Abstract
The aim of the study was to estimate the impact of oxygen depletion on macroinvertebrate community structure in benthic space. Macroinvertebrate assemblages and potential of dissolved oxygen (DO) consumption were investigated simultaneously in the plain rivers of the Ziya River Basin. The degree of DO depletion was represented by sediment oxygen demand (SOD) and DO, chemical oxygen demand (CODCr), and ammonia nitrogen (NH4 +-N) in the overlying water. The results showed an all-around hypoxia environment formed, and the values of DO, SOD, CODCr, and NH4 +-N were separately 0.11–4.03 mg L−1, 0.41–2.60 g m−2 day−1, 27.50–410.00 mg L−1, and 1.79–101.41 mg L−1. There was an abnormal macroinvertebrate assemblage, and only 3 classes, Insecta, Gastropoda, and Oligochaeta, were found, which included 9 orders, 30 families, and 54 genera. The biodiversity was at a low level, and Shannon-Wiener index was 0.00–1.72. SOD, and NH4 +-N had major impact on the macroinvertebrate community, and the former had negative effect on most taxa, for instance, Nais, Branchiura, Paraleptophlebia, etc., which were sensitive or had a moderate-high tolerance to pollution. NH4 +-N had both positive and negative impacts on benthic animals, for instance, Dicrotendipes, Gomphus, Cricotopus, etc., for the former, and Procladius, Limnodrilus, Hippeutis, etc., for the latter. They all had a moderate-high tolerance to pollution. It is significant to improve DO condition and macroinvertebrate diversity in river harnessing and management.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Dissolved oxygen (DO) is essential for survival of aquatic life. However, pollutants discharged into river channel can consume large amounts of oxygen during the process of form transformation, which results in hypoxia and a direct threat to aquatic organisms. Oxygen availability was proved to be an important factor in predicting aquatic invertebrate assemblages (Comte et al. 2010). Hypoxia induces changes in community structure and process (McNeil and Closs 2007; Ekau et al. 2010), behavior (Zhang et al. 2009; Riedel et al. 2014), and physiological features (Grieshaber et al. 1994; Urbina and Glover 2012) of aquatic organisms (fish, macroinvertebrate, zooplankton). Extreme lack of DO is likely to produce lethal effect on aquatic organisms (Nandan and Azis 1995; Rao et al. 2014), which threaten normal development of aquatic ecosystem.
The Hai River Basin (HRB), located in northern China, is one of China’s seven major river basins and is an important center of politics, economy, and culture. However, with the economic development speeding up, river pollution increases. Combined with water shortage due to local anthropogenic activities (Tang et al. 2015), the HRB has become one of the most polluted basins in China (Liu et al. 2010). There are eight sub-river basins in the HRB, of which the Ziya River Basin (ZRB) is the most polluted one. The ZRB is located in the southwest of the HRB and has more than 60 cities and counties, most of which gathers in plain area. The plain area includes Shijiazhuang city that is the provincial capital of Hebei and other major cities such as Xingtai city, Hengshui city, and Handan city. Accordingly, there is a large population in the plain area. Shijiazhuang city, Xingtai city, and Hengshui city have a population of 1.02 × 108, 7.10 × 106, and 4.34 × 106 people, respectively (National Bureau of Statistics 2010). As the economy develops, emission of pollutants increases and water pollution has become one of the major problems. Water and sediment are polluted simultaneously, and the proportion of blackwater river increases rapidly. Dry rivers and polluted rivers have dominated the ZRB (Zhu 2014). Water quality in most river sections is worse than grade V in the Ziya River and its main branch, Fuyang River (Li et al. 2012). Wastewater discharges affect the DO in water column (Antanasijevic et al. 2014). Especially in the plain area of the ZRB, ammonia nitrogen (NH4 +-N), which has great potential for oxygen consumption, has become the major excessive pollutant (Wang and Shan 2012).Oxygen depletion has become one of the prominent problems of rivers in the ZRB, which severely impacts aquatic organisms. Zhu claimed that aquatic animals and plants had almost become extinct in the ZRB (Zhu 2014). However, this is just a qualitative description and lacks support of quantitative research.
This study focused on DO consumption and macroinvertebrate assemblages in the plain rivers of the ZRB. We proposed the following hypotheses: (1) there was an oxygen-deprived benthic environment all around in plain rivers of the ZRB; (2) variables related with oxygen consumption had significant impact on community composition of benthic animals. Extreme hypoxic environment would support few pollution-tolerant species. Hypoxia condition was determined both in sediment and overlying water. We measured DO and two major substances of oxygen consumption, NH4 +-N and chemical oxygen demand (COD) of water and sediment oxygen demand (SOD). In parallel, macroinvertebrates were investigated.
Material and methods
Study area and sampling sites
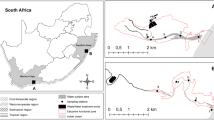
The ZRB, located in north China, is bounded on the north by the Daqing River Basin, on the west by the Taihang Mountain, on the east by the Bohai Sea, and on the south by the Zhangwei River Basin (Fig. 1). It stretches across Hebei Province, Shanxi Province, and Tianjin Municipality and supports 19 million populations. The basin has an area of 46,868 km2, 66 % and 34 % of which are mountain area and plain area respectively (The Hai River Basin (vol.1), 1997). This study focused on the plain area due to its heavy pollution and oxygen depletion. The basin, the area of northern temperate continental monsoon climate, has an annual average rainfall of 550 mm. Nevertheless, rivers with blackwater and odors have dominated the plain area of the basin, and oxygen depletion has become a prominent problem due to sewage discharge. Based on the previous study, NH4 +-N, an important substance consumed by DO, was highlighted as the main pollutant in the Ziya river system (Li et al. 2012; Zhao et al. 2014).
The Ziya river system has two big branches: the Hutuo River and the Fuyang River. Sampling sites were set in the Fuyang River and its important tributaries, considering that most river segments of the Hutuo River has dried up and even been abandoned in the whole years. Sampling sites were set based on the following principles: (1) considering the areas of the river systems and complexity of the river network, river systems with larger areas had more sampling sites, and fewer sites were set in river systems with small areas and simple river networks. (2) Sampling sites were set both on major and branch channels to study the differences and relationships between them. (3) Sites were set upstream and downstream of sewage outlets. (4) For those reaches with uniform characteristics, comparatively fewer sites were set. Twenty-four sampling sites were set in the plain rivers of the ZRB (Fig. 1), with rivers flowing down from Shijiazhuang city and Xingtai city. At the intersections of tributaries, sites were set specially.
Macroinvertebrates investigation
In August 2013, macroinvertebrates were sampled at each site. Although the rivers in the study area were mostly with slow flow and blackwater, macroinvertebrate sampling procedures were conducted in various habitat (run, riffle, pool, backwater, drop water, etc.). Quantitative and qualitative samplings were combined. Qualitative samples were taken using a standard D-net with 0.5-mm mesh. A surber with an area of 0.3 m × 0.3 m was used for quantitative sampling. Large rocks, woody debris, and leaves were scrubbed to dislodge organisms clinging to these substrates. There were at least four samples at a site. Samples were preserved with 10 % formalin in the field, and then transformed to 75 % ethanol in the laboratory. Organisms were sorted with forceps in white china plate. Samples were identified to the lower practicable taxonomic level (normally genus) possible.
Measurement of sediment oxygen demand
Sediment constitutes the main habitat for macroinvertebrate. However, polluted sediment has a high potential to consume DO. Sediment oxygen demand (SOD) has been found to contribute significantly to oxygen consumption in river and plays a primary role in the water quality (Hanes and Irvine 1966; James 1974; Hu et al. 2001). We measured SOD specially to determine the potential of sediment to consume DO. To ensure accuracy of laboratory experiment, two measurements were conducted. In August and October 2013, sediment samples were collected from the top 20 cm of the riverbed using a dredge sampler at each site. This was because the top 20 cm represent the most biologically active depositional layer for relatively slow flowing streams (Hickey 1988). At each site, three parallel samples were collected and then stored in polyethylene tubes with air isolating cock to protect sediments from oxidation during the preservation. Sediment samples were placed in a refrigerator at 8 °C (a representative hypolimnetic temperature). After delivery to the laboratory, the SOD of samples was measured as soon as possible.
A method of laboratory core incubation was applied. Sediment was incubated in cylindrical perspex cores with 6-cm internal diameter and 28-cm height. Sediment cylinders were 10 cm deep and with 14 cm overlying water. Sediment cores were incubated, at 25 °C in dark, for 24 h or longer until the DO concentration in the overlying water dropped to 2 mg L−1. The rate of oxygen consumption was calculated based on the slope along the DO versus time profiles and the area of the sediment-water interface. SOD was expressed in terms of grams per square meter per day (g O2 m−2 day−1). Mathematically, the SOD rate of core incubation can be formulated as
where
- SOD:
-
sediment oxygen demand, g m−2 d−1
- S:
-
slope of liner portion of usage curve, mg L−1 h−1
- V:
-
volume of the overlying water, litters
- A:
-
area of the core incubator, m2
The specific formula for the core incubator in this study was
Measurement of oxygen consumption in water
The characteristics of overlying water have inevitable effects on macroinvertebrate assemblages (Maul et al. 2004; Heatherly et al. 2007; Holland et al. 2015). During the field survey of macroinvertebrates and sediment, DO concentration in the overlying water was simultaneously measured using a portable DO analyzer (HACH/HQ30d, America). Water samples were collected in polythene bottles with 100 mL, acidified with sulfuric acid to pH < 2, and stored in car refrigerator at 2–5 °C. The COD was determined with the reactor digestion method (HACH/DR2800, America), and the ammonia was determined with the phenol-hypochlorite colorimetric method (State Environmental Protection Agency 2002).
Data processing
Analysis on macroinvertebrate community was conducted, and pollution levels of rivers were evaluated with DO, SOD, COD, and NH4 +-N as indicators followed by the investigation of their relationships. Microsoft Excel was used for drawing histogram and scatterplot. Map of sampling sites was produced by ArcGIS 10.0. Ordination analyses were performed using Canoco 5.0 which was a professional statistic software applied in ecology.
Results
Macroinvertebrate assemblages
The survey result showed that the macroinvertebrate community in the plain area of the Ziya river system had an abnormal structure. Benthic taxa were incomplete; only three classes, Insecta, Gastropoda, and Oligochaeta, appeared, while other taxa, such as Crustacea and Lamellibranchia disappeared. There were 9 orders, 30 families, and 54 genera. Aquatic insects were dominant in each taxon, of which Chironomidae was the major component. A total of 19 genera in Chironomidae were found, accounting for 35 % of the total taxa in the plain area of the basin (Table 1). The Ziya river system had a low density of macroinvertebrate generally. There was a large range of density which was from 11.11 to 3022.22 ind m−2 (Fig. 2). This was due to the differences of taxa that occurred between the sites. For instance, those sites with the most aquatic insects or Oligochaeta had a high density, while the sites with macroinvertebrate groups dominated by Gastropoda had a low density. The average and the median of the density were 328.31 ± 406.68 and 61.11 ind m−2, respectively. Similarly, the biomass was also at a low level, with an average of 0.40 ± 0.47 g m−2 and the median of 0.11 g m−2. The biomass had a wide range from 0.0009 to 2.57 g m−2 (Fig. 2). There were few taxa in each site, and obviously, those sites with Gastropoda had a large biomass, while the sites occupied by Chironomidae or Tubificidae had a small biomass.
Benthic community had an abnormal structure in the plain rivers of the Ziya river system. There were very low richness and low abundance at each site, coupled with a sparse distribution. It was often difficult to find benthic organisms. Several sampling processes for obtaining one sample and the communities were dominated by only one or two taxon. Shannon-Wiener index, which could simultaneously reflect biodiversity and evenness, was calculated for each site. The results showed that the plain rivers in the Ziya river system had a lower biodiversity. Values of the Shannon-Wiener index were from 0.00 to 1.72 (Table 2). There were up to nine sites with a “zero” level of biodiversity, and the reasons were as follows: (a) only one species was found in quantitative samples at site, such as S02, S06, S09, S13, and S14, and (b) no organisms were found at site, such as S10, S17, S18, and S21. The average and the median of the index value were respectively 0.62 ± 0.60 and 0.65, representing a low biodiversity.
Due to fewer species in quantitative samples, it was easy to determine the dominant species. We just applied the cumulative percentage of several species which had a relative large number of individuals in quantitative samples. The cumulative percentages of the species were above 70 %, 12 of which were 100 %(Table 2). This was because there were only one or two species that appeared in the quantitative samples, and these survivals dominated the corresponding locations obviously. The dominant taxa were mainly Chironomidae, followed by Tubificidae and Gastropoda. Adaptable or tolerable habitats for the dominant species were mainly lentic, polluted, eutrophic, hypoxia water, or sediments. For instance, Chironomus lived in lentic habitat and could bear high pollution; Radix preferred the sediments with rich organic matters and could tolerate anoxia; Tubificidae often survived in polluted and anoxic waters. Most dominant species had tolerance to moderate or high pollution, and the tolerance values were 3–7 or ≥7 (Table 2).
Oxygen content and consumption potential
Measurement of water column in situ showed that the DO in the Ziya river system was at a low level. The range of DO concentration was 0.11–4.03 mg L−1, and the average value was 1.35 ± 0.95 mg L−1. There were five sites with extremely low values of DO (<0.5 mg L−1). They were S09 (0.21 mg L−1), S10 (0.11 mg L−1), S15 (0.21 mg L−1), S19 (0.44 mg L−1), and S20 (0.17 mg L−1) (Fig. 3b). S10 and S15, located at the confluences of several branches, had low DO, indicating that convergence of pollutants did more contribution to oxygen depletion (Fig. 1). S19 and S20 were in the downstream location near the confluence and with a low level of DO. S09 was set in the reach that flows across a large area of rural space (Fig. 1), which received large amounts of household garbage and had a lot of potential to consume oxygen.
Pollutants in sediment and overlying water both contribute to DO consumption, and we measured both SOD and CODCr and NH4 +-N in the overlying water. Water column in the Ziya river system had high CODCr (Fig. 3b). There was a big range of CODCr, which was 27.50 to 410.00 mg L−1. The average and median of CODCr at the sites were 76.08 ± 80.46 and 54.50 mg L−1 respectively, indicating a high potential to consume oxygen during its transformation. Four sites, S13, S16, S19, and S23, which were with the CODCr contents even exceeded 100 mg L−1 (Fig. 3b). NH4 +-N had similar situation with CODCr. The range of NH4 +-N concentration was 1.79–101.41 mg L−1, and the average and the median were 27.41 ± 14.45 and 22.11 mg L−1 respectively. Three sites, S16, S19, and S23, had higher NH4 +-N, which were in accordance with the rule of CODCr (Fig. 3b).
Overall, SOD in summer was higher than in autumn, and the ranges of SOD were 0.41–2.60 g m−2 day−1 in summer and 0.22–1.23 g m−2 day−1 in autumn, respectively. Combining the summer and autumn, the average and the median of SOD were 0.97 ± 0.34 and 0.63 ± 0.24 g m−2 day−1(Fig. 3a). Most sites reflected a similar change trend of SOD from summer to autumn. Nevertheless, there were individual sites with a different law, which perhaps related with the pulse of pollution discharge. For instance, SOD of the site S18 was 2.60 g m−2 day−1 in summer, but 0.22 g m−2 day−1 in autumn (Fig. 3a). This was because S18 was just at the reach running through a village, and a pollution event occurred probably when sampling.
Impact of oxygen depletion on macroinvertebrate taxa
The relationship between macroinvertebrate community and oxygen consumption potential was investigated by statistical tests. Response data (macroinvertebrates) were first analyzed by DCA to decide the methods used for the following analysis. It was shown that the response data had a gradient of 6.8 SD units long, so linear method was not appropriate and unimodal method was chosen (Lepx and Smilauer 2003). Ordination of macroinvertebrates was conducted on the basis of genus abundance. The eigenvalues for the four axes were separately 0.9584, 0.7910, 0.6636, and 0.2242, and of which the sum of eigenvalues of the first two axes accounted for 66.34 %. The samples represented by the sites could be classified into four groups, and each group had its special macroinvertebrate taxa. The first three groups were S01-S14, S02-S11-S09, and S03-S06-S15, which were with three to six genera. The remaining samples with more genera constituted the fourth group (Fig. 4a).
Ordination of macroinvertebrate genera and environmental variables was conducted by CCA (Fig. 4b). The eigenvalues of four axes were respectively 0.7936, 0.4257, 0.2291, and 0.9233. The proportion of the sum of eigenvalues of the first two axes was 51.41 %. The four environmental variables, DO, SOD, CODCr, and NH4 +-N, were divided into two categories. The arrows represented that DO and SOD almost appeared as one, indicating that they had a close correlation. Similarly, the angle between the arrows characterized CODCr, and NH4 +-N was acute, and they were closely related. Conversely, the angle between the above two sets of variables were obtuse which showed a low correlation. SOD and NH4 +-N, represented by long arrows, had the main contribution to the distribution of macroinvertebrates, while CODCr had a smaller contribution which was shown as a short arrow.
Most genera were at the circle near the center, which was probably because three environmental variables were conducive to oxygen consumption except DO, and they did not prefer anoxia. Nevertheless, there were taxa, such as Alocinma and Lymnaeidae, distributed far from the center and arrows of environmental variables, indicating that these taxa had a low frequency of occurrence and were indifferent to oxygen consumption. Many taxa distributed along the positive direction of the arrow of NH4 +-N and were regarded as eutrophy lovers. These taxa included Dicrotendipes, Corixa, Veliidae, Hydrosous, Gomphus, Hydrobius, Cricotopus, Tanytarsus, Cladopelma, etc. There were minor groups of taxa distributed in the negative direction of the arrow of NH4 +-N, including Nais, Procladius, Psychodiae, Limnodrilus, Hippeutis, etc. Most of the macroinvertebrates were distributed in the negative direction of the arrow of SOD, revealing a greater effect of SOD on benthic life (Fig. 4b).
Discussion
Although there may be multiple types of pollutants, DO depletion was a significant problem for the plain rivers in the ZRB. We determined the potential of DO consumption from the perspective of sediment and the overlying water synchronously. The study results showed that DO, SOD, and NH4 +-N were the major factors contributing to the macroinvertebrate distribution and DO consumption. Liu et al. (2009) summarized sediment oxygen demand values in various systems from a literature review: river systems with substrate from clay loam to sandy loam had a 0–2 g m−2 day−1 (Liu et al. 2009). Based on our measurements, the sediment grain size of plain river in the ZRB was 10.21–60.07 μm (average 24.68 ± 12.12 μm), and the SOD was 0.41–2.60 g m−2 day−1 in summer and was 0.22–1.23 g m−2 day−1 in autumn. Compared with other studies, the river sediment had a high potential to consume DO in the plain area of ZRB.
According to the aquatic life water quality criteria of the US Environmental Protection Agency, aquatic organisms will fail to live if DO concentration was below 2.3 mg L−1. The range and the average of DO concentration of water column in the Ziya plain rivers were separately 0.11 –4.03 mg L−1 and 1.35 ± 0.95 mg L−1, indicating a low level. In this study, the range and the average of NH4 +-N concentration were respectively 1.79–101.41 mg L−1 and 27.41 ± 14.45 mg L−1, which far exceeded the class V guideline of NH4 +-N (2.0 mg/l) according to the national quality standards for surface waters, China (GB3838-2002). Compared with other studies on nitrogen-polluted rivers, plain rivers were extremely polluted in the ZRB. For instance, NH4 +-N concentration was 0.17–11.5 mg L−1 in the Haicheng River (China) (Bu et al. 2011), 0.003–0.285 mg L−1 in the Le′an River Catchment (China) (Gao et al. 2011), 0.07–2.67 mg L−1 in the Ebro River Basin (NE Spain) (Lassaletta et al. 2010), 0.097–0.157 mg L−1 in the Jadro River (Croatia) (Stambuk-Giljanovic 2006), and average 0.51 mg L−1 in the Qiantang river (China) (Wu et al. 2003).
There were simple macroinvertebrate communities under the condition of extreme DO depletion in both sediment and the overlying water. First, there were low biomass and density. Other similar researches were compared with our results. For instance, for a tributary to the Rio Grande in the upper Paraná basin, a human-impacted tropical river, density ranged from 0 to 7467 ind m−2, and biomass ranged from 0 to 1515 mg m−2 during the rainy season (Aguiar et al. 2015). Huo et al. surveyed macroinvertebrate community in Songhua River of different seasons, and density and biomass were respectively 48. 63 and 23.12 g m−2 (Huo et al. 2012). A tributary of the Yellow river, which was heavily polluted with total nitrogen, had a density and biomass of 1173.5 and 5430.1 mg m−2, respectively (Fu et al. 2010). Macroinvertebrate assemblages were investigated in the river system which was with heavy organic pollution in plain area of Shanghai city. The results showed that density declined from the city (8776.3 ind m−2) to the countryside (690.3 ind m−2) (Chen et al. 2013). The Haiyinqiao reach of Zhujiang river, which received large amounts of industrial discharge, had the maximum of density (305,280 ind m−2) and biomass (664.72 ind m−2) (Jiang et al. 2011). The results of our research showed that the biomass ranged from 0.0009 to 2.57 g m−2, and the density from 11.11 to 3022.22 ind m−2, indicating a low level compared with other similar studies.
Second, the dominant taxa were principally Chironomidae and Tubificidae, and most of them were tolerant to pollution and anoxia. Justus’s et al. (2014) research showed that those extremely tolerant taxa had a competitive advantage and would be successful when DO minima were <2 mg L−1; Glyptotendipes had a higher tolerance than Chironomus to anoxia (Justus et al. 2014). Irving et al. (2004) conducted a 10-day toxicity testing with Chironomus tentans larvae, showing that exposure to 1.2 ± 0.1 mg L−1 DO for 10 days did not affect C. tentans’ survival or growth and significant behavioral changes at 2.0 ± 0.1 mg L−1 DO or less (Irving et al. 2004). When oxygen content was decreased to 0.3–0.5 mg L−1, combined with sulfidic conditions, Tubifex showed much lower mortality and even better growth than L. hoffmeisteri did (Volpers and Neumann 2005). These studies provided assisted proofs for explaining the abnormal macroinvertebrate assemblages in our study area; under the condition of extreme DO depletion, there were few genera that survived, and high tolerance taxa dominated, such as Limnodrilus, Tubifex, Chironomus, Glyptotendipes.
Indeed, macroinvertebrate has been applied broadly in assessment of ecological status of aquatic ecosystems, such as Benthic Index of Biotic Integrity (B-IBI) in the USA (Kerans and Karr 1994), River Invertebrate Prediction and Classification System (RIVPACS) in the UK (Wright 1995), Australian River Assessment System (AUSRIVAS) in Australia (Coysh et al. 2000), and the Development and Testing of an Integrated Assessment System for the Ecological Quality of Streams and Rivers throughout Europe using Benthic Macroinvertebrates (AQEM) in the EU (Hering et al. 2004). Thus, abnormal community structure of macroinvertebrate indicated a poor ecological status. It is advisable to improve DO condition and macroinvertebrate diversity in river harnessing and management in the plain areas of the ZRB.
Conclusions
This study evaluated the impact of hypoxia environment on macroinvertebrate community structure, and the results showed the following:
-
1.
There was an all-around hypoxia environment and a high potential of DO consumption in the plain rivers of the ZRB. The value ranges of DO, SOD, CODCr, NH4 +-N were separately 0.11–4.03 mg L−1, 0.41–2.60 g m−2 day−1, 27.50–410.00 mg L−1, and 1.79–101.41 mg L−1.
-
2.
Three macroinvertebrate classes, Insecta, Gastropoda, and Oligochaeta, appeared in the plain rivers of the ZRB. These classes included 9 orders, 30 families, and 54 genera. Aquatic insects were the overwhelming majority of taxa. There was a low biodiversity, with Shannon-Wiener index of 0.00–1.72.
-
3.
SOD and NH4 +-N had major impact on macroinvertebrate community, and the former had negative effect on most taxa, for instance, Nais, Branchiura, Paraleptophlebia, etc. These taxa were sensitive or had a moderate-high tolerance to pollution. NH4 +-N had both positive and negative impacts on benthic animals, for instance, Dicrotendipes, Gomphus, Cricotopus, etc., for the former and Procladius, Limnodrilus, Hippeutis, etc., for the latter. These taxa had a moderate-high tolerance to pollution.
References
Aguiar ACF, Gücker B, Brauns M, Hille S, Boëchat IG (2015) Benthic invertebrate density, biomass, and instantaneous secondary production along a fifth-order human-impacted tropical river. Environ Sci Pollut Res 22:9864–9876
Antanasijevic D, Pocajtb V, Perić-Grujićb RM (2014) Modelling of dissolved oxygen in the Danube River using artificial neural networks and Monte Carlo simulation uncertainty analysis. J Hydrol 519:1895–1907
Bu HM, Meng W, Zhang Y (2011) Nitrogen pollution and source identification in the Haicheng River basin in Northeast China. Sci Total Environ 409(18):3394–3402
Chen XH, Gao W, Liu WL, Sun CJ, Kang LJ (2013) Characteristics of macrobenthos community structure and its relationship to environmental factors within a typical plain river network. Ecol Environ Sci (in Chinese) 22(8):1310–1316
Comte L, Lek S, de Deckere E, de Zwart D, Gevrey M (2010) Assessment of stream biological responses under multiple-stress conditions. Environ Sci Pollut Res 17:1469–1478
Coysh J, Nichols S, Ransom G, Simpson J, Norris R, Barmuta L, Chessman B (2000) AUSRIVAS macroinvertebrate bioassessment predictive modelling manual. Cooperative Research Centre for Freshwater Ecology, Canberra
Ekau W, Auel H, Poertner HO, Gilbert D (2010) Impacts of hypoxia on the structure and processes in pelagic communities (zooplankton, macro-invertebrates and fish). Biogeosciences 7(5):1669–1699
Fu XC, Ye L, Xu YY, Cai QH (2010) Survey of water quality and benthos in the Yellow River Basin. Ecol Sci (in Chinese) 29(1):001–007
Gao HY, Zhuang X, Zhang Q (2011) Temporal and spatial change of non-point source nitrogen pollution in Le′an river catchment of Poyang Lake. Resour Environ Yangtze Basin (in Chinese) 20(5):597–602
Grieshaber MK, Hardewig I, Kreutzer U, Poertner HO (1994) Physiological and metabolic responses to hypoxia in invertebrates. Rev Physiol Bioch P 125:43–147
Hanes N, Irvine RL (1966) Oxygen uptake rates of benthal systems by a new technique. Proceedings of the 21st Industrial Waste Conference: 468–479
Heatherly TII, Whiles MR, Royer TV, David MB (2007) Relationships between water quality, habitat quality, and macroinvertebrate assemblages in Illinois streams. J Environ Qual 36(6):1653–1660
Hering D, Moog O, Sandin L, Verdonschot PFM (2004) Overview and application of the AQEM assessment system. Hydrobiologia 516(1–3):1–20
Hickey CW (1988) Benthic chamber for use in rivers-testing against oxygen mass balances. J Environ Eng-Asce 114(4):1653–1660
Hilsenhoff WL (1998) Rapid field assessment of organic pollution. J T N Am Benthol Soc 7(1):65–68
Holland A, Duivenvoorden LJ, Kinnear SHW (2015) Effect of key water quality variables on macroinvertebrate and fish communities within naturally acidic wallum streams. Mar Freshwater Res 66(1):50–59
Hu WF, Lo W, Chun H, Sin SN, Yu PH (2001) Nutrient release and sediment oxygen demand in a eutrophic land-locked embayment in Hong Kong. Environ Int 26(5):369–375
Huo TB, Liu MH, Jiang ZF, Li Z, Ma B, Yu HX (2012) Macrobenthos community structure of macrobenthos and bioassessment of water quality in main stream of Songhua River. Chin J Appl Ecol 23(1):247–254
Irving EC, Liber K, Culp JM (2004) Lethal and sublethal effects of low dissolved oxygen condition on two aquatic invertebrates, Chironomus tentans and Hyalella azteca. Environ Toxicol Chem 23(6):1561–1566
James A (1974) The measurement of benthal respiration. Water Res 8(11):955–959
Jiang WX, Lai ZN, Peng SY, Gao Y, Yang WL, Pang SX (2011) Primary study of macroinvertebrate community structure in the pearl river Guangzhou portion. Environmental Monitoring in China 27(5):69–72
John CM, Yang LF, Tian LX (1994) Aquatic insects of China useful for monitoring water quality. Hohai University Press, Nanjing
Justus BG, Mize SV, Wallace J, Kroes D (2014) Invertebrate and fish assemblage relations to dissolved oxygen minima in lowland streams of southwestern Louisiana. River Res Appl 30(1):11–28
Kerans BL, Karr JR (1994) A Benthic Index of Biotic Integrity (B-IBI) for rivers of the Tennessee Valley. Ecol Appl 4:768–785
Lassaletta L, Garcia-Gomez H, Gimeno BS, Rovira JV (2010) Headwater streams: neglected ecosystems in the EU Water Framework Directive. Implications for nitrogen pollution control. Environ Sci Policy 13(5):423–433
Lenat DR (1993) A biotic index for the southeastern United States derivation and list of tolerance values, with criteria for assigning water-quality ratings. J N Am Benthol Soc 12(3):279–290
Lepx J, Smilauer P (2003) Multivariate analysis of ecological data using CANOCO. Cambridge University Press, Cambridge
Leunda PM, Oscoz J, Miranda R (2009) Longitudinal and seasonal variation of the benthic macroinvertebrate community and biotic indices in an undisturbed Pyrenean river. Ecol Indic 9:52–63
Li WZ, Li XY, Wang HL, Su JJ (2012) Spatial distribution of the main contaminations in aquatic environment in Fuyang River. Acta Sci Circumstantiae (in Chinese) 32(11):2814–2819
Liu XB, Li GF, Liu ZG, Guo WH, Gao NY (2010) Water pollution characteristics and assessment of lower reaches in Hai River Basin. Procedia Environ Sci 02:199–206
Liu WC, Chen WB, Kimura N (2009) Measurement of sediment oxygen demand to simulate dissolved oxygen distribution: case study in the Main Danshuei River Estuary. Environ Eng Sci 26(12):1701–1711
Liu YY, Zhang WZ, Wang YX (1979) Economic animals in China, freshwater mollusks. Science Press, Beijing
Maul JD, Farris JL, Milam CD, Cooper CM, Testa S, Feldman DL (2004) The influence of stream habitat and water quality on macroinvertebrate communities in degraded streams of northwest Mississippi. Hydrobiologia 518(1–3):79–94
McNeil DG, Closs GP (2007) Behavioural responses of a south-east Australian floodplain fish community to gradual hypoxia. Freshwater Biol 52(3):412–420
Nandan SB, Azis PKA (1995) Fish mortality from anoxia and sulphide pollution. J Hum Ecol 6(2):97–104
National Bureau of Statistics of the People’s Republic of China. Bulletin of the sixth national census of Hebei province in 2010
Rao YR, Howell T, Watson SB, Abernethy S (2014) On hypoxia and fish kills along the north shore of Lake Erie. J Great Lakes Res 40(1):187–191
Riedel B, Pados T, Pretterebner K, Schiemer L, Steckbauer A, Haselmair A (2014) Effect of hypoxia and anoxia on invertebrate behaviour: ecological perspectives from species to community level. Biogeosciences 11(6):1491–1518
Stambuk-Giljanovic N (2006) The pollution load by nitrogen and phosphorus in the Jadro River. Environ Monit Assess 123(1–3):13–30
State Environmental Protection Agency (2002) Method of monitoring and analysis of water and wastewater, 4th edn. China Environmental Science Press, Beijing
Tang WZ, Mao ZP, Zhang H, Shan BQ, Zhao Y, Ding YK (2015) Water resources: the prerequisite for ecological restoration of rivers in the Hai River Basin, northern China. Environ Sci Pollut Res 22:1359–1365
The Editorial Board of Hai river (1997) The Hai River Basin, vol 1. China Water & Power Pres, Beijing
Urbina MA, Glover CN (2012) Should I stay or should I go?: physiological, metabolic and biochemical consequences of voluntary emersion upon aquatic hypoxia in the scaleless fish Galaxias maculatus. J Comp Physiol B 182(8):1057–1067
Volpers M, Neumann D (2005) Tolerance of two tubificid species (Tubifex tubifex and Limnodrilus hoffmeisteri) to hypoxic and sulfidic conditions in novel, long-term experiments. Arch Hydrobiol 164(1):13–38
Wang BX, Yang LF (2004) A study on tolerance values of benthic macroinvertebrate taxa in eastern China. Acta Ecol Sin (in Chinese) 24(12):2768–2775
Wang C, Shan BQ (2012) The distribution of aerobic ammonia oxidizing microorganisms in Ziya River, Haihe Basin. Acta Sci Circumstantiae (in Chinese) 32(12):2943–2950
Wang JG, Huang HB, Yang MX (2003) Tolerance values of benthic macroinvertebrates and bioassessment of water quality in the Lushan Nature Reserve. Chin J Appl Environ Biol 9(3):279–284
Wright JF (1995) Development and use of a system for predicting macroinvertebrates in flowing waters. Austr J Ecol 20:181–197
Wu J, Yu ZM, Qian TM (2003) Studies on nitrogen pollution characteristics in Hangzhou section of the Qiantang river. Resour Environ Yangtze Basin (in Chinese) 12(6):552–556
Xing SW, Wang JC, Ding ZJ, Jiang YW (2012) Large benthonic invertebrates’ tolerance values and water quality evaluation in Liaoning Province. Environ Prot Sci (inChinese) 39(3):29–33
Zhang H, Ludsin SA, Mason DM, Adamack AT, Brandt SB, Zhang XS (2009) Hypoxia-driven changes in the behavior and spatial distribution of pelagic fish and mesozooplankton in the northern Gulf of Mexico. J Exp Mar Biol Ecol 381:S80–S91
Zhao W (2005) Aquatic biology. China Agriculture Press, Beijing
Zhao Y, Shan BQ, Zhang WQ, Wang C (2014) Forms and spatial distribution characteristics of nitrogen in Ziya River Basin. Environ Sci 35(1):143–149
Zhu YQ (2014) Ecological situation and governing method of plain rivers in the Ziya River Basin. Water Conserv Hebei Province in China 04:38–39
Acknowledgments
Financial support was provided by the “National Water Pollution Control and Management Technology Major Projects of China (2012ZX07203-006)”. Seven people participated in the field work, but we especially thank engineer Xuehong Kong. Thanks are due to Dr. Juguang Wang and his assistants from College of Fisheries of Huazhong Agricultural University for the identification of macroinvertebrates.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Ding, Y., Rong, N. & Shan, B. Impact of extreme oxygen consumption by pollutants on macroinvertebrate assemblages in plain rivers of the Ziya River Basin, north China. Environ Sci Pollut Res 23, 14147–14156 (2016). https://doi.org/10.1007/s11356-016-6404-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6404-z