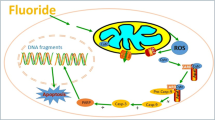
Abstract
Long-term occupational exposure to low level of fluoride can induce oxidative stress and apoptosis in many cells, including lymphocyte. However, the underlying mechanism remains unclear. Hence, this study was designed to explore the potential oxidative stress and apoptosis of long-term occupational exposure to low level of fluoride in aluminum smelter workers. A total of 120 aluminum smelter workers were recruited in control, low-, middle-, and high-fluoride exposure groups with 30 workers for each group. The peripheral blood samples were collected, centrifuged, and isolated to obtain serum and lymphocyte suspensions. The air and serum fluoride concentrations were detected by fluoride ion-selective electrode method. The lymphocytic apoptosis rate, DNA damage, oxidative stress, and mRNA levels of p53, Bcl-2, and Bax were assessed by Annexin V/PI staining, comet assay, attenuated total reflectance Fourier transform infrared spectroscopy and real-time polymerase chain reaction, respectively. Results showed that the air and serum fluoride concentrations of fluoride-exposed groups were higher than those of the control group (p < 0.05). Fluoride exposure might induce apoptosis, DNA damage and oxidative stress in a dose-dependent manner in lymphocytes (p < 0.05). The expression levels of p53 and Bax were increased with fluoride exposure in lymphocytes (p < 0.05), whereas the Bcl-2 expression was decreased but not significantly. Taken together, these observations indicate that long-term occupational exposure to low level of fluoride may lead to oxidative stress and induce apoptosis through the p53-dependent pathway in peripheral blood lymphocytes of aluminum smelter workers. Serum fluoride level may be the potential biomarker of fluoride exposure.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fluorine, a gaseous trace element, is an important natural and industrial environmental pollutant, which can also enter the human body and spread over the peripheral blood from drinking water, air or by skin contact (Manahan 2017; Whitford 1983). This element is used over several decades to prevent dental caries (Garcia et al. 2017). However, excessive fluoride exposure probably elicit the toxic effect in various tissues and organs, including the brain (Ge et al. 2018), reproductive system (Adedara et al. 2017), spleen (Ma et al. 2017), immune system (Singh et al. 2017), erythrocytes (Campos-Pereira et al. 2017), and lymphocytes (Liu et al. 2013). Notably, fluoride can be released from electrolytic aluminum industry during processing, although controlled within the hygienic standard (Søyseth et al. 2015). Nevertheless, long-term exposure to low level of fluoride could induce oxidative stress, genotoxicity, and apoptosis in many cell lines (Ribeiro et al. 2017), including lymphocytes (Jothiramajayam et al. 2014b), but the underlying mechanism remains unclear. Peripheral blood lymphocytes (PBLs) play an important role in immune function (Abbas et al. 2014; Xia et al. 2016). Therefore, in the present study, PBLs were isolated from the peripheral blood of aluminum smelter workers and used to investigate the underlying mechanism of fluoride-induced oxidative stress and apoptosis.
Apoptosis is a cell death mechanism characterized with cell shrinkage, membrane blebbing, chromatin condensation, oligonucleosomal DNA fragmentation, and apoptotic body formation (Sharma et al. 2017). Fluoride can inhibit cell viability and induce DNA damage as a result of apoptosis (Song et al. 2015; Suzuki et al. 2015). In addition, DNA damage can be caused by oxidative stress which leads to DNA modification and apoptosis (Muratori et al. 2015; Skipper et al. 2016). Similarly, fluoride may induce ameloblast apoptosis and dental fluorosis by oxidative stress and upregulate caspase pathway (Li et al. 2017). Therefore, in the present study, oxidative stress was evaluated by attenuated total reflectance Fourier-transform infrared (ATR-FTIR) spectroscopy (Barraza-Garza et al. 2016; Benseny-Cases et al. 2014; Schwarzbacherová et al. 2017; Simoncicova et al. 2018). ATR-FTIR is a novel technology which can detect subtle changes in the DNA/RNA, glycogen, protein, carbohydrate, lipid, and other substances in a specific wavelength (German et al. 2006). ATR-FTIR is an advanced technique in studying toxicological mechanisms due to its higher sensitivity and specificity than traditional toxicology research techniques (Pang et al. 2012). Oxidative stress is an important mechanism of fluoride-induced DNA damage in rat (Umarani et al. 2015). Notably, DNA damage induces apoptotic protein p53 phosphorylation and triggers apoptosis (Demoulin et al. 2015).
Tumor suppressor protein p53 is a nuclear transcription factor which induces apoptosis by upregulating the transcription of many proapoptotic genes and downregulating antiapoptotic genes (Zhang et al. 2015). Fluoride inhibits hepatocellular proliferation and induces apoptosis via p53 pathways in mice, thereby, indicating that fluoride induces apoptosis via p53 pathways in cells (Liu et al. 2018). Moreover, Bax is the main proapoptotic gene targeted by p53 which induces apoptosis (Liao et al. 2016). By contrast, the antiapoptotic protein Bcl-2, which is the antiapoptotic gene targeted by p53, can block intrinsic apoptosis by binding to Bax (Li et al. 2015). Fluoride exposure can increase the mRNA expression levels of Bax, decrease the mRNA expression level of Bcl-2, increase the expression rate of Bax/Bcl-2 and consequently induce apoptosis in PC12 cells (Liao et al. 2017). Nonetheless, the roles of DNA damage, oxidative stress and the changes of related apoptotic gene expression in fluoride induces PBLs apoptosis remain unclear.
Hence, we hypothesized that long-term exposure to low level of fluoride induces apoptosis via p53 pathway in PBLs of aluminum smelter workers, and serum fluoride levels may be a reliable peripheral biomarker for fluoride exposure. To address our hypothesis, firstly, environmental exposure and internal exposure concentrations of fluoride, including the air and serum fluoride concentrations, were determined by fluoride ion-selective electrode method. Secondly, PBLs apoptosis, DNA damage and oxidative stress were assessed by Annexin V/PI staining, comet assay and ATR-FTIR spectroscopy, respectively. Thirdly, the mRNA expression levels of p53, Bax, and Bcl-2 genes were assessed by real-time polymerase chain reaction (RT-PCR). Finally, on the basis of the results of the comprehensive study, we elucidated the underlying mechanisms of fluoride-induced apoptosis in PBLs.
Materials and methods
Subjects
A total of 120 male aluminum smelter workers were included in this study. According to different types of work, the workers were divided into four groups, including control, low-, middle-, and high-fluoride exposure groups, with 30 workers for each group. The main inclusion criteria for participation were workers with no underlying health problems, cardiovascular diseases, infection history, or X-ray exposure within the last 6 months. The exclusion criteria included a medication history and drug use in the past 2 weeks and unwillingness to participate in interviews. All participants completed a comprehensive questionnaire which included demographic information, smoking status, drinking status, occupational history, and disease history. This study was approved by the ethics and human subject committee of Guangxi Medical University (No. 20110120-5), and written informed consent was obtained from each participant.
Sample collection and fluoride concentration detection
Peripheral vein blood samples (5 mL) were collected from each worker during the same period. Normal heparin was used as anticoagulant. Serum was separated via centrifugation and stored in the refrigerator at 4 °C. PBLs were isolated from the remaining suspension by lymphocyte separation liquid, adjusted to 1 × 105 cells/mL by adding appropriate amount of RPMI1640 solution and stored in a refrigerator at 4 °C.
Air samples were collected from the workshops of electrolysis (high-exposed group), casting (middle-exposed group), thermoelectricity (low-exposed group), and mining plant (control group). Four sampling points (three replicates/point) were distributed equidistantly along the production line in each workshop and placed at 1.5 m above the ground. Air samples were collected in acetic acid-nitrocellulose microporous membranes with Gilian GilAir-5 air sampling pumps with an air flow of 5 L/min for 60 min. Air samples were collected three times a day at the same sampling point during the same time period for two successive days.
In order to characterize the exposure level, serum, and air fluoride concentrations were determined by fluoride ion selective electrode method.
Annexin V/PI staining
Apoptosis was determined using Annexin V/PI double-staining assay (KeyGen Biotech., Co., Ltd., Nanjing, China) by flow cytometry. As describing in the kit specification, the PBLs were resuspended with 500 μL of binding buffer and subsequently incubated with 5 μL of Annexin V and 5 μL of PI for 15 min in the dark. Live, early apoptotic, late apoptotic, and dead cells were detected by flow cytometry (CellQquest, BD Company, USA), and the results are shown by 2D plots. The total apoptosis rate was calculated by the following formula: total apoptosis rate (%) = early apoptosis rate + late apoptosis rate.
Comet assay
DNA damage, that is, DNA double-strand break, was assessed by comet assay (Botta et al. 2017). Briefly, 100 μL of PBL suspension was mixed with 100 μL of low-melting agarose and dropped onto comet slides covered with 1% normal melting agarose. Subsequently, the comet slides were placed at 4 °C, immersed in prechilled lysis buffer in the dark for 2 h and immersed in unwinding alkaline solution at 4 °C in the dark for 20 min. Electrophoresis was conducted in neutral comet assay buffer for 30 min (300 mA, 25 V) at room temperature. After electrophoresis, the slides were neutralized, dried and stained with 2 μg/mL ethidium bromide for 5 min in the dark. Digital images were captured with an Olympus BX61 fluorescence microscope. At least 100 cells were analyzed for each sample by using CASP software (CaspLab, Wroclaw, Poland). The comet rate, percentage of DNA in the tail, tail length, and the olive tail moment (OTM) were used as the parameters of DNA damage.
ATR-FTIR spectroscopy
ATR-FTIR spectroscopy was applied to examine changes in lymphocyte molecular biochemical structure as a result of their exposure to fluoride. As Pang described (Pang et al. 2012), PBLs suspension was fixed with 70% ethanol for 30 min. The supernatant was removed, and the suspension was applied to low-E reflective glass slides (1 cm × 1 cm). The slides were scanned by FTIR spectroscopy (100/FTIR Spectrometer, Perkin Elmer Corporation, USA). The derived spectrum was smoothened with OPUS6.5 software (Bruker Optics Corporation, USA). Amide I (1650 cm−1) was used as the benchmark prior to performing principal component analysis (PCA) and linear discriminant analysis (LDA) by Matlab R2012b (Math Works Corporation, USA).
RT-PCR
As Liao described (Liao et al. 2017), the primers for p53, Bcl-2, Bax, and Gapdh were described as follows: p53 forward, 5′-GTTTCCGTCTGGGCTTCTTG-3′ and p53 reverse, 5′-CACAACCTCCGTCATGTGCT-3′ (197 bp); Bcl-2 forward, 5′-ATCGCCCTGTGGATGACTGAG-3′ and Bcl-2 reverse, 5′-CAGCCAGGAGAA ATCAAACAGAGG-3′ (129 bp); Bax forward, 5′-GGACGAACTGGACAGTAACATGG-3′ and Bax reverse, 5′-GCAAAGTAGAAA AGGGCGACAAC-3′ (150 bp); and Gapdh forward, 5′-GCACCGTCAAGGCTGAGAAC-3′ and Gapdh reverse, 5′-TGGTGAAGACGCCAGTGGA-3′ (138 bp). The Gapdh level was used as internal standard to normalize the expression of target genes. RT-PCR was performed in a BIO-RAD CFX96 PCR System (Bio-Rad, USA) with SYBR® PrimeScript™ RT-PCR Kit II (Takara Biotech, Japan).
Statistical analysis
Data were analyzed using SPSS version 16.0 for Windows (SPSS Inc.). The results are presented as mean ± SD. Simple ANOVA, Dunnett’s T3, or χ2 test was used to assess the differences between control and fluoride-exposed groups. Pearson correlation was obtained to determine the linear relationship when the data were normally distributed; otherwise, Spearman’s rank correlation was used. Spectrum pretreatment and multivariable analysis (PCA and LDA) were performed using OPUS6.5, Matlab R2012b and SAS 9.4 Software (statistical analysis system). The significance level of data was set at p < 0.05.
Results
Demographic characteristics were similar among the workers
In order to well understand workers’ general situation, demographic characteristics were investigated by the structured questionnaire. As shown in Table 1, this study included 120 aluminum smelter workers, and all participants completed the structured questionnaire. No statistical difference was observed among the age, seniority, smoking, drinking, and education levels in each group (p > 0.05), indicating that possible confounding influence is balanced in each group.
Increased fluoride concentrations in air and serum
To determine the factory environment pollutant level and internal level, the air and serum fluoride concentrations were detected by fluoride ion selective electrode method, presented in Fig. 1. The air fluoride concentrations were higher in the fluoride-exposed groups than that in the control group (p < 0.05). The serum fluoride concentrations were increased in a dose-dependent manner with the increase in fluoride exposure levels. The serum fluoride concentrations were higher in the middle- and high-exposed groups than that in the control group (p < 0.05). Positive correlations were observed between serum fluoride concentrations and air fluoride concentrations (r = 0.650, p < 0.05). The results show that the serum fluoride concentration rises at different extent which might due to long-term exposure to fluoride in aluminum smelter workers.
The fluoride concentrations in the air and serum. Samples were determined by fluoride ion-selective electrode method. The results were analyzed with simple ANOVA. a Air fluoride concentrations, the air fluoride concentrations of control group were under detection limit (0.9 μg/m3), data represent mean ± SD, n = 24 for each group. b Serum fluoride concentrations, data represent mean ± SD, n = 30 for each group. *p < 0.05, as compared with the control group; #p < 0.05, as compared with the low-exposed group; +p < 0.05, as compared with the middle-exposed group
Fluoride induced apoptosis in PBLs of aluminum smelter workers
As increased serum fluoride was detected as environmental exposure level, PBLs might be influenced. So apoptosis level in PBLs was detected by Annexin V/PI double-staining assay. The apoptosis rate was slightly higher in the high-exposed group than that in the other groups (p < 0.05, Fig. 2). Positive correlations were observed between the apoptosis rate in PBLs and serum fluoride concentration of aluminum smelter workers (r = 0.331, p < 0.05). These results indicate that elevated apoptosis level in PBLs maybe attribute to long term-exposure to fluoride in aluminum smelter worker. Fluoride induced DNA damage in PBLs of aluminum smelter workers.
Fluoride induced apoptosis in PBLs of aluminum smelter workers. Samples were determined using Annexin V/PI double-staining assay by flow cytometry and the results were analyzed with simple ANOVA. a Total apoptosis of PBLs in each treated group. Data represent mean ± SD, n = 30 for each group. *p < 0.05, as compared with the control group; #p < 0.05, as compared with the low-exposed group; +p < 0.05, as compared with the middle-exposed group. b Representative experiment of apoptosis. Early apoptotic cells (AV+/PI−) were in the lower right quadrant and late apoptotic/necrotic cells (AV+/PI+) in upper right quadrant
DNA damage is increased as a result of apoptosis due to fluoride exposure, so the effect of fluoride on DNA damage was detected by comet assay. The DNA percentages in the tail and comet rate were higher in the middle- and high-exposed groups than those in other groups (p < 0.05, Fig. 3a and b, respectively). The tail length and OTM were higher in high-exposed group than those in the other groups (p < 0.05, Fig. 3b and c, respectively). The serum fluoride concentrations showed significant positive correlation with the percentage of DNA in the tail, tail length, OTM, and comet rate; and the correlation coefficients are 0.525, 0.494, 0.493, and 0.622 (all p < 0.05), respectively. Similar, positive correlations were observed between the comet rate and the apoptosis rate in PBLs of aluminum smelter workers (r = 0.709, p < 0.05). These results indicated that effect of fluoride on DNA damage in PBLs was obvious and might be related to the serum fluoride and apoptosis.
Fluoride induced DNA damage in PBLs of aluminum smelter workers. Samples were determined by comet assay. The results were analyzed with simple ANOVA and χ2 test. The percentage of DNA in the tail (a), tail length (b), olive tail moment (c), and comet rate (d) were used to evaluate DNA damage. The comet rate was equal to total number of comet cells in each group divided by total number of cells observed corresponding group. Data represent mean ± SD, n = 30 for each group. *p < 0.05, as compared with the control group; #p < 0.05, as compared with the low-exposed group; +p < 0.05, as compared with the middle-exposed group. e Representative experiment of comet assay. The comet images were obtained by epifluorescence microscopy (× 100)
ATR-FTIR analysis showed structural changes or content in DNA and amide in PBLs
Spectral analysis
As a novel method to detect subtle biochemical changes at sub-cytotoxic and sub-genotoxic concentrations, ATR-FTIR was applied to detect PBLs’ oxidative stress, including protein oxidation and DNA damage (Barraza-Garza et al. 2016; Benseny-Cases et al. 2014; Schwarzbacherová et al. 2017; Simoncicova et al. 2018). ATR-FTIR spectroscopy showed remarkable differences in the metabolic fingerprint between the control and fluoride-exposed groups. The peaks at 953, 993, 1020, 1065, 1121, 1400, 1255, and 1650 cm−1 were decreased with the increase in the concentration of fluoride (Fig. 4). Spectrum variations may suggest that the changes in the molecular structures or contents of cytoplasmic protein phosphorylation (953 cm−1), glycogen (993 and 1020 cm−1), DNA/RNA (1065 and 1121 cm−1), protein (1400 cm−1), amide III (1255 cm−1), and amide I (1650 cm−1) bands were determined by the presence of fluoride. These results indicated that hydrogen or conjugated bonds may be destroyed by fluoride in PBLs of aluminum smelter workers.
PCA and LDA analyses
To determine clearly the spectral features, multivariate dimensionality analysis was performed between PCA and LDA (Table 2). The cumulative contribution ratios of the top two principal components (PC) were PC 1 (92.93%) and PC 2 (3.68%) for 96.61% of the spectral features and information, which can represent the main biochemical changes in PBLs exposed to fluoride. The PC1 score was higher in the fluoride-exposed groups than that in the control group (p < 0.05, Table 2), the PC2 score also showed the same trend, but it was not significant. The 2D scattergram displaying the PCA-LDA scores in each group is shown in Fig. 5. The medians of the PCA-LDA1 scores were decreased in aluminum smelter worker following long-term exposure in the control, low-, middle-, and high-exposed groups (0.84, 0.10, −0.39, and − 0.78, respectively). The PC1 was mainly due to changes in the structure or content of DNA/RNA, glycogen, symmetric phosphate/DNA, and cytoplasmic protein in PBLs, these results were consistent with comet assay results. The PC2 mainly attributed to changes in amide I–III and the ratios of 1630 cm−1/1650 cm−1 were 0.97, 0.94, 0.91, and 0.89 in the high-, middle-, low-exposed groups, and control group, related to carbonyl groups(C=O) vibration and protein aggregation which closely related to protein oxidation. Thus, fluoride-induced oxidative stress in PBLs might due to PC1 and PC2.
Scatter gram of the PCA-LDA scores of the PBLs of fluoride-exposed workers. The spectrum data was analyzed by reducing dimension with PCA-LDA. n = 30 for each group. The black, blue, green, and red dashed lines represent the median of the PCA-LDA 1 scores of the high-, middle-, and low-exposed group and control group, respectively
Apoptosis-related gene expression levels rose in PBLs
In order to furtherly investigate fluoride-induced apoptosis, we detected apoptosis-related gene expression levels, including p53, Bax, and Bcl-2, with RT-PCR. Fluoride exposure slightly increases the expression levels of p53 and Bax in the PBLs of high-exposed workers, as compared with the other groups (p < 0.05, Fig. 6a and b). The Bcl-2 expression levels were decreased in fluoride-exposed groups compared with that in the control group, but the decrease was insignificant (Fig. 6c). The Bax/Bcl-2 expression ratio was higher in high-exposed group than those in other groups (p < 0.05, Fig. 6d). Moreover, significant correlations were observed between the expression levels of p53 and Bcl-2 or Bax (r = 0.959, r = −0.969, respectively; p < 0.01). The total apoptosis rate had a positive correlation to the expression levels of p53 and Bax (r = 0.991, r = 0.972, respectively; p < 0.01). Above results indicated that fluoride might induce apoptosis via p53 pathway in PBLs of aluminum smelter workers.
The mRNA expression of p53, Bax, and Bcl-2 genes in PBLs of fluoride-exposed workers. Samples were determined by RT-PCR. The results were analyzed with simple ANOVA. The levels of p53 (a), Bax (b), Bcl-2 (c), normalized by Gapdh mRNA, and the Bax/Bcl-2 ratio (d) are expressed as fold changes in figure. Data represent mean ± SD, n = 30 for each group. *p < 0.05, as compared with the control group; #p < 0.05, as compared with the low-exposed group; +p < 0.05, as compared with the middle-exposed group
Discussion
Oxidative stress and apoptosis of fluoride have received increased interest within the scientific community because of occupational exposure and aluminum fluoride used in aluminum smelter industry. However, the air fluoride concentration levels in the aluminum smelter industry are relatively low and controlled in accordance with the relevant standards. In addition, the increasing fluoride doses in air pollution cannot be ignored, especially in occupational environments. In consideration of the influence of fluoride on health results from a long-term accumulation process, the present study was designed to explore the potential oxidative stress and apoptosis in PBLs of aluminum smelter workers following long-term occupational fluoride exposure. We found that long-term occupational exposure to low levels of fluoride may lead to oxidative stress and induce apoptosis through the p53-dependent pathway in PBLs of aluminum smelter workers.
In this study, the air fluoride concentrations were higher in the fluoride-exposed groups than that in the control group and decreased with the increase in distance from the electrolysis workshop (high-fluoride exposure group). Nevertheless, all concentrations were less than 0.5 mg/m3 and controlled in accordance with the relevant standards (Zhang and Cao 1996). Therefore, the potential oxidative stress and apoptosis of long-term occupational exposure to low level of fluoride were carried out in PBLs of aluminum smelter workers. Consequently, the serum fluoride concentrations were increased significantly in a dose-dependent manner with the increase in fluoride exposure levels. Positive correlations were observed between serum fluoride concentrations air fluoride concentrations. These results indicated that serum fluoride levels may be the potential biomarker of occupational fluoride exposure, and the peripheral blood may be one of the primary regions where fluoride accumulates. It is noteworthy that fluoride exposure may also mediate oxidative stress and apoptosis in peripheral blood cells (Jothiramajayam et al. 2014a; Rocha-Amador et al. 2011). Similarly, our study showed that PBLs were sensitive to fluoride exposure. The total apoptosis rate was increased in a dose-dependent manner with the increase in fluoride exposure levels in PBLs. Positive correlations were observed between the apoptosis rate in PBLs and serum fluoride concentrations. These results suggested that the raised serum fluoride levels may play a role in elevating apoptosis level in PBLs of aluminum smelter worker following long-term fluoride exposure.
Apoptosis is a normal physiological activity of the body that maintains environmental stability (Redza-Dutordoir and Averill-Bates 2016). Nonetheless, significantly high or low apoptosis can exert a negative effect on the body (Sharma et al. 2017). DNA damage is increased as a result of apoptosis due to fluoride exposure (Song et al. 2015; Suzuki et al. 2015). Therefore, we further investigated the underlying mechanism of potential fluoride-induced oxidative stress and apoptosis in PBLs. Our study showed that DNA strand breaks in PBLs may be caused by fluoride exposure. Positive correlations were observed between apoptosis and DNA strand breaks which suggested that DNA strand breaks may be a reason for fluoride-induced apoptosis in PBLs. Notably, oxidative damage is an important mechanism of fluoride-induced DNA damage (Muratori et al. 2015; Skipper et al. 2016). In the present study, ATR-FTIR was used to evaluate the oxidative damage and genotoxicity by detecting subtle changes in cytoplasmic protein phosphorylation, glycogen, DNA/RNA, protein, amide III, and amide I (Martin et al. 2007; Pang et al. 2012; Schwarzbacherová et al. 2017). ATR-FTIR spectroscopy results showed that the peaks at 953, 993, 1020, 1065, 1121, 1400, 1255, and 1650 cm−1 were decreased with the increased fluoride-exposure concentration. The spectral variations may suggest that the changes in the molecular structures or contents of cytoplasmic protein phosphorylation (953 cm−1), glycogen (993 and 1020 cm−1), DNA/RNA (1065 and 1121 cm−1), protein (1400 cm−1), amide III band (1255 cm−1), and amide I (1650 cm−1) bands were determined by the presence of fluoride; these results indicated that hydrogen or conjugated bonds may be destroyed in different degrees (Pang et al. 2012). Additionally, to determine the spectral features, multivariate dimensionality analysis was performed between PCA and LDA. The medians of the PCA-LDA1 scores were higher in the control group than those in the fluoride-exposed groups, thereby indicating that PC 1 was mainly due to the changes in the structure or content of DNA/RNA, glycogen, symmetric phosphate/DNA and cytoplasmic protein in PBLs (Trevisan et al. 2010). The PC2 mainly attributed to changes in amide I–III which represented carbonyl groups(C=O) stretching vibration and protein aggregation that closely related to protein oxidation (Schwarzbacherová et al. 2017; Simoncicova et al. 2018). Fluoride can induce oxidative stress and genotoxicity in PBLs. Hence, oxidative stress may be an important mechanism of fluoride-induced DNA damage and apoptosis in PBLs of aluminum smelter worker.
Previous studies supported our findings by showing that fluoride exposure can induce apoptosis and change the mRNA expression levels of p53, Bcl-2, or Bax in different cell types (Liao et al. 2017; Liu et al. 2018; Tu et al. 2018). Apoptotic protein p53 can be accumulated and activated, which can also trigger cell cycle arrest and apoptosis induced by DNA damage (Karimian et al. 2016). In our study, the increasing mRNA expression levels of p53 were correlated with the apoptosis and comet rate in fluoride-exposed groups. Results suggested that apoptotic protein p53 may be involved in the mechanism of fluoride-induced apoptosis in PBLs. Apoptotic protein p53 mediates apoptosis by upregulating the transcription of many proapoptotic genes and downregulating antiapoptotic genes (Zhang et al. 2015). Bcl-2 family members, that is, downstream proteins of p53 pathways, exhibit crucial functions in apoptosis, and they can be divided into antiapoptotic and proapoptotic proteins according to their structural and functional characteristics (Moldoveanu et al. 2014; Siddiqui et al. 2015). The mechanism of antiapoptotic protein Bc1-2 may prevent the activation of apoptosis-related genes by inhibiting DNA transcription (Upadhyay et al. 2003). Alternatively, such mechanism inhibits the functions of apoptosis-related genes (Upadhyay et al. 2003). The Bax function is opposite to that of Bcl-2, and Bax can form heterodimers with Bcl-2 (Moldoveanu et al. 2014). Thus, Bcl-2 is inactivated, thereby promoting apoptosis. The present results showed that fluoride exposure may slightly increase the mRNA expression levels of Bax, decrease the mRNA expression levels of Bcl-2 and increase the expression of Bax/Bcl-2 ratio in PBLs. These results are consistent with the findings reported in the literature (Liao et al. 2017). The Bax/Bcl-2 ratio may be used as the evaluating index to determine the apoptosis process (Aboutaleb et al. 2015; Zhang et al. 2014). Moreover, significant correlations were observed between the expression levels of p53 and Bcl-2 or Bax. The total apoptosis rate was positively associated with the expression levels of p53 and Bax. These results suggested that the fluoride-induced apoptosis of PBLs may be mediated with the upregulated expression levels of p53, Bax, and Bax/Bcl-2 ratio.
In conclusion, these cell experiments were carried out in fluoride-exposed working population. Therefore, the results may indicate the toxic effects of fluoride on the body. Evidence presented herein showed that oxidative stress may mediate fluoride-induced apoptosis through a p53-dependent pathway in PBLs. Our results also demonstrated that the expression levels of p53, Bax, and Bax/Bcl-2 ratio were increased significantly with the increase in fluoride exposure levels in PBLs. Long-term occupational exposure to low level of fluoride might also cause genotoxicity, oxidative stress, DNA damage and apoptosis in PBLs. Taken together, these findings suggested that in vivo occupational exposure to fluoride may resulted in oxidative damage and induced apoptosis through a p53-dependent pathway in PBLs. p53-dependent apoptosis mediated by DNA damage may play a role in fluoride-induced apoptosis of PBLs. Furthermore, serum fluoride levels may be a potential biomarker of fluoride exposure.
References
Abbas AK, Lichtman AH, Pillai S (2014) Cellular and molecular immunology E-book. Elsevier Health Sciences, Philadelphia, pp 544
Aboutaleb N, Shamsaei N, Khaksari M, Erfani S, Rajabi H, Nikbakht F (2015) Pre-ischemic exercise reduces apoptosis in hippocampal CA3 cells after cerebral ischemia by modulation of the Bax/Bcl-2 proteins ratio and prevention of caspase-3 activation. J Physiol Sci 65:435–443
Adedara IA, Olabiyi BF, Ojuade TD, Idris UF, Onibiyo EM, Farombi EO (2017) Taurine reverses sodium fluoride-mediated increase in inflammation, caspase-3 activity, and oxidative damage along the brain–pituitary–gonadal axis in male rats. Can J Physiol Pharmacol 95:1019–1029
Barraza-Garza G, Castillo-Michel H, de la Rosa LA, Martinez-Martinez A, Perez-Leon JA, Cotte M, Alvarez-Parrilla E (2016) Infrared spectroscopy as a tool to study the antioxidant activity of polyphenolic compounds in isolated rat enterocytes. Oxidative Med Cell Longev 2016:9245150
Benseny-Cases NR, Klementieva O, Cotte M, Ferrer I, Cladera J (2014) Microspectroscopy (μFTIR) reveals co-localization of lipid oxidation and amyloid plaques in human Alzheimer disease brains. Anal Chem 86:12047–12054
Botta L, Brunori F, Tulimieri A, Piccinino D, Meschini R, Saladino R (2017) Laccase-mediated enhancement of the antioxidant activity of propolis and poplar bud exudates. ACS Omega 2:2515–2523
Campos-Pereira FD, Lopes-Aguiar L, Renosto FL, Nogueira GAS, Costa EFD, Barbieri Pulz R, Silva-Zacarin ECM, Oliveira CA, Pigoso AA, Severi-Aguiar GDC (2017) Genotoxic effect and rat hepatocyte death occurred after oxidative stress induction and antioxidant gene downregulation caused by long term fluoride exposure. Chem Biol Interact 264:25–33
Demoulin B, Hermant M, Castrogiovanni C, Staudt C, Dumont P (2015) Resveratrol induces DNA damage in colon cancer cells by poisoning topoisomerase II and activates the ATM kinase to trigger p53-dependent apoptosis. Toxicol in Vitro 29:1156–1165
Garcia RI, Gregorich SE, Ramos-Gomez F, Braun PA, Wilson A, Albino J, Tiwari T, Harper M, Batliner TS, Rasmussen M, Cheng NF, Santo W, Geltman PL, Henshaw M, Gansky SA (2017) Absence of fluoride varnish–related adverse events in caries prevention trials in young children, United States. Prev Chronic Dis 14:E17
Ge Y, Chen L, Yin Z, Song X, Ruan T, Hua L, Liu J, Wang J, Ning H (2018) Fluoride-induced alterations of synapse-related proteins in the cerebral cortex of ICR offspring mouse brain. Chemosphere 201:874–883
German MJ, Hammiche A, Ragavan N, Tobin MJ, Cooper LJ, Matanhelia SS, Hindley AC, Nicholson CM, Fullwood NJ, Pollock HM, Martin FL (2006) Infrared spectroscopy with multivariate analysis potentially facilitates the segregation of different types of prostate cell. Biophys J 90:3783–3795
Jothiramajayam M, Sinha S, Ghosh M, Nag A, Jana A, Mukherjee A (2014a) Sodium fluoride promotes apoptosis by generation of reactive oxygen species in human lymphocytes. J Toxicol Environ Health A 77:1269–1280
Jothiramajayam M, Sinha S, Ghosh M, Nag A, Jana A, Mukherjee A (2014b) Sodium fluoride promotes apoptosis by generation of reactive oxygen species in human lymphocytes. J Toxic Environ Health A 77:1269–1280
Karimian A, Ahmadi Y, Yousefi B (2016) Multiple functions of p21 in cell cycle, apoptosis and transcriptional regulation after DNA damage. DNA Repair 42:63–71
Li X, Huang J-M, Wang J-N, Xiong X-K, Yang X-F, Zou F (2015) Combination of chrysin and cisplatin promotes the apoptosis of Hep G2 cells by up-regulating p53. Chem Biol Interact 232:12–20
Li W, Jiang B, Cao X, Xie Y, Huang T (2017) Protective effect of lycopene on fluoride-induced ameloblasts apoptosis and dental fluorosis through oxidative stress-mediated caspase pathways. Chem Biol Interact 261:27–34
Liao J-M, Cao B, Deng J, Zhou X, Strong M, Zeng S, Xiong J, Flemington E, Lu H (2016) TFIIS. h, a new target of p53, regulates transcription efficiency of pro-apoptotic bax gene. Sci Rep 6:23542
Liao Q, Zhang R, Wang X, Nian W, Ke L, Ouyang W, Zhang Z (2017) Effect of fluoride exposure on mRNA expression of cav1. 2 and calcium signal pathway apoptosis regulators in PC12 cells. Environ Toxicol Pharmacol 54:74–79
Liu J, Cui H, Peng X, Fang J, Zuo Z, Wang H, Wu B, Deng Y, Wang K (2013) Dietary high fluorine induces apoptosis and alters Bcl-2, Bax, and caspase-3 protein expression in the cecal tonsil lymphocytes of broilers. Biol Trace Elem Res 152:25–30
Liu H, Luo Q, Cui H, Deng H, Kuang P, Lu Y, Fang J, Zuo Z, Deng J, Li Y (2018) Sodium fluoride causes hepatocellular S-phase arrest by activating ATM-p53-p21 and ATR-Chk1-Cdc25A pathways in mice. Oncotarget 9:4318
Ma Y, Zhang K, Ren F, Wang J (2017) Developmental fluoride exposure influenced rat's splenic development and cell cycle via disruption of the ERK signal pathway. Chemosphere 187:173–180
Manahan S (2017) Environmental chemistry. CRC Press, Boca Raton
Martin FL, German MJ, Wit E, Fearn T, Ragavan N, Pollock HM (2007) Identifying variables responsible for clustering in discriminant analysis of data from infrared microspectroscopy of a biological sample. J Comput Biol 14:1176–1184
Moldoveanu T, Follis AV, Kriwacki RW, Green DR (2014) Many players in BCL-2 family affairs. Trends Biochem Sci 39:101–111
Muratori M, Tamburrino L, Marchiani S, Cambi M, Olivito B, Azzari C, Forti G, Baldi E (2015) Investigation on the origin of sperm DNA fragmentation: role of apoptosis, immaturity and oxidative stress. Mol Med 21:109–122
Pang W, Li J, Ahmadzai AA, Heppenstall LD, Llabjani V, Trevisan J, Qiu X, Martin FL (2012) Identification of benzo[a]pyrene-induced cell cycle-associated alterations in MCF-7 cells using infrared spectroscopy with computational analysis. Toxicology 298:24–29
Redza-Dutordoir M, Averill-Bates DA (2016) Activation of apoptosis signalling pathways by reactive oxygen species. Biochimica et Biophysica Acta (BBA)-Molecular Cell Research 1863:2977–2992
Ribeiro DA, Yujra VQ, da Silva VHP, Claudio SR, Estadella D, de Barros Viana M, Oshima CTF (2017) Putative mechanisms of genotoxicity induced by fluoride: a comprehensive review. Environ Sci Pollut Res 24:15254–15259
Rocha-Amador DO, Calderón J, Carrizales L, Costilla-Salazar R, Pérez-Maldonado IN (2011) Apoptosis of peripheral blood mononuclear cells in children exposed to arsenic and fluoride. Environ Toxicol Pharmacol 32:399–405
Schwarzbacherová V, Wnuk M, Lewinska A, Potocki L, Zebrowski J, Koziorowski M, Holečková B, Šiviková K, Dianovský J (2017) Evaluation of cytotoxic and genotoxic activity of fungicide formulation Tango® super in bovine lymphocytes. Environ Pollut 220:255–263
Sharma A, Sharma R, Singh SP, Khinchi M (2017) A brief review on apoptosis. Asian J Pharm Res Dev 5:1–10
Siddiqui WA, Ahad A, Ahsan H (2015) The mystery of BCL2 family: Bcl-2 proteins and apoptosis: an update. Arch Toxicol 89:289–317
Simoncicova J, Kalinakova B, Kovacik D, Medvecka V, Lakatos B, Krystofova S, Hoppanova L, Paluskova V, Hudecova D, Durina P, Zahoranova A (2018) Cold plasma treatment triggers antioxidative defense system and induces changes in hyphal surface and subcellular structures of Aspergillus flavus. Appl Microbiol Biotechnol 102:6647–6658
Singh R, Hussain MA, Kumar J, Kumar M, Kumari U, Mazumder S (2017) Chronic fluoride exposure exacerbates head kidney pathology and causes immune commotion in Clarias gariepinus. Aquat Toxicol 192:30–39
Skipper A, Sims JN, Yedjou CG, Tchounwou PB (2016) Cadmium chloride induces DNA damage and apoptosis of human liver carcinoma cells via oxidative stress. Int J Environ Res Public Health 13:1–10
Song GH, Huang FB, Gao JP, Liu ML, Pang WB, bin Li W, Yan XY, Huo MJ, Yang X (2015) Effects of fluoride on DNA damage and caspase-mediated apoptosis in the liver of rats. Biol Trace Elem Res 166:173–182
Søyseth V, Henneberger P, Virji MA, Bakke B, Kongerud J (2015) Construction of a job exposure matrix to dust, fluoride, and polycyclic aromatic hydrocarbons in the Norwegian aluminum industry using prediction models. Ann Occup Hyg 59:1106–1121
Suzuki M, Bandoski C, Bartlett JD (2015) Fluoride induces oxidative damage and SIRT1/autophagy through ROS-mediated JNK signaling. Free Radic Biol Med 89:369–378
Trevisan J, Angelov PP, Patel II, Najand GM, Cheung KT, Llabjani V, Pollock HM, Bruce SW, Pant K, Carmichael PL, Scott AD, Martin FL (2010) Syrian hamster embryo (SHE) assay (pH 6.7) coupled with infrared spectroscopy and chemometrics towards toxicological assessment. Analyst 135:3266–3272
Tu W, Zhang Q, Liu Y, Han L, Wang Q, Chen P, Zhang S, Wang A, Zhou X (2018) Fluoride induces apoptosis via inhibiting SIRT1 activity to activate mitochondrial p53 pathway in human neuroblastoma SH-SY5Y cells. Toxicol Appl Pharmacol 347:60–69
Umarani V, Muvvala S, Ramesh A, Lakshmi B, Sravanthi N (2015) Rutin potentially attenuates fluoride-induced oxidative stress-mediated cardiotoxicity, blood toxicity and dyslipidemia in rats. Toxicol Mech Methods 25:143–149
Upadhyay D, Panduri V, Ghio A, Kamp DW (2003) Particulate matter induces alveolar epithelial cell DNA damage and apoptosis: role of free radicals and the mitochondria. Am J Respir Cell Mol Biol 29:180–187
Whitford GM (1983) Fluorides: metabolism, mechanisms of action and safety. Dental Hygiene 57:16
Xia G, Sun X, Zheng X, Wang J (2016) Decreased expression of programmed death 1 on peripheral blood lymphocytes disrupts immune homeostasis in peripartum cardiomyopathy. Int J Cardiol 223:842–847
Zhang Y, Cao SR (1996) Coal burning induced endemic fluorosis in China. Fluoride 29:207–211
Zhang X, Bi L, Ye Y, Chen J (2014) Formononetin induces apoptosis in PC-3 prostate cancer cells through enhancing the Bax/Bcl-2 ratios and regulating the p38/Akt pathway. Nutr Cancer 66:656–661
Zhang Y, Li Q, Shu Y, Wang H, Zheng Z, Wang J, Wang K (2015) Induction of apoptosis in S180 tumour bearing mice by polysaccharide from Lentinus edodes via mitochondria apoptotic pathway. J Funct Foods 15:151–159
Acknowledgements
The authors would like to thank the volunteers who participated in this study.
Funding
This work was financially sponsored by the Scientific Research Foundation of the Education Department of Guangxi Province (Grant No. 2002316 and KY2015ZD058) and the National Natural Science Foundation (Grant No. 81602888).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Wen, P., Wei, X., Liang, G. et al. Long-term exposure to low level of fluoride induces apoptosis via p53 pathway in lymphocytes of aluminum smelter workers. Environ Sci Pollut Res 26, 2671–2680 (2019). https://doi.org/10.1007/s11356-018-3726-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-3726-z