm for another 24 h, centrifuged and filtered, and the filtrate was analyzed for P as described above. The amount of P desorbed was calculated as the difference between the equilibrium and original concentrations after and before desorption, respectively. An exponential growth model with 2 and 3 parameters was used to describe the amount of P desorbed from the sediments. The two parameter exponential growth model is as follows:
where Pdes (mmol kg−1) is amount of P desorbed from the sediments, and a (mmol kg−1) and b (kg mmol−1) are constants related to desorption potential and the desorption rate, respectively.
The three-parameter exponential growth model displayed in (Eq. 7) takes into account the initial P concentration (P0; which includes the P that is free or loosely adsorbed onto the surfaces of the sediment), a, the coefficient of desorption potential, and b, the desorption rate constant, as in Eq. 6:
The retention capacity (Pret), quantitatively defined in (Eq. 8), is the amount of P adsorbed irreversibly onto the adsorbent surfaces and is the difference between adsorbed and desorbed P in the current experiment:
The P retention rate constant, K r, can be obtained by plotting Pads against Pret (Pads − Pdes), in which K r ≤ 1.
2.6 Data processing and statistical analysis
The data were subjected to ANOVA analysis and stepwise multiple regression with the PROC NLIN model, and the use of quadratic, Gauss–Newton, Marquardt, and exponential models for goodness of fit correlations (SAS, version 9.1.3, 2004).
3 Results and discussion
3.1 Phosphorus adsorption
Similar trends (Fig. 2) in P adsorption were displayed by all sediments, i.e., at low P concentrations, P adsorbed increased slowly with increasing P, but at a certain P concentration, P adsorbed increased progressively more strongly until a fairly high constant rate of increase had been attained. Although all sediments displayed this general pattern, the amount of P adsorbed at a given P concentration differed among the various sediments. Over the whole P concentration range, P adsorption capacity followed the order: estuary > wetland > river ≥ canal ≥ marine > lake sediments.
Within the initial low P concentration range, a linear relationship existed between the equilibrium P concentration and the amount of P adsorbed for sediments with both low and high P adsorption capacities (Fig. 3), from which EPC0 and NAP were derived from the X value when zero adsorption occurred and Y value as the intercept, respectively. In this P concentration range, both canal and lake sediments had greater NAP and EPC0 values than the other sediments (Table 3). This result demonstrated that sediments from the wetland, marine, river, or estuary had higher potential in P adsorption than either the canal or lake sediment since the P adsorption capacity is adversely controlled by NAP and EPC0 (Froelich 1988; Zhang and Huang 2007).
Linear relationship between equilibrium and adsorbed P concentrations for wetland and canal sediments, i.e., Pads = KPe − NAP where Pads is the adsorbed P, K (slope) is a constant, Pe is the equilibrium P concentration, and NAP (x-intercept) is the amount of P that had been adsorbed by the sediment prior to the experiment. The EPC0 is given by the Pe = NAP/K when Pads = 0, i.e., the equilibrium P concentration where neither adsorption nor desorption occurs
Within the whole P concentration range, both Freundlich and Langmuir adsorption isotherms were used to describe the relationship between the equilibrium P concentration and the amount of adsorbed P (Fig. 4 and Table 3). In general, the Freundlich equation provided a better fit to the data than the Langmuir. Based on higher values for n and K f in the Freundlich isotherm, marine and estuary sediments showed a higher P adsorption potential than the other sediments. For wetland sediments the relationship between the amount of P adsorbed and the equilibrium P concentration as determined by the Langmuir and linear isotherms was quite similar. For the estuary sediment, the results obtained with Langmuir and Freundlich isotherms were generally in a good agreement. Further the results obtained with the Langmuir isotherm indicated that estuary sediment has the highest Pmax and Kl values, but the relationship was relatively weak (low R 2). The two-surface Langmuir equation fitted the P adsorption data well for all sediments.
Because the parameters derived from the two-surface Langmuir equation (Table 4) differ greatly among the sediments, several different mechanisms might be involved in determining the intensity of P adsorption. For example, the estuary sediment, which showed a high Pmax with the simple Langmuir equation, had the largest b1 value (high affinity maximum for P adsorption) while in the marine sediments (see Table 4), the highest b2 (low affinity maximum for P adsorption) and largest exponential factor (n) were found with the Freundlich equation (see Table 3). For the wetland sediment, high- and low-affinity P adsorption maxima were both relatively low, whereas the k1 (high-affinity P bonding energy) and k2 values (low-affinity P bonding energy) were the highest among all the sediments. The main reason to include the amount of P adsorbed on both axes of the two-surface approach is to decrease the variability of the data (Harter 1984; Schulthess and Dey 1996). This was effective for the marine sediments but was not obvious in the others. Instead, when Pe/Pads vs. Pe was plotted for all the sediments, two straight lines each with a good fit to the data points were obtained (Fig. 5).
Graphs of the two-surface Langmuir isotherm for the amounts of P adsorbed by sediments sourced from various sites in Florida. The two-surface Langmuir isotherm equation is: \( {{\hbox{P}}_{\rm{ads}}} = \frac{{{b_1}{K_1}{P_e}}}{{1 + {k_1}{P_e}}} + \frac{{{b_2}{k_2}{P_e}}}{{1 + {k_2}{P_e}}} \), where b1 and b2 are the high-affinity and low-affinity maxima of P sorption, respectively, and k1 and k2 are related to high-affinity and low-affinity bonding energies of P sorption, respectively. Two straight lines can be obtained from a plot of Pads/Pe vs. Pads if the data are a good fit to the isotherm equation
The greater intensity of P adsorption by the estuary than lake sediment implies that major differences in physicochemical characteristics exist probably due to a larger fraction of fine particles in the former than the latter (Table 5). This might be true because estuarine water velocity is substantially lower towards the mouth allowing most fine particles to settle to the bottom, whereas in the channelized Kissimmee River as it enters Lake Okeechobee, the velocity is much greater than that of the estuary. However, the particle size distributions of estuary and lake sediments proved to be quite similar (see Table 5). Some other rather complicated factors and their interactions, such as ion exchange capacity, organic matter content, Ca and Mg, amorphous or poorly crystalline complexes of Fe and Al, etc., might play an important role in P adsorption behaviors. Because sediments are highly heterogeneous in chemical composition and physical properties, the relationship to P adsorption behavior is not very close. The high P adsorption capacity of the estuary sediment might be an example of the effects of multiple or unknown factors.
The high P adsorption in the marine sediment might be related to an abundance of CaCO3 which has an especially high affinity for P resulting in Ca-bound P being the dominant form in the marine environments (up to 56% of total and 96% of inorganic P, Koch et al. 2001). Similar results have been reported by others (Stumm and Leckie 1970; De Kanel and Morse 1978; Kitano et al. 1978), who concluded that the formation of inorganic solid-phase P reservoirs in marine carbonate sediments is driven by P chemisorption onto calcite and aragonite followed by coprecipitation to form apatite. In aquatic systems rich in Ca, such as canal sediments in Water Conservation Area-3 of the Everglades, the Ca-bound P can constitute as much as 87% of total P (Wang and Li 2009). Reddy et al. (1995) also found a significant correlation between P adsorption capacity and the concentration of reactive Ca in stream sediments in the Lake Okeechobee Basin. In addition to Ca, Mg-bound P may play an important role in aquatic ecosystems with abundant quantity of Mg, such as in the waters from the marine and canal in this experiment (see Table 2). According to Pant and Reddy (2001) based on their P adsorption experiment with estuarine sediments under different redox conditions, Mg as MgCO3 might play a dominant role over Ca in binding P. They explained that the mass ratio of Ca to Mg was high, and the system was relatively dominated with CaCO3 compared with MgCO3, which left Mg available for adsorbing P (Pant and Reddy 2001). However, because Ca and Mg have quite similar function and mechanism in binding P and they coexist under the natural environment in most cases and it is hard to separate one from the other, most people refer using Ca- and Mg-bound P instead of Ca-P or Mg-P (Reddy and DeLaune 2008).
The high P adsorption potential of wetland sediments may result from abundant hydrated oxides of Fe and Al as evidenced by the extraordinarily high concentrations extracted with 1 mol L−1 NH4-oxalate relative to the other sediments (see Table 1) confirming the findings of Reddy et al. (1995) who demonstrated a high correlation between P adsorption potential and oxalate extractable Fe and Al in Everglade sediments. Moreover, sediments enriched with amorphous and poorly crystalline forms of Fe can act as an excellent reservoir for P by adsorbing excess P (Hingston et al. 1974; Krom and Berner 1980; Loeppert and Inskeep 1996). It has been widely accepted that aerobic conditions with Fe(III) dominated to promote P adsorption by lake sediments and anoxic conditions with Fe(II) dominated to favor P release from the sediments to the overlying water since several decades ago (Einsele 1936, 1938; Mortimer 1941). Dramatic improvements in P adsorption capacity of marine sediments by the addition of amorphous Fe oxides have been observed by many investigators (Sundby et al. 1992; Jensen et al. 1995; Slomp et al. 1998; Schwertmann and Cornell 2000). However, some researchers observed that in certain marine sediments the overall P adsorption behavior was governed by the content of exchangeable phosphate rather than Fe oxides, and the Fe oxides became more important only in sediments that were poor in P (Zhang and Huang 2007). Clearly many factors can play a crucial role in P adsorption in such heterogeneous systems. For example, P adsorption is affected significantly by organic matter (Reddy et al. 1995), Fe- and Al-organic matter complexes (Syers et al. 1973; Traina et al. 1986), and reduction of Fe(III) oxyhydroxides to more soluble Fe(II) forms under anaerobic conditions (Patrick and Khalid 1974; Pant and Reddy 2001). It has been found by a number of scientists (Caraco et al. 1993; Urban 1994; Lamers et al. 1998; Zak et al. 2006) that sulfate (SO4 2−) plays an important role in enhancing P desorption of various sediments in the natural aquatic system mainly by the formation of Fe reducing sulfide (S2−) through SO4 2− reduction. As a result, soluble Fe(II) becomes insoluble FeS and formerly immobile phosphate is released. On the other hand, organic matter in the sediments may act as electron donor and Fe(III) and SO4 2− as electron acceptors to improve P mobilization (Holmer and Storkholm 2001; Zak et al. 2006). However, there seems to be no enough evidence in a short period (24 h) to display such influence of SO4 2− in the current studied sediments although the SO4 2− concentration from the marine water was very high (see Table 2).
Under certain circumstances, oxygen status in the natural aquatic system may play an important role for P adsorption. For example, a long-standing paradigm of oxygen controls the P release from sediments has been established by pioneer researchers (Einsele 1936, 1938; Mortimer 1941). Some studies with strong correlation results support this paradigm (Beutel and Horne 1999; Grochowska and Gawronska 2004) but contrary results have been found by many other researchers (Garrell et al. 1977; Gachter and Wehrli 1998; Schauser and Chorus 2007), which has casted doubt on the universal validity that oxygen controls the P release from sediments. Therefore, Hupfer and Lewandowski (2008) concluded that the temporal existence of a thin oxidized sediment surface layer could affect only fluctuations of the temporary P pool at the sediment surface but not the long-term P retention.
Comparing results from fresh samples, effect of oxygen status on P adsorption by these air dried sediment samples used in this experiment might not be important because Fe(II) in the sediments must be transformed to Fe(III) after dehydration, which should make the results from various sediment origins comparable. For instance, the sediments from seasonal wetland have relatively better aeration than the sediments from lake under deep water, which would result in complicated comparisons if fresh samples were used in such an experiment. However, there must be some differences between laboratory results and the natural aquatic conditions that rather complicated reactions, such as chemical, biological, and their interactions, always happen.
The empirical coefficients of the Freundlich, Langmuir, and Langmuir two-surface isotherms determined for the various sediments in the dehydrated status (see Table 3) pertain only to the capability of each to adsorb the reactive P that was added to the solution. These coefficients which are lower than those of the other sediments reflect the relatively low affinity of P for the canal and lake sediments. Both canal and lake sediments have high Pe and NAP values due to their prolonged contact with ambient waters containing relatively high concentrations of reactive P (see Table 2). Because the lake drains via the canal into the Everglades, the P concentration of these waters was assumed to be similar, indicating that mineral particle surfaces of the lake and canal sediments are likely to be highly occupied by phosphate. In contrast, the relatively “clean” sediments (estuary, marine, and wetland) are likely to possess more unoccupied surface sites of mineral or metal oxides available for P adsorption than in the other sediments helping to explain the relatively low P adsorption capacities of canal and lake sediments. In addition, canal and lake sediments are complex arrangements of many components related to the upstream runoff and velocity caused by heavy rainfall, such as tropical storms or hurricanes often happen in this subtropical region, which might be another reason to display a lower adsorption capacity compared to the other sediments.
3.2 Phosphorus desorption
Phosphorus desorption from all sediments by adding Cl− (KCl) to replace adsorbed P increased at an exponential rate with increasing amounts of adsorbed P (Fig. 6). This seems to be a reliable generalization because there was an excellent data fit to the three-parameter growth model (Table 6) used to describe desorption kinetics. However, the quantity of P desorbed by Cl− was mainly in an exchangeable form adsorbed on the surface of mineral or metal oxide particles, and this fraction of P is considered to be most reactive, which can influence the overlying water quality substantially (Reddy and DeLaune 2008). Initial equilibrium P concentrations (P0) for all sediments (see Table 6) were negative and relatively low except for the lake and canal sediments, which means that P0 had little influence on P desorption. Therefore, there was a good fit of the P desorption data with the two-parameter exponential growth model as well for all these sediments.
Desorption of adsorbed P from different sediments sourced from various sites in Florida by the addition of 20 mmol L–1 KCl. The graphs were obtained by fitting the experimental data to a 3-parameter growth model of desorption kinetics: Pdes = P0 + aExp(bPads) where Pdes is the amount of P desorbed, P0 is concentration of P the initial P concentration, a is the coefficient of desorption potential, b is the desorption rate constant and Pads is the amount of P that had been adsorbed by the sediment
Both the estuary and wetland sediments had low P desorption potential (“a” values; see Table 6), whereas for the lake and wetland sediments, the values were 167- and 15-fold greater than that for the estuary. Thus the lake sediment had a relatively high P desorption potential. The same results, visually depicted in Fig. 6, showed that the lake sediment underwent the greatest and the estuary sediment the least P desorption by this neutral salt of KCl. Therefore, the adsorbed P in the estuary sediment seemed to be more stable than that in the other sediments.
The reason resulted in such differences of P desorption among various sediments is not quite clear from the current experiment, the capacity of positive charges on the sediment surface with low pH, such as the sediments from the estuary with pH as low as 5.8 in the 0–5 cm layer (see Table 1), definitely help to retain surface adsorbed P. However, this cannot explain other sediments, such as the canal sediments (pH 7.8) as compared with the lake sediments (pH 7.6). We believe that the adsorption and desorption capacity of P with different sediments is a rather complex consequence of multiple factors and their interactions, e.g., pH value, electrical conductivity, mineral or metal oxide type, particle size in related to the total surface area, organic matter, etc.
3.3 Phosphorus retention potential
The capacity of each sediment to retain P was obtained by plotting Pret (Pads − Pdes) against Pads, which proved to be a perfect linear relationship for each of the sediments (Fig. 7). The level of adsorbed P retention was surprisingly high for each of the studied sediments; the fraction not being displaced by Cl− was as high as >95% for both the estuary and wetland sediments and >85% for lake and canal sediments (Table 7). In order to obtain the retention level as described above, Pant and Reddy (2001) found cases where it was necessary to force the intercept through zero whereas in the current study, all intercept (α) were negligible, i.e., < <0.01 mmol kg−1 (see Table 7, Fig. 7).
P retention behavior of different sediments sourced from various sites in Florida. Each graphs displays the amounts of previously adsorbed P remaining on each sediment after being desorbed with 20 mmol L–1 KCl. Retention is described by the following equation: Pret = K rPads + α. The P retention rate constant, K r, was obtained by plotting Pads against Pret (Pads − Pdes), where K r ≤ 1
The P retention seems to be closely related to the density of vacant sites on the sediment particle surfaces. Estuary, marine, river, and wetland sediments, being relatively clean, have ample surface sites with positive charges available to adsorb the exchangeable form of P. In contrast, the corresponding sites on canal and lake sediments are largely occupied because of their long history of exposure to relatively high concentrations of P in the ambient waters. This result seems to have a good agreement with other researchers (Lin and Banin 2005; Lin et al. 2009) who concluded that long-term pollution of the river (Daliao river in China) lead to the decrease of P adsorption capacity of the river sediment or the top sandy soil in the basin of the river and the increase of P release potential. In aquatic ecosystems, P is distributed between the solid and liquid phases as a result of rather complex physical, chemical, and biological reactions involved in the P adsorption–desorption cycle. Even under the experimental conditions, the P adsorption and desorption behaviors differ greatly among various sediment types with their specific characteristics, which mostly result from the predominated or multiple attributes. For example, the low pH value in the estuary and river sediments, high electrical conductivity and abundant CaCO3 content in the marine sediments, extremely high Fe and Al in the wetland sediments (see Table 1) might be predominated factors in governing their P retention capacities.
Adsorption of P from ambient water onto sediment sinks is desirable to improve overlain water quality. On the other hand, P release from sediments by desorption is an important source of P contamination as internal loading of P to the overlying water column. P release can be evaluated by means of an adsorption experiment in which no P or very little P is added. This would be similar to the above study with the low range of P concentrations for determining the linear isotherm. Although there might be some differences in the natural ecosystems where much more complicated interactions occurred between biotic and abiotic components as compared to the current experiment with air-dried sediment samples, the results may provide some fundamental understanding about the behaviors of P adsorption and desorption among various sediments. In this instance, P desorption occurred when the equilibrium P concentration was lower than that of P adsorbed onto the sediment. Such releases of P are usually low since only exchangeable P is adsorbed or desorbed when exchanges occur between the solid and liquid phases. However, in the natural aquatic ecosystems, chemical, biological reactions, and their interactions in most cases are often involved, which may influence the overlying water quality considerably. To evaluate the double role of sediments as P sinks or sources to the overlying water, both adsorption and desorption processes are important. Overall, the retention of P depends not only on adsorption and desorption capacities determined mainly by the sediment types with their specific characteristics but more importantly, the environmental conditions prevailing in the sedimentation process, such as pH value, mineral and metal oxide particles, oxygen status, organic matter content and redox conditions, etc., may greatly affect the retention capacity of P to these sediments.
4 Conclusions
In spite of the relatively small number of sediment samples from the large and diverse ecosystems involved in this study, P adsorption, desorption, and retention behaviors of the sediment were quite distinct from each other. Lake and canal sediments had greater native adsorbed P and equilibrium P concentrations than the other sediments. These distinct properties of lake and canal sediments may be related to their complex physical, chemical, and biological reactions, such as precipitation, metal (Fe and Al) oxide formation, Ca-, Mg-bound, oxidation and reduction under aerobic and anaerobic conditions, etc., at the solid–liquid interface in the ambient waters containing relatively high P concentrations over prolonged periods.
The Freundlich isotherm was superior to the simple Langmuir isotherm in describing P absorption behavior of the various sediments but a better fit with the two-surface Langmuir equation revealed enhanced P adsorption capacities of the estuary, marine, and wetland sediments. The desorption of P from various sediments fitted an exponential growth model well showing that the estuary and wetland sediments had greater abilities to retain adsorbed P than the other sediments, which might be attributed to the low pH value for the estuary sediments and extraordinary high content of Fe and Al in the wetland sediments.
The desorption solution with the strength as high as 20 mmol L−1 Cl− used cannot fully replace all the adsorbed P from the sediment particle surfaces although Cl− as a neutral salt has been widely adopted to replace exchangeable P. The result implies that all these studied sediments have a substantial retention intensity with at least 85% of adsorbed P being retained against desorption. The results indicate that although all these studied sediments can still adsorb and retain P, control P external loading to these aquatic systems has become a critical challenge under the natural environmental conditions as a long-term strategy. In addition, under the natural environment, the P adsorption and retention capacities might not be as high as in the current experiment with air-dried samples, especially for the sediments always under the water, such as from lake, marine, or estuary, etc., where Fe(II) rather than Fe(III) might be a predominated form, which has a relatively lower stability and weaker P retention capacity as compared with Fe(III) oxide. The lake and canal sediments have displayed a relatively low P adsorption and retention capacity, which implies the importance in controlling the external loading in order to maintain their ecological and environmental functions. Although wetland, estuary, and marine sediments seem to have a potential in retaining P from external or internal loading from the present experiment, those aquatic systems are very vulnerable, subtle rise of P in the system may cause a serious consequence of ecosystem alteration, which has evidently occurred in the Florida Everglades wetland, where the ecosystem has changed substantially due to the P increase in the water body, e.g., invasive plants replacing native ones, food chain changes for aquatic fauna, wading birds and animals, etc. This is especially exacerbated in the area adjacent to the Everglades agricultural area, where nonpoint pollution has been identified as a major contribution, and the restoration of such deteriorated ecosystems undergoing (Comprehensive Everglades Restoration Plan) would cost billions of dollars and take several decades.
References
Beutel MW, Horne AJ (1999) A review of the effects of hypolimetic oxygenation on lake and reservoir water quality. Lake Reserv Manag 15:285–297
Caraco NF, Cole JJ, Likens GE (1993) Sulfate control of phosphorus availability in lakes. Hydrobiol 253:275–280
Conley DJ, Smith WM, Cornwell JC, Fisher TR (1995) Transformation on particle-bound phosphorus at the land sea interface. Estuar Coast Shelf Sci 40:161–176
Craft CB, Richardson CJ (1993) Peat accretion and phosphorus accumulation along a eutrophication gradient in the northern Everglades. Biogeochem 22:133–156
De Kanel J, Morse JW (1978) The chemistry of orthophosphate uptake from seawater on to calcite and aragonite. Geochim Cosmochim Acta 42:465–476
Diaz OA, Daroub SH, Stuck JD, Clark MW, Lang TA, Reddy KR (2006) Sediment inventory and phosphorus fractions for water conservation area canals in the Everglades. Soil Sci Soc Am J 70:863–871
Einsele W (1936, 1938) Cited by Hupfer M, Jewandowski J (2008) Oxygen controls the phosphorus release from lake sediments—a long-lasting paradigm in limnology. Int Rev Hydrobiol 93:415–432
Fisher MM, Bremner M, Reddy RK (1992) A simple, inexpensive piston corer for collecting undisturbed sediment/water interface profiles. J Paleolimn 7:157–161
Froelich PN (1988) Kinetic control of dissolved phosphate in natural rivers and estuaries: a primer on the phosphate buffer mechanism. Limnol Oceanogr 33:649–668
Froelich PN, Bender ML, Luedtke NA, Heath GR, DeVries T (1982) The marine phosphorus cycle. Am J Sci 282:474–511
Gachter R, Wehrli B (1998) Ten years of artificial mixing and oxygenation: no effect on the internal phosphorus loading of two eutrophic lakes. Environ Sci Technol 32:3659–3665
Garrell MH, Confer JC, Kirschner D, Fast AW (1977) Effects of hypolimnetic aeration on nitrogen and phosphorus in a eutrophic lake. Water Res 13:343–347
Gibbs RJ (1981) Sites of river-derived sedimentation in the ocean. Geology 9:77–80
Gomez E, Durillon C, Gofes G, Picot B (1999) Phosphate adsorption and release from sediments of brackish lagoons: pH, O2 and loading influence. Wat Res 33:2437–2447
Grochowska J, Gawronska H (2004) Restoration effectiveness of a degraded lake using multi-year artificial aeration. Polish J Environ Stud 13:671–681
Harter RD (1984) Curve-fit errors in Langmuir adsorption maxima. Soil Sci Soc Am J 48:749–752
Haunert DE, Steward J (1994) Surface water improvement and management plan for the Indian River Lagoon. South Florida Water Management District, West Palm Beach
Hingston FJ, Posner AM, Quirk JP (1974) Anion adsorption by goethite and gibbsite, 2. Desorption of anions from hydrous oxide surfaces. J Soil Sci 25:16–26
Holford ICR, Wedderburn RWM, Mattingly GEG (1974) A Langmuir two-surface equation as a model for phosphate adsorption by soils. J Soil Sci 25:242–255
Holmer M, Storkholm P (2001) Sulfate reduction and sulfur cycling in lake sediments: a review. Freshw Biol 46:431–451
Hupfer M, Lewandowski J (2008): Oxygen controls the phosphorus release from lake sediments- a long lasting paradigm in limnology. Review Hydrobiology, 93:415–432.
Jensen HS, Mortensen PB, Andersen FØ, Rasmussen E, Jensen A (1995) Phosphorus cycling in coastal marine sediments, Aarhus Bay, Denmark. Limnol Oceanogr 40:908–917
Kitano Y, Okumura M, Idogaki M (1978) Uptake of phosphate ions by calcium carbonate. Geochem J 12:29–37
Koch MS, Benz RE, Rudnick DT (2001) Solid-phase phosphorus pools in highly organic carbonate sediments of northeastern Florida Bay. Estuar Coast Shelf Sci 52:279–291
Krom MD, Berner RA (1980) Adsorption of phosphate in anoxic marine sediments. Limnol Oceanogr 25:797–806
Lamers LPM, Tomassen HBM, Roelofs JGM (1998) Sulfate-induced eutrophication and phytotoxicity in freshwater wetlands. Environ Sci Technol 32:199–205
Lebo ME (1991) Particle-bound phosphorus along an urbanized coastal plain estuary. Mar Chem 34:225–246
Lin C, Banin A (2005) Effect of long-term effluent reacharge on phosphate sorption by soils in a wastewater reclamation plant. Water Air Soil Pollut 164:257–273
Lin C, Wang Z, He M, Li Y, Liu R, Yang Z (2009) Phosphorus sorption and fraction characteristics in the upper, middle and low reach sediments of the Daliao river systems, China. J Hazard Mat 170:278–285
Loeppert RH, Inskeep WP (1996) Iron. In: Sparks DL (ed) Methods of soil analysis, Part 3, SSSA Book Series 5. Soil Science Society of America Inc, Wisconsin, pp 639–664
Meybeck M (1982) Carbon, nitrogen, and phosphorus transport by world rivers. Am J Sci 282:401–450
Miller WP, Miller DM (1987) A micro-pipette method for soil mechanical analysis. Commun Soil Sci Plant Anal 18:1–15
Morel FMM, Hering JG (1993) Principles and applications of aquatic chemistry. Wiley, New York, pp 320–570
Mortimer CH (1941) The exchange of dissolved substances between mud and water in lakes. J Ecol 29:280–329
Olson RV, Ellis R Jr (1982) Iron. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis, Part 2-chemical and microbiological properties, 2nd edn. Soil Science Society of America Inc, Wisconsin, pp 301–312
Paludan C, Morris JT (1999) Distribution and speciation of phosphorus along a salinity gradient in intertidal marsh sediments. Biogeochem 45:197–221
Pant HK, Reddy KR (2001) Phosphorus sorption characteristics of estuary sediments under different redox conditions. J Environ Qual 30:1474–1480
Patrick WH Jr, Khalid RA (1974) Phosphate release and sorption by soils and sediments: effect of aerobic and anaerobic conditions. Science 186:53–55
Phirman D (2000) The ecology of Florida Bay. www.restudy.org/flabay.htm
Posner AM, Bowden JW (1980) Adsorption isotherms: should they be split? J Soil Sci 31:1–10
Reddy KR, DeLaune RD (2008) Biogeochemistry of Wetlands: science and applications. CRC Press, Taylor and Francis Group, pp 335–339
Reddy KR, DeLaune RD, DeBusk WF, Koch MS (1993) Long-term nutrient accumulation rates in the Everglades. Soil Sci Soc Am J 57:1147–1155
Reddy KR, Diaz OA, Scinto LJ, Agami M (1995) Phosphorus dynamics in selected wetlands and streams of the Lake Okeechobee basin. Ecol Eng 5:183–207
Reddy KR, Wang Y, DeBusk WF, Fisher MM, Newman S (1998) Forms of soil phosphorus in selected hydrologic units of the Florida Everglades. Soil Sci Soc Am J 62:1134–1147
Richardson CJ (1985) Mechanisms controlling phosphorus capacity in freshwater wetlands. Science 228:1424–1427
SAS Institute (2004) SAS/STAT user’s guide, version 9.1.3, Cary, NC
Schauser I, Chorus I (2007) Assessment of internal and external lake restoration measures for two Berlin lakes. Lake Reserv Manag 23:366–376
Schwertmann U, Cornell RM (2000) Iron oxides in the laboratory: preparation and characterization. Wiley-VCH, Weinheim
Schulthess CP, Dey DK (1996) Estimation of Langmuir constants using linear and nonlinear least squares regression analyses. Soil Sci Soc Am J 60:433–442
SFWMD (1992) Surface water improvement and management plan for the Everglades: supporting information document. South Florida Water Management District, West Palm Beach
Shuman LM (1975) The effect of soil properties on zinc adsorption by soils. Soil Sci Soc Am Proc 39:454–458
Slomp CP, Malschaert JFP, van Raaphors WT (1998) The role of adsorption in sediment–water exchange of phosphate in North Sea continental margin sediments. Limnol Oceanogr 43:832–846
Sposito G (1982) On the use of the Langmuir equation in the interpretation of “adsorption” phenomena: II. The “two-surface” Langmuir equation. Soil Sci Soc Am J 46:1147–1152
St Johns River Water Management District (2008) St Johns river—fast facts. http://www.nbbd.com/godo/StJohns.html
Stumm W, Leckie JO (1970) Phosphate exchange with sediments: its role in the productivity of surface waters. In Advances in water pollution research, Pergamon Press, NY, pp 1–16
Sundby B, Gobeil C, Silverberg N, Mucci A (1992) The phosphorus cycle in coastal marine sediments. Limnol Oceanogr 37:1129–1145
Syers JK, Harris RF, Armstrong DE (1973) Phosphate chemistry in lake sediments. J Environ Qual 2:1–14
Traina SJ, Sposito G, Hesterberg D, Kafkafi U (1986) Effects of pH and organic acids on orthophosphate solubility in an acidic, montmorillonitic soil. Soil Sci Soc Am J 50:45–52
Urban NR (1994) Retention of sulfur in lake sediments. In: Baker LA (ed) Environmental chemistry of lakes and reservoirs. Adv Chem Ser 237:323–369
Wagner SC, Hanson DJ, Olness A, Voorhess WD (1998) A volumetric inorganic carbon analysis system. Soil Sci Soc Am J 62:690–693
Walter LM, Morse JW (1984) Reactive surface area of skeletal carbonates during dissolution: effect of grain size. J Sediment Petrol 54:1081–1090
Wang Q, Li Y (2009) Phosphorus distribution and fractionation in wetland and canal sediments of Water Conservation Area 3 in Florida Everglades. Water Resour Res (submitted)
Woodward-Clyde (1994) Uses of the Indian River Lagoon. Indian River Lagoon National Estuary Program, Melbourne
Yoo JH, Ro HM, Coi WJ, Yoo SH, Han KH (2006) Phosphorus adsorption and removal by sediments of a constructed marsh in Korea. Ecol Eng 27:109–117
Zak D, Kleeberg A, Hupfer M (2006) Sulfate-mediated phosphorus mobilization in riverine sediments at increasing sulfate concentration, River Spree, NE Germany. Biogeochem 80:109–119
Zhang JZ, Huang X (2007) Relative importance of solid-phase phosphorus and iron on the sorption behavior of sediments. Environ Sci Tech 41:2789–2795
Zwolsman JJG (1994) Seasonal variability and biogeochemistry of phosphorus in the Scheldt Estuary, southwest Netherlands. Estuar Coast Shelf Sci 39:227–248
Acknowledgments
The authors are deeply grateful to Mrs. G. Yu and Ms. L. Rosado, Mr. N. Campbell of the Tropical Research and Education Center, IFAS, University of Florida; Mrs. Y. Wang of the Soil and Water Science Department, IFAS, University of Florida; Dr. Z. He at the Indian River Research and Education Center, IFAS, University of Florida; and Mr. W. Perry of Everglades National Park and Dry Tortugas National Park. All of these colleagues provided invaluable assistance in sediment sampling and/or in performing the chemical analyses.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Marcel van der Perk
Rights and permissions
About this article
Cite this article
Wang, Q., Li, Y. Phosphorus adsorption and desorption behavior on sediments of different origins. J Soils Sediments 10, 1159–1173 (2010). https://doi.org/10.1007/s11368-010-0211-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-010-0211-9