Abstract
Speed-of-processing abilities decline with age yet are important in performing instrumental activities of daily living. The useful field of view, or Double Decision task, assesses speed-of-processing and divided attention. Performance on this task is related to attention, executive functioning, and visual processing abilities in older adults, and poorer performance predicts more motor vehicle accidents in the elderly. Cognitive training in this task reduces risk of dementia. Structural and functional neural correlates of this task suggest that higher-order resting state networks may be associated with performance on the Double Decision task, although this has never been explored. This study aimed to assess the association of within-network connectivity of the default mode network, dorsal attention network, frontoparietal control network, and cingulo-opercular network with Double Decision task performance, and subcomponents of this task in a sample of 267 healthy older adults. Multiple linear regressions showed that connectivity of the cingulo-opercular network is associated with visual speed-of-processing and divided attention subcomponents of the Double Decision task. Cingulo-opercular network and frontoparietal control network connectivity is associated with Double Decision task performance. Stronger connectivity is related to better performance in all cases. These findings confirm the unique role of the cingulo-opercular network in visual attention and sustained divided attention. Frontoparietal control network connectivity, in addition to cingulo-opercular network connectivity, is related to Double Decision task performance, a task implicated in reduced dementia risk. Future research should explore the role these higher-order networks play in reduced dementia risk after cognitive intervention using the Double Decision task.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The cognitive trajectory of healthy aging has been well observed and documented to reveal a prototypical pattern of cognitive decline [1,2,3]. Speed-of-processing abilities are particularly vulnerable to age-related cognitive decline and may underlie cognitive performance in other domains sensitive to aging such as working memory and reasoning [4,5,6,7]. These cognitive processes are necessary to maintain instrumental activities of daily living in aging [8, 9]. Therefore, it is important to accurately measure speed-of-processing abilities in aging. The useful field of view (UFOV) task is comprised of hierarchical subtests assessing an individual’s ability to quickly process visual information while also using divided and selective attention [10,11,12]. Ultimately, the task asks an individual to correctly discriminate between 2 objects in the center of the visual field, while also attending to the correct location of a target object among distractor objects in the periphery. Task difficulty is manipulated by shortening presentation times of stimuli and increasing the number of distractors. Performance on the UFOV test in aging is related to performance in attention, executive functioning, memory, and visual processing [12]. Importantly, performance on the UFOV test is also predictive of the number of motor vehicle accidents in older adults, and UFOV performance on subtests 2 and 3 has been related to timed and untimed tasks of daily living in older adults [10, 13, 14].
The UFOV test was adapted and made commercially available by POSIT Science Brain HQ (www.brainhq.com), titled the Double Decision task. The Double Decision task is a redesigned and commercialized version of the UFOV test that incorporates crucial elements of the classic UFOV test into one assessment. Therefore, performance on the Double Decision task directly measures UFOV test performance. UFOV performance will henceforth be referred to as “Double Decision” performance, including when referencing previous literature where “UFOV” term was used.
Given the Double Decision task’s robust relationship with cognitive domains in aging and daily functional abilities, it has served as a prime target for cognitive interventions aimed at altering the trajectory of cognitive decline. The Advanced Cognitive Training for Independent and Vital Elderly (ACTIVE) study was one of the first of its kind to investigate the longitudinal efficacy of 6 weeks of cognitive training intervention in a large (n = 2,832) sample of older adults [15, 16]. Pivotal findings from this intervention trial showed that Double Decision task training was associated with improved speed-of-processing and reduced risk of dementia at a 10-year follow-up [16, 17]. Additional findings from the ACTIVE intervention study revealed reduced medical expenditures for individuals who underwent speed-of-processing training at 1- and 5-year follow-up and better physical functioning at 5-year follow-up [18, 19]. Despite clear evidence of cognitive and functional abilities associated with Double Decision task performance, neural correlates of Double Decision task performance have not been well studied.
A recent study exploring structural neural correlates of Double Decision task performance found that in a sample of older adults, poorer Double Decision task performance was associated with reduced cortical thickness in multiple cortical areas, including the posterior frontal lobe, right temporal lobe, and superior and inferior parietal gyri [20]. Another study exploring structural brain correlates found cortical thickness in dorsolateral prefrontal cortex, intraparietal sulcus, precuneus, and inferior parietal lobes to be related to Double Decision task performance [21]. Only one study has explored resting state functional connectivity associated with Double Decision training, and found increased connectivity of brain areas associated with executive functioning and visual processing after training [22]. These structural and functional correlates of Double Decision task performance and training overlap with functional brain networks related to aging.
Yeo and colleagues [23] described 7 distinct cerebral resting state networks (RSNs), or anatomically distinct brain regions with correlated fluctuations of blood-oxygen-level-dependent (BOLD) functional magnetic resonance imaging (fMRI) signal when at rest. From these networks, the default mode network (DMN), dorsal attention network (DAN), frontoparietal control network (FPCN), and cingulo-opercular network (CON) have been termed “high-order” RSNs due to their association with advanced cognitive processes and are linked to cognitive changes in aging [24, 25]. The DMN is involved in self-referential processing and episodic memory retrieval and has garnered much attention in aging research due to the reliable connectivity changes in pathological and non-pathological aging and diminished cognitive functioning [26,27,28,29]. The FPCN, CON, and DAN are also related to higher-order cognitive functions such as mediating attention and correlation between other networks, and have also been shown to decline with age [25, 30, 31]. For example, Grady and colleagues [30] found reduced FPCN between-network connectivity in older age and stronger FPCN between-network connectivity relating to better memory performance, regardless of age. This suggests that FPCN connectivity may be indicative of cognitive control needed in memory formation. Additionally, findings from Hausman and colleagues [25] demonstrated that stronger CON connectivity is related to better NIH Toolbox fluid cognition scores, specifically episodic memory, attention, and executive function tasks in an aging population.
Structural and functional neural correlates of Double Decision performance overlap with brain regions involved in these RSNs. For example, cortical thinning of brain areas in FPCN (dorsolateral prefrontal cortex), DAN (intraparietal sulcus), and DMN (precuneus and inferior parietal lobes) is associated with Double Decision task performance [21]. Moreover, resting state functional connectivity increased in brain areas that comprise the CON (between the anterior insula and anterior cingulate cortex) and FPCN (between the dorsal lateral prefrontal cortex and somatosensory cortex) after Double Decision training [22]. These findings suggest that RSN connectivity may be associated with Double Decision task performance and that this association may be specific to certain higher-order RSNs, although no study has explored RSN connectivity associated with Double Decision task performance. Studying the association of Double Decision task performance with higher-order RSNs may reveal RSNs key to reducing risk of dementia, subsequently illuminating neural targets to augment cognitive interventions (i.e., through non-invasive brain stimulation). Additionally, it is imperative in furthering our understanding of RSNs involved in crucial cognitive functions of healthy aging.
The Double Decision task fundamentally requires visual perceptual abilities and divided attention. Recent research suggests that connectivity of the CON is indicative of visual processing speed abilities and its connectivity may predict visual processing gains after an alertness training in healthy older adults [32, 33]. Divided attention abilities may play a role in episodic memory encoding performance in older adults, and may predict amount of falls later in life [34, 35]. Divided attention performance has not been well characterized in higher-order RSNs in a healthy aging human population, although task-based functional MRI and rodent studies show recruitment of the prefrontal cortex and thalamus [36, 37].
These processes are fundamental components that may contribute to the efficacy of the Double Decision task. By exploring the association of higher-order RSN connectivity in speeded visual perception and Divided Attention tasks alone, we may better understand if the connectivity of one central RSN is crucial across these cognitive processes, or if the integration of these two components into one task (Double Decision task) elicits a unique pattern of association within higher-order resting state networks. These findings may also suggest specific RSN connectivity that is important in reduced dementia risk.
We therefore assessed the relationship of speeded visual perception, divided attention, and the Double Decision task performance in four higher-order RSNs (DMN, FPCN, CON, and DAN) in a large sample of healthy older adults. We predicted that visual perception and divided attention fundamental tasks will have a targeted association with CON, as this RSN is involved in attention and alertness [38, 39]. Then, due to Double Decision’s complexity and combination of visual perception and divided attention, we predicted that additional RSNs involved in decision-making and cognitive control would be recruited (FPCN and DAN), consistent with areas of increased functional connectivity after Double Decision training [22]. Additionally, we hypothesized that Double Decision task performance would not be associated with the DMN connectivity, despite DMN connectivity being associated with progression to Alzheimer’s dementia [40]. While functional connectivity of brain areas included in the DMN increased after Double Decision training, we hypothesize that Double Decision performance at a baseline timepoint would not associate with DMN connectivity due to the DMN being a “task-negative” network [22].
Materials and methods
Participants
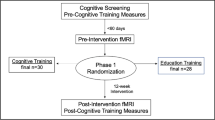
Participants in this study were part of an ongoing National Institute on Aging-funded Phase III double-blinded randomized clinical trial, Augmenting Cognitive Training (ACT; (NCT028511) [41]. Participants were recruited at the University of Florida and the University of Arizona via local research registries, community outreach, community agencies, newspaper advertisements, public service announcements, mailings, and posted flyers. Eligible participants needed to be right-handed and within the age range of 65–89; have no history of neurological disorders (including brain/head injury and dementia), major psychiatric illnesses, or formal diagnosis of cognitive impairment; and needed to have no contraindications to MRI. More details regarding inclusion/exclusion criteria may be found in Woods et al. [41]. Cognitive status was screened by performance on a comprehensive neuropsychological battery. Participants were not eligible if they performed 1.5 standard deviations below normative scores on the National Alzheimer’s Coordinating Center (NACC) Uniform Data Set (UDS-III) in any cognitive domain of general cognition, memory, visuospatial functioning, executive functioning/working memory, or language [42]. All participants provided written informed consent approved by the Institutional Review Boards at the University of Arizona and the University of Florida and research was carried out in accordance with institutional guidelines and the Declaration of Helsinki. Double Decision task performance and functional magnetic resonance imaging (fMRI) were performed within 60 days of each other.
POSIT science BRAIN HQ outcome measures
The Hawk Eye task, Divided Attention task, and Double Decision task were administered as part of a larger computerized POSIT Science BRAIN HQ assessment module. All tasks were administered at moderate difficulty. The outcome variable used will be the log10 transformed averaged presentation time of correct trials for the Hawk Eye (visual perception task) and Double Decision tasks, and log2 transformed averaged presentation time of correct trials for the Divided Attention task. Outcome variables are log transformed for normality. Presentation times of stimuli adapt based on the accuracy of the participant’s previous answer, with an intended accuracy criterion of 80%. Accurate answers result in faster stimuli presentation times on the next trial, so smaller overall outcome scores reflect faster presentation times. The adaptation of stimuli presentation time is on a log scale. Therefore, at slower presentation times, the adaptive time increment is longer, and at faster presentation times, the adaptive time increment is shorter. The Divided Attention stimuli presentation adaptation is on a log2 scale, and Hawk Eye and Double Decision stimuli presentation adaptation is on a log10 scale. Therefore, the Divided Attention task has shorter overall time adaptation increments. Differences in time increments of stimuli presentation across tasks depend on the nature of the task. Response times of participants are not part of outcome measures, as reaction time is not of interest.
Hawk eye task (visual perception)
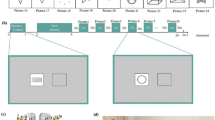
The participant is shown a screen as seen in Fig. 1, panel A, for varying amounts of time. A group of birds with varying spacing will appear in the periphery on the screen. After the stimuli in the periphery disappear, the participant is asked to identify where a target hawk was located on the screen among distractor birds. In Fig. 1, panel A, the target hawk is identified as having a different wing and head color, and different tail shape. There are 25 total trials.
Divided attention task
The participant is shown a screen as seen in Fig. 1, panel B, for varying amounts of time. Figures vary by shape, fill, and color. The participant is asked to press the left key if the figures match on color, and press the right key if the figures do not match on color. There are 60 total trials.
Double decision task
The participant is shown a screen as seen in Fig. 1, panel C, for varying amounts of time. After the target objects in the screen disappear, participants are asked to correctly identify which object was presented in the center of the screen (truck or van) while also correctly locating a simultaneously presented target object in the periphery among distractors (Route 66 sign). There are 25 total trials.
Image acquisition
Participants underwent a 60-min MRI scan with structural (including T1 image) and echo-planar functional imaging. Resting state fMRI data were collected via a 3-Tesla Siemens Magnetom Prisma scanner with a 64-channel head coil at the Center for Cognitive Aging and Memory at the University of Florida, and a 3-Tesla Siemens Magnetom Skyra scanner with a 32-channel head coil at the University of Arizona. Both study sites used identical scanning procedures and sequences, including reducing head motion through the use of foam padding and the use of earplugs to reduce scanner noise. During the 6-min resting state functional image acquisition, participants were asked to rest with their eyes open while looking at a fixation cross. The sequence protocol is as follows: number of volumes = 120, repetition time (TR) = 3,000 ms, echo time (TE) = 30 ms, flip angle = 70°, voxel size = 3.0 × 3.0 × 3.0 mm, 44 slices, field of view (FOV) = 240 × 240 mm. For normalization, a 3-min T1-weighted 3D magnetization-prepared rapid gradient echo image was collected. The sequence protocol is as follows: TR = 1,800 ms, TE = 2.26 ms, flip angle = 8°, voxel size = 1.0 × 1.0 × 1.0 mm3, 176 slices; FOV = 256 × 256 mm.
Image pre-processing and resting state network extraction
Functional images were pre-processed and analyzed through a MATLAB R2019b functional connectivity toolbox “CONN Toolbox,” version 18b (www.nitrc.org/projects/conn, RRID: SCR_009550) and SPM 12 [43, 44]. The default CONN pre-processing pipeline was applied and is as follows: functional realignment, slice-timing correction, segmentation and normalization, and smoothing using a Gaussian kernel of 8-mm full width half maximum (FWHM). Movement outlier scans were identified through global blood-oxygen-level-dependent (BOLD) signal and subject motion via the Artifact Rejection Toolbox (ART). Framewise displacements of 0.9 mm or more, or BOLD signal change of ± 5 standard deviations, were flagged as outliers.
In addition to the core pre-processing steps, the CONN Toolbox applies a default denoising pipeline. Noise factors are estimated from BOLD signal and removed from each voxel using ordinary least squares regression. This pipeline implements an anatomical component-based correction procedure (aCompCor) that identifies five different noise components from white matter and cerebrospinal spaces [45]. Additionally, subject motion parameters (12 noise components; 3 translations, 3 rotations, and their first-order derivatives) and scrubbing (removal of scans identified through the outlier identification step in pre-processing) steps are implemented [46, 47]. A temporal band pass filter was applied to remove frequencies below 0.008 Hz or above 0.09 Hz to isolate slow frequency fluctuations and reducing frequencies due to psychological factors, head motion, or random noise.
Quality control plots for each individual were manually assessed, which displays a comparison of the distribution of functional connectivity between randomly selected pairs of voxels before and after denoising. Scrubbing, outlier identification through the ART tool, and motion parameters were included as covariates when deriving RSN connectivity values.
Publicly available parcellations of regions of interest (ROIs) comprising resting state networks were utilized [23]. Within-network functional connectivity of the DMN, DAN, FPCN, and CON was extracted via the CONN Toolbox by calculating the Fisher z-transformed bivariate correlations between ROI-ROI BOLD time-series within each RSN. The CONN Toolbox then computes average within-network connectivity by calculating the mean of pairwise correlations of every possible ROI-ROI combination in each RSN. This results in one variable taken as a proxy of mean within-network connectivity for a specific RSN. Figure 2 displays the centroid point of each ROI included in each RSN [25].
Visualization of ROI-ROI connections. A Anterior, B superior, C right hemisphere. Color depicts distinct networks [25]
Participants were excluded from analyses if invalid scans due to motion resulted in less than 4 min of useable resting state data, or resulted in less than 80 useable volumes [48,49,50]. Participants were also excluded if their resulting RSN connectivity was determined to be an outlier (> + 3 Z-score). Out of 283 participants recruited, 9 were excluded from this analysis due to use of an improper imaging head coil, 5 participants were excluded due to excessive in scanner movement, and 2 participants were excluded due to outlier resting state network variables. This resulted in a final sample of 267 cognitively healthy older adults (mean age = 71.68, standard deviation = 513; Table 1) from the University of Florida (UF; n = 170) and the University of Arizona (UA; n = 97) used for analysis.
Statistical analysis
Demographic and POSIT assessment data were managed using REDCap electronic data capture tools hosted at the University of Florida [51, 52]. All analyses were performed on SPSS version 25. UF and UA demographic factors were compared and then combined. To assess the relationship of the Hawk Eye task, Divided Attention task, and Double Decision task performance with DMN, DAN, FPCN, and CON, multiple independent linear regressions were performed. Hawk Eye task, Divided Attention task, and Double Decision task performance was predicted from mean within-network connectivity of specified RSNs, controlling for scanner, sex, age, and education, resulting in 4 linear regressions per cognitive task. Assumptions of all linear regressions (i.e., normality of residuals) were confirmed.
Results
Table 2 displays complete output of RSN results controlling for covariates, and Fig. 3 displays scatterplots of regression output for each RSN.
Regressions of cingulo-opercular (CON) and frontoparietal control (FPCN) network connectivity and POSIT measures with 95% confidence intervals (n = 267 for all panels). Colors match network color scheme in Fig. 2. A Hawk Eye and CON; B Divided Attention and CON; C Double Decision and CON; D Double Decision and FPCN; r2 reflects variance explained from the partial correlation between resting-state network connectivity and POSIT assessment performance; β = standardized beta; x-axis = residuals of resting state network connectivity controlling for scanner, sex, education, and age; y-axis = log transformed POSIT performance
Hawk Eye task
Within-network connectivity of CON predicted Hawk Eye performance, in that faster performance associated with greater connectivity (β = − 0.138, p < 0.05). DAN, DMN, or FPCN connectivity did not significantly associate with Hawk Eye task performance. Age also predicted Hawk Eye performance in all RSN regressions, in that older age resulted in slower performance (p < 0.001). Sex, education, or scanner did not significantly associate with Hawk Eye performance. RSN results are visually depicted in Fig. 3, panel A.
Divided attention task
Within-network connectivity of CON predicted Divided Attention task performance, in that faster performance associated with greater connectivity (β = − 0.125, p < 0.05). DAN, DMN, or FPCN connectivity did not significantly associate with Divided Attention task performance. Age also predicted Divided Attention performance in all RSN regressions, in that older age resulted in slower performance (p < 0.001). Sex, education, or scanner did not significantly associate with Hawk Eye performance. RSN results are visually depicted in Fig. 3, panel B.
Double decision task
Within-network connectivity of the CON predicted Double Decision task performance, in that faster performance associated with greater connectivity (β = − 0.121, p < 0.05). Within-network FPCN connectivity also predicted Double Decision task performance in the same direction and at almost identical magnitude (β = − 0.122, p < 0.05). DMN nor DAN connectivity significantly predicted Double Decision task performance. Age and sex predicted Double Decision performance in all RSN regressions (p < 0.001; p < 0.05, respectively), in that older age and female sex associated with slower Double Decision performance. Higher education significantly associated with faster Double Decision performance (p < 0.05) only with CON in the regression model, and was trending when in FPCN, DMN, and DAN regression models. Scanner did not significantly associate with Double Decision performance. RSN results are visually depicted in Fig. 3, panels C and D.
Discussion
Performance on the Double Decision task, which targets speed-of-processing and divided attention, is related to amount of motor vehicle accidents and ability to perform activities of daily living in older adults [13, 53, 54]. Additionally, training in the Double Decision task successfully improves speed-of-processing and physical functioning, and reduces risk of dementia and medical expenditures in older adults over a span of 5–10 years [15,16,17,18,19]. Despite this task’s clear importance in altering the trajectory of cognitive decline and improving functional abilities in older adults, few studies have assessed the neural correlates associated with this task. Moreover, no study has assessed the relationship between Double Decision task performance and higher-order RSNs that are involved in dementia risk and executive performance in aging [24,25,26,27, 30, 31]. This study is the first to investigate the association of four higher-order resting state networks (FPCN, CON, DAN, and DMN) with Double Decision task performance in a sample of healthy older adults, as well as tasks assessing fundamental components that comprise the Double Decision task (visual processing (Hawk Eye task) and Divided Attention task).
Higher-order resting state networks in visual speed processing and divided attention
Tasks targeting fundamental components of the Double Decision task (speeded visual processing and divided attention) associated with within-network connectivity of the CON. Better performance on the Hawk Eye task, targeting speeded visual processing, was related to stronger CON connectivity. Central brain regions that comprise the CON include the anterior insula and the anterior cortex [23, 55]. Connectivity of the CON is thus related to sustained alertness and speed of stimulus detection [38, 39]. Ruiz-Rizzo and colleagues [33] identified the connectivity of a central hub within the CON to be related to visual speed processing performance in aging. Our findings confirm the role CON connectivity plays in sustained attention and visual speed processing in an older adult population.
CON connectivity was also related to Divided Attention task performance, although with relatively weaker association compared to the Hawk Eye task. The Divided Attention task does require sustained alertness, but also requires suppression of irrelevant task information (i.e., identical figure shape or fill). The insula, another key brain area included in the CON, may act as a central hub to filter and identify salient stimuli [55, 56]. Indeed, increased connectivity of key areas in the CON (anterior cingulate cortex, anterior insula, and frontal operculum) has been associated with identification of target auditory stimuli among distractors [21, 57], confirming CON’s role in performance monitoring, irrelevant stimuli suppression, and adaptive control. Taken together, these findings are consistent with the role of CON as a network involved in sustained alertness and selective attention, while also adding to the body of literature showing CON’s involvement in episodic memory and executive function in healthy aging [25, 57].
A unique component of the Hawk Eye and Divided Attention task is speeded stimulus presentation. This suggests that CON may also play a role in the speeded component of visual and sustained attention in a healthy aging population. This is important, as speed-of-processing has a unique association with age-related decline in cognitive functions and activities of daily living [54, 58].
DAN and FPCN are considered “task-positive” networks, meaning they play a role in attentionally demanding and externally oriented tasks [39, 59]. However, neither of these networks appeared to be associated with performance on Hawk Eye (visual speed) or Divided Attention tasks in our healthy aged sample. While the independent function of the CON is sometimes challenging to isolate due to its co-activation with FPCN and DAN, the sustained attention required for the Hawk Eye and Divided Attention tasks may be highlighting the unique role of the CON. Sadaghiani and D’Esposito [39] demonstrated that the CON was only involved in intrinsically maintained tonic alertness and suppression of irrelevant stimuli, while DAN and FPCN are uniquely important in phasic alertness, on an event-by-event basis. Therefore, the sustained attention and stimuli suppression required to perform Hawk Eye and Divided Attention tasks may elicit a distinct association with CON connectivity, although this hypothesis was not statistically tested in this study. The lack of association between these tasks and the DMN is congruent with the current understanding of the DMN as an internally focused network that is suppressed during externally oriented tasks [60].
Higher-order resting state networks in double decision
When fundamental components are combined into one task, FPCN connectivity becomes important in task performance in addition to CON connectivity. As hypothesized, both FPCN and CON connectivity were significantly associated with Double Decision task performance, in that faster performance related to higher connectivity of these networks. This is the first study that has explored the role of higher-order resting state networks in the Double Decision task in a sample of healthy older adults. These findings align with a recent study assessing brain area connectivity changes after Double Decision training in older adults [22], who found that after training, there was an increase in connectivity among brain areas involved in FPCN and CON. Our findings expand upon the information on brain region connectivity in the Double Decision task, and suggest that specific brain networks are involved.
Our findings also contribute to a larger body of research exploring neural correlates of the Double Decision task. Cortical thickness in areas of the FPCN was related to subtests 1, 2, and 3 of Double Decision performance [20, 21], suggesting that in addition to structural correlates, FPCN connectivity also plays an important role in Double Decision task performance. Connectivity of the dorsolateral prefrontal cortex with the intraparietal sulcus (central nodes in the FPCN) is implicated in cognitive status of older adults, and frontoparietal functional disconnection occurs in mild cognitive impairment [61]. Thus, networks involved in Double Decision task performance are also involved in higher-level decision-making, executive functioning, and overall cognitive status.
The literature regarding the neural correlates of the Double Decision task and CON is inconsistent. Schmidt and colleagues [21] did not find the cortical thickness of CON brain regions to be associated with Double Decision task performance, but a more recent study showed that cortical thickness in key areas of CON does relate to better Double Decision task performance [20]. Our findings imply that CON plays at least a functional role in Double Decision task performance. The data used for this study and Kraft et al.’s [20] study were drawn from the same central database. It may be the case that our sample of older adults is starting to experience some cognitive decline and therefore is recruiting additional brain areas to functionally compensate for structural deterioration to maintain task performance. Previous research has shown that structurally atrophic brain areas will attempt to increase connectivity among themselves to preserve function in an attempt to compensate for structural damage, particularly in frontal brain areas [62].
DAN and DMN connectivity were not associated with Double Decision task performance, contrary to our hypothesis that DAN would play an important role. This is surprising, given DAN’s role in visual distractor suppression, orienting visual attention, and diminished connectivity with aging [63,64,65]. However, previous research shows higher DAN connectivity was observed only when attending to one target, rather than two simultaneous targets as in the Double Decision task [65]. The lack of DAN association suggests that the Double Decision task may tap into attention processes exclusively targeting sustained attention and sustained divided attention that may be dissociable from targeted attention processes involving the DAN.
Despite the implication of DMN connectivity in Alzheimer’s dementia risk, and Double Decision training reducing dementia risk, DMN connectivity was not related to Double Decision task performance. DMN is typically regarded as a “task-negative” network; therefore, it was not expected that Double Decision task performance would associate with DMN connectivity. However, these findings do suggest that the integrity of the FPCN may be playing a role in dementia risk reduction. In addition to its role in divided attention, FPCN is also involved in DMN and DAN network modulation and anticorrelation and has been shown to selectively engage with DAN during externally oriented tasks while simultaneously disengaging with DMN, and vice versa [66]. This results in an anticorrelation between DMN and DAN that is important in efficient cognitive functioning, and also tends to decline with aging [31, 67, 68]. Furthermore, recent work from Ewers and colleagues [69] has shown that the segregation, or the balance of between-network to within-network connectivity, is important in resilience of cognitive decline in individuals with Alzheimer’s disease. Given these findings, future work should focus on FPCN’s role in cognitive trajectory after cognitive training in the Double Decision task.
Limitations and future directions
Our study is not without limitations and important considerations in generalizing conclusions. First and foremost, the racial/ethnical distribution of the current sample is predominantly Caucasian (88%) with an average education level reflecting a bachelor’s degree. Therefore, findings from this study are not entirely generalizable to Black/African American, Hispanic, or Asian American individuals. For example, previous research suggests patterns of resting state connectivity differ among racial/ethnic groups of non-Hispanic White, non-Hispanic Black, and Hispanic older adults [70], and there is increasing evidence that experiencing chronic, life-long discrimination impacts patterns of brain functional connectivity [71, 72]. The United States Census reported only 32.1% of Americans had obtained a bachelors’ degree or higher in the years 2015–2019 (https://www.census.gov/quickfacts/fact/table/US/AGE135219#AGE135219). Therefore, our sample predominantly reflects an exclusive portion of the American population that may have higher levels of cognitive reserve, another factor known to alter cognitive functioning and resting state network patterns [68, 73]. Future studies need to assess the relationship of higher-order RSN connectivity with speed-of-processing tasks and tasks of divided attention in a population that is more reflective of US demographic distributions and also in populations that are most at risk for dementia [74].
Training in the Double Decision task results in reduced dementia risk; however, this study did not assess longitudinal change of RSN connectivity. Findings from this study contribute to the understanding of the role of CON and FPCN in attention, speeded visual processing, and divided attention in healthy aging at one timepoint. To understand the role these RSNs play in dementia risk and maintenance of cognitive and functional status, future studies could focus on RSN connectivity change after training in the Double Decision task. Key brain areas of the FPCN and CON include prefrontal cortices, which are prime targets for non-invasive brain stimulation (i.e., transcranial direct current stimulation; tDCS). As tDCS has been shown to improve cognitive abilities in older adults, future studies could also explore the ability of tDCS to alter or increase FPCN and CON connectivity after Double Decision training [75]. Given FPCN’s role in inter-network modulation, and the importance of network segregation in cognitive functioning, future research should also study the association of these tasks with between-network connectivity. Exploring between-network connectivity could more comprehensively address the role these resting state networks may play in task performance. Lastly, individuals included in these analyses were cognitively healthy. Extending these analyses to include individuals with mild cognitive impairment could greatly benefit our understanding of higher-order RSNs in aging and cognition.
Conclusions
Findings from this study contribute to the growing body of literature aimed at assessing the role of higher-order resting state networks and crucial cognitive functions in aging. Connectivity of the CON is related to speeded visual processing and divided attention in a healthy aging population. FPCN connectivity, in addition to CON connectivity, is related to a task of combined speeded visual processing and divided attention, which is implicated in reduced dementia risk. This points to the importance of the FPCN in complex tasks of attention, and highlights future directions focused on FPCN connectivity and altered trajectory of cognitive decline.
Data and materials availability
The data analyzed in this study is subject to the following licenses/restrictions: data are managed under the data sharing agreement established with NIA and the parent R01 clinical trial Data Safety and Monitoring Board in the context of an ongoing Phase III clinical trial (ACT study, R01AG054077). All trial data will be made publicly available 2 years after completion of the parent clinical trial, per NIA and DSMB agreement. Requests for baseline data can be submitted to the ACT Publication and Presentation (P&P) Committee and will require submission of a data use, authorship, and analytic plan for review by the P&P committee (ajwoods@phhp.ufl.edu). Requests to access these datasets should be directed to ajwoods@ufl.edu.
Code availability
Not applicable.
References
Parkin AJ, Java RI. Deterioration of frontal lobe function in normal aging: influences of fluid intelligence versus perceptual speed. Neuropsychology. 1999;13:7.
Salthouse TA. Selective review of cognitive aging. J Int Neuropsychol Soc. 2010;16:754–60. https://doi.org/10.1017/S1355617710000706.
Verhaeghen P, Salthouse TA. Meta-analyses of age-cognition relations in adulthood: estimates of linear and nonlinear age effects and structural models. Psychol Bull. 1997;122:19.
Clay OJ, Edwards JD, Ross LA, Okonkwo O, Wadley VG, Roth DL, et al. Visual function and cognitive speed of processing mediate age-related decline in memory span and fluid intelligence. J Aging Health. 2009;21:547–66. https://doi.org/10.1177/0898264309333326.
Ebaid D, Crewther SG, MacCalman K, Brown A, Crewther DP. Cognitive processing speed across the lifespan: beyond the influence of motor speed. Front Aging Neurosci 2017;9. https://doi.org/10.3389/fnagi.2017.00062.
Reuter-Lorenz PA, Festini SB, Jantz TK. "Executive functions and neurocognitive aging." In Handbook of the psychology of aging, pp. 67-81. Academic Press, 2021.
Salthouse TA. Decomposing age correlations on neuropsychological and cognitive variables. J Int Neuropsychol Soc. 2009;15:650–61. https://doi.org/10.1017/S1355617709990385.
Cahn-Weiner DA, Boyle PA, Malloy PF. Tests of executive function predict instrumental activities of daily living in community-dwelling older individuals. Appl Neuropsychol. 2002;9:187–91. https://doi.org/10.1207/S15324826AN0903_8.
Johnson JK, Lui L-Y, Yaffe K. Executive function, more than global cognition, predicts functional decline and mortality in elderly women 2007:15.
Aust F, Edwards JD. Incremental validity of useful field of view subtests for the prediction of instrumental activities of daily living. J Clin Exp Neuropsychol. 2016;38:497–515. https://doi.org/10.1080/13803395.2015.1125453.
Ball KK. Clinical visual perimetry under-estimates peripheral field problems in older adults. Clin Vis Sci. 1990;5:113–25.
Woutersen K, Guadron L, van den Berg AV, Boonstra FN, Theelen T, Goossens J. A meta-analysis of perceptual and cognitive functions involved in useful-field-of-view test performance. J Vis. 2017;17:11. https://doi.org/10.1167/17.14.11.
Ball KK, Owsley C, Sloane ME, Roenker DL, Bruni JR. Visual attention problems as a predictor of vehicle crashes in older drivers. Invest Ophthalmol Vis Sci. 1993;34:3110–23.
Clay OJ, Wadley VG, Edwards JD, Roth DL, Roenker DL, Ball KK. cumulative meta-analysis of the relationship between useful field of view and driving performance in older adults: current and future implications. Optom Vis Sci. 2005;82:724–31. https://doi.org/10.1097/01.opx.0000175009.08626.65.
Ball K, Berch DB, Helmers KF, Jobe JB, Leveck MD, Marsiske M, et al. Effects of cognitive training interventions with older adults: a randomized controlled trial. JAMA. 2002;288:2271. https://doi.org/10.1001/jama.288.18.2271.
Rebok GW, Ball K, Guey LT, Jones RN, Kim H-Y, King JW, et al. Ten-year effects of the advanced cognitive training for independent and vital elderly cognitive training trial on cognition and everyday functioning in older adults. J Am Geriatr Soc. 2014;62:16–24. https://doi.org/10.1111/jgs.12607.
Edwards JD, Xu H, Clark DO, Guey LT, Ross LA, Unverzagt FW. Speed of processing training results in lower risk of dementia. Alzheimers Dement Transl Res Clin Interv. 2017;3:603–11. https://doi.org/10.1016/j.trci.2017.09.002.
Ross LA, Sprague BN, Phillips CB, O’Connor ML, Dodson JE. The impact of three cognitive training interventions on older adults’ physical functioning across 5 years. J Aging Health. 2018;30:475–98. https://doi.org/10.1177/0898264316682916.
Wolinsky FD, Mahncke HW, Kosinski M, Unverzagt FW, Smith DM, Jones RN, et al. The ACTIVE cognitive training trial and predicted medical expenditures. BMC Health Serv Res. 2009;9:109–109. https://doi.org/10.1186/1472-6963-9-109.
Kraft JN, O’Shea A, Albizu A, Evangelista ND, Hausman HK, Boutzoukas E, et al. structural neural correlates of double decision performance in older adults. Front Aging Neurosci. 2020;12:278. https://doi.org/10.3389/fnagi.2020.00278.
Schmidt EL, Burge W, Visscher KM, Ross LA. Cortical thickness in frontoparietal and cingulo-opercular networks predicts executive function performance in older adults. Neuropsychology. 2016;30:322–31. https://doi.org/10.1037/neu0000242.
Ross LA, Webb CE, Whitaker C, Hicks JM, Schmidt EL, Samimy S, et al. The effects of useful field of view training on brain activity and connectivity. J Gerontol Ser B. 2019;74:1152–62. https://doi.org/10.1093/geronb/gby041.
Yeo BT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011;106:1125–65. https://doi.org/10.1152/jn.00338.2011.
Geerligs L, Renken RJ, Saliasi E, Maurits NM, Lorist MM. A brain-wide study of age-related changes in functional connectivity. Cereb Cortex. 2015;25:1987–99. https://doi.org/10.1093/cercor/bhu012.
Hausman HK, O’Shea A, Kraft JN, Boutzoukas EM, Evangelista ND, Van Etten EJ, et al. The role of resting-state network functional connectivity in cognitive aging. Front Aging Neurosci. 2020;12:177. https://doi.org/10.3389/fnagi.2020.00177.
Andrews-Hanna JR, Snyder AZ, Vincent JL, Lustig C, Head D, Raichle ME, et al. Disruption of large-scale brain systems in advanced aging. Neuron. 2007;56:924–35. https://doi.org/10.1016/j.neuron.2007.10.038.
Damoiseaux JS, Beckmann CF, Arigita EJS, Barkhof F, Scheltens Ph, Stam CJ, et al. Reduced resting-state brain activity in the “default network” in normal aging. Cereb Cortex. 2008;18:1856–64. https://doi.org/10.1093/cercor/bhm207.
Greicius MD, Krasnow B, Reiss AL, Menon V. Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc Natl Acad Sci. 2003;100:253–8. https://doi.org/10.1073/pnas.0135058100.
Greicius MD, Srivastava G, Reiss AL, Menon V. Default-mode network activity distinguishes Alzheimer’s disease from healthy aging: evidence from functional MRI. Proc Natl Acad Sci. 2004;101:4637–42. https://doi.org/10.1073/pnas.0308627101.
Grady C, Sarraf S, Saverino C, Campbell K. Age differences in the functional interactions among the default, frontoparietal control, and dorsal attention networks. Neurobiol Aging. 2016;41:159–72. https://doi.org/10.1016/j.neurobiolaging.2016.02.020.
Shaw EE, Schultz AP, Sperling RA, Hedden T. functional connectivity in multiple cortical networks is associated with performance across cognitive domains in older adults. Brain Connect. 2015;5:505–16. https://doi.org/10.1089/brain.2014.0327.
Penning MD, Ruiz-Rizzo AL, Redel P, Müller HJ, Salminen T, Strobach T, et al. alertness training increases visual processing speed in healthy older adults. Psychol Sci 2021:095679762096552. https://doi.org/10.1177/0956797620965520.
Ruiz-Rizzo AL, Sorg C, Napiórkowski N, Neitzel J, Menegaux A, Müller HJ, et al. Decreased cingulo-opercular network functional connectivity mediates the impact of aging on visual processing speed. Neurobiol Aging. 2019;73:50–60. https://doi.org/10.1016/j.neurobiolaging.2018.09.014.
Anderson ND, Iidaka T, Cabeza R, Kapur S, McIntosh AR, Craik FIM. The effects of divided attention on encoding- and retrieval-related brain activity: a PET study of younger and older adults. J Cogn Neurosci. 2000;12:775–92. https://doi.org/10.1162/089892900562598.
Verghese J, Buschke H, Viola L, Katz M, Hall C, Kuslansky G, et al. Validity of divided attention tasks in predicting falls in older individuals: a preliminary study. J Am Geriatr Soc. 2002;50:1572–6. https://doi.org/10.1046/j.1532-5415.2002.50415.x.
Johnson JA, Zatorre RJ. Neural substrates for dividing and focusing attention between simultaneous auditory and visual events. Neuroimage. 2006;31:1673–81. https://doi.org/10.1016/j.neuroimage.2006.02.026.
Wimmer RD, Schmitt LI, Davidson TJ, Nakajima M, Deisseroth K, Halassa MM. Thalamic control of sensory selection in divided attention. Nature. 2015;526:705–9. https://doi.org/10.1038/nature15398.
Coste CP, Kleinschmidt A. Cingulo-opercular network activity maintains alertness. Neuroimage. 2016;128:264–72. https://doi.org/10.1016/j.neuroimage.2016.01.026.
Sadaghiani S, D’Esposito M. Functional characterization of the cingulo-opercular network in the maintenance of tonic alertness. Cereb Cortex. 2015;25:2763–73. https://doi.org/10.1093/cercor/bhu072.
Serra L, Cercignani M, Mastropasqua C, Torso M, Spanò B, Makovac E, et al. Longitudinal changes in functional brain connectivity predicts conversion to Alzheimer’s disease. J Alzheimers Dis. 2016;51:377–89. https://doi.org/10.3233/JAD-150961.
Woods AJ, Cohen R, Marsiske M, Alexander GE, Czaja SJ, Wu S. Augmenting cognitive training in older adults (The ACT Study): design and methods of a phase III tDCS and cognitive training trial. Contemp Clin Trials. 2018;65:19–32. https://doi.org/10.1016/j.cct.2017.11.017.
Weintraub S. UDS-III Norms. 2017.
Penny WD, Friston KJ, Ashburner JT, Kiebel SJ, Nichols TE. Statistical parametric mapping: the analysis of functional brain images. Elsevier; 2011.
Whitfield-Gabrieli S, Nieto-Castanon A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2012;2:125–41. https://doi.org/10.1089/brain.2012.0073.
Behzadi Y, Restom K, Liau J, Liu TT. A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. Neuroimage. 2007;37:90–101. https://doi.org/10.1016/j.neuroimage.2007.04.042.
Friston KJ, Williams S, Howard R, Frackowiak RSJ, Turner R. Movement-Related effects in fMRI time-series: Movement Artifacts in fMRI. Magn Reson Med. 1996;35:346–55. https://doi.org/10.1002/mrm.1910350312.
Power JD, Mitra A, Laumann TO, Snyder AZ, Schlaggar BL, Petersen SE. Methods to detect, characterize, and remove motion artifact in resting state fMRI. Neuroimage. 2014;84:320–41. https://doi.org/10.1016/j.neuroimage.2013.08.048.
Parkes L, Fulcher B, Yücel M, Fornito A. An evaluation of the efficacy, reliability, and sensitivity of motion correction strategies for resting-state functional MRI. Neuroimage. 2018;171:415–36. https://doi.org/10.1016/j.neuroimage.2017.12.073.
Satterthwaite TD, Elliott MA, Gerraty RT, Ruparel K, Loughead J, Calkins ME, et al. An improved framework for confound regression and filtering for control of motion artifact in the preprocessing of resting-state functional connectivity data. Neuroimage. 2013;64:240–56. https://doi.org/10.1016/j.neuroimage.2012.08.052.
Van Dijk KRA, Sabuncu MR, Buckner RL. The influence of head motion on intrinsic functional connectivity MRI. Neuroimage. 2012;59:431–8. https://doi.org/10.1016/j.neuroimage.2011.07.044.
Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L, et al. The REDCap consortium: Building an international community of software platform partners. J Biomed Inform. 2019;95:103208–103208. https://doi.org/10.1016/j.jbi.2019.103208.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–81. https://doi.org/10.1016/j.jbi.2008.08.010.
Owsley C, Ball K, Sloane ME, Roenker DL, Bruni JR. Visual/cognitive correlates of vehicle accidents in older drivers. Psychol Aging. 1991;6:403–15. https://doi.org/10.1037/0882-7974.6.3.403.
Owsley C, Sloane M, McGwin G Jr, Ball K. Timed instrumental activities of daily living tasks: relationship to cognitive function and everyday performance assessments in older adults. Gerontology. 2002;48:254–65. https://doi.org/10.1159/000058360.
Seeley WW, Menon V, Schatzberg AF, Keller J, Glover GH, Kenna H, et al. Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci. 2007;27:2349–56. https://doi.org/10.1523/JNEUROSCI.5587-06.2007.
Menon V, Uddin LQ. Saliency, switching, attention and control: a network model of insula function. Brain Struct Funct. 2010;214:655–67. https://doi.org/10.1007/s00429-010-0262-0.
Vaden KI, Kuchinsky SE, Cute SL, Ahlstrom JB, Dubno JR, Eckert MA. The cingulo-opercular network provides word-recognition benefit. J Neurosci. 2013;33:18979–86. https://doi.org/10.1523/JNEUROSCI.1417-13.2013.
Salthouse TA, Ferrer-Caja E. What needs to be explained to account for age-related effects on multiple cognitive variables? Psychol Aging. 2003;18:91–110. https://doi.org/10.1037/0882-7974.18.1.91.
Grady CL, Protzner AB, Kovacevic N, Strother SC, Afshin-Pour B, Wojtowicz M, et al. A multivariate analysis of age-related differences in default mode and task-positive networks across multiple cognitive domains. Cereb Cortex. 2010;20:1432–47. https://doi.org/10.1093/cercor/bhp207.
Anticevic A, Cole MW, Murray JD, Corlett PR, Wang X-J, Krystal JH. The role of default network deactivation in cognition and disease. Trends Cogn Sci. 2012;16:584–92. https://doi.org/10.1016/j.tics.2012.10.008.
Liang P, Wang Z, Yang Y, Jia X, Li K. Functional disconnection and compensation in mild cognitive impairment: evidence from DLPFC connectivity using resting-state fMRI. PLoS ONE. 2011;6: e22153. https://doi.org/10.1371/journal.pone.0022153.
Vieira BH, Rondinoni C, Garrido Salmon CE. Evidence of regional associations between age-related inter-individual differences in resting-state functional connectivity and cortical thinning revealed through a multi-level analysis. Neuroimage. 2020;211: 116662. https://doi.org/10.1016/j.neuroimage.2020.116662.
Bagarinao E, Watanabe H, Maesawa S, Mori D, Hara K, Kawabata K, et al. Reorganization of brain networks and its association with general cognitive performance over the adult lifespan. Sci Rep. 2019;9:11352. https://doi.org/10.1038/s41598-019-47922-x.
Hopfinger JB, Buonocore MH, Mangun GR. The neural mechanisms of top-down attentional control. Nat Neurosci. 2000;3:284–91. https://doi.org/10.1038/72999.
Lanssens A, Pizzamiglio G, Mantini D, Gillebert CR. Role of the dorsal attention network in distracter suppression based on features. Cogn Neurosci. 2020;11:37–46. https://doi.org/10.1080/17588928.2019.1683525.
Gao W, Lin W. Frontal parietal control network regulates the anti-correlated default and dorsal attention networks. Hum Brain Mapp. 2012;33:192–202. https://doi.org/10.1002/hbm.21204.
Avelar-Pereira B, Bäckman L, Wåhlin A, Nyberg L, Salami A. Age-related differences in dynamic interactions among default mode, frontoparietal control, and dorsal attention networks during resting-state and interference resolution. Front Aging Neurosci. 2017;9:152. https://doi.org/10.3389/fnagi.2017.00152.
Franzmeier N, Göttler J, Grimmer T, Drzezga A, Áraque-Caballero MA, Simon-Vermot L, et al. Resting-state connectivity of the left frontal cortex to the default mode and dorsal attention network supports reserve in mild cognitive impairment. Front Aging Neurosci. 2017;9:264. https://doi.org/10.3389/fnagi.2017.00264.
Ewers M, Luan Y, Frontzkowski L, Neitzel J, Rubinski A, Dichgans M, et al. Segregation of functional networks is associated with cognitive resilience in Alzheimer’s disease. Brain 2021:awab112. https://doi.org/10.1093/brain/awab112.
Turney IC, Chesebro AG, Rentería MA, Lao PJ, Beato JM, Schupf N, et al. APOE ε4 and resting-state functional connectivity in racially/ethnically diverse older adults. Alzheimers Dement Diagn Assess Dis Monit. 2020;12: e12094. https://doi.org/10.1002/dad2.12094.
Clark US, Miller ER, Hegde RR. Experiences of discrimination are associated with greater resting amygdala activity and functional connectivity. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3:367–78. https://doi.org/10.1016/j.bpsc.2017.11.011.
Han SD, Lamar M, Fleischman D, Kim N, Bennett DA, Lewis TT, et al. Self-reported experiences of discrimination in older black adults are associated with insula functional connectivity. Brain Imaging Behav. 2020. https://doi.org/10.1007/s11682-020-00365-9.
Pietzuch M, King AE, Ward DD, Vickers JC. The influence of genetic factors and cognitive reserve on structural and functional resting-state brain networks in aging and Alzheimer’s disease. Front Aging Neurosci. 2019;11:30. https://doi.org/10.3389/fnagi.2019.00030.
Chen C, Zissimopoulos JM. Racial and ethnic differences in trends in dementia prevalence and risk factors in the United States. Alzheimers Dement Transl Res Clin Interv. 2018;4:510–20. https://doi.org/10.1016/j.trci.2018.08.009.
Indahlastari A, Hardcastle C, Albizu A, Alvarez-Alvarado S, Boutzoukas EM, Evangelista ND, et al. A systematic review and meta-analysis of transcranial direct current stimulation to remediate age-related cognitive decline in healthy older adults. Neuropsychiatr Dis Treat. 2021;17:971–90. https://doi.org/10.2147/NDT.S259499.
Acknowledgements
We would like to thank all of our participants for their time and research assistants for their hard work and instrumental role in making this manuscript possible.
Funding
We would like to acknowledge support by the National Institute of Aging/National Institutes of Health (T32AG020499, K01AG050707, R01AG054077, P30AG019610, and T32AG061892), the University of Florida Center for Cognitive Aging and Memory Clinical Translational Research, the state of Arizona and Arizona Department of Health Services, the McKnight Brain Research Foundation, and the National Heart, Lung, and Blood Institute (T32HL134621).
Author information
Authors and Affiliations
Contributions
Cheshire Hardcastle wrote the first draft of the manuscript under the mentorship of Adam J. Woods, and all authors commented on and approved previous versions. Material preparation and data collection were performed by Jessica Kraft, Alejandro Albizu, Hanna K. Hausman, Nicole D. Evangelista, Emanuel M. Boutzoukas, Andrew O’Shea, Emily J. Van Etten, Pradyumna H. Bharadwaj, Hyun Song, and Samantha Smith. Cheshire Hardcastle, Jessica Kraft, and Hanna Hausman contributed to data analysis and processing. Eric Porges, Steven T. DeKosky, Georg A. Hishaw, Samuel S. Wu, Michael Marsiske, Ronald Cohen, and Gene E. Alexander contributed to study conception and design. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The studies involving human participants were reviewed and approved by the University of Florida Institutional Review Board and the University of Arizona Institutional Review Board. The patients/participants provided their written informed consent to participate in this study.
Consent to participate
Freely-given, informed consent to participate in the study was obtained from all study participants.
Consent for publication
The participants provided informed consent regarding publishing their data.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Hardcastle, C., Hausman, H.K., Kraft, J.N. et al. Higher-order resting state network association with the useful field of view task in older adults. GeroScience 44, 131–145 (2022). https://doi.org/10.1007/s11357-021-00441-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-021-00441-y