Abstract
Microorganisms are considered a genetic resource with great potential for achieving sustainable development of agricultural areas. The objective of this research was to determine the effect of microorganism application forms on the production of biomass, gas exchange, and nutrient content in upland rice. The experiment was conducted under greenhouse conditions in a completely randomized design in a factorial 7 × 3 + 1, with four replications. The treatments consisted of combining seven microorganisms with three application forms (microbiolized seed; microbiolized seed + soil drenched with a microorganism suspension at 7 and 15 days after sowing (DAS); and microbiolized seed + plant sprayed with a microorganism suspension at 7 and 15 DAS) and a control (water). Treatments with Serratia sp. (BRM32114), Bacillus sp. (BRM32110 and BRM32109), and Trichoderma asperellum pool provided, on average, the highest photosynthetic rate values and dry matter biomass of rice shoots. Plants treated with Burkolderia sp. (BRM32113), Serratia sp. (BRM32114), and Pseudomonas sp. (BRM32111 and BRM32112) led to the greatest nutrient uptake by rice shoots. Serratia sp. (BRM 32114) was the most effective for promoting an increase in the photosynthetic rate, and for the greatest accumulation of nutrients and dry matter at 84 DAS, in rice shoots, which differed from the control treatment. The use of microorganisms can bring numerous benefits of rice, such as improving physiological characteristics, nutrient uptake, biomass production, and grain yield.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In Brazil, rice can be grown in two types of ecosystems: the lowland and upland conditions with supplemental irrigation or rainwater only (Silva et al. 2010). In upland ecosystem, rice is usually planted in rotation with soybeans, in the renewal of degraded pastures, or as the first crop in opening up new areas for agriculture (Santos et al. 2006). The production of upland rice is growing in importance due to the reduction of irrigation water availability for irrigated lowland rice crops (Nascente et al. 2013).
Among the alternatives for cultivating upland rice, there is the no-tillage system (NTS), which is characterized by maintaining straw on the soil surface in order to provide reduced soil temperature as well as increased levels of soil organic matter and biological activity (D’andréa et al. 2004; Nascente et al. 2013, 2016). However, rice in the NTS has a slow initial growth rate and is more subject to competition imposed by weeds (Nascente et al. 2016). Thus, the start-up and the initial seedling development are important characteristics and need to improve for establishing rice crops in the NTS.
The increases in soil organic matter content in the NTS are one of the main sources of nutrients and energy for an extraordinarily diverse community of soil microorganisms (Ferreira and Martin-Didonet 2012), including bacteria and fungi (Baldani et al. 2000). Rhizodeposition refers to the portion of the soil where the total carbon from the roots are transferred into soil, comprising exudates (smaller molecules) and secretions (larger molecules such as enzymes), lysates of dead cells, and mucilages. This rich deposition has a high microbial activity, given the abundance of microorganisms present in the soil surface layer (Spaepen et al. 2009). Constituents of the microflora, the plant growth-promoting rhizobacterias (PGPR), have important molecules within their cell constitution, such as flagellin (protein constituent of flagella) and lipopolysaccharides (LPS, cell membrane constituents) (Kaschuk et al. 2010). These components are recognized by the plant as metabolism activators. Thus, the benefits of the interaction between PGPR and plants can be direct, such as growth promotion, since some bacterial groups produce phytohormones, such as indole acetic acid (IAA), cytokinins, and gibberellins, solubilize phosphorus, and produce siderophores (Spaepen et al. 2009; Ahemad and Kibret 2014). Among the indirect benefits, there is the protection against pathogens, such as the induction of resistance and direct antagonism (Filippi et al. 2011).
These PGPRs are basic components of food webs and play crucial and unique roles in the agricultural production system (Anees et al. 2010). Several bacterial genera are currently being studied as biostimulants and biofertilizers, such as Azospirillum strain AbV5 (Isawa et al. 2010), Gluconacetobacter (Muthukumarasamy et al. 2005), Herbaspirillum, Burkholderia (Baldani et al. 2000), Pseudomonas (Yao et al. 2010), Bacillus, Serratia, Paenibacillum, Enterobacter, and Klebsiella (Spaepen et al. 2009). Thus, the association between PGPR and many crops, such as sugarcane, Saccharum officinarum (Lopes 2013); corn, Zea mays (Dartora et al. 2013); common beans, Phaseolus vulgaris (Martins 2013); eucalyptus, Eucalyptus grandis (Moreira and Araújo 2013); and rice, Oryza sativa (Souza Júnior et al. 2010), resulted in plant growth promotion without nitrogen fertilizer supplementation, in addition to reducing the occurrence of diseases.
Research focusing on PGPRs, which naturally occurs as potential biostimulants for rice plants, has shown promising results from an agricultural point of view (Isawa et al. 2010). During studies conducted at Embrapa Arroz e Feijão, promising rhizobacteria isolates (BRM32113, BRM32111, BRM32114, BRM32112, BRM32110, and BRM32109) were selected, because they have shown promise mainly due to their increased biomass production and disease suppression (Filippi et al. 2011; Silva et al. 2012; França et al. 2015).
Additionally, during studies conducted at the Federal Rural University of Amazon, four Trichoderma asperellum isolates were selected and tested as growth promoters and biocontrol agents, both in the greenhouse and in the field (Silva et al. 2012; Ferrari et al. 2013; França et al. 2015). However, there is still little information about what effects these bioagents (rhizobacterias isolates and T. asperellum) have on the gas exchange and nutrition of rice plants in upland regions. Therefore, this paper brings innovation due to making comparison of bioagents’ beneficial effects in promoting the growth of upland rice, which is done by assessing the health of the plant (gas exchange and nutritional status).
The hypothesis of this study is that the previously selected bioagents, when applied, either via seed or soil/foliar spray, significantly affect the development of rice plants, which is reflected in gas exchange, biomass production, and nutrient concentration. The objective was to determine the effect of different methods of applying microorganisms, previously identified as growth inducers, on the production of biomass dry matter, gas exchange, and nutrient content of upland rice plants.
Materials and methods
Environment characterization
The experiment was conducted in a greenhouse at the Embrapa Arroz e Feijão research center, Santo Antônio de Goiás, GO, Brazil. We used soil from the arable layer (0–0.20 m) of a clay loam (kaolinitic, thermic Typic Haplorthox) acidic soil from a Brachiaria brizantha pasture that had been in use for 20 years, with 377, 260, and 363 g kg−1 of sand, silt, and clay, respectively. The chemical characteristics of the soil were determined according to the methods described by Claessen (1997). The results were pH (H2O) = 5.8; hydrolytic acidity = 46.4 mmolc kg−1; Ca2+ = 11.61 mmolc kg−1; Mg2+ = 1.38 mmolc kg−1; Al3+ = 1.0 mmolc kg−1; H+ + Al3+ = 25.0 mmolc kg−1; P = 0.2 mg kg−1; K+ = 45 mg kg−1; Cu2+ = 2.1 mg kg−1; Zn2+ = 0.2 mg kg−1; Fe3+ = 14.3 mg kg−1; Mn2+ = 6.0 mg kg−1; total exchangeable bases = 14.14 mmolc kg−1, total N = 0.9 g kg−1; and soil organic matter = 17.93 g kg−1.
Three weeks before the rice planting, pots were filled with 7 kg of soil fertilized with 70 mg dm−3 of N (urea), 400 mg dm−3 P2O5 (simple superphosphate), and 200 mg dm−3 of K2O (potassium chloride). Soil moisture, throughout the experiment, was monitored daily by weighing the pots. The evapotranspirated moisture was replaced when it reached 85%, increasing it to 100% of the field capacity.
Experimental design and treatments
The experimental design was completely randomized in a factorial 7 × 3 + 1, with four replications. The treatments consisted of combining seven microorganisms, six rhizobacteria isolates and a T. asperellum pool with three application form. The rhizobacterial isolates (BRM 32113, 32111, 32112, 32114, 32110, and 32109) are described in Table 1, and they are currently stored and preserved in the Multifunction Microorganisms and Fungi collection from the Embrapa Arroz e Feijão. The T. asperellum pool is composed of four isolates T-06, T-09, T-12, and T-52, which were isolated from rhizospheric soils of reforested and native forest areas in the Amazon, taxonomically identified by Ferrari et al. (2013), and currently stored and preserved in the Fungal Culture Collection of the Plant Protection Laboratory at the Federal University Rural of Amazon.
There were three application forms: (1) seed—microbiolized seed; (2) seed-soil—microbiolized seed + soil drenched with microorganism suspension at 7 and 15 days after sowing (DAS); and (3) seed-plant—microbiolized seed + plant sprayed with microorganism suspension at 7 and 15 DAS. The control treatment consisted of no microbiolized seed and soil drenched and plant sprayed with only water without any microorganism.
Seed microbiolization
The bacterial suspensions were prepared with water, from cultures that had been growing for a 24-h period on solid medium 523 (Kado and Heskett 1970), at 28 °C, and the concentration was set in a spectrophotometer to A540 = 0.5 (108 UFC). The rice seeds were immersed in each suspension, and the control seeds were immersed in water for a period of 24 h under constant agitation at 25 °C.
Each isolate of T. asperellum pool was grown in a Petri dish containing potato dextrose agar (PDA) for 5 days and bioformulated as described by Silva et al. (2012). The seed treatment was performed at concentrations of 10 g of powdered (Silva et al. 2012) T. asperellum per 1 kg of seed (Filippi et al. 2011; Silva et al. 2012). The concentration of the biological suspension was 108 conidia ml−1.
-
Soil drench: 100 ml of suspension of each treatment, all the bacterial isolates (108 CFU), the T. asperellum pool (108 conidia. ml−1), and water drenched the trial soil at 7 and 15 DAS.
-
Plant spray pulverization: 30 ml of suspension of each treatment, all the bacterial isolates (108 CFU), the T. asperellum pool (108 conidia ml−1), and water were sprayed on the plants, using a manual backpack sprayer, at a constant pressure provided by a CO2 pressure source and a conical nozzle type (TX-VS2), at 7 and 15 DAS.
Rice plant management
Ten rice seeds were sown per pot (mutant genotype 07SEQCL441 CL was derived from a Primavera variety and was resistant to Imazapyr + Imazapic herbicide) on September 10, 2015. Plant emerged on September 15, 2015. Ten days after germination, we thinned the shoots and kept three plants per pot. At the beginning of the rice tillering stage, a topdressing fertilization (1 g of ammonium sulfate per pot) was performed. Weed control was performed manually, and there was no incidence of any insects or diseases.
Assessments
Gas exchange
The gas exchange and sheet width (cm) were sampled from the rice plants. Therefore, we measured photosynthetic rate, A (μmol CO2 m−2 s−1); transpiration rate, E (mmol H2O m−2 s−1); stomata conductance, gs (mol H2O m−2 s−1); internal CO2 concentration, Ci (vpm); and leaf temperature, Tleaf (°C), determined by a portable gas meter in the infrared region IRGA (LCpro, ADC BioScientific), from 08:00 to 10:00 am, at 33 and 66 DAS.
Samples were taken in the middle third of the first fully expanded leaf (top to base) during the two evaluation periods. The equipment was set to use concentrations of 370–400 mol mol−1 CO2 in the air, which is the reference condition used in the IRGA photosynthesis chamber. The photon flux density photosynthetic active (PPFD) used was 1200 μmol [quanta] m−2 s−1. The minimum equilibration time set for performing the reading was 2 min.
Biomass production
The shoots of rice plants were collected for biomass assessment 84 days after seed emergence, when 50% of plants were in full flower stage. Thus, in each treatment, the plants were dried at 65 °C, until constant weight, and weighed to determine the dry matter of the biomass.
Concentration of nutrients in rice plants
Following the collected plants’ drying and weighing, aliquots were taken which were grounded and an analysis of the nutrient content (N, P, K, Ca, Mg, Fe, Zn, Cu, and Mn) was performed in the rice shoots in accordance with recommendations set out by Malavolta et al. (1997).
Statistical analysis
All data were analyzed using analysis of variance, and F probability test was performed. The microorganism species and their application forms were considered fixed effects. Blocks and all block interactions were considered random effects. A comparison of means was performed with Tukey’s test (p ≤ 0.05). Dunnett’s test was performed at a significance of p ≤ 0.05 to compare the treatment without microorganism (control) with each treatment with microorganism application. We used the SAS statistical package (SAS 1999).
Results
Physiological characterization of the gas exchange process
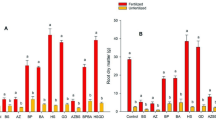
The gas exchanges were more pronounced when rice plants were treated with growth-promoting microorganisms (Table 2). The photosynthetic rate (A) was significantly higher than that in the control, especially for isolates BRM32110 (16.5%), T. asperellum pool (15.2%), BRM32109 (13.5%), BRM32112 (7.9%), and BRM32114 (6.4%). The transpiration rate (E) values of rice plants treated with the isolate BRM32113 were similar to the control treatment. Plants treated with the isolates BRM32110, T. asperellum pool, BRM32109, and BRM32111 presented the highest E. Regarding stomata conductance (gs), plants treated with the isolate BRM32110 presented the highest values and differed statistically from plants treated with isolates BRM32113 and BRM32112, as well as the control treatment (Table 2). There was no statistical difference among plants treated with isolates and the control for internal CO2 concentration (Ci) and leaf temperature (Tleaf).
The three different microorganism application forms did not affect leaf width, photosynthesis, stomata conductance, and leaf temperature (Table 2). Moreover, the application form affected transpiration, which was higher in the microbiolized seed and sprayed plant treatments, both statistically different from only microbiolized seed treatment. On the other hand, internal CO2 concentration (Ci) was also affected by application form; the highest values were observed in plants from microbiolized seeds only.
Shoot dry biomass
Concerning shoot dry biomass, there were differences observed among treatments (Table 3). The isolate BRM32114 provided the largest increase (21.53 g), which differed from the control (18.54 g). The isolate BRM32111 provided a reduction in rice biomass (11.24 g) and was also different from the control treatment. The remaining isolates did not provide significant increases in biomass production, based on the control treatment.
The application form of the three isolates (BRM32111, BRM32114, and BRM32112) did not affect the dry matter accumulation of rice plant shoots (Table 4). On the other hand, dry matter was higher in rice plants when the isolates BRM32113 and T. asperellum pool were applied by seed-soil or by seed-plant. The isolate BRM32110 applied by seed-soil provided a lower shoot dry matter than that applied by the seed. Similar results were observed from the isolate BRM32109, when applied by seed-plant; it provided lower biomass values, statistically different from the seed only treatment. In all three application forms, the isolate BRM32114 provided the highest dry biomass values, while the isolate BRM32111 provided the lowest biomass values of rice shoots.
Nutrient content in rice plants
Regarding nutrient levels in treated rice plants, microorganisms and application form had no interaction for K, Mg, and Fe absorption (Table 5). Thus, all three application forms and isolates did not affect K content in rice plants. On the other hand, rice plants treated with the isolate BRM32112 increased Mg absorption, which was different from plants treated with isolates BRM32113 and BRM32111. Regarding Fe absorption, rice shoots treated with the isolate BRM32111 presented the highest levels of this micronutrient and was different from isolates BRM32113, BRM32114, BRM32110, and BRM32109. Mg contents were higher in plants when microorganisms were applied by seed and by soil than by seed only application. The highest values for Fe content were obtained when isolates were applied in the seed only or by seed-plant.
Regarding levels of N, P, Ca, Cu, Mn, and Zn nutrients, the statistical analyses revealed a significant interaction between microorganisms and application form (Table 6). Thus, the isolate BRM32111 applied only by seeds presented the highest N contents in rice plants and differed from other isolates. The isolate BRM32109, applied by seed and by soil, allowed the highest N contents, and isolates BRM32111 and BRM32110 applied by seed and by plant provided the statistically highest contents of N. However, for the majority of isolates (BRM32113, BRM32111, BRM32114, BRM32112, and T. asperellum pool), it was observed that seed application form provided the best results of N content in the rice shoots.
Higher levels of P were observed in the rice plant shoots when the isolate BRM32111 was applied to the seed. On the other hand, rice plants presented the lowest level of this nutrient when treated by the isolate BRM32109 (Table 6). The seed-soil application of isolates BRM32112, BRM32109, BRM32114, and T. asperellum pool provided the highest values of P contents, different from treatment with isolates BRM32113, BRM3211, and BRM32110. The isolate BRM32112 applied by seed-plant application provided the highest values and differed from isolates BRM32113, BRM32114, BRM32110, BRM32109, and T. asperellum pool. Isolates BRM32113, BRM32111, BRM32114, BRM32110, and T. asperellum pool applied by the seed provided the highest P content in rice plants and isolates BRM32112 and BRM32109 when applied by seed-plant and seed-soil.
T. asperellum pool applied in the seed provided the highest levels of Ca in the rice plant shoots, which differed from the isolates BRM32113 and BRM32112 (Table 6). When the isolate BRM32109 was applied by seed-soil, plants obtained the highest values of Ca, which was different from isolates BRM32113, BRM32111, BRM32114, BRM32112, and T. asperellum pool. When the isolate BRM32110 was applied by seed-plant, higher levels of Ca in rice plant shoots were obtained, differing from isolates BRM32113, BRM32111, BRM32109, and T. asperellum pool. The application of BRM32113, BRM32111, BRM32109, and T. asperellum pool only by seed was more effective to raise Ca content in the rice plant shoots. In plants treated with isolates BRM32112 and BRM32110, Ca contents were similar for both application forms, by seed-soil and by seed-plant, while for the isolate BRM32114, the best result was obtained by seed-plant application.
For Cu, all isolates provided similar levels in the rice plant shoots when isolates were applied by the seed (Table 6). However, the isolate BRM32111 was the most efficient and different from others when applied by seed-soil. The isolate BRM32112, applied by seed-plant, allowed the best results differing from isolates BRM32114, BRM32109, and T. asperellum pool.
Plants treated with the isolate BRM32111 allowed the highest content of Mn in rice plant shoots when the application was by the seed, which differed from isolates BRM32113, BRM32110, and BRM32109 (Table 6). The isolate BRM32111 also provided the highest values and differed from the other bioagents, when applied by seed-soil. For the seed-plant application, plants treated by isolate BRM32113 provided the best results differing from isolates BRM32109 and T. asperellum pool. The application of isolates only in seeds resulted in higher Mn content in rice plants, when treated with isolates BRM32113, BRM32114, BRM32112, and T. asperellum pool, while, for the isolate BRM32111, it was the seed-soil application, and for isolates BRM32110 and BRM32109, it was the seed-soil or seed-plant application that provided the highest Mn content in the rice plant shoots.
The isolate BRM32110 provided the highest Zn content in the rice plant shoots, when the application was made only by the seed, and was different from the isolate BRM32113 (Table 6). The highest values were obtained with the T. asperellum pool, which differed from the microorganisms BRM32113 and BRM32111. In the seed-soil application, the highest levels of Zn were found in the plants treated with the isolate BRM32110, which differed from all other five isolates, when the application was seed-soil or seed-plant. For isolates BRM32113, BRM32111, BRM32110, BRM32109, and T. asperellum pool, the best form of application to allow high Zn uptake is microbiolization of rice seeds. In plants treated with the isolate BRM32114, the highest values of Zn in rice shoots were found in the seed-soil application, and for those treated with the isolate BRM32112, the highest were found in the seed-plant application.
While comparing the contents of nutrients in the control, the Ca absorption by rice plants was reduced in the treatments with isolates BRM32113 and BRM32111 (Table 5). On the other hand, the Mn absorption was increased when the rice plants were treated with the bioagent BRM32111. In addition, the highest absorption of N was obtained when the rice plants were treated with isolates BRM32113, BRM32111, and BRM32109.
Discussion
The application forms, on average, showed similar results. Then, the application only in the seeds would be enough to provide effects of the microorganisms on upland rice plants development. It should be easier and cheaper to the farmers once they need only this application during plant development. Results also demonstrated that the treated rice plants had a more efficient gas exchange process, which includes photosynthesis, stomata conductance, and transpiration, when compared with the non-treated plants (control plants). These results corroborate our research hypothesis; the microorganisms utilized positively affected the physiological parameters of the rice plants and could have significant effects on biomass production and crop yields. The ability to photosynthesize, and the ability of the stomata to drive water in a vapor form (from leaf to atmosphere) in a given time (stomata conductance), has been intensively studied since these parameters determine the initial process for the biomass production. Bacillus sp. isolate (BRM32110) provided the highest photosynthetic rate and stomata conductance to rice plants and did not differ from Serratia sp. (BRM32114), Bacillus sp. isolate (BRM32109), and T. asperellum pool. This suggests that these microorganisms positively influenced the stomata opening and closing, which directly affected the process of gas exchange and contributed to a greater accumulation of biomass. Concerning the internal CO2 concentration, there were no differences among the plants, both treated and untreated, indicating that CO2 concentration was the same as in plants that are correctly irrigated, regardless of the presence of plant growth-promoting microorganisms. Souza et al. (2015) observed increases of up to 176% of the content of total sugars in rice plants after seed-soil application of isolates BRM32113 (Burkholderia sp.), BRM32111 (Pseudomonas sp.), and T. asperellum pool. The physiological effects caused by microorganisms on plant growth include changes in many processes in which range from the dynamics of gas exchange (photosynthesis, transpiration, and stomata conductance), dry matter accumulation, and nutrients to the water-soil-plant-atmosphere relationships (Auge 2001; Barzana et al. 2012).
Microorganisms can provide direct stimulus to plant growth by producing plant hormones or similar substances (Bandara et al. 2006; Guo et al. 2008; Pérez-García et al. 2011), or indirect stimulation by increasing the photosynthetic rate, as was shown in this study. Corroborating this information, the isolate BRM32114 (Serratia sp.) from the rhizosphere of rice plants (Table 1) improved the photosynthetic rate in 6.4% and the production of biomass to the order of 16.12%, considering all application forms and biomass in 19.14%, when it was applied only in seeds. Besides, this isolate was the only one that differed from the control in plant biomass production. Thus, the isolate BRM32114 (Serratia sp.) was considered the best microorganism to promote increases in rice shoot biomass in this research.
Regarding the nutritional status of the plants, it was observed that the plants treated with the isolate BRM32111 (Pseudomonas sp.), when applied to the seed, had the highest levels of N, P, Mn, and Fe, when compared to the other bioagents. However, these results may be related to the concentration effect (Fageria et al. 2011), since the plants treated with this isolate BRM32111 (Pseudomonas sp.) had the lowest biomass dry weight (24.11 g, Table 3). The isolate BRM32110 (Bacillus sp.), when only the seed was applied, was highlighted to increase contents of Cu and Zn in rice shoots, while the other Bacillus sp. isolate BRM32109, when seed-soil was applied, increased the levels of P and Ca in the rice shoots. Based on these results, it seems that microorganisms provided rice shoots an increased nutrient uptake. Microorganisms can also provide indirect plant growth stimulation by improving the availability and absorption of nutrients (Pérez-García et al. 2011; Zhang et al. 2011.). Thus, it can be inferred that, with the exception of K and Mg, most of the microorganisms studied during our research provided rice shoots a significant increase in nutrient content. These results suggest that both increased photosynthetic rate and an increase in the content of nutrients in rice plants may have produced a direct effect on biomass production, which has a significant effect on a crop yield (Fageria 2009; Fageria et al. 2011).
Rêgo et al. (2014) reported that rice seeds treated with isolates T. asperellum pool, BRM32113 (Burkholderia sp.), and BRM32111 (Pseudomonas sp.) showed changes in root architecture by increasing the length of the roots, an expansion of the cortex, increased by 2% in aerenchyma spaces, an increase in diameter of the vascular cylinder, and an increase in the volume of the root and thereby confirmed that treated plants are more efficient in absorbing nutrients. Subsequently, Martins (2015) identified and characterized the genera of respective rhizobacteria (Table 1), indicating that the isolates BRM32113 (Burkholderia sp.) and BRM32114 (Serratia sp.) produce AAI, cellulase, and siderophores. In the same study, isolates BRM32111 (Pseudomonas sp.) and BRM32112 (Pseudomonas sp.) were identified and characterized, showing that they produce cellulose, phosphatase, and siderophores. This information helps to partly explain the higher biomass gain in plants treated with the isolate BRM32114 (Serratia sp.) (by seed application) and in plants treated with the isolate BRM32113 (Burkholderia sp.) (by seed-plant application). In addition, plants treated with the isolates BRM32111 and BRM32112 (both Pseudomonas sp.) (solubilizing phosphate) showed the highest levels of P and Fe.
Species of the genus Trichoderma are naturally found in almost all soil types and act against pathogens, using different action mechanisms; they also promote beneficial effects, such as plant growth (Shoresh et al. 2005; Perazzolli et al. 2008; Vinale et al. 2008). The T. asperellum pool used in this study is formed by isolated T-6, T-9, T-12, and T-52, which were selected and characterized by Silva et al. (2012), indicating that they are phosphate solubilizers and cellulose degraders and, in vitro, reduced the mycelial growth of Rhizoctonia solani.
During our research, we tested seven potential bioagents, which were previously identified as antagonists and inducers of resistance to the majority of rice pathogen (Filippi et al. 2011; Silva et al. 2012; Pereira Filho 2013). This paper presents the comparison of bioagents’ beneficial effects in promoting the growth of rice as a novelty, which is done by assessing the health of the plant (gas exchange and nutritional status).
Based on the results, it appears that overall, the application form did not influence the results and then, only the application on the seeds could provide effects on the plant development. Besides, the use of bioagents can be a sustainable alternative for increasing the biomass production of rice plants, which is a characteristic that is positively correlated to a crop yield (Alvarez et al. 2012). This study also indicated that the isolates BRM32114 (Serratia sp.), the both Bacillus sp. BRM32110 and BRM32109, and the T. asperellum pool are the most promising to use on a commercial scale, since it excelled in promoting better performance of upland rice plants, besides being effective antagonists to the main rice pathogens (França et al. 2015; Souza et al. 2015; Martins 2015). Also, in general, these isolates provided higher nutrient concentration (Tables 4 and 5) in the rice plants, which indicates that their presence encourages the availability of nutrients in the soil and, consequently, the uptake of these by plants (Table 4). It is worth mentioning that the isolate BRM32114 (Serratia sp.) was the best bioagent, either to stimulate a higher photosynthetic rate (both in the vegetative and reproductive stage) or to promote greater accumulation of dry matter at 84 DAS in the rice plants, independent of application form, and because it was the only isolate that provided increases in the biomass production of rice that differed from the control (no microorganism application), further studies are needed to better understand the processes that are involved in bioagent-plant interaction, such as evaluating the efficiency of these bioagents under field conditions, quantifying key elements such as plant hormones and enzymes involved in physiological mechanism of growth induction and promoters of disease resistance, and defining the morpho-anatomical characteristics of the isolates during their interaction with the plant.
Conclusion
Overall, the application forms of microorganisms evaluated showed similar results. Then, we recommend only the application on the rice seeds. Microorganism application positively and differently affected the growth parameters evaluated in rice plants. Rice plants treated with isolates Serratia sp., Bacillus sp. (BRM32114, BRM32110, BRM32109), and T. asperellum pool provided, on average, the highest photosynthetic rate values and dry matter biomass of rice shoots. Plants treated with Burkholderia sp., Serratia sp., and Pseudomonas sp. isolates (BRM32113, BRM32114, BRM32111, and BRM32112) led to the greatest nutrient uptake by the rice shoots. However, it is complicated to use a mixture of microorganism under field conditions. Therefore, among the evaluated microorganisms, we selected the isolate BRM32114 (Serratia sp.) for future studies in field conditions, since it promoted, in rice shoots, the highest photosynthetic rate (both in vegetative and reproductive stage), for the greatest accumulation of nutrients and dry matter at 84 DAS, and for differing from the control in the characteristic of dry biomass production.
References
Ahemad M, Kibret M (2014) Mechanisms and applications of plant growth promoting rhizobacteria: current perspective. J King Saud Uni 26:1–20
Alvarez RCF, Crusciol CAC, Nascente A, Rodrigues JD, Habermann G (2012) Gas exchange rates, plant height, yield components, and productivity of upland rice as affected by plant regulators. Pesqui Agropecu Bras 47:1455–1461
Anees M, Edel-Hermann V, Steinberg C (2010) Buildup of patches caused by Rhizoctonia solani. Soil Biol Biochem 42:1661–1672
Auge RM (2001) Water relation drought and VA mycorrhizal symbiosis. Mycorrhiza 11:3–42
Baldani VLD, Baldani JI, Döbereiner J (2000) Inoculation of rice plants with the endophytic diazotrophs Herbaspirillum seropedicae and Burkholderia spp. Biol Ferti Soils 30:485–491
Bandara WM, Seneviratne G, Kulasooriva SA (2006) Interactions among endophytic bacteria and fungi: effects and potentials. J Biosciences 31:645–650
Barzana G, Aroca R, Paz JÁ, Chaumont F, Martinez-Ballest MC, Carvajal M, Ruiz-Lozano JM (2012) Arbuscular mycorhhizal symbiosis increases relative apoplastic water flow in roots of the host plant under both well-watered and drought stress conditions. Ann Bot-London 109:1009–1017
Claessen MEC (1997) Manual for methods of soil analysis, 2nd edn. Embrapa Solos, Rio de Janeiro
D’andréa AF, Silva MLN, Curi N, Guilherme LRG (2004) Estoque de carbono e nitrogênio e formas de nitrogênio mineral em um solo submetido a diferentes sistemas de manejo. Pesqui Agropecu Bras 39:179–186
Dartora J, Guimarães VF, Marini D, Sander G (2013) Adubação nitrogenada associada à inoculação com Azospirillum brasilense e Herbaspirillum seropedicae na cultura do milho. Rev Bras Eng Agríc Ambiental 17:1023–1029
Fageria NK (2009) The use of nutrients in crop plants. Boca Raton, Florida
Fageria NK, Baligar VC, Jones CA (2011) Growth and mineral nutrition of field crops, 3rd edn. Boca Raton, Florida
Ferrari, A.; Batista, T. F. V.; Montenegro; Delalibera Junior, I. (2013) Diversidade de Trichoderma spp. coletados em solo na base petrolífera “Pedro de Moura” em Coari-AM. In: XIII Siconbiol, Bonito, v. 1, p.1
Ferreira EPB, Martin-Didonet CCG (2012) Mulching and cover crops effects on the soil and rhizosphere-associated bacterial communities in field experiment. J Agric Sci Technol 14:671–681
Filippi MCC, Silva GB, Silva-Lobo VL, Cortes MMCB, Moraes AJG, Prabhu AS (2011) Leaf blast (Magnaporthe oryzae) suppression and growth promotion by rhizobacteria on aerobic rice in Brazil. Biol Control 58:160–166
França SKS et al (2015) Biocontrol of sheath blight by Trichoderma asperellum in tropical lowland rice. Agron Sustain Dev 35:317–324
Guo B, Wang Y, Sun X, Tang K (2008) Bioactive natural products from endophytes: a review. Appl Biochem Micro 44:136–142
Isawa T, Yasuda M, Awasaki H, Minamisawa K, Shinozaki S, Nakashita H (2010) Azospirillum sp. strain B510 enhances rice growth and yield. Microbes Environ 25:58–61
Kado CJ, Heskett MG (1970) Selective media for isolation of Agrobacterium, Corynebacterium, Erwinia, Pseudomonas and Xanthomonas. Phytopathology 60:969–976
Kaschuk G, Alberton O, Hungria M (2010) Three decades of soil microbial biomass studies in Brazilian ecosystems: lessons learned about soil quality and indications for improving sustainability. Soil Biol Biochem 42:1–13
Lopes ML (2013) Estudo do polimorfismo cromossômico em S. cerevisiae (linhagem PE- 2) utilizada no processo industrial de produção de etanol. Tese, Universidade Estadual Julio de Mesquita Filho
Malavolta E, Vitti GC, Oliveira AS (1997) Avaliação do estado nutricional de plantas: princípios e aplicações. Piracicaba
Martins AS (2013) Desenvolvimento do feijão-comum tratado com Bacillus subtilis. Dissertação, Universidade Federal de Lavras
Martins BEM (2015) Caracterização morfológica, bioquímica e molecular de isolados bacterianos antagonistas a Magnaporthe oryzae. 80 f. Dissertação, Universidade Federal de Goiás
Moreira ALL, Araújo FF (2013) Bioprospecção de isolados de Bacillus spp. como potenciais promotores de crescimento de Eucalyptus urograndis. Rev Árvore 37:933–943
Muthukumarasamy R, Cleenwerck I, Revathi G, Vadivelu M, Janssens D, Hoste B, Kang G, Park K, Young S, Tongmin S, Caballero-Mellado J (2005) Natural association of Gluconacetobacter diazotrophicus and diazotrophic Acetobacter peroxydans with wetland rice. Syst Appl Microbiol 28:277–286
Nascente AS, Crusciol CAC, Cobucci T (2013) The no-tillage system and cover crops—alternatives to increase upland rice yields. Eur J Agron 45:124–131
Nascente AS, Lacerda MC, Lanna AC, Filippi MCC, Silva DM (2016) Cover crops can affect soil attributes and yield of upland rice. Aust J Crop Sci 10:176–184
Perazzolli M, Dagostin S, Ferrari A, Elad Y, Pertot I (2008) Induction of systemic resistance against Plasmopara viticola in grapevine by Trichoderma harzianum T39 and benzothiadizole. Biol Control 47:228–234
Pereira Filho CR (2013) Seleção e aplicação de rizobactérias promotoras de crescimento e indutoras de resistência sistêmica em arroz (Oryza sativa) de terras altas ( 2013). Dissertação, Universidade Federal de Goiás
Pérez-García A, Romero D, De Vicente A (2011) Plant protection and growth stimulation by microorganisms: biotechnological applications of Bacillus in agriculture. Curr Opin Biotech 22:187–193
Rêgo MCF, Ilkiu-borges F, Filippi MCC, Gonçalves, LA, Silva GB (2014) Morphoanatomical and Biochemical Changes in the Roots of Rice Plants Induced by Plant Growth-Promoting Microorganisms. J Botany 1--10. doi:10.1155/2014/818797
Santos AB, Stone LF, Vieira NRA (2006) A cultura do arroz no Brasil. Embrapa Arroz e Feijão, Santo Antônio de Goiás
Shoresh M, Yedidia I, Chet I (2005) Involvement of jasmonic acid/ethylene signaling pathway in the systemic resistance induced in cucumber by Trichoderma asperellum T203. Phytopathology 95:76–84
Silva VAC et al (2010) Comportamento de cultivares de arroz de terras altas na Zona da Mata de Pernambuco. Rev Bras Eng Agríc Ambiental 14:1030–1037
Silva JC, Torres DB, Lustosa DC, Filippi MCC, Silva GB (2012) Rice sheath blight biocontrol and growth promotion by Trichoderma isolates from the Amazon. Rev Ciênc Agrár 55:243–250
Sousa T (2014) Indução de crescimento e resistência à brusone em plantas de arroz por ação de biopromotores e adubação silicatada. Dissertação, Universidade Federal Rural da Amazônia
Souza Júnior IT, Moura AB, Schafer JT, Corrêa BO, Gomes CB (2010) Biocontrole da queima-das-bainhas e do nematoide-das-galhas e promoção de crescimento de plantas de arroz por rizobactérias. Pesqui Agropecu Bras 45:1259–1267
Souza ACA, Sousa TP, Cortês MVB, Rodrigues FÁ, Silva GB, Filippi MCC (2015) Enzyme-induced defense response in the suppression of rice leaf blast (Magnaporthe Oryzae) by silicon fertilization and bioagents. Int J Res Stud Biosci 3:22–32
Spaepen S, Vanderleyden J, Okon Y (2009) Plant growth-promoting actions of rhizobacteria. Adv Bot Res 51:284–320
Vinale F, Sivasithamparam K, Ghisalbert EL, Mara R, Barbetti MJ, Li H, Woo SL, Lorito M (2008) A novel role for Trichoderma secondary metabolites in the interactions with plants. Physiol Mol Plant P 72:80–86
Yao LX, Wu ZS, Zheng YY, Kaleem I, Li C (2010) Growth promotion and protection against salt stress by Pseudomonas putida Rs-198 on cotton. Eur J Soil Biol 46:49–54
Zhang YF, He LY, Chen ZJ, Wang QY, Qian M, Sheng XF (2011) Characterization of ACC deaminase producing endophytic bacteria isolated from copper-tolerant plants and their potential in promoting the growth and copper accumulation of Brassica napus. Chemosphere 83:57–62
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Yi-ping Chen
Rights and permissions
About this article
Cite this article
Nascente, A.S., de Filippi, M.C.C., Lanna, A.C. et al. Biomass, gas exchange, and nutrient contents in upland rice plants affected by application forms of microorganism growth promoters. Environ Sci Pollut Res 24, 2956–2965 (2017). https://doi.org/10.1007/s11356-016-8013-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-8013-2