Abstract
Purpose
The present study provides a review on sea-use impacts and how they are handled in life cycle assessments (LCA). It aims at defining the impact pathways for occupation and transformation impacts on marine ecosystems due to human activities (constructions, fishing, aquaculture, navigation).
Methods
First, a review was performed on human interventions leading to environmental impacts in marine areas and on additional fishery-related impact categories used in LCA of seafood, in order to identify the main methodological deficiencies existing in LCA of seafood products. Second, the sea-use impact category has been defined, by detailing the human interventions leading to impacts on the marine environment and which should be accounted for in LCA. Subsequently, the identification and description of the possible impact pathways linking activities and interventions to impact categories are carried out at endpoint and midpoint levels. This assessment has been based on a review of existing methods of land use, and suggests the use of certain indicators, which could be available for different types of marine activities and ecosystems.
Results
This study highlights the needs to account for impacts of human activities due to sea use. Additional indicators have often been added in LCA of seafood, to assess the impacts of seafloor destruction and biomass removal. By extending the scope to other activities than fisheries, many interventions lead to impacts on marine ecosystems: biomass removal and benthic construction, invasive species release, shading, artificial habitat creation, noise, turbidity, and changes in original habitat availability. The impact pathway definition and the identification of the most relevant methods for sea use highlighted the need to assess impacts on ecosystem services (life support functions, global material cycling, and detoxification of pollutants) and on biodiversity as well as biotic resource depletion.
Conclusions
A consensus for biotic resource depletion assessment still needs to be found despite recent innovative proposals. For the sea-use impact assessment, methods using species-area relationships, as well as methods focusing on ecosystem services, appear particularly relevant. In a context of strong marine resource overexploitation, and limited marine biodiversity data, the deficiencies in biomass production capability (provisioning services) could be the first stage of sea-use development.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Life cycle assessment (LCA) tends to be exhaustive for the impacts it aims to assess in order to identify options to limit pollution transfers and resource depletion. However, as identified by Munkung and Gheewala (2007), Pelletier et al. (2007), and Thrane et al. (2009), there is a need in method development to specifically assess (1) the impacts of seafood products for comparison between terrestrial and aquatic food products, (2) fish stock depletion, and (3) seafloor damage, among other objectives. These insufficiencies limit the identification capacity of LCA in a context of biotic marine resources overexploitation (FAO 2010) and of increased disturbance of marine ecosystems due to human activities (Halpern et al. 2008). These latter trends might even worsen with the growing demand of fish for food and particularly of products from aquaculture (FAO 2010). This situation could be aggravated by the rapid development of new offshore activities (e.g., wind farms, as the most used technology to date, followed by wave and tidal energy devices, expected to increase worldwide in the near future), for which environmental impact studies still need to be developed or further deepened (Inger et al. 2009).
The present work provides a review on sea-use impacts due to occupation or transformation of marine areas and on the manner in which they are handled in LCA. To this aim, Section 2 of this paper summarizes the human interventions leading to environmental impacts on the marine environment which should be accounted for in LCA, as well as the fishery-related indicators already used or proposed for LCA of seafood. In Section 3, this paper reviews the impact pathways linking human interventions due to sea-use and environmental impacts. An inventory is made of possible and relevant indicators, for every kind of marine activity and ecosystem, and the possible methods evaluating sea-use impact assessment are identified. These methods have been inspired from the land-use impact assessment methods and from the additional fishery-related indicators used for LCA of seafood previously identified.
2 Impacts due to sea use and their assessment in LCA
2.1 Human interventions leading to environmental impacts due to sea use
By analogy with land use, a sea-use impact category should account for physical impacts due to occupation or transformation of marine areas. According to the literature, the main interventions leading to impacts have been sorted into eight categories. They have been summarized in Table 1 according to human activities and sea compartments. These interferences can affect both the pelagic and the benthic zones (i.e., the water column or the seafloor, respectively). They include biomass removal, invasive species release, seafloor destruction, artificial habitat creation, shading, turbidity (and sedimentation), noise, and changes of the surface or volume of original habitat.
The emissions of pollutants and eutrophic substances into the environment due to marine activities have already been assessed through existing midpoint and endpoint impact categories and therefore have been excluded from Table 2. For instance, the damage due to greenhouse gas emissions, should typically be assessed using the pathway from the midpoint impact “climate change” category to the endpoint “ecosystems” and “human health” areas of protection, as has been performed for terrestrial ecosystems. The same applies for oceanic or abiotic resources extraction (sediments, polymetallic nodules, oil, etc.).
Biotic resources use
One of the major interventions on marine ecosystems is the biotic resources uptake caused by fishing activities (Halpern et al. 2008; Pauly et al. 2005). It affects the ecosystems by direct effects on exploited stocks, both in pelagic and benthic ecosystems (Pauly et al. 2005). Stocks are often beneath their regeneration threshold worldwide: half is fully exploited, while a quarter is being overexploited, depleted, or is recovering from depletion (FAO 2010). The overexploitation of fisheries also leads to indirect effects, with modifications along the whole food chain: a decline of the average trophic level of global landings are observed, which implies that the ecosystem is likely to increasingly rely on fish that originate from the lower part of marine food webs (FAO 2010). Resources uptake also occurs for other activities: extensive aquaculture and harvesting of seaweed imply a reduction of available nutrients, possibly leading to competition in oligotrophic areas. For offshore animal aquaculture, this uptake of nutrients for feed occurs to a certain degree according to the type of animal farmed and to the type of aquaculture: it can be high for plankton-filtering species like mussels and oysters, but close to null for intensive aquaculture of carnivore fish species kept in cages, with external feeding.
Invasive species
Invasive species are one of the main drivers of biodiversity loss (Nelson 2005). It is one of the consequences of long-distance shipping lanes, mostly due to ballast water discharge from liquid-transportation ships, as well as organisms stuck to the ships’ hulls (Halpern et al. 2008). The International Convention for the Control and Management of ship’s Ballast Water and Sediments (BWM Convention) was adopted in 2004, under the influence of the International Maritime Organization (IMO 2004). It is assumed that this Convention can considerably reduce the risks of the introduction of future ballast water-mediated species, although it is still considered not powerful enough and was only ratified by 30 countries in 2005 (Gollasch et al. 2007; IMO 2011). Unexpected escaping of non-native or genetically selected strains of cultivated species is equally an issue (Halpern et al. 2008).
Seafloor destruction
In addition to biotic resources depletion, destructive fishing (using trawls or dredges) destroys the seafloor. Kaiser et al. (2002) estimated that about 75 % of the shelf area is trawled worldwide every year. The installation of pipelines and associated structures on the seafloor can affect benthic habitats, from physical occupation to conversion of the seafloor. Pipeline laying can disturb shellfish beds, hard-bottomed habitats, and submerged aquatic vegetation (Johnson et al. 2008). Seafloor destruction also occurs when explosives are used in shallow water.
Artificial habitat creation
Marine activities can also lead to positive effects on the environment by creation of artificial habitats (Inger et al. 2009). Artificial reefs are submerged structures deliberately placed on the seabed to mimic natural reefs (Baine 2001). Life in natural reefs is more diverse with more trophic levels than artificial ones. However, artificial reefs allow for higher abundance, its intensity depending on the complexity of their structure, such as the quantity of holes contained in the reef (Hackradt et al. 2011). To a certain extent, any submerged structure can be assumed to play this role of artificial habitat creation, with a growing intensity when structures are highly convoluted. The gain of habitat will also depend on the original habitat, being higher in soft seabeds than in hard ones (Moura 2010).
Shading
Human activities can lead to shading: overwater structures create shade, which reduces the light levels beneath the structure. It can reduce prey organism abundance and the complexity of the habitat by reducing aquatic vegetation and phytoplankton abundance (Johnson et al. 2008). This occurs when any floating opaque construction, moored within the photic zone or at its surface, prevents primary production by photosynthesis (in the benthic and/or pelagic zones). This can also be an issue for aquaculture, particularly for seaweed cultivation (Roesijadi et al. 2008).
Turbidity and sedimentation
The resuspension of bottom sediments in coastal waters results from both naturally occurring (e.g., storms, waves, etc.) and anthropogenic forces (e.g., trawling, dredging, etc.). The latter have become increasingly important during recent decades (Ruffin 1998). Turbidity and sedimentation may also result from discharge of terrestrial wastes, especially soil excavation, or from storm water discharge, which can be abnormally turbid and rich in suspended-sediments, due to anthropogenic activities on land. Increased turbidity, due to suspended particles, may decrease light penetration in the water column, to the extent that submerged aquatic vegetation cannot photosynthesize anymore. The major impact that can be estimated is therefore related to shading. Other effects related to turbidity include the saturation of filter-feeding animals by particles above a certain density and size.
Noise
There are many natural sounds in the ocean, including rain, wind, marine life, ice, and seismic activity (McCarthy 2004). However, nowadays, anthropogenic noises in the sea are rising, originating from land-based activities (helicopters, airplanes), navigation, icebreaking activities, hovercraft, dredging, anchored constructions (e.g., tunnel boring, drilling, and marine explosions) and several ultra- or infra-sound emissions emanating mostly from onboard equipment (McCarthy 2004). It is still hard to quantify with precision the effects of underwater noise on marine life. It has been estimated to be low for the benthos (Pauly et al. 2005). However, studies have shown that powerful ultra-sonic or sonic emissions by long range sonars or air-gun seismic prospection can reduce growth and reproduction in a variety of marine organisms and can possibly be a cause of death for marine mammals (McCarthy 2004).
Original habitat loss
If seafloor use is maintained over a long period (constructions), the quantity of available virgin habitat decreases (although there is a creation of a new habitat, see below). This decrease can either occur for surfaces (in benthic habitats) or for volumes (in pelagic habitats), depending on the construction’s height and position. A local decrease in volume of the pelagic zone also occurs when floating constructions and any other activity using floating material in the pelagic zone (like aquaculture or floating wind power installation) have been set up.
To conclude, many disturbances in marine ecosystems exist due to human transformation and occupation of the sea. They induce negative impacts on ecosystems, but, in some cases, can also play a positive role (such as the creation of artificial habitats). These impacts can differ between the seafloor and the pelagic zone and some of them can originate from land-based activities. Amongst this set of interventions, some authors have already addressed the consequences of fisheries and aquaculture.
2.2 Current LCA indicators for seafood and products from aquaculture
Regarding LCA of aquatic products, additional fishery-related categories are often added to conventional and generic impact categories to assess some of the impacts previously reviewed (see Table 2 and Avadi and Fréon (2013) for a review). These categories mainly focus on seafloor damage and renewability of the biotic resources. They include seafloor impacts, Net Primary Production used along the whole food chain (NPPuse), bycatches and stock destructions.
Seafloor destruction
The seafloor impact of trawling and/or creeling was quantified in several studies (Ramos et al. 2011; Ziegler et al. 2011; Ziegler and Valentinsson 2008) using a methodology developed by Nilsson and Ziegler (2007) to spatially analyze demersal fishing effort data. It allowed the assessment of biological impacts on the benthic habitats due to fishing activities. The disturbance intensity was quantified according to the location of fishing activities (using GIS) and to the frequency at which the area was swept by fishing gear. These data were coupled with marine habitat recoverability data (from the MarLIN database (Marine Biological Association of the UK 2004)), mainly depending on the substratum types of the habitat. Subsequently, the impact could be quantified for different fisheries, in proportion to the habitat type affected by trawls.
In other studies only the trawled areas in square meter were quantified, without any damage assessment (Ellingsen and Aanondsen 2006; Ziegler et al. 2009, 2011). Some authors made a distinction between seafloor trawled in oxygenated areas (i.e., corresponding to benthic communities in the photic zone) and seafloor trawled in oxygen-depleted areas (i.e., mainly corresponding to colonies of sulfur and nitrogen-reducing bacteria). In their key framework, Mila i Canals et al. (2007) suggested that physical impacts of fishing should be assessed from both “natural environment” and “resource” perspectives.
Net primary production (NPP)
used by exploiting terrestrial or marine areas, humans can modify the quantity of biomass in an ecosystem (Haberl and Weisz 2007), either by:
-
(1)
Changing its biotic production potential (BPP), measured as NPP, which flows in kilogram of carbon per square meter and per year), or
-
(2)
Harvesting parts of the biomass produced, measured as NPPuse (quantities in kilogram of carbon) (Aubin et al. 2006, 2009), also named primary production required, to sustain the fishery by Libralato et al. (2008) and Pauly and Christensen (1995).
Certain authors consider for the second case the human appropriation of NPP (HANPP, kilogram of carbon per square meter and per year) to express flows instead of quantities (Haberl and Weisz 2007). This indicator is especially useful for LCA applied to aquaculture, where both terrestrial and aquatic feeds are used (Papatryphon et al. 2004). It allows the expression of direct impacts on the availability of the biotic resources and indirectly on biodiversity through food web perturbations. Biomass uptakes are mainly expressed in carbon equivalent (kilogram, Ceq), taking into account trophic levels. This approach shows the mass of carbon of the required primary biomass, including losses occurring during trophic level conversions. Although the term NPP should include respiration, it is not always clear whether authors do really consider it or merely use gross primary production. In fisheries science studies, the indirect effects of the removal of the lower trophic levels on the higher levels can be accounted for (Libralato et al. 2008): if certain herbivore fish are removed from the sea, a certain amount of carnivore fish would not develop, and so on along the whole food chain. For simplicity, LCA in this field are restricted to direct NPPuse and do not include long-term effects on ecosystem NPP levels (Pelletier et al. 2007).
Bycatches
Bycatches (including discards at sea and retained incidental catches) have also been quantified in LCA of seafood in terms of (1) kilograms of undersized catches or catches of non-commercial species, using discard mortality estimates according to fishing techniques (Ziegler and Valentinsson 2008), (2) bycatch or discard rate, which is the bycatch (or discards) to landing ratio (Ziegler et al. 2003, 2009, 2011), (3) discard rates ratio (a ratio of ratio) which is the ratio between the fishery discard rate and the worldwide discard rate (Vázquez-Rowe et al. 2012), and (4) quantity of avoided production due to discards (Ziegler et al. 2003) to account for the indirect effects of discards. According to the key framework from Mila i Canals et al. (2007), these discards and their associated mortality as well as the fishing mortality of target species should be considered, from a “resource” perspective, as a depletion of biotic resources. Quantifying direct NPPuse related to landings and discards is a first step in this depletion assessment.
Stock destruction
In a context of severe overfishing, it appears important to quantify these impacts in relation with their renewability. Some new suggestions for the Biotic Natural Resource Depletion (BNRD) assessment have been brought forth by Emanuelson et al. (2012) and Langlois et al. (2012) including midpoint impact categories for the former authors. They have been based on the Maximum Sustainable Yield (MSY) and associated values of biomass and fishing mortality, which are commonly used references for fish stock status assessment. This is the highest yield in fish production that can be sustained on a long term when stocks are exploited. It results from the assumption that fish production can be increased up to a certain level by increasing the fishing effort. Beyond the MSY level, the renewal of the resource (by reproduction and body growth) cannot keep pace with the removal caused by fishing, and a further increase in the exploitation level would lead to a reduction in landings (Graham 1935; Schaefer 1954). The estimation of fishing rates that correspond to this level has been widely debated for many years because it is based on the assumption that production in the ecosystem can reach a maximum (Larkin 1977) and because single species stock assessment methods do not seem suitable alone for a sustainable management of marine resources (Botsford et al. 1997). However, MSY and the associated reference points are still the most common values used to assess stocks and manage fisheries, even if not used by all management agencies (Ricard et al. 2012). The method proposed by Langlois et al. (2012) allows the calculation of the BNRD impact in a potential time of regeneration, whereas Emanuelson et al. (2012) suggested a quantification of the biomass that would not be produced in the future due to the current overexploitation.
Some other indicators have been proposed, with a lesser applicability in LCA for a generic purpose. To assess impacts of overfishing on the stocks, the mean size of target catch has been used by Ziegler et al. (2009 and 2011) although this indicator cannot be extended for comparisons between different species. The use of the fisheries in balance (FiB) index has also been proposed by Ramos et al. (2011). It aims at identifying the fishing down marine food webs phenomenon, which suggests that when fish species at the top of the trophic chain are overexploited, there is an increase in the capture of species lower than the trophic level (Pauly et al. 1998). It considers the total exploitation of the ecosystem by fisheries. Hence, it cannot be associated to a functional unit expressed in landings of individual species. Apart from these indicators of overfishing, local ecological impact categories have recently been proposed by Ford et al. (2012) to assess impacts of nutrient release and impacts on biodiversity. They suggest that the number of escaped fishes, the number of disease outbreaks, the parasite abundance on farms, and the percent reduction in wild fish survival could be used as indicators of biodiversity aspects. These indicators are specific to aquaculture.
To conclude, several indicators quantifying biotic production and its sustainability for both biotic-exploited resources and seafloor quality exist for the environmental assessment of seafood and products from aquaculture. These approaches are in agreement with the recommendations proposed by Mila i Canals et al. (2007), suggesting to account for discards and physical impacts of seafloor destruction from a resource and/or natural environment perspective. Nevertheless, impact pathways for marine use or biotic resource depletion have not yet been defined and cause-effect chains still need to be established.
3 Methodological perspectives from existing methods of land-use impact assessment
3.1 Review of impact pathways developed for land-use impact assessment and perspectives for sea use
For terrestrial ecosystems, the impacts of occupation and transformation of space for human activities have been assessed through the land-use impact category. A sea-use impact category has been proposed (Langlois et al. 2011), by analogy with land use, assessing transformation and occupation impacts of marine habitats. The framework for the land-use impact assessment set up by Mila i Canals et al. (2007) could be applicable to marine habitats to take into account the impacts of construction, fishing, navigation, and aquaculture, as also mentioned in Koellner et al. (2013b). At midpoint level, the amount of land used is the simplest index currently applied (Goedkoop et al. 2009; Guinée et al. 2001). It consists in considering land as an entire resource, eventually containing several categories of land (urban, agricultural, or natural) to introduce a quality difference between different types of use (Goedkoop et al. 2009).
For the Area of Protection (AoP) dealing with “natural resources” (endpoint level), depletion of soil has been proposed, combining the local soil reserves and the solar energy needed to regenerate the lost soil (Núñez et al. 2013). For the AoP dealing with “ecosystem quality,” a decrease in quality within a terrestrial ecosystem can be assessed with many ecosystem products and services being taken into account. In operational methods implemented in LCA software products, the quality index used to quantify the impacts of land use is often biodiversity, as an endpoint category (Bare 2002; Goedkoop et al. 2009; Goedkoop and Spriensma 2001). Firstly, biodiversity has a strong positive effect on the major land ecosystem services (which are a growing concern since the Millenium Ecosystem Assessment established their importance in 2005), and secondly, habitat changes, loss, or degradation are one of the main anthropogenic drivers of biodiversity loss (Millenium Ecosystem Assessment 2005). According to the key framework for terrestrial land use impact assessment from Mila i Canals et al. (2007), impacts on biodiversity and on three major ecosystem services should be integrated within land use. These three services include: (1) provisioning ecosystem services (BPP) (2) supporting, and (3) regulating services (including carbon sequestration potential, freshwater regulation potential, water purification potential, and erosion regulation potential). Apart from biodiversity, characterization factors have been calculated for other impact pathways. Soil ecological functions have been regionally assessed by Saad et al. (2011) through the GaBi software (Beck et al. 2010). Recently, erosion regulation potential and BPP have also been assessed and expressed as NPP depletion. This approach was applied regionally and at the worldwide scale by Núñez et al. (2013) and Pfister et al. (2011), respectively. Muller-Wenk and Brandao (2010) also proposed methods for carbon sequestration potential assessment. All these recent enhancements for land-use impact assessment begin to seal the robust framework defined by Mila i Canals et al. (2007), after many years of debate.
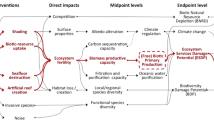
In the case of sea use, impact pathways linking the interventions described in the previous section to midpoint and endpoint levels can be summarized in the same way as has been done for land use (Fig. 1). This is stimulated by the terrestrial land-use impact pathways from Koellner et al. (2013a).
Marine ecosystem products and services are (1) global material cycling (through carbon, nitrogen, phosphorus cycles, etc.), (2) support of world fisheries and aquatic ecosystems, and (3) transformation and detoxification of pollutants (Diaz et al. 2005). It would be awkward to give priority to only one of these services. For this reason, the possibility to assess marine-use impacts through a proxy affecting all of these services, such as changes in marine biodiversity, has been considered in this paper. Other possibilities to assess alternative impact pathways (especially impacts on marine BPP functions, which are particularly altered) are also considered. Koellner et al. (2013a) suggested accounting for every ecosystem service at midpoint level, and combining them at endpoint level. Thus, impacts on (1) climate regulation, (2) biotic primary production, and (3) oceanic water purification should also be included within the sea-use impact pathways at midpoint level. They have been detailed in the following section, in addition to the assessment of the biotic natural resource depletion and the biodiversity damage potential.
3.2 Impact assessment of biotic natural resource depletion
Potentially depleted marine resources include commercial fished or harvested biomass. Their availability for future generations can be assessed under the AoP of “natural resources”. In this area of protection, only abiotic resource depletion has presently been accounted for in operational methods. In this case, it is expressed at the endpoint level as the surplus energy needed to make the resource available at some point in the future in EcoIndicator99 (Goedkoop and Spriensma 2001) and Impact2002+ (Jolliet et al. 2003) or in monetary units in ReCiPe (Goedkoop et al. 2009). There is no consensus yet for this impact assessment and only recommendations have yet been formulated (Udo de Haes et al. 2002).
From a global point of view, resource depletion is defined as “the decrease of availability of the total reserve of potential functions of resources, due to the use beyond their rate of replacement,” considering both non-renewable and renewable resources (ILCD 2010). Different options have been proposed, considering the depletion or the scarcity of resources as assumed in the definition (Guinée et al. 2001), or according to an intrinsic property as exergy suggested by Wagendorp et al. (2006) and Dewulf et al. (2007). To assess the depletion of biotic resources according to their scarcity, several methods have been identified (Heijungs et al. 1997) which account either for the size of their population (or reserve), or for a restoration time, at the species or ecosystem scale. In the field of LCA of fisheries, measures based on resource renewability, through the MSY values, have lately been proposed by Emanuelson et al. (2012) and Langlois et al. (2012) and belong to this category (see part 2.2). For the case of an intrinsic property, energy, and/or exergy could be used for the biotic resource depletion impact assessment as proposed by Bastianoni (2002), Núñez et al. (2013) in a terrestrial context.
To conclude, there is a strong need for a consensus in the field of natural resources depletion assessment, especially as this is a major concern in marine ecosystems.
3.3 Impact assessment of biodiversity damage potential
Biodiversity covers a very large perimeter, including genetic, species, and ecosystem diversity (UNEP 1993). It has been widely studied in LCA (Curran et al. 2011), either as a proxy for the area of protection “ecosystem” or, more rarely, at midpoint level (Jeanneret et al. 2008).
Biodiversity can be directly assessed in LCA at the endpoint level as “biodiversity damage potential” using a species-area relationship in operational methods (Goedkoop et al. 2009; Goedkoop and Spriensma 2001).
It has been developed in many terrestrial LCA papers for European countries (Koellner 2000, 2002; Koellner et al. 2004; Koellner and Scholz 2008; Weidema and Lindeijer 2001), South-East Asian countries (Schmidt 2008), North-American countries (Geyer et al. 2010) and more recently, at a broader scale (de Baan et al. 2013). This relationship expresses that larger areas are more likely to enclose more diverse types of habitat and therefore should include more species. Therefore, it allows the quantification of a loss of biodiversity associated with a decrease of the available surface of an ecosystem (called regional damage on species richness). It also allows the measurement of local damage on species richness, due to a change in land cover. Most of the time, it is based on species richness of vascular plants. Other taxons could be used (Koellner and Scholz 2008), but these imply data limitations. This type of assessment requires the collection of data for species-area relationship, specific from both the eco-regions and the types of use. Some studies focusing particularly on species-area relationships in marine ecosystems do exist (Neigel 2003). Nevertheless, according to the Millenium Ecosystem Assessment, to this day, marine diversity has not been sufficiently documented: indeed, at the species-scale only the diversity of the Chondrichthyan class has been studied in detail (Nelson 2005).
Other methods have also been developed. The Agroscope Reckenholz-Tänikon Research Station (ART) produced a method for the integration of biodiversity loss for agricultural production, called SALCA-Biodiversity. This is a scoring method of species damage evaluation based on expert knowledge and existing data (Jeanneret et al. 2008). It can be very useful as a complement to the LCA approach although only the agricultural stage can be analyzed. This type of scoring can also be found for fishing activities, through the SeaFood Watch, a consumer guide, provided by the Marine Stewardship Council (Wallace 2000). Indicators represent the quality of benthic habitats, the status of wild stocks for the fished species and bycatches, and the role of those species in the food chain. Their levels of disturbance are related to fishing practices and management effectiveness to obtain a final aggregated score of the ecosystem functioning. The choice of this kind of method for sea-use impact assessment would imply the calculation of characterization factors with requirements for high levels of expert knowledge. Subsequently, the method would be extended to other activities than fisheries. Another possibility may be an assessment based on genetic diversity. However, this has never been developed in LCA, and does not seem relevant for the sea-use impact assessment since it is almost unknown in marine ecosystems at a global scale (Nelson 2005).
In contrast, an assessment based on biodiversity at the ecosystem scale, expressing scarcity or vulnerability appears particularly relevant for marine ecosystems, where many activities are concentrated on coastal areas which are now considered as some of the most degraded and vulnerable ecosystems in the world (Agardy and Alder 2005). Classifications including biogeographic criteria can be used as ecosystem-level measurement of biodiversity (Costello 2009). For terrestrial ecosystems, there have been suggestions to directly express the scarcity and vulnerability of the ecosystems using this type of classification (Michelsen 2007; Michelsen et al. 2012; Weidema and Lindeijer 2001). Another possibility would be to indirectly express scarcity and vulnerability, using the species-area relationship as in ReCiPe (Goedkoop et al. 2009) and in EcoIndicator99 (Goedkoop and Spriensma 2001), because it expresses a loss of biodiversity due to a decrease in land availability. In the case of marine ecosystems, the use of province classifications would be more relevant, as species-area relationships are not well developed. It is worth noting that the boundary between biodiversity at the ecosystem scale and the area of protection “natural resources” is quite hard to define. Scarcity at the ecosystem scale could rather express natural resource depletion, as performed by Langlois et al. (2012) in the field of LCA of fisheries, where they associated primary production and ecosystem classification to quantify biomass scarcity in a given area.
3.4 Impact assessment of other ecosystem services
The framework of terrestrial land use from Mila i Canals et al. (2007) combines a spatial and a temporal dimension. It is generic and allows the assessment of all kinds of ecosystem services. It requires defining a quality index specific from the ecosystem service that is to be assessed, and whose values could be compared from one activity to another. These values might be significantly different from the state the comparison is to be made with, and may vary with time after a certain time of restoration, before reaching a new steady state.
Biomass production capability
To assess impacts of transformation and occupation on ecosystem life support functions, many possibilities exist for a quality index related to biomass production capability. In terrestrial ecosystems, the change in organic carbon in the soil relates to a range of soil properties responsible for soil resilience and fertility, making it a robust indicator of Biotic Production Potential (BPP) changes induced by land use (Brandão and Mila i Canals 2012). In marine ecosystems, life support functions are not always directly related to seafloor properties because most of NPP rather originates from the pelagic zone (Charpy-Roubaud and Sournia 1990)). Thus, an indicator describing the quality of the seafloor would not be fully relevant to assess BPP and it seems more relevant to choose an indicator in relation with the production itself.
NPP is a growth-based indicator for various life support functions (potential for food production, biochemical substance, and energy cycles). It expresses a biological production capacity, depending on the type of ecosystem (due to productivity variations with geographical characteristics, such as latitude, circulation, illumination…) and on the type of activities (e.g., shadow floating structures, degraded seafloor for coastal areas). It is calculated as the difference between total carbon fixed by photosynthesis and respiration. Several authors have suggested to use this net carbon uptake by the ecosystem as quality index for the terrestrial land use impact assessment (e.g., Blonk et al. 1997; Lindeijer 2000; Pfister et al. 2011; Weidema and Lindeijer 2001). One of the reasons for this choice is the data availability, as well as the objectivity of its measurement (Zhang et al. 2010).
Besides this indicator, another possibility to express the life support capability of the ecosystems is to measure the free Net Primary Production (fNPP). It expresses the amount of biomass remaining for nature’s functioning and life support functions. Calculation of fNPP is the subtraction of the biomass uptake from humans to the total potential of productivity in the considered area (NPP minus HANPP).
Some authors suggested using fNPP as a quality index for terrestrial land-use impact assessment (Ii et al. 2008; Lindeijer et al. 2002; Nakagawa et al. 2002). NPP mainly addresses the biomass available for human life, whereas fNPP is rather an indicator for non-human life support. As the sea is being heavily exploited for the extraction of biomass from wildlife, this indicator seems particularly relevant for the sea-use impact assessment.
Quantifying variations in NPP and HANPP within the framework developed by Mila i Canals (2007) allows the assessment of activities inducing variations in biotic production potential (shading, artificial habitat effect, benthic destruction including by some bottom gears), as well as activities related to biotic resource removal (certain aquaculture activities and fishing). It can be used for both benthic and pelagic systems, thus allowing to consider the three dimensions of the oceanic realms; this contrasts with the species-area relationship, which is more adapted to benthic communities. In this way, comparisons between terrestrial and marine ecosystem exploitation could also be easily performed, favored by a similar range of primary production in both ecosystems (Geider et al. 2001). Moreover, in marine ecosystems, biodiversity and production are related (Libralato et al. 2008), thus reinforcing the idea that the impact pathway associated to production should be a priority for further calculations of sea-use characterization factors.
The most commonly highlighted drawbacks for indicators related to NPP in the literature for terrestrial ecosystems, are that productivity is not only influenced by the ecosystem (soil) quality, but also by the use of fertilizers and pesticides, climate, management… (Weidema and Lindeijer 2001). In marine ecosystems, the addition of chemicals is only at an experimental stage because oceans are highly dispersive, and enterprises to physically modify ecosystems have been extremely limited. As management and climate can be categorized through the typology of use and of ecozones, respectively, it appears that the limitations existing in the terrestrial context do not occur for marine ecosystems. Nevertheless, contradiction with the impact category of eutrophication could be underlined: an emission of eutrophic molecules can be assessed as a negative impact from the eutrophication point of view, whereas it can be considered to be a positive one from the production point of view. Impact categories in LCA have been created in order to assess the extent of unbalance at the global scale. It is thus relevant to separately account for phenomena of eutrophication on one hand and lack of biomass in the oceans on the other hand. Another drawback for an indicator related to ecosystem production (such as fNPP) is that it would not always be able to properly reflect imbalance within the trophic food chain (“fishing down the food web”) as observed through fish catches, with a possible drift towards overfishing of the lowest trophic levels. A way to avoid this could be to take into account the indirect effects of biomass uptake (Libralato et al. 2008).
In a broader context than LCA, the use of production as an ecosystem functioning indicator is not new and has already been performed by Libralato et al. (2008). HANPP has been suggested as indicator for Materials and Energy Flow Analysis (Haberl and Weisz 2007) and as a marine footprint in several studies (Parker and Tyedmers 2011). In the context of LCA, the present review on LCA applied to seafood products shows the interest of expressing the human uptake using NPPuse. Mattila et al. (2011) have also put forward this term as one of the possible indicators of land use within the input–output method. The NPP-based methods should be regionalized, following the recommendations from a recent consensual publication, assembling many authors, and mentioning the need for a regionalization of land use impacts (Koellner et al. 2013b). The further development of a sea use category should account for these recommendations as well, in order to be compatible with the future improvements of land-use impact assessment.
Carbon sequestration potential and filtration and purification potential
Impacts on marine ecosystem fertility imply consequences on the free biotic primary production, but also on other ecosystem services: carbon sequestration potential, and filtration and purification potential. Marine carbon sequestration potential can be interpreted at midpoint level as climate regulation. Firstly, organic carbon is sequestrated by biomass (Falkowski et al. 1998). Nevertheless, this carbon sequestration can be compared to the one of the less than 10-year-old agricultural products. BSI (2011) recommended not to consider this sequestration because of its short-time scale. Secondly, NPP strongly influences the accumulation of organic carbon in sediments (Calvert 1987). For the conversion of carbon into carbonates, the relationship is not a direct one (carbonate dissolution or decrease in its rate of formation are mainly due to acidification). Climate regulation is also ensured by the albedo, which can vary with the coverage of the sea surface (in relation with the intervention of shading).
Living organisms also perform water purification (Diaz et al. 2005). The marine microbial community has a critical role in detoxification—filtering water, reducing effects of eutrophication, and degrading toxic hydrocarbons. All life in the ocean being directly or indirectly dependent on the intensity of primary production (except for anecdotic cases of chemosynthesis), water purification could be positively linked to marine primary production.
4 Conclusions
This study highlights the need to account for impacts of human activities due to sea use and offers some perspectives in this respect. Additional indicators have often been added in the LCA of seafood to assess impacts of seafloor destruction and biomass removal. Extending the scope to other activities than fisheries, it appears that many human interferences lead to impacts on marine ecosystems: biomass removal, invasive species release, seafloor destruction, artificial habitat creation, shading, increase in turbidity, sedimentation and noise emissions, and changes in original habitat availability. The impact pathway definition and the identification of the most relevant methods for sea use highlight the need to assess impacts on ecosystem services and on biodiversity, at the midpoint and endpoint levels respectively, as well as biotic resource depletion.
A consensus on the manner to account for biotic resource depletion still needs to be found, despite recent innovative suggestions on this topic At endpoint level, the methodology developed by Koellner and Scholz (2008) using a species-area relationship appears particularly relevant in the case of sea use, especially because it is widely used for land-use impact assessment. Only a lack of data could limit the development of characterization factors using this method. At midpoint level, the method developed by Mila i Canals et al. (2007) to assess the impacts of transformation and occupation on ecosystem services is the most consensual one (ILCD 2011). In a context of strong overexploitation of the marine resources, it appears particularly relevant to express the deficiencies in available biomass for ecosystem functioning (provisioning services). Data on marine biodiversity being scarce, the methodology developed by Mila i Canals et al. (2007) applied to provisioning services presently appears to be the most convenient in a first approach to assess impacts of sea use. It would be particularly efficient for quantifying the benefits of direct consumption of marine animal proteins in comparison to products from aquaculture. Finally, measurement of free BPP in the oceans in a context of overexploitation seems particularly relevant.
References
Agardy T, Alder J (2005) Coastal systems. In: Ecosystems and human well-being: current state and trends. pp 513–549. http://www.maweb.org/documents/document.288.aspx.pdf. Accessed 25 Apr 2012.
Aubin J, Papatryphon E, Van der Werf HMG et al (2006) Characterisation of the environmental impact of a turbot (Scophthalmus maximus) re-circulating production system using life cycle assessment. Aquaculture 261(4):1259–1268. doi:10.1016/j.aquaculture.2006.09.008
Aubin J, Papatryphon E, Van der Werf HMG, Chatzifotis S (2009) Assessment of the environmental impact of carnivorous finfish production systems using life cycle assessment. J Clean Prod 17(3):354–361. doi:10.1016/j.jclepro.2008.08.008
Avadi A, Fréon P (2013) Life cycle assessment of fisheries: a review for fisheries scientists and managers. Fish Res 143:21–38. doi:10.1016/j.fishres.2013.01.006
Baine M (2001) Artificial reefs: a review of their design, application, management and performance. Ocean Coast Manage 44:241–259. doi:10.1016/S0964-5691(01)00048-5
Bare JC (2002) Traci. J Ind Ecol 6:49–78. doi:10.1162/108819802766269539
Bastianoni S (2002) Use of thermodynamic orientors to assess the efficiency of ecosystems: a case study in the Lagoon of Venice. TheScientificWorldJOURNAL 2:255–260. doi:10.1100/tsw.2002.88
Beck T, Bos U, Wittstock B et al. (2010) LANCA® Land use indicator value calculation in life cycle assessment—method report. pp 67. http://www.lbp-gabi.de/files/lanca_website.pdf. Accessed 28 Mar 2011
Blonk H, Lindeijer E, Broers J (1997) Towards a methodology for taking physical degradation of ecosystems into account in LCA. Int J Life Cycle Assess 2:91–98. doi:10.1007/BF02978766
Bosma R, Anh PT, Potting J (2011) Life cycle assessment of intensive striped catfish farming in the Mekong Delta for screening hotspots as input to environmental policy and research agenda. Int J Life Cycle Assess 16:903–915. doi:10.1007/s11367-011-0324-4
Botsford LW, Castilla JC, Peterson CH (1997) The management of fisheries and marine ecosystems. Science 277:509–515. doi:10.1126/science.277.5325.509
Brandão M, Mila i Canals L (2012) Global characterisation factors to assess land use impacts on biotic production. Int J Life Cycle Assess 1–10. doi: 10.1007/s11367-012-0381-3
BSI (2011) Specification for the assessment of the life cycle greenhouse gas emissions of goods and services. pp 38. http://www.bsigroup.com/upload/Standards%20&%20Publications/Energy/PAS2050.pdf Accessed 7 August 2012
Calvert SE (1987) Oceanographic controls on the accumulation of organic matter in marine sediments. Geol Soc, Lond, Spec Publ 26:137–151. doi:10.1144/GSL.SP.1987.026.01.08
Charpy-Roubaud C, Sournia A (1990) The comparative estimation of phytoplanktonic, microphytobenthic and macrophytobenthic primary production in the oceans. Mar Microb Food Webs 4:31–57
Costello M (2009) Distinguishing marine habitat classification concepts for ecological data management. Mar Ecol-Prog Ser 397:253–268. doi:10.3354/meps08317
Curran M, De Baan L, De Schryver A et al (2011) Toward meaningful end points of biodiversity in life cycle assessment. Environ Sci Technol 45:70–79. doi:10.1021/es101444k
de Baan L, Alkemade R, Koellner T (2013) Land use impacts on biodiversity in LCA : a global approach. Int J Life Cycle Assess 18(6):1216–1230. doi:10.1007/s11367-012-0412-0
Dewulf J, Bösch ME, Meester BD et al (2007) Cumulative Exergy Extraction from the Natural Environment (CEENE): a comprehensive life cycle impact assessment method for resource accounting. Environ Sci Technol 41:8477–8483. doi:10.1021/es0711415
Diaz S, Tilman D, Fargione L (2005) Biodiversity regulation of ecosystem services. In: Ecosystems and human well-being: current state and trends. pp 297–329. http://www.millenniumassessment.org/documents/document.280.aspx.pdf. Accessed 08 Aug 2010.
Efole Ewoukem T, Aubin J, Mikolasek O et al (2012) Environmental impacts of farms integrating aquaculture and agriculture in Cameroon. J Clean Prod 28:208–214. doi:10.1016/j.jclepro.2011.11.039
Ellingsen H, Aanondsen A (2006) Environmental impacts of wild caught cod and farmed salmon—a comparison with chicken. Int J Life Cycle Assess 11:60–65. doi:10.1065/lca2006.01.236
Emanuelson A, Ziegler F, Pihl L et al. (2012) Overfishing, overfishedness and wasted potential yield: new impact categories for biotic resources in LCA. In; 8th International Conference on LCA Food 2012 - Life Cycle Assessment in the Agri-Food Sector. Saint-Malo (France), pp 511–516
Emanuelsson A, Flysjö A, Thrane M, Ndiaye V, Eichelsheim JL, Ziegler F (2008) Life cycle assessment of southern pink shrimp products from Senegal. In: In: 6th International Conference on Life Cycle Assessment in the Agri-Food Sector, Zurich., pp 1–9
Falkowski PG, Barber RT, Smetacek V (1998) Biogeochemical controls and feedbacks on ocean primary production. Science 281:200–206. doi:10.1126/science.281.5374.200
FAO (2010) Part 1: World review of fisheries and aquaculture. In: State of world fisheries and aquaculture, 2010. FAO, Rome, Italy, pp 3–89
Ford JS, Pelletier NL, Ziegler F et al (2012) Proposed local ecological impact categories and indicators for life cycle assessment of aquaculture. J Ind Ecol 16:254–265. doi:10.1111/j.1530-9290.2011.00410.x
Geider RJ, Delucia EH, Falkowski PG et al (2001) Primary productivity of planet earth: biological determinants and physical constraints in terrestrial and aquatic habitats. Glob Change Biol 7:849–882. doi:10.1046/j.1365-2486.2001.00448.x
Geyer R, Stoms DM, Lindner JP et al (2010) Coupling GIS and LCA for biodiversity assessments of land use. Int J Life Cycle Assess 15:454–467. doi:10.1007/s11367-010-0170-9
Goedkoop M, Spriensma R (2001) The Eco-Indicator 99. A damage oriented method for life cycle impact assessment. Methodology Report. PRé Consultants, Amersfoort, pp 132
Goedkoop M, Heijungs R, Huijbregts M et al. (2009) ReCiPe 2008, a life cycle impact assessment method which comprises harmonised category indicators at the midpoint and the endpoint level; first edition Report I. Den Haag, pp125
Gollasch S, David M, Voigt M et al (2007) Critical review of the IMO international convention on the management of ships’ ballast water and sediments. Harmful Algae 6:585–600. doi:10.1016/j.hal.2006.12.009
Graham M (1935) Modern theory of exploiting a fishery, and application to North Sea trawling. ICES J Mar Sci 10:264–274. doi:10.1093/icesjms/10.3.264
Guinée J, Gorrée M, Heijungs R et al. (2001) Life cycle assessment—An operational guide to the ISO standards. p11. http://media.leidenuniv.nl/legacy/new-dutch-lca-guide-part-1.pdf. Accessed 30 March 2011
Haberl H, Weisz H (2007) The potential use of the Materials and Energy Flow Analysis (MEFA) framework to evaluate the environmental costs of agricultural production systems and possible applications to aquaculture. In: Comparative assessment of the environment costs of aquaculture and other food production sectors: methods of meaningful comparisons. FAO, Rome (Italy), pp 97–119
Hackradt CW, Félix-Hackradt FC, García-Charton JA (2011) Influence of habitat structure on fish assemblage of an artificial reef in southern Brazil. Mar Environ Res 72:235–247. doi:10.1016/j.marenvres.2011.09.006
Halpern BS, Walbridge S, Selkoe KA et al (2008) A global map of human impact on marine ecosystems. Science 319:948–952. doi:10.1126/science.1149345
Heijungs R, Guinée J, Huppes G (1997) Impact categories for natural resources and land use. CML report 138. https://openaccess.leidenuniv.nl/dspace/bitstream/1887/8070/1/11_500_002.pdf. Accessed 10 August 2010
Ii R, Yamaguchi K, Okada A et al. (2008) Land use damage assessment and its application on resource extraction and waste landfill impact categories in LIME2. New Energy and Industrial Technology Development Organization of Japan. http://www.pacific.co.jp/thesis/item/environment_59.pdf. Accessed 02 February 2011
ILCD (2010) International Reference Life Cycle Data System (ILCD) Handbook - Framework and requirements for LCIA models and indicators. European Commission - Joint Research Centre - Institute for Environment and Sustainability, Luxembourg. lct.jrc.ec.europa.eu/…/ILCD34 Handbook-LCIA-Framework-requirements-online-12March2010.pdf. Accessed 07 July 2012.
ILCD (2011) Recommendations for life cycle impact assessment in the European context. pp 143. http://lct.jrc.ec.europa.eu/pdf-directory/Recommendation-of-methods-for-LCIA-def.pdf. Accessed 29 October 2012.
IMO (2004) International convention for the control and management of ship’s ballast water and sediments. http://www.imo.org. Accessed 23 Nov 2011.
IMO (2011) Status of Conventions summary. http://www.imo.org/About/Conventions/StatusOfConventions/Pages/Default.aspx. Accessed 23 November 2011.
Inger R, Attrill MJ, Bearhop S et al (2009) Marine renewable energy: potential benefits to biodiversity? An urgent call for research. J Appl Ecol 46:1145–1153. doi:10.1111/j.1365- 2664.2009.01697.x
Jeanneret P, Baumgartner DU, Knuchel RF, Gaillard G (2008) A new LCIA method for assessing impacts of agricultural activities on biodiversity (SALCA-Biodiversity). In: Life Cycle Assessment in the Agri-Food Sector. Thomas Nemecek and Gérard Gaillard, Zurich, Switzerland, pp 34–39
Jerbi MA, Aubin J, Garnaoui K et al (2011) Life cycle assessment (LCA) of two rearing techniques of sea bass (Dicentrarchus labrax). Aquacult Eng 46:1–9. doi:10.1016/j.aquaeng.2011.10.001
Johnson M, Boelke C, Chiarella L, et al. (2008) Impacts to marine fisheries habitat from nonfishing activities in the Northeastern United States. pp 322. http://www.fpir.noaa.gov/Library/HCD/NOAA%20Technical%20Memo%20NMFS-NE-209.pdf. Accessed 19 Oct 2012
Jolliet O, Margni M, Charles R et al (2003) IMPACT 2002+: A new life cycle impact assessment methodology. Int J Life Cycle Assess 8:324–330. doi:10.1007/BF02978505
Kaiser MJ, Collie JS, Hall SJ et al (2002) Modification of marine habitats by trawling activities: prognosis and solutions. Fish Fisheries 3:114–136. doi:10.1046/j.1467-2979.2002.00079.x
Koellner T (2000) Species-pool effect potentials (SPEP) as a yardstick to evaluate land-use impacts on biodiversity. J Clean Prod 8:293–311. doi:10.1016/S0959-6526(00)00026-3
Koellner T (2002) Land use in product life cycles and its consequences for ecosystem quality. Int J Life Cycle Assess 7:130–130. doi:10.1007/BF02978857
Koellner T, Scholz R (2008) Assessment of land use impacts on the natural environment. Part 2: generic characterization factors for local species diversity in Central Europe. Int J Life Cycle Assess 13:32–48. doi:10.1065/lca2006.12.292.2
Koellner T, Hersperger AM, Wohlgemuth T (2004) Rarefaction method for assessing plant species diversity on a regional scale. Ecography 27:532–544. doi:10.1111/j.0906-7590.2004.03832.x
Koellner T, De Baan L, Beck T et al (2013a) UNEP-SETAC guideline on global land use impact assessment on biodiversity and ecosystem services in LCA. Int J Life Cycle Assess 18:1188–1202. doi:10.1007/s11367-013-0579-z
Koellner T, De Baan L, Beck T et al (2013b) Principles for life cycle inventories of land use on a global scale. Int J Life Cycle Assess 18:1203–1215. doi:10.1007/s11367-012-0392-0
Langlois J, Hélias A, Delgenes JP, Steyer JP (2011) Review on land use considerations in life cycle assessment: methodological perspectives for marine ecosystems, Towards Life Cycle Sustainability Management. Finkbeiner, M., Berlin, pp 85–96
Langlois J, Fréon P, Delgenes JP et al. (2012) Biotic resources extraction impact assessment in LCA of fisheries. In: 8th International Conference on Life Cycle Assessment in the Agri-Food Sector. Saint-Malo (France), pp 517–522
Larkin PA (1977) An epitaph for the concept of maximum sustained yield. T Am Fish Soc 106:1–11. doi:10.1577/1548-8659(1977)106<1:AEFTCO>2.0.CO;2
Libralato CM, Tudela S et al (2008) Novel index for quantification of ecosystem effects of fishing as removal of secondary production. Mar Ecol-Prog Ser 355:107–129. doi:10.3354/meps07224
Lindeijer E (2000) Biodiversity and life support impacts of land use in LCA. J Clean Prod 8:313–319. doi:10.1016/S0959-6526(00)00025-1
Lindeijer E, Kok I, Eggels P, Alfers A (2002) Improving and testing a land use methodology in LCA. Including case-studies on bricks, concrete and wood. pp 131. http://en.scientificcommons.org/18266238. Accessed 01 Sept 2010
Marine Biological Association of the United Kingdom (2004) Marine Life 1 Information network: biology and sensitivity key information sub-programme. http://www.marlin.ac.uk/. Accessed 25 Oct 2012
Mattila T, Seppälä J, Nissinen A, Mäenpää I (2011) Land use impacts of industries and products in the Finnish economy: a comparison of three indicators. Biomass Bioenerg 35:4781–4787. doi:10.1016/j.biombioe.2011.02.052
McCarthy E (2004) International regulation of underwater sound: establishing rules and standards to address ocean noise pollution. Springer, pp 287
Michelsen O (2007) Assessment of land use impact on biodiversity. Int J Life Cycle Assess 13:22–31. doi:10.1065/lca2007.04.316
Michelsen O, Cherubini F, Stromman A (2012) Impact assessment of biodiversity and carbon pools from land use and land use changes in LCA, exemplified with forestry operations in Norway. J Ind Ecol 16:231–242. doi:10.1111/j.1530-9290.2011.00409.x
Mila i Canals L, Bauer C, Depestele J et al (2007) Key elements in a framework for land use impact assessment within LCA. Int J Life Cycle Assess 12:5–15. doi:10.1065/lca2006.05.250
Millennium Ecosystem Assessment (2005) Ecosystems and human well-being: biodiversity synthesis. World Resources Institute, Washington, DC. pp 86. http://www.millenniumassessment.org/documents/document.354.aspx.pdf. Accessed 25 April 2012
Moura ACB (2010) Experimental study of the macrobenthic colonisation and secondary production in the artificial reefs of Algarve coast. Universidade do Algarve
Muller-Wenk R, Brandao M (2010) Climatic impact of land use in LCA-carbon transfers between vegetation/soil and air. Int J Life Cycle Assess 15:172–182. doi:10.1007/s11367-009-0144-y
Munkung R, Gheewala SH (2007) Use of Life Cycle Assessment (LCA) to compare the environmental impacts of aquaculture and agri-food products. In: Comparative assessment of the environment costs of aquaculture and other food production sectors: methods of meaningful comparisons. FAO, Rome (Italy), pp 87–96
Nakagawa A, Ii R, Abe K et al (2002) Development of life-cycle impact assessment method for land use. Construction of the framework of the method and calculation of the damage factors by NPP. EnvironSyst Res 30:109–118
Neigel JE (2003) Species-area relationships and marine conservation. Ecol Appl 13:138–145. doi:10.1890/1051-0761(2003)013[0138:SARAMC]2.0.CO;2
Nelson GC (2005) Biodiversity. In: ecosystems and human well-being: current state and trends. Rashid Hassan, Robert Scholes, Neville Ash, Washington, Covelo, London, pp 77–122.
Nilsson P, Ziegler F (2007) Spatial distribution of fishing effort in relation to seafloor habitats in the Kattegat, a GIS analysis. Aquat Conserv Mar Freshwat Ecosyst 17:421–440. doi:10.1002/aqc.792
Núñez M, Antón A, Muñoz P, Rieradevall J (2013) Inclusion of soil erosion impacts in LCA on a global scale: application to energy crops in Spain. Int J Life Cycle Assess 18(4):755–767. doi:10.1007/s11367-012-0525-5
Papatryphon E, Petit J, Kaushik SJ, Van der Werf HMG (2004) Environmental 1 impact assessment of salmonid feeds using life cycle assessment (LCA). Ambio 33:316–323. doi:10.1579/0044-7447-33.6.316
Parker RWR, Tyedmers PH (2011) Uncertainty and natural variability in the ecological footprint of fisheries: a case study of reduction fisheries for meal and oil. Ecol Indic. doi:10.1016/j.ecolind.2011.06.015
Pauly D, Christensen V (1995) Primary production required to sustain global fisheries. Nature 374:255–257. doi:10.1038/374255a0
Pauly D, Christensen V, Dalsgaard J et al (1998) Fishing down marine food webs. Science 279:860–863. doi:10.1126/science.279.5352.860
Pauly D, Bakun A, Authors C, Christensen V (2005) Marine fisheries systems. in: ecosystems and human well-being: current state and trends. pp 477–511. http://www.millenniumassessment.org/documents/document.287.aspx.pdf. Accessed 25 Aug 2010.
Pelletier NL, Ayer NW, Tyedmers PH et al (2007) Impact categories for life cycle assessment research of seafood production systems: review and prospectus. Int J Life Cycle Assess 12:414–421. doi:10.1065/lca2006.09.275
Pfister S, Bayer P, Koehler A, Hellweg S (2011) Environmental impacts of water use in global crop production: hotspots and trade-offs with land use. Environ Sci Technol 45:5761–5768. doi:10.1021/es1041755
Ramos S, Vázquez-Rowe I, Artetxe I et al (2011) Environmental assessment of the Atlantic mackerel (Scomber scombrus) season in the Basque Country. Increasing the timeline delimitation in fishery LCA studies. Int J Life Cycle Assess 16:599–610. doi:10.1007/s11367-011-0304-8
Ricard D, Minto C, Jensen OP, Baum JK (2012) Examining the knowledge base and status of commercially exploited marine species with the RAM legacy stock assessment database. Fish Fish 13(4):380–38. doi:10.1111/j.1467-2979.2011.00435.x
Roesijadi G, Copping AE, Husemann MH et al. (2008) Techno-economic feasibility analysis of offshore seaweed framing for bioenergy and biobased products. pp 115. http://www.scribd.com/doc/16595766/Seaweed-Feasibility-Final-Report. Accessed 19 Oct 2012
Ruffin KK (1998) The persistence of anthropogenic turbidity plumes in a shallow water estuary. Estuar Coast Shelf S 47:579–592. doi:10.1006/ecss.1998.0366
Saad R, Margni M, Koellner T et al (2011) Assessment of land use impacts on soil ecological functions: development of spatially differentiated characterization factors within a Canadian context. Int J Life Cycle Assess 16:198–211. doi:10.1007/s11367-011-0258-x
Schaefer MB (1954) Some aspects of the dynamics of populations important to the management of the commercial marine fisheries. Bull IATTC 1:27–56. doi:10.1139/f57-025
Schmidt JH (2008) Development of LCIA characterisation factors for land use impacts on biodiversity. J Clean Prod 16:1929–1942. doi:10.1016/j.jclepro.2008.01.004
Thrane M (2004) Environmental impacts from Danish Fish Products—hot spots and environmental policies. Aalborg University, Denmark, PhD Dissertation
Thrane M (2006) LCA of Danish fish products. New methods and insights. Int J Life Cycle Assess 11(1):66–74
Thrane M, Ziegler F, Sonesson U (2009) Eco-labelling of wild-caught seafood products. J Clean Prod 17:416–423. doi:10.1016/j.jclepro.2008.08.007
Udo de Haes H, Finnveden G, Goedkoop M et al. (2002) Life-cycle impact assessment: striving towards best practice. Society of Environment Toxicology and Chemistry (SETAC), pp 249
UNEP (1993) Text of the convention on biological diversity. United Nations, Rio de janeiro.http://www.cbd.int/convention/text/. Accessed 05 Apr 2011
Vázquez-Rowe I, Moreira MT, Feijoo G (2012) Inclusion of discard assessment indicators in fisheries life cycle assessment studies. Expanding the use of fishery-specific impact categories. Int J Life Cycle Assess 17(5):535–549
Wagendorp T, Gulinck H, Coppin P, Muys B (2006) Land use impact evaluation 1 in life cycle assessment based on ecosystem thermodynamics. Energy 31:112–125. doi:10.1016/j.energy.2005.01.002
Wallace S (2000) Seafood watch. http://www.montereybayaquarium.org/cr/cr_seafoodwatch/sfw_aboutsfw.aspx?c=ln. Accessed 14 Dec 2011
Weidema BP, Lindeijer E (2001) Physical impacts of land use in product life cycle assessment. pp 52.http://www.lca-net.com/files/gaps9.pdf. Accessed 01 Sept 2010
Zhang Y, Singh S, Bakshi BR (2010) Accounting for ecosystem services in life cycle assessment, Part I: a critical review. Environ Sci Technol 44:2232–2242. doi:10.1021/es9021156
Ziegler F, Valentinsson D (2008) Environmental life cycle assessment of Norway lobster (Nephrops norvegicus) caught along the Swedish west coast by creels and conventional trawls—LCA methodology with case study. Int J Life Cycle Assess 13:487–497. doi:10.1007/s11367-008- 0024-x
Ziegler F, Nilsson P, Mattsson B, Walther Y (2003) Life cycle assessment of frozen cod fillets including fishery-specific environmental impacts. Int J Life Cycle Assess 8:39–47. doi:10.1007/BF02978747
Ziegler F, Eichelsheim JL, Emanuelsson A et al (2009) Life cycle assessment of southern pink shrimp products from Senegal. FAO, Rome (Italy)
Ziegler F, Emanuelsson A, Eichelsheim JL et al (2011) Extended life cycle assessment of southern pink shrimp products originating in Senegalese artisanal and industrial fisheries for export to Europe. J Ind Ecol 15:527–538. doi:10.1111/j.1530-9290.2011.00344.x
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Rainer Zah
Rights and permissions
About this article
Cite this article
Langlois, J., Fréon, P., Steyer, JP. et al. Sea-use impact category in life cycle assessment: state of the art and perspectives. Int J Life Cycle Assess 19, 994–1006 (2014). https://doi.org/10.1007/s11367-014-0700-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11367-014-0700-y