Abstract
The annual water level regulating of the Three Gorges Reservoir prolonged the submerged duration (from 2 to 8 months) and resulted in the reversal of natural flood rhythms (winter submerged). These changes might alter plant community characteristics in the water level fluctuation zone (WLFZ). The aim of this study was to determine the plant community characteristics in the WLFZ and their responses to the environmental factors (i.e., annual hydrological regulation, topographic characteristics, soil physical properties and soil nutrients). The height, coverage, frequency and biomass of each plant species and the soil properties at each elevation zone (150, 155, 160, 165 and 170 m) were measured from March to September in 2010. Univariate two-factor analysis and redundancy analysis (RDA) were used to analyze the spatial and temporal variations of the community characteristics and identify the key environmental factors influencing vegetation. We found that 93.2 % of the species analysed were terrestrial vascular plants. Annual herbs made up the highest percentage of life forms at each altitude. The differences in the species number per square metre, the Shannon–Wiener diversity index and the biomass of vegetation demonstrated statistical significance with respect to sampling time but not elevation. The most dominant species at altitudes of 150, 155, 160, 165 and 170 m were Cynodon dactylon, Cyperus rotundus, Digitaria sanguinalis, Setaria viridis and Daucus carota, respectively. The concentrations of soil nutrients appeared to be the lowest at an altitude of 150 m, although the differences with respect to elevation were not significant. The results of the RDA indicated that the key factors that influenced the species composition of vegetation were elevation, slope, pH and the concentration of soil available phosphorus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
River systems around the globe are increasingly affected by damming to satisfy increasing demands for hydroelectricity, flood mitigation and reliable water supplies (O’Connor 2001). The impoundment of dams can significantly alter river flow, shorelines, sediment and aquatic fauna and can result in the loss of original riparian vegetation (Merritt and Wohl 2006; New and Xie 2008; Willison et al. 2013). Alteration of hydrological regimes due to the operation of reservoirs may be the main mechanism affecting shoreline vegetation (Nilsson and Berggren 2000; Ye et al. 2013b). Other factors (e.g., flooding, soil nutrients and soil contamination) influenced by dams also have the potential to change the surrounding plant community (Aerts et al. 2003). However, there is very little information about the vegetation responses to annual hydrological regime regulation, topographic characteristics, soil physical properties or soil nutrients in the impoundment areas of reservoirs.
The Three Gorges Dam (TGD), the largest hydropower project in the world, was initiated in 1994. The first impoundment occurred in June 2003, and the water level of the Three Gorges Reservoir (TGR) rose from approximately 60 to 135 m above the original riverbank. The second impoundment occurred in October 2006, and the water level rose to 156 m. The Three Gorges Dam reached its design-maximum reservoir water level of 175 m (574 ft) for the first time on October 26, 2010, which resulted in the formation of a water level fluctuation zone (WLFZ) with an area of 302.0 km2. The WLFZ is the exposed region between the high and low water levels along the bank of a reservoir (Wantzen et al. 2008). Compared with other reservoirs, the WLFZ of the TGR has a longer submergence period. The zones at altitudes of 150 and 170 m are submerged for 8 months and nearly 2 months, respectively. In addition, the hydrological regime of the TGR is the exact opposite of the natural flood rhythms of the Yangtze River (Fan et al. 2012). The water level is maintained at 145 m in summer (from June to September) and at 175 m in winter (from October to December).
Before the implementation of the TGR, the main vegetation types in the zone below an elevation of 175 m were trees (e.g., Pinus massoniana and Cupressus funebris), shrubs (e.g., Vitex negundo, Securinega suffruticosa and Myricaria laxiflora) and herbs (Imperata cylindrica, Arthraxon hispidus and Cynodon dactylon) (Chen et al. 2008). The filling of the TGR may have resulted in the loss of pre-existing vegetation and the formation of novel plant communities. However, several studies have focused only on plant species composition (Wang et al. 2012), heavy metal (Ye et al. 2013a), soil major nutrients (Ye et al. 2013b) and eco-friendly utilisation mode of resources (Yuan et al. 2013) in the WLFZ. Studies of other physical and chemical soil properties have not been reported, and little is known about the WLZF of the TGR. The variation in vegetation in the WLFZ is caused by complex interactions involving the hydrological regime, topographic characteristics, soil nutrients and contamination. Most plants in the WLFZ of plain reservoirs such as lakes are hydrophytes or helophytes because of the relatively short submergence time and low water level variations (Crivelli and Grillas 1995; Miranda and Hodges 2000). The plants in the WLFZ of valley-dammed reservoirs in the southwest of China, such as the Ertan reservoir in Sichuan province, could not survive because of the steep slopes. The WLFZ of the TGR has complicated topographical characteristics and a special hydrological regulating regime. For the WLFZ managers, further studies on the important factors influencing the vegetation may contribute to greater understanding and better prediction of the successional trends of the plant community, which may be essential for managing the plant community, protecting the WLFZ habitat and restoring the WLFZ ecosystem function.
Our specific objectives were as follows: (1) to confirm the plant community characteristics in the WLFZ after implementation of the TGR, (2) to confirm how much nitrogen and phosphorus is released by the death and the decomposition of vegetation after winter submergence and (3) to determine the effects of annual hydrological regime regulation and soil properties on the plant community in the WLFZ.
Study area and methods
Study area
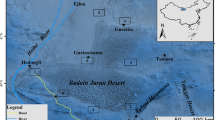
The Xiaojiang River is the tributary with the largest area of the WLFZ in the TGR in China (Fig. 1). The main stream belonging to the TGR is 117.5 km in length. The WLFZ in the Xiaojiang River is located at 30°49′–31°41′ N, 107°56′–108°54′ E. The area of the WLFZ is 48.02 km2, which accounts for 15.9 % of the total WLFZ in the TGR. The average annual temperature is 18.6 °C. The average annual precipitation is 1,100–1,500 mm, most of which occurs between April and October. The soil types in the five transects from the Qukou transect to the Shuangjiang transect are yellow soil, yellow soil, red soil, red soil and red soil. The duration of submergence is higher at lower elevations. For example, the submerged time at altitudes of 150, 155, 160, 165 and 170 m were 256, 200, 147, 116 and 57 days, respectively, between October 2009 and June 2010.
Vegetation sampling
Vegetation surveys were performed along five transects (Fig. 1) between March and September in 2010. The width of each transect ranged from 30 to 60 m. These surveys were performed during flood-regulation periods. The water level fluctuated primarily between 145 and 150 m. The water level only rose to 172.5 m (566 ft) between 2008 and 2009. Therefore, we divided each transect into five elevation zones (150, 155, 160, 165 and 170 m). Three square vegetation sampling plots (1 × 1 m) were located at 10- or 20-m intervals along each elevation zone of each transect. The water levels in each of the months from March to September were 156.7, 154.4, 154.4, 147.5, 150.9, 153.1 and 162.4 m, respectively. Due to the submersion of some elevation zones in different months, the number of plots in the months from March to September was 45, 60, 60, 75, 45, 45 and 30, respectively. A total of 390 plots were sampled. We measured the height, coverage, frequency and above-ground biomass of each plant species in each plot. Based on the plant life forms, the plants were sorted into five groups: perennial herbs, annual herbs, climbing plants, shrubs and trees. Based on their dependence on water, the plants were classified into three groups: hydrophytes, mesophytes and xerophytes. Plant samples were oven-dried at 65 °C and analysed for dry weight. The nitrogen and phosphorus concentrations in 36 species were analysed (Bao 2005).
Environmental factors
The slopes of the transects were measured with an inclinator and were classified into six grades (I through VI) : <5, 6–15, 16–25, 26–35, 35–45 and >45°. After plants had been removed, the soil depth (SD) was measured with a soil auger (Arbeláez and Duivenvoorden 2004). The soil density (0–20 cm) was investigated using the cutting ring method. Three random topsoil samples (0–20 cm) were collected and then mixed to form a composite sample at each elevation zone of each transect. A total of 120 mixed samples were sealed in plastic bags and brought to the laboratory. These soil samples were dried at 70 °C for chemical analysis. We measured the soil pH and contents of soil organic matter (SOM), total nitrogen (TN), available nitrogen (AN), total phosphorus (TP), available phosphorus (AP), total potassium (TK) and available potassium (AK), using methods described by Bao (2005).
Data analysis
The relative height (RH), relative coverage (RC), relative frequency (RF) and relative biomass (RB) of each plant species were expressed as the percentages of the height, coverage, frequency and biomass, respectively, of all species recorded. The sum of the above four relative variables of a species was called the importance value index (IVI = RH + RC + RF + RB) (Jerry et al. 2002; Zhang 2011). The relative IVI of each plant species was expressed as the percentage of the IVI for all species recorded. The IVI, which ranged from 0 to 4, represented the relative importance or dominance of a plant species in a plot zone. We calculated the number of species (R) per square metre for each plot and the Shannon–Weiner diversity index (H′): \( {H}^{\prime}=-\sum\limits_{i=1}^s {{P_i}\ln {P_i}} \), where P i is the relative IVI of the ith species (i = 1 to s) and s is the total number of species. Dominance was used to describe the degree of predominance of a plant species in an ecological community. Y i = P i × f i , where Y i is the dominance of the ith species at the elevation of the transects, P i is the relative IVI of the ith species, and f i is the frequency of the ith species. Dominant species were defined as those with dominance values ≥0.02 (Peng et al. 2011; Zhang 2011).
A univariate two-factor analysis of variance of the general linear model was performed to determine whether there was a significant interaction effect between sampling time (months) and elevation. The significances of differences in the means of species richness, fresh weight, coverage and soil properties were compared using Tukey’s test. When comparing the soil nutrients in the different transects, the mixed samples from all elevations were used as replicates for each transect. The statistical analyses described above were performed using SPSS 16.0.
A preliminary detrended correspondence analysis of the IVI of the species at each elevation of each transect revealed that the gradient lengths were <3.0, indicating that the majority of the species exhibited linear responses to underlying environmental variation, which justified the use of linear multivariate methods. Thus, we identified the key environmental factors influencing the vegetation by redundancy analysis (RDA) using the Canoco 4.5 software package (Leps and Smilauer 2003; Michael et al. 2012). The scaling of the ordination focused on inter-species correlations, and a Monte Carlo permutation test based on random permutations was conducted to test the significance of the eigenvalues of the first canonical axis. The species data were log-transformed to reduce the effects of extreme values, and the environmental data were square-root transformed (Leps and Smilauer 2003; Gazer 2011).
Results
Species composition
A total of 146 vascular plant species were identified, belonging to 116 genera of 38 families. The plant families with the largest number of species in the studied area were Asteraceae (N = 25, 17.1 %), Poaceae (N = 24, 16.4 %), Fabaceae (N = 10, 6.8 %), Polygonaceae (N = 8, 5.5 %), Cyperaceae (N = 5, 3.4 %) and Lamiaceae (N = 5, 3.4 %). A total of 107 dicot (73.3 %), 36 monocot (24.7 %) and 3 fern (2.0 %) species were recorded in the sampling sites.
The life forms present at the highest percentage at each altitude were annual herbs. The percentages of annual herbs at the altitudes of 150, 155, 160, 165 and 170 m were 71.4, 68.1, 57.0, 53.0 and 56.0 %, respectively. The less frequent life forms (mostly at the altitude of 170 m) were climbing plants (Ficus tikoua, Paederia scandens, Vitis amurensis, Pueraria phaseoloides and Cayratia japonica), shrubs (Boehmeria nivea, Lespedeza davidii, Lespedeza cuneata and V. negundo) and trees (Morus alba, Albizia kalkora and Broussonetia papyrifera).
We found that 93.2 % of the plant species present were terrestrial vascular plant species (N = 136) in the study sites, whereas only 10 hydrophytes or helophytes—Equisetum arvense, E. debile, Polygonum hydropiper, Oryza sativa, Ranunculus sceleratus, Alternanthera philoxeroides, Oenanthe javanica, Centella asiatica, Beckmannia syzigachne and Ludwigia prostrata—were identified in the abandoned pits, ponds and paddy fields.
Dominant species
The vegetation distribution and the IVI of the dominant plant species varied with elevation (Table 1). The dominant species exhibiting the highest IVI values at the altitudes of 150, 155, 160, 165 and 170 m were C. dactylon (1.42 ± 0.66), Cyperus rotundus (0.81 ± 0.67), Digitaria sanguinalis (0.62 ± 0.67), Setaria viridis (0.55 ± 0.69) and Daucus carota (0.97 ± 0.42), respectively. The IVI of X. sibiricum, C. dactylon, C. rotundus and P. hydropiper were negatively correlated with elevation, whereas the IVI of Artemisia annua and D. carota were positively correlated with elevation.
Plant diversity
The species number per square metre (R) ranged from 3 to 23, with an average of 10, in the WLFZ. The mean R was the highest during July (12.5 ± 4.3) and was lower in June, May, April, August, September and March in descending order (Fig. 2a). The mean R at the altitude of 165 m was the highest (10.7 ± 4.8) and was lower at the altitudes of 170, 160, 155 and 150 m in descending order (Fig. 2b). The significance values for sampling time and for the interaction of sampling time and elevation, but not for elevation alone, were <0.05. Therefore, the differences in R exhibited statistical significance with respect to sampling time (F = 14.285, p < 0.01) and sampling time × elevation (F = 2.687, p < 0.05) but not with respect to elevation.
The Shannon–Wiener diversity index (H′) values ranged from 1.64 to 3.41, with an average of 2.93, in the WLFZ. The average H′ was highest (3.18.0 ± 0.11) during July and was lower in May, June, April, August, September and March in descending order (Fig. 2c). The average H′ was highest (3.12 ± 0.11) at the altitude of 170 m and was lower at 165, 160, 155 and 150 m in descending order (Fig. 2d). However, there were no significant differences in H′ with respect to sampling time, elevation or sampling time × elevation.
Plant biomass
The fresh weight of vegetation ranged from 5.1 to 3,513.0 g m−2, with an average of 627.2 g m−2. The average fresh weight was highest (1,294.2 ± 774.3 g m−2) during September and was lower in August, June, July, May, April and March in descending order (Fig. 3a). The average fresh weight was highest (721.9 ± 614.5 g m−2) at the altitude of 170 m and was lower at 165, 150, 160, 165 and 155 m in descending order (Fig. 3b). Differences in the fresh weight were significant with respect to sampling time (F = 12.77, p < 0.01) but not with respect to elevation or sampling time × elevation. The fresh weight (N = 120, r = −0.227, p < 0.05) and the coverage (N = 120, r = −0.429, p < 0.001) were also highly negatively correlated with the slope (Fig. 4a, b). The dry weight of vegetation varied from 4.8 to 920.5 g m−2, with an average of 162.1 g m−2, from March to September in 2010. Differences in the dry weight also demonstrated statistical significance with respect to sampling time (F = 11.018, p < 0.01) but not elevation or sampling time × elevation.
Environmental factors
The soil depth ranged from 7.6 to 73.0 cm, with an average of 29.91 cm. Differences in the soil depth (F = 9.874, p < 0.01) and soil density (F = 3.176, p < 0.05) were significant with respect to elevation. The average soil depth was greatest at slopes <5°. Differences in the soil depth were significant with respect to slope (F = 9.293, p < 0.01; Fig. 4c). The soil density ranged from 0.79 to 1.9 g m−3, with an average of 1.35 g m−3. The average soil density was highest at slopes of >45°. Differences in the soil density were significant with respect to slope (F = 2.664, p < 0.05; Fig. 4d). The average concentrations of SOM, TN, AN, TP, AP, TK and AK, but not soil pH, were lowest at the altitude of 150 m, although the differences with respect to elevation were not significant (Table 2). The differences in the average concentrations of SOM, TN, AN, TP and TK, but not pH, AP and AK, were significant with respect to sampling time. Differences in the concentrations of all soil nutrients were significant with respect to the different transects investigated.
The RDA of species and environmental variables
The ordination analysis revealed the environmental gradient in the area. The results of the RDA showed that elevation was the best explanatory variable (Table 3, Fig. 5). The conditional effects indicated the following: (1) the contributions of elevation (F = 8.29, p < 0.01), slope (F = 2.46, p < 0.05), pH (F = 2.27, p < 0.05) and AP (F = 1.95, p < 0.05) were significant during the forward selection; (2) the contributions of the remaining variables were not significant. The RDA results also showed that there was a strong correlation between vegetation and environmental factors, with species–environment correlations of 0.979 on the first axis and 0.808 on the second axis. The eigenvalues for the first four RDA axes were 0.287, 0.099, 0.087 and 0.071. The cumulative percentage variance of species–environment relations of the first axis and second axis were 40.4 and 54.3 %, respectively. The sum of all the RDA canonical eigenvalues was 0.711.
Ordination diagram showed the result of RDA analysis of the important value index of species and environment variables. SOM Soil organic matter, TN total nitrogen, AN available nitrogen, TP total phosphorus, AP available phosphorus, TK total potassium, AK available potassium. Only species shown were those with >3 % fit. Ar an, Artemisia annua; Al ph, Alternanthera philoxeroides; Ac au, Acalypha australis; Ar ar, Artemisia argyi; Ar hi, Arthraxon hispidus; Bi pi, Bidens pilosa; Ca ab, Carpesium abrotanoides; Cl ch, Clinopodium chinense; Co ca, Comnyza canadensis; Cy da, Cynodon dactylon; Cy ro, Cyperus rotundus; Da ca, Daucus carota ; Di sa, Digitaria sanguinalis; Ec pr, Eclipta prostrate; Ec cr, Echinochloa crusgali; Gn af, Gnaphalium affine; Ma ja, Mazus japonicas; Se vi, Setaria viridis; Tr pe, Trigonotis peduncularis; Ox co, Oxalis corniculata; Po hy, Polygonum hydropipe; Vi sa, Vicia sativa; Xa si, Xanthium sibiricum
Nitrogen and phosphorus contents in 36 species
The nitrogen and phosphorus contents in the different species varied (Fig. 6). The nitrogen contents in the plants ranged from 5.02 (Rumex acetosa) to 34.75 g kg−1 (Chenopodium ambrosioides), with an average of 13.04 g kg−1. The phosphorus contents in the plants ranged from 1.62 (Hemarthria altissima) to 5.90 g kg−1 (Eclipta prostrata), with an average of 3.86 g kg−1. The dry weights varied from 176.9 to 556.9 g m−2, with an average of 307.6 g m−2 in September 2010. Therefore, the nitrogen and phosphorus released from the vegetation that died and decomposed during submergence varied from 0.89 to 7.3 g m−2 and from 0.29 to 3.29 g m−2 respectively.
Discussion
Characteristics of vegetation in relation to environmental factors in the WLFZ
Among the different environmental variables (topographic and soil variables) studied, elevation was found to be the main factor that influenced the distribution and the important value index of species in the WLFZ. After the filling of the TGR, the original vegetation, especially trees and shrubs, could not survive because they could not tolerate the winter submergence and summer drought (New and Xie 2008). In the present study, we found that a few shrubs (B. nivea, L. davidii, L. cuneata and V. negundo) and trees (M. alba, A. kalkora and B. papyrifera) only appeared at the altitude of 170 m. The main vegetation type at the five elevations was herbs. The dominant species with the highest important value index at the altitudes of 150 and 155 m were perennial herbaceous plants (C. dactylon and C. rotundus). However, the dominant species at elevations >160 m were annual herbs, including D. sanguinalis and D. carota. The previous studies have also reported that C. dactylon was the dominant species at lower elevations, whereas Comnyza canadensis was dominant at an elevation of 170 m in the WLFZ in the TGR (Wang et al. 2012; Ye et al. 2013b). A number of studies have also shown that relatively flood-tolerant species are found mainly at lower elevations and that relatively flood-sensitive species are found mainly at higher elevations (Van Eck et al. 2004; Lesnssen and De Kroon 2004). This species distribution pattern might be caused by several synthetic attributes (e.g., the inundation water depth, the tolerant flooding capacity, the life form, the dispersal mode and the inter-specific competition).
Several perennial herbs, such as C. dactylon, C. rotundus and H. altissima, are able to survive the submerged period because of their dormant tubers or rhizomes. These species may have evolved morphological, physiological and biochemical adaptations to oxygen deficiency. For example, C. dactylon, C. rotundus and Rumex palustris develop higher underground biomass, photosynthetic capacity and amylase activity (Peña-Fronteras et al. 2009; Hong et al. 2011; Jie et al. 2012). Recently, considerable progress has been made in terms of understanding the molecular aspects governing these responses to flooding. Oxygen-sensing mechanisms in Arabidopsis and rice have been found to be mediated by an N-end rule pathway. SUB1A, a gene responsible for tolerance of complete submergence, was found only in some low-yielding rice varieties in India and Sri Lanka (Gibbs et al. 2011; Francesco et al. 2011). Because C. dactylon and C. rotundus are able to tolerate complete submergence for 7 months, they may also have underlying molecular mechanisms that will be discovered in the future.
The annual herbs of the WLFZ at >160 m, such as X. sibiricum, B. pilosa, E. prostrata, C. canadensis and P. hydropiper, had short life cycles (Kolar and Lodge 2001). In addition, the exposed time of the WLFZ at >160 m was at least 200 days. Thus, these species may rapidly attain maturity before being submerged. In addition, seed dispersal by wind, water, animals and humans may be an important factor (Merritt and Wohl 2006). For example, the seeds of the dominant plants, such as C. canadensis and Bidens pilosa, are dispersed by wind (Dauer et al. 2006).
Slope is also a key factor influencing plant community characteristics. In general, the steeper the slope of the WLFZ, the greater the amount of soil loss was resulting from erosion by water waves and rainfall (Marden et al. 2005). This effect is a potential threat to the existing and growing plants. Several measures for soil and water conservation should be considered in vegetation restoration efforts.
A total of 136 terrestrial vascular plant species were found in the study sites. In general, hydrophytes or helophytes could not survive because of continuous exposure of the WLFZ in summer. Only a few hydrophytes or helophytes were found in the abandoned pits, ponds and paddy fields that held water. Seeds, tubers, or rhizomes of the most herbaceous terrestrial plants could sprout and grow quickly once the WLFZ was exposed. Therefore, the pattern of herbaceous terrestrial plants being the most abundant life form might not vary during the early stages of succession.
Soil properties and their influence factors in the WLFZ
The pH of the soil was found to be highest at the altitude of 150 m, with an average of 8.29. In general, the pH of soil increases with increasing soil moisture, especially for acidic soil types (Huang 2000). Flooding simulation experiments have shown that the soil pH of acidic soil might increase after flooding events (Sun et al. 2007; Valenzuela-Encinas et al. 2009). Because the submergence period is the longest at the elevation of 150 m, it is likely that the soil pH increases with soil moisture, indicating that the soil type is most likely acidic.
The concentrations of nitrogen and phosphorus appeared to be lowest at the elevation of 150 m, although the differences with elevation were not significant. The long-term submergence in winter and the high frequency of floods in summer may result in this pattern, as soil nutrients may be released when submerged and soil that serves as a nutrient source may be scoured by repeated flooding. Qiu and McComb (1996) reported that the concentration of total N was reduced after long-term submergence. However, different inundation periods and water depths in the WLFZ did not play a significant role in current soil development (Wang et al. 2012). In addition, the variation in available phosphorus was greater than of the variation in total phosphorus. Soil nutrients, especially available nutrients, are also affected by the parent types of soil, plant uptake and rainfall (Bardgett and Wardle 2003; Knoepp and Vose 2007). For example, the soil nitrogen contents in different vegetation restoration types are higher in shrub soils than in herb soils (Ye et al. 2012). The concentration of dissolved reactive phosphorus increases significantly with increasing rainfall intensity (Shiqaki et al. 2007).
Practical implications
The filling of the TGR resulted in loss of the original vegetation, especially trees and shrubs. The current plant species with high economic value should be chosen in the process of vegetation restoration. For example, the flood-tolerant edible hydrophytes in a dike-pond system may provide economic returns for farmers (Willison et al. 2013). The chemical constituents (xanthanolides, kaurene glycosides and essential oils) in X. sibiricum and C. rotundus have anti-oxidant, anti-bacterial, anti-inflammatory and anti-tumour properties (Han et al. 2007; Jin et al. 2011). The sesquiterpene artemisinin, derived from A. annua, has received attention due to its anti-malarial properties and its potential for curing some cancers (Arsenault et al. 2008). The acetone extract from A. annua possesses good insecticidal properties (Khosravi et al. 2011). M. alba has been reported to be a potential source of protein for ruminant livestock (Doran et al. 2007). Therefore, the above-described plants should be prioritised in vegetation restoration efforts.
In winter inundating season, the nitrogen and phosphorus released from the vegetation in the WLFZ ranged from 0.89 to 7.3 g m−2 and from 0.29 to 3.29 g m−2, respectively. The death and decomposition of vegetation may result in secondary pollution during submergence. Harvesting herbs with high nitrogen and phosphorus contents might effectively remove nitrogen and phosphorus from the WLFZ. To avoid secondary pollution, this vegetation should be harvested before it is submerged. The harvesting of several economically valuable herbs may reduce the cost of vegetation restoration. The economic and ecological benefits of harvesting of several plant species, such as M. alba, H. altissima and C. dactylon, should be considered.
However, there are many uncertainties that cannot be answered by our study because the WLFZ is only 2 years old, and the plants and soil in the WLFZ are likely to continue to be in stages of acclimation. Therefore, long-term investigations and observations should be continued in this area to identify and monitor changes in the characteristics of the plant communities and soil properties.
Conclusions
A total of 146 species of vascular plants were found in the WLFZ. There were no significant differences in the number of species per square metre, the biomass or the biodiversity index at different elevations. The dominant species of plants varied with elevation. The dominant species, exhibiting the highest important value index, at the altitudes of 150, 155, 160, 165 and 170 m were C. dactylon, C. rotundus, D. sanguinalis, S. viridis and D. carota, respectively. Soil depth and soil density were significantly different at different elevations, but differences in soil pH values, the concentrations of SOM, TN, AN, TP, AP, TK and AK were not. Therefore, different inundation periods and water depths may only result in different dominant species at different elevations and may not play a significant role in the chemical properties of the soil. In winter inundating season, the released nitrogen and phosphorus from the vegetation varied from 0.89 to 7.3 g m−2 and from 0.29 to 3.29 g m−2 respectively. The results of the RDA indicated that the main factors influencing the vegetation characteristics were the elevation, the slope, the pH and the concentration of soil available phosphorus.
References
Aerts R, de Caluwe H, Beltman B (2003) Is the relation between nutrient supply and biodiversity co-determined by the type of nutrient limitation? Oikos 101:489–498
Arbeláez MV, Duivenvoorden JF (2004) Patterns of plant species composition on Amazonian sandstone outcrops in Colombia. J Veg Sci 15:181–188
Arsenault PR, Wobbe KK, Weathers PJ (2008) Recent advances in artemisinin production through heterologous expression. Curr Med Chem 15:2886–2896
Bao SD (2005) Analysis of soil chemistry. Chinese Agriculture Press, Beijing, pp 211–405, p25-16
Bardgett RD, Wardle AD (2003) Herbivore-mediated linkages between aboveground and belowground communities. Ecology 84:2258–2268
Chen WL, Jiang MX, Zhao CM, Tian ZQ (2008) Plants and vegetation in the valley of the Three Gorges Reservoir. China Waterpower Press, Beijing, pp p28–p78
Crivelli AJ, Grillas P (1995) Responses of vegetation to a rise in water level at Kerkini Reservoir (1982–1991), a Ramsar Site in Northern Greece. Environ Manage 19:417–430
Dauer JT, Mortensen DA, Humston R (2006) Controlled experiments to predict horseweed (Conyza canadensis) dispersal distances. Weed Sci 54:484–489
Doran MP, Laca EA, Sainz RD (2007) Total tract and rumen digestibility of mulberry foliage (Morus alba), alfalfa hay and oat hay in sheep. Anim Feed Sci Tech 138:239–253
Fan Y, Liu WW, Wang J, Liao L, Wang Y (2012) Riparian vegetation’s responses to the new hydrological regimes from the three gorges project: clues to revegetation in reservoir water-level-fluctuation zone. Acta Ecol Sin 32:89–98
Francesco L, Kosmacz M (2011) Oxygen sensing in plants is mediated by an N-end rule pathway for protein destabilization. Nature 479:419–422
Gazer MH (2011) Vegetation composition and floristical diversity in date palm orchards of Central Saudi Arabia. Acta Bot Hung 53:111–112
Gibbs DJ, Lee SC, Isa NM (2011) Homeostatic response to hypoxia is regulated by the N-end rule pathway in plants. Nature 479:415–418
Han T, Li HL, Zhang QY, Han P, Zheng HC, Rahman K, Qin LP (2007) Bioactivity-guided fractionation for anti-inflammatory and analgesic properties and constituents of Xanthium strumarium L. Phytomedicine 14:825–829
Hong M, Guo QS, Nie BH, Kang Y, Pei SX, Jin JQ, Wang XF (2011) Responses of Cynodon dactylon population in hydro-fluctuation belt of Three Gorges Reservoir area to flooding-drying habitat change. Ying Yong Sheng Tai Xue Bao 22:2829–2835
Huang CY (2000) Agrology. Chinese Agriculture Press, Beijing, pp p171–p178
Jerry RB, Paul LR, Michael B (2002) Patterns of tree dominance in coniferous riparian forests. Forest Ecol Manag 166:311–329
Jie SL, Fan DY, Xie ZQ, Zhang XY, Xiong GM (2012) Features of leaf photosynthesis and leaf nutrient traits in reservoir riparian region of Three Gorges Reservoir, China. Acta Ecol Sin 32:1723–1733
Jin JH, Lee DU, Kim YS, Hyun PK (2011) Anti-allergic activity of sesquiterpenes from the rhizomes of Cyperus rotundus. Arch Pharm Res 34:223–228
Khosravi R, Sendi JJ, Ghadamyari M, Yezdani E (2011) Effect of sweet wormwood Artemisia annua crude leaf extracts on some biological and physiological characteristics of the lesser mulberry pyralid, Glyphodes pyloalis. J Insect Sci 156:1–13
Knoepp JD, Vose JM (2007) Regulation of nitrogen mineralization and nitrification Southern Appalachian ecosystems: separating the relative importance of biotic vs. abiotic controls. Pedobiologia 51:89–97
Kolar CS, Lodge DM (2001) Progress in invasion biology: predicting invaders. Trends Ecol Evol 16:199–204
Lenssen JPM, De Kroon H (2004) Abiotic constraints at the upper boundaries of two Rumex species on a freshwater flooding gradient. J Ecol 93:138–147
Leps J, Smilauer P (2003) Multivariate analysis of ecological data using CANOCO. Cambridge University Press, New York, pp 13–15, 50–51, 70–217
Marden M, Rowan D, Phillips C (2005) Stabilising characteristics of New Zealand indigenous riparian colonising plants. Plant Soil 278:95–105
Merritt DM, Wohl EE (2006) Plant dispersal along rivers fragmented by dams. River Res Appl 22(1):1–26
Michael K, Stéphane D, Francescode B, Jan J, Robin JP, Barbara S, Wilfried T, Sandra L (2012) Assessing species and community functional responses to environmental gradients: which multivariate methods? J Veg Sci 23:805–821
Miranda LE, Hodges KB (2000) Role of aquatic vegetation coverage on hypoxia and sunfish abundance in bays of a eutrophic reservoir. Hydrobiologia 427:51–57
New T, Xie ZQ (2008) Impacts of large dams on riparian vegetation: applying global experience to the case of China’s Three Gorges Dam. Biodivers Conservn 17:3149–3163
Nilsson C, Berggren K (2000) Alterations of riparian ecosystems caused by river regulation. BioScience 50(9):783–792
O’Connor TG (2001) Effect of small catchment dams on downstream vegetation of a seasonal river in semi-arid African savanna. J Appl Ecol 38:1314–1325
Peña-Fronteras JT, Villalobos MC, Baltazar AM, Merca FE, Ismail AM, Johnson DE (2009) Adaptation to flooding in upland and lowland ecotypes of Cyperus rotundus, a troublesome sedge weed of rice: tuber morphology and carbohydrate metabolism. Ann Bot 103:295–302
Peng X, Xie QL, Chen SB, Huang XL, Qiu JB, Zhong W, Guan WC (2011) The community distribution pattern of intertidal macrozoobenthos and the responses to human activities in Yueqing Bay. Acta Ecol Sin 31:954–963
Qiu S, McComb AJ (1996) Drying-induced stimulation of ammonium release and nitrification in reflooded lake sediment. Mar Freshwater Res 47:531–536
Shiqaki F, Sharpley A, Prochnow LI (2007) Rainfall intensity and phosphorus source effects on phosphorus transport in surface runoff from soil trays. Sci Total Environ 373:334–343
Sun L, Chen S, Chao L, Sun T (2007) Effects of flooding on changes in Eh, pH and speciation of cadmium and lead in contaminated soil. Bull Environ Contam Toxicol 79:514–518
Valenzuela-Encinas C, Neria-Gonzalez I, Alcantara-Hernandez RJ, Estrada-Alvarado I, Dendooven L, Marsch R (2009) Changes in the bacterial populations of the highly alkaline saline soil of the former lake Texcoco (Mexico) following flooding. Extremophiles 13:609–621
Van Eck WHJM, Van de Steeg HM, Blom CWPM, De Kroon H (2004) Is tolerance to summer flooding correlated with distribution patterns in river floodplains? A comparative study of 20 terrestrial grassland species. Oikos 107:393–405
Wang YC, Lei B, Zhang S (2012) Differences in vegetation and soil characteristics at different water-level altitudes in the drawdown areas of Three Gorges Reservoir area. J Lake Sci (in Chinese) 24:206–212
Wantzen KM, Rothhaupt KO, Cantonati M (2008) Ecological effects of water-level fluctuations in lakes: an urgent issue. Hydrobiologia 613:1–4
Willison JHM, Li RX, Yuan XZ (2013) Conservation and ecofriendly utilization of wetlands associated with the Three Gorges Reservoir. Environ Sci Pollut Res. doi:10.1007/s11356-012-1438-3
Ye C, Cheng XL, Zhang YL, Wang ZX, Zhang QF (2012) Soil nitrogen dynamics following short-term revegetation in the water level fluctuation zone of the Three Gorges Reservoir, China. Ecol Eng 38:37–44
Ye C, Li SY, Zhang YL, Tong XZ, Zhang QF (2013a) Assessing heavy metal pollution in the water level fluctuation zone of China’s Three Gorges Reservoir using geochemical and soil microbial approaches. Environ Monit Assess 185(1):231–240
Ye C, Zhang KR, Deng Q, Zhang QF (2013b) Plant communities in relation to `flooding and soil characteristics in the water level fluctuation zone of the Three Gorges Reservoir, China. Environ Sci Pollut Res 20(3):1794–1802
Yuan XZ, Zhang YW, Liu H, Xiong S, Li B, Deng W (2013) The littoral zone in the Three Gorges Reservoir, China: challenges and opportunities. Environ Sci Pollut Res. doi:10.1007/s11356-012-1404-0
Zhang JT (2011) Quantitative ecology. Science Press, Beijing, pp p15–p30
Acknowledgements
This study is supported by the National Natural Science Foundation of China (grant no. 50979061), Major Science and Technology Program for Water Pollution Control and Treatment (grant no. 2013ZX07104-004) and the Key Technologies R&D Program (grant no. 2012BAC06B04). We also thank Dr. Qingfeng Wang, Xiaojie Pan, Xiaojuan Chen and Xi Zou for the comments on early draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Zhihong Xu
Rights and permissions
About this article
Cite this article
Zhang, Z., Wan, C., Zheng, Z. et al. Plant community characteristics and their responses to environmental factors in the water level fluctuation zone of the three gorges reservoir in China. Environ Sci Pollut Res 20, 7080–7091 (2013). https://doi.org/10.1007/s11356-013-1702-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-013-1702-1