Abstract
The Juarez Valley is an important agricultural region in northern Mexico, conveniently organized into three modules (I to III). For decades, their soils have been exposed to organochlorine pesticides (OCPs) and also have been irrigated with wastewaters, which may contain heavy metals. Nowadays, there is very limited information regarding the presence of OCPs and heavy metals in these soils. Thus, the aim of this study was to diagnose these soils for OCPs and heavy metal content by using gas chromatography coupled with electron micro-capture detector and atomic absorption spectrometry, respectively. The results indicated that 4,4′-dichlorodiphenyldichloroethylene and 4,4′-dichlorodiphenyltrichloroethane were primarily disseminated across the three modules since they were found in 100% and 97% of the analyzed soils, respectively. According to international regulations, none of the determined OCP concentrations are out of the limits. Additionally, the Cu, Zn, Fe, Pb, and Mn were found in all sampled soils from the three modules. The highest concentration of Fe was found in module II (1902.7 ± 332.2 mg kg−1), followed by Mn in module III (392.43 ± 74.43 mg kg−1), Zn in module I (38.36 ± 26.57 mg kg−1), Pb in module II (23.48 ± 6.48 mg kg−1), and Cu in module I (11.04 ± 3.83 mg kg−1) (p ≤ 0.05). These values did not exceed the limits proposed by international standards. The Cd was detected in most of the analyzed soils and all their values, with an average of 2 mg kg−1, surpassed the Mexican standards (0.35 mg kg−1). This study has mapped the main OCPs and heavy metals in the Juarez Valley and can serve as a starting point to further monitor the behave of xenobiotics. Since these recalcitrant compounds might be bio-accumulated in biological systems, further analytical methods, as well as remediation techniques, should be developed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Juarez Valley is located in the state of Chihuahua, Mexico, bordering to the north by TX, USA (105° 30′ and 106° 30′ west longitude, and parallels 30° 56′ and 31° 45′ north latitude). It includes the municipalities of Juarez, Guadalupe, and Praxedis G. Guerrero, with a total extension of 25,456 ha. The climate of this region is very dry with annual rainfall and temperatures of 200–300 mm and 17 °C, respectively. Nevertheless, it should be noted that temperatures below − 5 °C in extreme winter or above 40 °C in hot summer might be reached (INEGI 2003). The Valley is an important agricultural region where, historically, crops like wheat, pepper, tomato, onion, alfalfa, pecan, pistachio, and cotton have been grown. The intensive production of these crops has entailed the use of agrochemicals such as organochlorinated pesticides (OCPs) or heavy metal–based compounds (Udeigwe et al. 2015).
During many decades, the OCPs have also been widely used in international soils to eliminate pests and their occurrence have been reported. For example, soils in China were comprehensively explored to determine the incidence of these types of pollutants (Zhang et al. 2012). Similarly, several studies reported the presence of these compounds in agricultural soils of Pakistan, India, and, Italy, among others countries (Mahmood et al. 2014; Chakraborty et al. 2015; Qu et al. 2016).
Due to the effectiveness of the pioneer 4,4′-dichlorodiphenyltrichloroethane (DDT) as a pesticide, other chlorinated substances such as isodrin, dieldrin, endosulfan, hexachlorobenzene (HCB), and hexachlorocyclohexane (HCH), were synthesized and extensively applied in agricultural soils (Ming 2011). These pesticides are classified as persistent organic pollutants because of their chemical composition. In comparison with many other “naturally” occurring substances, these materials are more resistant to chemical, biochemical, and photochemical degradation; thus, they have a relatively long half-life (Li 2018). Unfortunately, the OCPs can be accumulated in the adipose tissue, kidneys, liver, blood, brain, and spleen (Liu et al. 2017). Studies have provided evidence to associate these pesticides with diverse health issues in humans, such as metabolic syndrome, obesity, and type 2 diabetes (Salihovic et al., 2016; Rosenbaum et al. 2017). The OCPs have also been found in crocodiles that show signs of feminization and atrophy of their reproductive organs (Raloff 1995) and in birds whose eggs have shown changes in shell thickness (King et al. 2003).
These and many other deleterious effects led to the prohibition of these substances by the Stockholm Convention Treaty in 2004 (Stockholm Convention 2004). Although controls in the use of DDT were applied in the USA since the 1970s (US EPA 2016), it is reported that DDT was used in Mexico until 2000 (Díaz-Barriga et al. 2012), while others, like endosulfan and aldrin, are still used in Mexican agricultural lands to treat crops such as corn, cotton, and beans. Notably, the use of these latter compounds is restricted or prohibited in several countries that adhere to the Stockholm Convention (Tsai 2010).
Heavy metals are defined as elements that have a density greater than 5 g cm−1. Some of them are soluble in water when they are presented in their ionic forms, which may lead to their absorption by living organisms (Ming 2011). Inside cells, heavy metals interfere with a variety of metabolic processes that can promote cancer development (Vella et al., 2017) and neurological disorders (Yorifuji et al. 2011). Elements like zinc (Zn) and iron (Fe), considered essentials because of their function to human beings, can have also adverse effects when they are ingested in high concentrations. For instance, there is evidence that consumption of Fe in higher doses is linked with hypertriglyceridemia and insulin resistance in mice (Dongiovanni et al. 2013); Zn is associated with pancreatic issues in animals (Carpenter et al. 2004; Carreira et al. 2011).
The excess of heavy metals in environmental matrices such as soil and water has been linked with human activities. Particularly, the prevalence of metals in several agricultural soils is highly associated with the use of agrochemical fertilizers or antifungal/bactericides (Tan et al. 2018). In countries such as Pakistan, China, Korea, New Zealand, and Iran, the occurrence of mercury (Hg), Cr, Fe, Ni, Cd, Pb, Zn, Cu, As, Co, and Mn has been reported (Afzal et al. 2012; Xiao et al. 2017; Zang et al. 2017; Kwon et al. 2017; Martin et al., 2017; Naghipour et al. 2018).
To the best of the authors’ knowledge, there are no reports regarding the presence of OCPs in the soils from the Juarez Valley and the studies on heavy metals in this area are still very limited. Therefore, the aim of this work was to perform an analysis on the soils of the region to diagnose their compounds and element architecture.
Materials and methods
Sample collection and processing
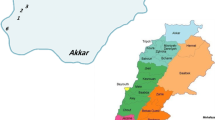
Soil samples (0–30 cm deep) were collected in June 2018, following the criteria of the Mexican Norm NMX-AA-132-SCFI-2006. Administratively, the Juarez Valley is divided into three modules (Fig. 1) and 10 composite samples were taken for each module (Table 1). The soil samples were sieved (mesh size no. 10) and stored in paper bags to be transported to the laboratory.
Pesticide analysis by GC-μECD
Pesticide determination was done following the method reported by Cantú-Soto et al. (2011). First, 0.5 g of soil samples was dispersed in a mortar with 0.6 g of Al2O3 (aluminum oxide) (J.T. Baker, Pittsburgh, PA, USA). Then, the samples were purified by incorporating them into a 5-mL glass syringe packed with 2.6 g of Al2O3 and glass fiber at the bottom; the organic compounds were eluted with 40 mL of hexane GC grade (Honeywell, NJ, USA). The eluted was collected in a 50-mL conical tube where the hexane was evaporated to dryness at 40 °C using gaseous N2, and the concentrate was reconstructed with 100 μL of hexane. A gas chromatograph with Network GC system (Agilent Technologies 7890A, Palo Alto, CA, USA), equipped with an electron micro-capture detector (μECD) with a DB-5 column (30 m in length × 0.25 mm i.d., the thickness of 0.25 μm), was used for the analysis. The column temperature was initiated at 110 °C for 1 min, then it was increased to 280 °C at a rate of 15 °C min−1; the final temperature was maintained for 2 min. The temperatures of the injector and detector were 270 °C and 340 °C, respectively. Helium was used as carrier gas at 2.3 mL min−1. A negative control (0.5 g of Al2O3) and a positive control of soil fortified with 100 μL of a 500-μg kg−1 mix of pesticides of interest: DDT, 4,4′-dichlorodiphenyldichloroethylene (DDE), 4,4′-dichlorodiphenyldichloroethane (DDD), aldrin, dieldrin, endosulfan, HCB, isodrin, lindane, methoxychlor, and mirex (PESTANAL® Sigma-Aldrich, St. Louis, MO, USA) were also analyzed. The method was validated by analyzing the following parameters: linearity, recovery percentages, and limits of quantification and detection (Table 2).
Determination of heavy metals by atomic absorption spectrometry
Soil samples were subjected to chemical digestion before the instrumental analysis, following the method reported by Meza-Figueroa et al. (2007), with some modifications. Briefly, 2 g of each sample was mixed with 50 mL of 0.5 N HCl (hydrochloric acid) (Reactivos Meyer, Ciudad de Mexico, Mexico) in 250-mL beakers to extract bioavailable metals. This process was carried out for 24 h at room temperature with sporadic agitation. A negative (0.5 N HCl) sample and a positive sample of fortified soil with 0.5 ppm of a mix of metals of interest, Fe, Mn, Cu, Cd, Pb, and Zn (Merck, Darmstadt, Germany), were also processed. After the digestion process, samples were filtered (Whatman #42), and the liquid phase was brought to a volume of 100 mL with 0.5 N HCl. Samples and controls were analyzed by flame atomic absorption spectrometry using an Agilent 240 FS spectrometer (Agilent Technologies 7890A, Palo Alto, CA, USA), equipped with hollow-cathode lamps. Argon was used as the carrier gas. The method was validated by analyzing the following parameters: recovery percentages, linearity, and limits of quantification and detection as previously reported (Cota-Ruiz et al. 2018).
pH and electrical conductivity analysis
The pH and electric conductivity (EC) measurements were done following the criteria of the Mexican norm NOM-021-SEMARNAT-2000. Soil pH was determined by shaking 10 g of soil with 20 mL of distilled water in plastic containers for 5 min at room temperature, following by a non-shaking incubation of 10 min. The operation was repeated two more times. The pH analysis was done using a potentiometer (HANNA model HI 207, Wooncocket, RI, USA). EC was done by mixing 10 g of soil and 50 mL of distilled water, at the same above-mentioned conditions. After this period, the analysis was done using an EC meter (Thermo model Orion 3 star, Waltham, MA, USA).
Statistical analysis
To evaluate differences in pesticides and heavy metal content among the different modules, an ANOVA analysis was performed. Tukey’s HSD was used to distinguish mean significant differences (p ≤ 0.05). When data did not follow a normal distribution, they were Box-Cox transformed. Descriptive statistical parameters were also estimated for pH and CE. All the analyses were done with IBM SPSS statistics version 23 for Windows (Armonk, NY, USA).
Results and discussion
Pesticides in soils of the Juarez Valley
The concentration of DDE, DDT, isodrin, endosulfan, and DDD for modules I, II, and III of the Juarez Valley soils are shown in Table 3. The agrochemical DDE was fully detected in all sampled sites, registering average values of 4.86 ± 6.2 μg kg−1, 18.27 ± 31.04 μg kg−1, and 5.90 ± 4.81 μg kg−1, for modules I, II, and III, respectively. These mean values did not significantly differ among them (p ≤ 0.05). Meanwhile, the chemical DDT was identified in all analyzed sites from modules I and II, and in 90% of the sampled soils in module III, with average values of 0.83 ± 0.56 μg kg−1, 19.08 ± 53.71 μg kg−1, and 1.67 ± 0.79 μg kg−1, respectively. These mean values were not significantly different (p ≤ 0.05). Additionally, isodrin was found in 60% of the sampled sites in module I (12.29 ± 3.6 μg kg−1). However, it was only identified in one site in module II (0.71 μg kg−1), and no presence was detected in module III. Contrarily, endosulfan was encountered in 60% of the sample sites (1.44 ± 0.4 μg kg−1) for module III, while it was detected in only one site in module I (1.19 μg kg−1) and also in one point at module II (10.76 μg kg−1). Finally, the agrochemical DDD was detected only in two sites in module II (24.37 and 0.42 μg kg−1) and in one site in module III (0.84 μg kg−1).
The presence of OCP compounds in Mexican agricultural soils has been previously reported in the literature. For instance, Wong et al. (2008) described the presence of OCPs in agricultural soils in three Mexican states: Chiapas, Tabasco, and Veracruz. They found residues of DDT in almost all the sampled sites, Chiapas being the state that had the highest concentrations (an average of 360 μg kg−1), in comparison with Veracruz (6.5 μg kg−1) and Tabasco (0.17 μg kg−1). In the Yaqui and Mayo Valleys, Cantú-Soto et al. (2011) found DDT, DDE, DDD, and endosulfan at 17.9, 11.2, 1.6, and 6.7 ppm, respectively. These two regions in the state of Sonora, Mexico, perform very intensive agriculture activities that have been recognized as the cradle of the “Green Revolution.” In another study where soils from northern and central Sonora were analyzed, Leal et al. (2014) reported residues of DDT, DDE, DDD, endosulfan, and isodrin, with averages of 2.86, 14.63, 2.22, 4.35, and 3.30 μg kg−1, respectively. These values are similar to those reported in the present study. Only one study was found that analyses the presence of OCPs in agricultural areas (Díaz-Barriga et al. 2012) in the state of Chihuahua, Mexico. This study specified that the maximum amounts of DDT and DDE were 0.79 mg kg−1 and 0.642 mg kg−1, respectively. These values are greater than those found in the present study.
The presence of pesticides such as DDT, endosulfan, HCH, methoxychlor, chlordane, dieldrin, endrin, and aldrin have also been found in soils from China (Zhou et al. 2013; Han et al. 2017), India (Chakraborty et al. 2015; Kulangaravalappil and Chenicherry 2018), Pakistan (Mahmood et al. 2014), Romania (Tarcau et al. 2013), and Italy (Qu et al. 2016); this demonstrates that agricultural lands polluted with OCPs is a widespread issue. The differences between the amounts of OCPs in different agricultural regions, and even within the same region, are closely related to dissipation and volatilization processes, as these parameters directly influence the persistence of pesticides in soils. In addition, these processes might be affected by weather conditions, such as temperature and humidity (Zhang et al. 2012).
The OCPs can be accumulated in agricultural lands by their direct application (Wong et al. 2008) and also by the use of treated wastewater containing OCPs (Sun et al. 2009). In the case of the Juarez Valley, it has been published that some treated water contains OCPs, such as DDT and DDE (Palomo-Rodríguez et al. 2013). Inopportunely, these waters are mixed with waters of the Rio Grande River and then used to irrigate crops. Although the concentrations of the OCPs reported in this research were found to be within the FAO regulations (FAO 2000), it should not be discounted that they might bio-accumulate in crops (Adeyeye and Osibanjo 1999). On the other hand, even at low-environmental amounts, they may elicit deleterious cell responses leading to diseases or cell malfunctions (Androutsopoulos et al. 2013). In addition, these compounds could infiltrate into groundwater, affecting human and animal health (Grondona et al. 2019). Moreover, there is evidence about the possible incorporation of pesticides into the food chain due to their bioaccumulation in some vegetables (Zhang et al. 2015).
Heavy metals in soils of the Juarez Valley
The heavy metals Cu, Zn, Fe, Pb, Mn, and Cd examined in this study were found at different concentrations in the three analyzed modules of the Juarez Valley (Fig. 2). The content of Cu in module I (11.04 ± 3.83 mg kg−1) was higher than that in module II (7.79 ± 3.32 mg kg−1) and module III (6.2 ± 1.51 mg kg−1) (Fig. 2(A)) (p ≤ 0.05). The obtained values for the module I are similar to the values previously reported in soils belonging to this district (Cota Ruiz et al. 2019). In the latter study, the authors reported an average value of 20.06 ± 3.39 mg of Cu per kg of soil. In module I, it was also found that the concentration of Zn was significantly higher (38.36 ± 26.57 mg kg−1) compared with the soils from module II (23.29 ± 18.94 mg kg−1) and module III (11.80 ± 2.68 mg kg−1) (Fig. 2(B)) (p ≤ 0.01). A recent investigation conducted by Adagunodo et al. (2018) reported that Nigerian soils associated with agricultural activities had the presence of Cr, Cd, Ni, V, Cu, Co, Sb, Pb, and Zn, with Zn being the one that exhibited the higher concentrations, with values of up to 61.30 mg kg−1, similar to the values reported in this study for agricultural soils (Fig. 2(B)). Additionally, Martin et al. (2017) found Cd, Cr, Cu, Hg, Ni, As, Pb, and Zn in agricultural soils from New Zealand, where Zn showed some of the highest concentrations with values up to 163 mg kg−1. Remarkably, all tested soils in the current study presented concentrations of Cu and Zn within the permissible ranges established by the Mexican regulation and by the World and Health Regulation (WHO) standards.
The highest values of Fe and Pb were found in module II, registering quantities of 1902.7 ± 332.2 mg kg−1 (p ≤ 0.01) and 23.48 ± 6.48 mg kg−1 (p ≤ 0.01), respectively (Fig. 2(C, D)). These values did not exceed the limits found in Mexican laws nor the established limits by WHO. Studies performed in different Mexican agricultural lands have also reported the presence of these and other heavy metals. For instance, Flores-Magdaleno et al. (2011) found that agricultural soils from the state of Hidalgo, in the central region of the country, had concentrations of heavy metal(oid)s Pb, Ni, Cd, As, Cr, and Hg up to > 2.5, < 1.40, < 0.20, > 0.10, > 0.016, and > 0.010 mg kg−1, respectively. A similar case was reported in Nigeria by Adagunodo et al. (2018), where they found that agricultural soils exhibited Pb in concentrations up to 43.89 mg kg−1. Another study performed by González et al. (2012), in an agricultural area in the state of Zacatecas, Mexico, located in the north-central region, found that Pb presented the highest concentrations (> 600 mg kg−1) compared with other analyzed heavy metals (González et al. 2012; Kamunda et al. 2016). In a study regarding the Juarez Valley, Cota-Ruiz et al. (2019) sampled four soils belonging to module I of this current study and they did not detect the presence of Zn nor Pb. These differences can be explained in terms of distance and location between their sampling sites and the ones presented in this study, since module I has an extension of over 3000 ha (INEGI 1999).
Additionally, the most elevated concentration of Mn was found in module III (392.43 ± 74.43 mg kg−1), compared with module II (326.96 ± 75.17 mg kg−1) and module I (231.55 ± 28.21 mg kg−1) (Fig. 2(E)) (p ≤ 0.01). Interestingly, it has been reported that Mn and Fe oxides may adsorb other heavy metals such as As and Cd (Suda and Makino 2016; Xu et al. 2017). Thus, the soils from module III could have more possibilities to reduce the heavy metal uptake by agricultural crops. However, functional experiments to demonstrate this are needed. In this study, none of the registered values for Mn concentration exceeded international standards.
The levels of Cd did not significantly differ among the tested modules (Fig. 2(F)) (p ≤ 0.05). The mean Cd levels in the three modules did surpass the values allowed by Mexican law (0.35 mg kg−1) based on crop tolerance, but not the values specified by the WHO (3 mg kg−1) for agricultural soils (Kamunda et al. 2016). It is important to note that in some cases, in the three modules, results exceeded the permissible values specified by WHO. In consequence, some of these agricultural soils represent a threat since Cd may accumulate in crops.
In the current study, the analyzed soils were classified as neutral to slightly alkaline and as saline to highly saline (Table 4). The pH of soils may induce the acquisition of heavy metals. For instance, it was previously reported that the transfer of Cd, Hg, Pb, Hg, Cu, and Zn from soil to shoots in grains of rice (Oryza sativa) was increased when the soils were acidic (Mao et al. 2019). This latter finding could indicate that the heavy metals in the soils of the Juarez Valley cannot be easily integrated into the trophic networks. However, plants like Acacia tortilis, Calotropis procera, Convovalus sp., Prosopis juliflora, Salsola sp., Ochradenus baccatus, Convolvulus sp., and Pergularia tomentosa are capable of absorbing As, Cd, Cu, Pb, and Zn from alkaline soils (Al-Farraj and Al-Wavel, 2007). Thus, many other variables, such as salinity, type of soil, plant species, among others, affect the mobility of heavy metals in soils (Acosta et al. 2011). Further heavy metal translocation studies using agricultural crops should be performed to determine their consequences to the living systems.
Conclusions
DDE and DDT were the most widespread agrochemicals in the Juarez Valley since they were found in 100% and 97% of the tested soils. Isodrin prevalence was higher in module I, as 60% of the sampled soils presented the pesticide. Meanwhile, endosulfan was detected in 60% of the soils in module III. The lowest occurrence was exhibited by DDD since it was detected in 20% and 10% of the tested soils from modules II and II, respectively. Although the concentration of all analyzed pesticides did not exceed the international regulations, their potential adverse effects cannot be discounted until more studies on biological systems have been done. As for heavy metals, Fe and Mn presented the highest concentrations in comparison with the rest of their analyzed counterparts (p ≤ 0.05). The heavy metals Fe, Mn, Zn, Pb, and Cu were found in all tested soils; however, they did not surpass the limits specified by Mexican legislation nor the ones by the WHO. The content of Cd in some analyzed soils did exceed the limits proposed by Mexican and international regulations. Given the physicochemical properties of the soils in the Valley, along with the potential adverse effects of xenobiotics to biological systems, more studies are needed to monitor the concentrations and to determine the fate of these compounds in the environment.
References
Acosta JA, Jansen B, Kalbitz K, Faz A, Martínez-Martínez S (2011) Salinity increases mobility of heavy metals in soils. Chemosphere 85:1318–1324. https://doi.org/10.1016/j.chemosphere.2011.07.046
Adagunodo TA, Sunmonu LA, Emetere ME (2018) Heavy metals’ data in soils for agricultural activities. Data Brief 18:1847–1855. https://doi.org/10.1016/j.dib.2018.04.115
Adeyeye A, Osibanjo O (1999) Residues of organochlorine pesticides in fruits, vegetables and tubers from Nigerian markets. Sci Total Environ 231:227–233. https://doi.org/10.1016/S0048-9697(99)00067-4
Afzal M, Shabir G, Iqbal S, Mustafa T, Khan Q, Khalid Z (2012) Assessment of heavy metal contamination in soil and groundwater at Leather Industrial Area of Kasur, Pakistan. Clean-Soil Air Water 42:1133–1139. https://doi.org/10.1002/clen.201100715
Al-Farraj AS, Al-Wavel MI (2007) Heavy metals accumulation of some plant species grown on mining area at Mahad AD’Dahab, Saudi Arabia. J Appl Sci 7:1170–1175. https://doi.org/10.3923/jas.2007.1170.1175
Androutsopoulos V, Hernandez AF, Leiesivuori J, Tsatsakis AM (2013) A mechanistic overview of health associated effects of low levels of organochlorine and organophosphorous pesticides. Toxicology 307:89–94. https://doi.org/10.1016/j.tox.2012.09.011
Cantú-Soto EU, Meza-Montenegro MM, Valenzuela-Quintanar AI, Félix-Fuentes A, Grajeda-Cota P, Balderas-Cortes JJ, Osorio-Rosas CL, Acuña-García G, Aguilar-Apodaca MG (2011) Residues of organochlorine pesticides in soils from the Southern Sonora, Mexico. Bull Environ Contam Toxicol 87:556–560. https://doi.org/10.1007/s00128-011-0353-5
Carpenter JW, Andrews GA, Beyer WN (2004) Zinc toxicosis in a free-flying trumpeter swan (Cygnus buccinator). J Wildl Dis 40:769–774. https://doi.org/10.7589/0090-3558-40.4.769
Carreira V, Gadsen BJ, Harrison TM, Braselton E, Fitzgerald S (2011) Pancreatic atrophy due to zinc toxicosis in two African ostriches (Struthio camelus). J Zoo Wildl Med 42:304–308. https://doi.org/10.1638/2010-0146.1
Chakraborty P, Zhang G, Li J, Sivakumar A, Jones K (2015) Occurrence and sources of selected organochlorine pesticides in the soil of seven major Indian cities: assessment of air-soil exchange. Environ Pollut 204:74–80. https://doi.org/10.1016/j.envpol.2015.04.006
Cota-Ruiz K, Hernández-Viezcas JA, Varela-Ramírez A, Valdés C, Núñez-Gastélum JA, Martínez-Martínez A, Delgado-Rios M, Peralta-Videa JR, Gardea-Torresdey JL (2018) Toxicity of copper hydroxide nanoparticles, bulk copper hydroxide, and ionic copper to alfalfa plants: A spectroscopic and gene expressionstudy. Environ Pollut 243:703–712. https://doi.org/10.1016/j.envpol.2018.09.028
Cota-Ruiz K, Lopez de los Santos Y, Hernandez-Viezcas JA, Delgado-Rios M, Peralta-Videa JR, Gardea-Torresdey JL (2019) A comparative metagenomic and spectroscopic analysis of soils from an international point of entry between the US and Mexico. Environ Int 123:558–566. https://doi.org/10.1016/j.envint.2018.12.055
Díaz-Barriga F, Trejo-Acevedo A, Betanzos AF, Espinosa-Reyes G, Alegría-Torres JA, Pérez IN (2012) Assessment of DDT and DDE levels in soil, dust, and blood samples from Chihuahua, Mexico. Arch Environ Contam Toxicol 65:351–358. https://doi.org/10.1007/s00244-011-9700-0
Dongiovanni P, Ruscica M, Rametta R, Recalcati S, Steffani L, Gatti S, Girelli D, Cairo G, Magni P, Fargion S, Valenti L (2013) Dietary iron overload induces visceral adipose tissue insulin resistance. Am J Pathol 182:2254–2236. https://doi.org/10.1016/j.ajpath.2013.02.019
FAO (2000) Assessing soil contamination. A reference manual. http://www.fao.org/3/X2570E/X2570E00.htm.
Flores-Magdaleno H, Mancilla-Villa O, Mejía-Saenz E, Olmedo-Bolaños M, Bautista-Olivas L (2011) Heavy metals in agricultural soils and Irrigation wastewater of Mixquiahuala, Hidalgo, Mexico. Afr J Agric Res 6:5505–5511. https://doi.org/10.5897/AJAR11.414
González O, Gómez-Bernal J, Ruíz-Huerta A (2012) Plants and soil contamination with heavy metals in agricultural areas of Guadalupe, Zacatecas, Mexico. In: Srivastra J (ed) Environmental Contamination. In Tech, Shanghai, China, pp 41–42
Grondona SI, Gonzalez M, Martínez DE, Massone HE, Miglioranza KS (2019) Assessment of organochlorine pesticides in phreatic aquifer of Pampean Region, Argentina. Bull Environ Contam Toxicol 102:544–549. https://doi.org/10.1007/s00128-019-02584-3
Han Y, Mo R, Yuan X, Zhong D, Tang F, Ye C, Liu Y (2017) Pesticide residues in nut-planted soils of China and their relationship between nut/soil. Chemosphere 180:42–47. https://doi.org/10.1016/j.chemosphere.2017.03.138
INEGI (1999) Estudio hidrológico del Estado de Chihuahua. National Institute of Statistics and Geography. Ciudad de Mexico, Mexico
INEGI (2003) Síntesis de Información Geográfica del Estado de Chihuahua. National Institute of Statistics and Geography. Ciudad de Mexico, Mexico
Kamunda C, Mathuthu M, Madhuku M (2016) Health risk assessment of heavy metals in soils from Witwatersrand Gold Mining Basin, South Africa. Int J Environ Res Public Health 13:663–673. https://doi.org/10.3390/ijerph13070663
King K, Zaun B, Schotborgh M, Hurt K (2003) DDE-Induced eggshell thinning in White-faced ibis: a continuing problem in the Western United States. Southwest Nat 48:356–364
Kulangaravalappil N, Chenicherry S (2018) Fate and distribution of organochlorine insecticides (OCIs) in Palakkad soil, India. Sust Environ Res 28:179–185. https://doi.org/10.1016/j.serj.2018.01.007
Kwon JC, Nejad Z, Jung M (2017) Arsenic and heavy metals in paddy soil and polished rice contaminated by mining activities in Korea. Catena 148:92–100. https://doi.org/10.1016/j.catena.2016.01.005
Leal SSD, Valenzuela QAI, Gutiérrez CML, Bermúdez AMC, García HJ, Aldana MML, Grajeda CP, Silveira GMI, Meza MMM, Palma DSA, Leyva GGN, Camarena GBO, Valenzuela NCP (2014) Residuos de plaguicidas organoclorados en suelos agrícolas. Terra Latinoam 32:1–11
Li Z (2018) Health risk characterization of maximum legal exposures for persistent organic pollutant (POP) pesticides in residential soil: An analysis. J Environ Manag 205:163–173. https://doi.org/10.1016/j.jenvman.2017.09.070
Liu O, Wang Q, Xu C, Shao W, Zhang C, Liu H, Jiang Z, Gu A (2017) Organochloride pesticides impaired mitochondrial function in hepatocytes and aggravated disorders of fatty acid metabolism. Sci Rep 7:46339–46350. https://doi.org/10.1038/srep46339
Mahmood A, Malik RN, Li J, Zhang G (2014) Human health risk assessment and dietary intake of organochlorine pesticides through air, soil and food crops (wheat and rice) along two tributaries of river Chenab, Pakistan. Food Chem Toxicol 71:17–25. https://doi.org/10.1016/j.fct.2014.05.008
Mao C, Song Y, Chen L, Ji J, Li J, Yuan X, Yango Z, Ayoko G, Frost R, Theiss F (2019) Human health risks of heavy metals in paddy rice based on transfer characteristics of heavy metals from soil to rice. Catena 175:339–348. https://doi.org/10.1016/j.catena.2018.12.029
Martin AP, Turnbull RE, Rissmann CW, Rieger P (2017) Heavy metal and metalloid concentrations in soils under pasture of southern New Zealand. Geoderma Reg 11:18–27. https://doi.org/10.1016/j.geodrs.2017.08.005
Meza-Figueroa D, de la O-Villanueva M, de la Parra ML (2007) Heavy metal distribution in dust from elementary schools in Hermosillo, Sonora, México. Atmos Environ 41:276–288. https://doi.org/10.1016/j.atmosenv.2006.08.034
Ming Y (2011) Environmental toxicology: biological and health effects of pollutants. CRC Press, Florida
Naghipour D, Davoud S, Tghavi K (2018) Data of heavy metals in soil and ground wáter at kiwi gardens of Amlash in Guilan Province, Iran. Data Brief 18:1556–1561. https://doi.org/10.1016/j.dib.2018.04.046
Palomo-Rodríguez M, Anaya SA, Rivera GM, Martínez RJG, Reyes GA (2013) Plaguicidas organoclorados en agua residual sometida a un proceso de dilución al mezclarse con fuentes alternativas de abastecimiento. Agrofaz 13:67–72
Qu C, Albanese S, Chen W, Lima A, Doherty AL, Piccolo A, Arienzo M, Qi S, de Vivo B (2016) The status of organochlorine pesticides contamination in the soils of the Campanian Plain, southern Italy, and correlations with soil properties and cancer risk. Environ Pollut 216:500–511. https://doi.org/10.1016/j.envpol.2016.05.089
Raloff J (1995) Beyond Estrogens. Why unmasking hormone-mimicking pollutants proves so challenging. Sci News 148:44–47. https://doi.org/10.2307/3979492
Rosenbaum PF, Weinstock RS, Silverstone AE, Sjödin A, Pavuk M (2017) Metabolic syndrome is associated with exposure to organochlorine pesticides in Anniston, AL, United States. Environ Int 108:11–21. https://doi.org/10.1016/j.envint.2017.07.017
Salihovic S, Ganna A, Fall T, Broeckling CD, Perenni JE, van Bavel B, Lind PM, Ingelsson E, Lind L (2016) The metabolic fingerprint of p,p′-DDE and HCB exposure in humans. Environ Int 88:60–66. https://doi.org/10.1016/j.envint.2015.12.015
Stockholm Convention (2004) Stockholm Convention on persistent organic pollutions (POPs). http://www.pops.int/TheConvention/Overview/TextoftheConvention/tabid/2232/Default.aspx.
Suda A, Makino T (2016) Functional effects of manganese and iron oxides on the dynamics of trace elements in soils with a special focus on arsenic and cadmium: a review. Geoderma 270:68–75. https://doi.org/10.1016/j.geoderma.2015.12.017
Sun K, Zhao Y, Gao B, Lio X, Zhang Z, Xing B (2009) Organochlorine pesticides and polybrominated diphenyl ethers in irrigated soils of Beijing, China: levels, inventory and fate. Chemosphere 77:1199–1205. https://doi.org/10.1016/j.chemosphere.2009.09.016
Tan W, Gao Q, Deng C, Wang Y, Lee W-L, Hernández-Viezcas JA, Peralta-Videa JR, Gardea-Torresdey JL (2018) Foliar exposure of Cu (OH)2 nanopesticide to basil (Ocimum basilicum): variety-dependent copper translocation and biochemical responses. J Agric Food Chem 66:3358–3366. https://doi.org/10.1021/acs.jafc.8b00339
Tarcau D, Cucu-Man S, Burukova J, Klanova J, Covaci A (2013) Organochlorine pesticides in soil, moss and tree-bark from North-Eastern Romania. Sci Total Environ 456–457:317–324. https://doi.org/10.1016/j.scitotenv.2013.03.103
Tsai WT (2010) Current status and regulatory aspects of pesticides considered to be persistent organic pollutants (POPs) in Taiwan. Int J Environ Res Public Health 7:3615–3627. https://doi.org/10.3390/ijerph7103615
Udeigwe TK, Teboh JM, Eze PN, Stietiya MH, Kumar V, Hendrix J, Mascagni JHJ, Ying T, Kandakji T (2015) Implications of leading crop production practices on environmental quality and human health. J Environ Manag 151:267–279. https://doi.org/10.1016/j.jenvman.2014.11.024
US EPA (2016) DDT regulatory history: a brief survey (to 1975). U.S. Environmental Protection Agency, Washington, D.C.
Vella V, Malaguarnera R, Lappano R, Maggilioni M, Belfiore A (2017) Recent views of heavy metals as possible risk factors and potential preventive and therapeutic agents in prostate cancer. Mol Cell Endocrinol 457:57–72. https://doi.org/10.1016/j.mce.2016.10.020
Wong F, Alegria H, Jantunen L, Bidleman T, Salvador-Figueroa M, Gold-Bouchot G, Ceja-Moreno V, Waliszewsky S, Infanzo R (2008) Organochlorine pesticides in soils and air of southern Mexico: chemical profiles and potential for soil emissions. Atmos Environ 42:7737–7745. https://doi.org/10.1016/j.atmosenv.2008.05.028
Xiao R, Wang S, Li R, Wang J, Zhang Z (2017) Soil heavy metal contamination and health risks associated with artisanal gold mining in Tongguan, Shaanxi, China. Ecotoxicol Environ Saf 141:17–24. https://doi.org/10.1016/j.ecoenv.2017.03.002
Xu X, Chen C, Wang P, Kretzschmar R, Zhao FJ (2017) Control of arsenic mobilization in paddy soils by manganese and iron oxides. Environ Pollut 231:37–47. https://doi.org/10.1016/j.envpol.2017.07.084
Yorifuji T, Tsuda T, Inoue S, Takao S, Harada M (2011) Long-term exposure to methylmercury and psychiatric symptoms in residents of Minamata, Japan. Environ Int 37:907–913. https://doi.org/10.1016/j.envint.2011.03.008
Zang F, Wang S, Nan Z, Nan Z, Ma J, Zhang Q, Chen Y, Li Y (2017) Accumulation, spatio-temporal distribution, and risk assessment of heavy metals in the soil-corn system around a polymetallic mining area from the Loess Plateau, Northwest China. Geoderma 305:188–196. https://doi.org/10.1016/j.geoderma.2017.06.008
Zhang A, Chen Z, Ahrens L, Liu W, Li Y-F (2012) Concentrations of DDTs and enantiomeric fractions of chiral DDTs in agricultural soils from Zhejiang Province, China, and correlations with total organic carbon and pH. J Agric Food Chem 60:8294–8301. https://doi.org/10.1021/jf3024547
Zhang A, Luo E, Sun J, Xiao H, Liu W (2015) Distribution and uptake pathways of organochlorine pesticides in greenhouse and conventional vegetables. Sci Total Environ 505:1142–1147. https://doi.org/10.1016/j.scitotenv.2014.11.023
Zhou Q, Wang J, Meng B, Cheng J, Lin G, Chen J, Zheng D, Yu Y (2013) Distribution and sources of organochlorine pesticides in agricultural soils from central China. Ecotoxicol Environ Saf 93:163–170. https://doi.org/10.1016/j.ecoenv.2013.03.029
Acknowledgments
J.A.N-G is grateful to Universidad Autónoma de Ciudad Juárez for providing facilities for the realization of this study. S.R-C wish to thank CONACYT for the master degree scholarship granted.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Philipp Gariguess
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Núñez-Gastélum, J.A., Hernández-Carreón, S., Delgado-Ríos, M. et al. Study of organochlorine pesticides and heavy metals in soils of the Juarez valley: an important agricultural region between Mexico and the USA. Environ Sci Pollut Res 26, 36401–36409 (2019). https://doi.org/10.1007/s11356-019-06724-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-06724-4