Abstract
Oak wilt disease and deer browsing have depleted the canopy and forest floor of secondary Quercus serrata and Quercus variabilis forests in Japan. Small-scale clear-cuttings enhance sprouting, and are, therefore, recommended for restoration of the damaged forests. However, most damaged forests are composed of mature stands, and it is not clear whether they would sprout successfully when cut. Moreover, little is known about the regeneration of forests damaged by both oak wilt disease and deer browsing. To determine the appropriate treatment for restoration of damaged Q. serrata and Q. variabilis forests, we established a study site with three different treatments (10 × 30 m each) in Kyoto City: clear-cut and fenced, removed dead oak trees and fenced, and removed dead oak trees and unfenced. After 1-year treatment, the number of species and individual saplings tended to increase at the lower slope of each treatment site with dominance of Ilex pedunculosa. The largest species and individual numbers were observed at the clear-cut fenced site, where pioneer species such as Mallotus japonicus were found. A few seedlings of Q. serrata were found at each treatment site, and no Q. variabilis was recorded. The emergence of Q. serrata seedlings was explained by their location at the slope and not by the treatment. Moreover, many sprouts of Q. serrata and Q. variabilis were observed at the clear-cut fenced site. Although further monitoring is needed, clear-cutting and fencing appear to be an effective method for the rehabilitation of depleted forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The secondary Quercus serrata Murray and Quercus variabilis Blume forests in Japan are now under threat with the depletion of the canopy and forest floor caused by oak wilt disease and deer browsing. Japanese oak wilt disease has been increasing since the 1990s (Kuroda et al. 2012) and damages due to this disease have been reported from 32 of the 47 prefectures in Japan (Ministry of Agriculture Forestry and Fisheries of Japan 2018). The damaged secondary forests are distributed close to residential areas, and they were used for firewood and charcoal production (Kuroda and Yamada 1996; Nagashima 2017). However, after progress in energy production took place, those forests became abandoned, leading them to grow larger and older (e.g., average trees with 30–40 cm of diameter at breast height). It is known that the ambrosia beetle, Platypus quercivorus, which carries the pathogenic fungus Raffaelea quercivora, the direct cause of oak wilt disease, successfully propagates in large trunks (Kobayashi and Ueda 2005; Kinuura and Kobayashi 2006). Thus, the abovementioned forests became an ideal place for the ambrosia beetle to propagate, which resulted in a mass mortality of large trees in the canopy layer.
The negative impact of Japanese sika deer, Cervus nippon, also has been increasingly observed for the last 2 decades, as its distribution expanded and population increased (Takatsuki 2009). Deer grazing and browsing decrease shrub and herb layer coverage (Fujiki et al. 2010), which prevents regeneration for diverse desirable species, including oak trees (Ward et al. 2018). In areas where deer occur, shade-tolerant and unpalatable species became predominant (Takatsuki 2009; Itô 2016). Moreover, soil erosion is also increasingly observed (Furusawa et al. 2003; Chu et al. 2010; Osawa et al. 2015). This might reduce the capacity of forests to conserve soil and water (Hashimoto and Fujiki 2014). Those changes in the forests might decline the forest resilience, the ability to recover to or maintain Quercus forests.
In fact, several studies implied the low resilience of Quercus forests damaged by oak wilt disease or the forest damaged both by oak wilt disease and deer browsing. Gaps caused by the mortality of oak trees can improve the light condition incidence on the forest floor, and might promote regeneration of tree species (Suzuki 2013; Itô 2016). However, few studies reported on the regeneration possibility of oak trees in the gaps created by the mass tree mortality. Instead, studies suggest the possibility of replacement of trees to shade-tolerant species in the canopy (Nakajima and Ishida 2014), and inhibition of regeneration by dense shrub layers covered by evergreen broadleaved species (Itô et al. 2009) or dense forest floor vegetation covered by bamboos (Itô et al. 2011). Studies on structural changes and regeneration possibilities for forests damaged both by oak wilt disease and deer browsing are scarce. Itô (2015) indicated that the regeneration species in forest gaps were limited to unpalatable species, such as Symplocos prunifolia Siebold & Zucc. and Triadica sebifera (L.) Small, and to the originally abundant shade-tolerant evergreen broadleaved species, such as Cleyera japonica Thunb. and Eurya japonica Thunb.
On the other hand, forest management might enhance the resilience of forests to both oak wilt disease and deer browsing. To reduce mass mortality by oak wilt disease, and to reestablish the oak forests, small-scale clear-cutting that enhances sprouting is recommended (Kuroda et al. 2009, 2012). However, most damaged stands are mature, and it is not clear whether they would sprout successfully after cut (Itô 2013). Establishment of deer-proof fences in gaps generated by oak wilt disease might also enhance regeneration. For example, establishing 4 m2 fenced quadrats, Obora et al. (2013) indicated that the coverage of shrub species, such as Rubus species and Miscanthus sinensis Andersson became dominant, whereas few tree species were observed. Nagashima (2017) also established 4 m2 fenced quadrats in a forest damaged both by oak wilt disease and deer browsing. They found that plots on the lower side of slopes with brighter light conditions showed possibility for Q. serrata regeneration. However, an appropriate treatment to enhance the resilience of and the recovery of Quercus forest remains to be developed.
To determine the appropriate treatment for Quercus forest restoration, we established three different treatment sites: clear-cut and fenced, removed standing dead oak trees and fenced, and removed standing dead oak trees and unfenced. Each of the treatment sites comprised 300 m2, and were located in Kyoto City. This study aims to understand the early-stage vegetation recovery after those three treatments, and to discuss the regeneration possibility of each treatment for rehabilitation of depleted Quercus forests.
Methods
Study site
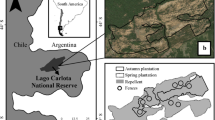
For the study site, we chose a west-facing slope (35°09′N, 135°78′E) on the east side of the “Sakura-no-mori” valley at the Takaragaike Park in Kyoto City (Fig. 1). The slope angle was 27°, with brown forest soil. Takaragaike Park is formed by a hilly urban forest (Takaragaike hill), with an area of 96.25 ha. The climate is temperate, with an annual mean temperature of 15.9 °C and annual precipitation of 1491 mm, averaged from 1981 to 2010 (Japan Meteorological Agency 2017). Takaragaike hill used to be utilized for firewood and charcoal production, and raking of fallen leaves were also conducted actively (Hirayama et al. 2011). At that time, a Pinus densiflora Sieb. Et Zucc. forest covered the entire hill (Morishita and Ando 2002). Since the late 1960s, though, the forest started to be abandoned because the infrastructure of gas distribution had been established and demands placed on forests for energy production diminished (Kyoto City 2011). In addition, pine wilt disease expanded in the 1980s and resulted in the mass mortality of P. densiflora. Afterwards, the forest changed to secondary deciduous forest, dominated by Q. serrata and Q. variabilis (Morishita and Ando 2002). Nonetheless, the oak wilt disease had rapidly expanded since 2004 when it was first reported in the northern of Kyoto City. Mass mortality broke out in Quercus forests, and at the Takaragaike hill and it was reported in 2010 (Yoshii and Kobayashi 2016). Additionally, Japanese sika deer also started invading the area since 2010, and in consequence, the forest floor vegetation rapidly diminished. The forest of the west-facing slope of “Sakura-no-mori” at the Takaragaike Park was not the exception, and in this area, the forest consists of Quercus trees which survived the oak wilt disease and by gaps generated by dead trees, a typical vegetation of Takaragaike hill. The Takaragaike Park is frequently used nowadays for recreation by the citizens, and thus a reestablishment of the Quercus forest is strongly needed. Furthermore, Rhododendron reticulatum D. Don ex G. Don is a dominant species in the shrub layer of Quercus forests. It is especially important for restoration methods, because of its appeal to the park visitors (showy flowers in spring), but also because it was heavily damaged by deer browsing.
Field survey
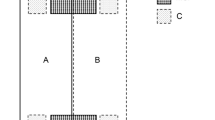
To determine the appropriate treatment for restoration of the damaged Quercus forests, we established three treatment sites (A–C) of 10 m × 30 m along with the study site slope (Fig. 1), where the site conditions (slope angle and soil condition) were similar. The light conditions of the three treatment sites were also similar (0.30–0.63 of relative photosynthetic photon flux density (rPPFD) measured in 2014, p = 0.545, Kruskal–Wallis test). For site A, 3 standing dead oak trees [one Q. variabilis and two Q. serrata; diameter at breast height and height (DBH): 12.0–20.8 cm] were removed and the area was fenced to prevent deer invasion. For site B, one standing dead Q. serrata tree (DBH: 36.0 cm, located at the boundary of site C) was removed but area was not fenced. For site C, clear-cut was conducted and was fenced. The standing dead trees were removed to understand its effect on vegetation recovery and to avoid risks of dead trees falling and harming citizens visiting the Takaragaike Park. The treatments were conducted in November 2015. Each treatment site was further subdivided into upper, middle and lower 10 m × 10 m subplot, based on its location at the slope. Prior to the treatment application, tree census (species name, DBH) was conducted for trees taller than 1.2 m in 2014 to understand the species composition of the sites. For sites A and B, tree census (species name, DBH) was also conducted for all the trees taller than 0.5 m in April 2016 to understand the species composition after treatment application. Nagashima (2017) reported a previous study on seedling establishment at the same area, where she censused and mapped trees in 2014 (also species names, DBH). To update the tree location map of Nagashima (2017), we checked the location of removed and new labeled trees. At site C, the location of the stumps was confirmed based on the tree numbers of the location map of Nagashima (2017).
Regeneration was assessed by three aspects: new recruited saplings at the shrub layer, seedling germination and sprouted stems (from the stumps in the clear-cut site C). Individual and species numbers of new recruited saplings that reached the shrub layer (taller than 0.5 m and lower than 2.0 m) were investigated in each subplot in October 2016. To monitor the species and numbers of individuals of current-year seedlings, and their survival from April to October 2016, we further established five 1 m × 1 m quadrats at the four corners and at the middle of each 10 m × 10 m subplot. Sprouts from the stump at site C were also monitored from April to October 2016, and the species, the number of sprouted stumps, the number of sprouted stems of each stump, diameter at ground height, and the height of the highest sprouted stem for each stump were observed. To assess the light condition in each subplot, we measured the photosynthetic photon flux density (PPFD) using a quantum sensor (LI-190R, Li-Cor Inc., Lincoln, Nebraska) at ground level at the center of each 1 m × 1 m quadrat in October 2016. To calculate the relative photosynthetic photon flux density (rPPFD), one sensor was placed in an open space of the “Sakurano-mori,” valley to measure the PPFD every 10 s, where data was automatically stored in a data logger (LI-1500G, Li-Cor Inc., Lincoln, Nebraska). Hemispherical canopy photographs were taken at the center of each subplot at the height of 50 cm, using a Nikon D3200 with a Sigma 10 mm F2.8 EXDC fisheye lens.
Data analysis
Vegetation after treatment application
Based on the tree census of April 2016, the ratio of accumulated basal area for each species of trees taller than 1.2 m was calculated per subplot to understand the dominant species. For the clear-cut site C, the ratio of accumulated basal area was also calculated using the tree census data before treatment, carried out in 2014. The updated location maps of the remaining trees on sites A and B, and the stumps of site C were established using the geographic information system (ArcGIS ver.10.4, ESRI, Redlands, California).
Regeneration situation
The number of individuals and the number of species of new recruited saplings per subplots were calculated based on the field data, and the tendency among treatments and the plot location (upper, middle, and low) were interpreted. The species composition was measured by calculating the ratio of number of individuals for each species.
For germinated seedlings, total number of individuals and species per subplots were calculated by accumulating the numbers of the five quadrats in each subplot. The differences among treatments and plot location were examined accordingly. The field data for October were used for the calculations. Based on the total number of individuals per species of each subplot, the Morisita similarity index (Morisita 1959) was calculated and used for cluster analysis to classify the subplots with the same species composition. The cluster analysis was applied using the average linkage method based on Euclidian distance. Each of the vegetation types derived from the cluster analysis was defined based on the ratio between the number of individuals per species to the total number of individuals on each subplot. A detrended correspondence analysis (DCA) was also implemented to interpret the relationship among the vegetation groups, light conditions and plot locations. The relationship between the derived axes and light condition (mean rPPFD and mean canopy openness) was examined by calculating the Pearson’s correlation coefficient. The relationship with the plot location was examined by calculating the Spearman’s rank correlation coefficient. The rPPFD was calculated by dividing the sampled PPFD (data taken from the 1 m × 1 m quadrat) with the control PPFD (data taken from the open space at the corresponding time). The mean rPPFD of each subplot was then calculated from the values of the five quadrats included in each subplot. The software CanopOn2 (Takenaka 2009) was used to calculate the canopy openness from the hemispherical canopy photographs, and the mean canopy openness was calculated from the values of the five quadrats of each subplot. Cluster analysis and DCA were performed using PC-ORD version 4.0 (McCune and Mefford 1999). According to the recommendation of PC-ORD (McCune and Mefford 1999), after-the-fact coefficient of determination between relative Euclidean distance in the unreduced species space and Euclidean distance in the ordination space was utilized as the contribution ratio of the derived axes of DCA.
To estimate whether stumps successfully sprouted, we measured (1) the ratio of sprouted stumps to the total stumps, (2) the mean number of sprouted stems per stumps, and (3) the mean maximum height of sprouted stems, per each species and by combining the data of the three subplots in October. The relationship between sprouted Q. serrata and Q. variabilis stumps and their DBH before cutting were also examined.
Results
Vegetation after treatment application
The dominant tree species of all the subplots in sites A and B after treatments were Q. serrata and Q. variabilis. For the shrub species, the dominant species were R. reticulatum, Evodiopanax innovans (Siebold & Zucc.) Nakai and Lyonia ovalifolia (Wall.) Drude (Fig. 2). Before clear-cut in site C, the upper and middle plots had the same composition, whereas the lower plot was dominated by Prunus grayana Maxim., followed by Q. serrata. Figure 3 shows the location of trees taller than 1.2 m in sites A and B, and the stumps which used to be taller than 1.2 m before clear-cutting in site C. Larger number of trees were found in site C, compared to sites A and B, especially for the shrub species in the upper to middle plots. Rhododendron reticulatum, L. ovalifolia, and E. japonica tended to occur in the upper to middle plots in every site, with stems usually taller than 1.2 m, compared to the lower plots.
Regeneration situation
New recruited saplings at the shrub layer
The number of new recruited saplings at the shrub layer was generally lower for the upper plots, increasing towards the lower plots for all treatment sites. This tendency was especially clear for the clear-cut site C, where 14, 55 and 140 (from upper to lower plot) new recruited saplings were recorded (Fig. 4a). For site B, however, the middle plot showed the largest number of saplings. Regarding treatments, the clear-cut site C tended to show the largest number of saplings followed by sites B and A. Compared to sapling numbers, species numbers also indicated the same tendency. Species number increased as the plot position lowered at the slope, and site C tended to have the largest species number among the plots located at same level on different treatments (Fig. 4a). The main species of new recruited saplings was Ilex pedunculosa Miq., which occurred in all the subplots, occupying 50–100% to the total sapling number (Fig. 4b). A large number of saplings of I. pedunculosa were originated from sprouts. Rhododendron reticulatum saplings were observed only at the upper plot of site B (one individual) and at the middle plot of site C (four individuals). Mallotus japonicus (L.f.) Müll.Arg. and Vaccinium oldhamii Miq. were observed in the middle and lower plots of site C, but not at other treatment sites.
Seedlings
Similar to the new recruited saplings, the number of seedlings also showed a tendency to increase from upper towards lower plots within treatments, except for site C, where the middle plot showed the largest number of seedlings (Fig. 5a). Comparing treatments, site C indicated the largest number of seedlings, whereas sites A and B did not show clear differences. The number of species also increased from upper towards lower plots in sites B and C. For site A, however, the upper plot showed a larger number compared to the middle plot. Similar to seedling number, site C tended to have the largest species number among the different treatment sites.
Cluster analysis identified three vegetation groups (Fig. 6). Group I consisted of the upper and lower plots of site A, and middle and lower plots of site B. Although there were some differences between the plots of site A and B, such as the proportion of L. ovalifolia, its most common species were Q. serrata, E. innovans, and Pourthiaea villosa E.Pritz (Fig. 5b). The mean canopy openness of each plot ranged from 15.14 to 30.24%, and the rPPFD ranged from 0.07 to 0.21 (Table 1). Group II was composed by the upper plot of site B and all the three plots of site C. Its dominant species was M. japonicus. The mean canopy openness of each plot ranged from 23.82 to 50.74% and the rPPFD ranged from 0.19 to 0.35 (Table 1). Group III consisted uniquely of the middle plot of site A, and it was dominated by I. pedunculosa. The mean canopy openness was 16.90% and the rPPFD was 0.08 (Table 1). Quercus serrata seedlings were mostly observed at the middle to lower plots, regardless of treatments, but one seedling was found in the upper plot of site A (which did not have any in the middle; Table 2). Contrastingly, only one seedling of Q. variabilis was found at the lower plot of site B.
Vegetation groups were successfully produced by DCA ordination (Fig. 7). Species scores increased in the order of M. japonicus, E. innovans and L. ovalifolia in axis 1 (contribution ratio: 28.0%); thus, vegetation groups were ordered as II and I along axis 1. Ilex pedunculosa showed lower score in axis 2 (contribution ratio: 35.7%, the cumulative contribution ratio of the two axes: 63.7%), which separated group III from the other groups. Axis 1 had a significant negative correlation with canopy openness (− 0.80, p < 0.01) and rPPFD (− 0.82, p < 0.01). However, no significant relationships could be found with axis 2, and the factor dividing group III was unclear. Quercus serrata and Q. variabilis showed relatively high scores for axis 1, which suggests that the seedlings germinated in shaded place. It also showed high scores for axis 2, but the reason for this observation was not clear.
Result of DCA ordination. A-U the upper plot of site A, A-Mid the middle plot of site A, A-L the lower plot of site A, B-U the upper plot of site B, B-Mid the middle plot of site B, B-L the lower plot of site B, C-U the upper plot of site C, C-Mid the middle plot of site C, C-L the lower plot of site C. *Only the main species are sited in the figure
Regeneration by sprouting in site C
Overall, three out of the seven stumps of Q. serrata in site C subplots sprouted (42.9% of stem sprouting; Table 3). Mean number of sprouted stems per stumps was 20 (Table 3). The data of DBH before cutting was available for five of the seven stumps of Q. serrata, including for two of the sprouted stumps. For the remaining two, tree number was lost and identification was then unavailable. The DBH of sprouted stumps before cutting was of ca. 25 cm and that of non-sprouted stumps ranged from 12 to 32 cm (Table 4). Sixty-eight stems sprouted from the single stump of Q. variabilis, which DBH was 27 cm before cutting. Both sprouts of Q. serrata and Q. variabilis showed a rapid growth, reaching the average maximum height of 117.7 and 143.9 cm, respectively (Table 3). The main shrub species, R. reticulatum and L. ovalifolia, showed more than 80% in sprouting rate, together with the evergreen shrubs E. japonica and C. japonica (Table 3). The former two deciduous shrubs tended to reach more than 50 cm in average maximum height, but the latter two evergreen shrubs only reached about 20 cm. Ilex pedunculosa which was the dominant species at the shrub layer, showed a lower ratio of sprouted stumps compared to the two evergreen species (66.7%), but a rapid growth of up to 60 cm (Table 3).
Discussion
The number of recruited saplings to the shrub layer and the number of seedlings were larger at site C, the clear-cut site. In addition, the number between sites A and site B did not show large differences, especially for the seedling assessment. The same tendency was observed for the species number of saplings and seedlings. This indicates that the treatment of clear-cut and fencing contributed to an increase in the numbers of individuals and species. Contrastingly, deer-proof fencing did not influence the numbers of individuals and species at sites in which dead oak trees were removed. Recently, Suzuki (2013) and Suzuki and Ito (2014) mentioned that ground vegetation was recovered in gaps on fenced and unfenced plots, but on plots with or without deer fencing changes were not observed. The improved light condition created by clear-cutting, therefore, was recognized as the main driver of the increase in the number of individuals and species of saplings and seedlings. The larger number of trees found before treatment at site C, compared to sites A and B (Fig. 3), could be a factor that contributed to the larger number of saplings and seedlings observed in site C. However, its contribution seems to be low at this early-stage of vegetation recovery, because the main species were different; E. japonica, R. reticulatum, and L. ovalifolia for trees taller than 1.2 m before treatment, and I. pedunculosa for saplings and M. japonicas for seedlings.
The light condition may also determine the vegetation types for seedlings. The light condition was more intense in group II, followed by I or III according to the average canopy openness and rPPFD. Those two estimates also showed significant negative correlation with the axis 1 of the DCA. Group II, defined as having the brightest light condition, was dominated by M. japonicas, and included three subplots of site C. One reason for this might be that germination of the seed bank takes place in areas where light condition is improved by clear-cutting, as reported for studies using clear-cut sites and gaps of Q. serrata forest (Matsuura et al. 2004; Suzuki 2013). The main species in groups I and III were observed within or at the surrounding subplots in the upper layer (Fig. 3). Thus, studied species were considered to germinate after seed dispersal within the study site. Evodiopanax innovans, one of the main species observed in group I, is known to germinate in less bright conditions than those of clear-cut sites (Yamase 2000), which supports our result. The canopy openness and rPPFD of subplots assigned to groups I and III were similar. Relationships with axis two were not found; therefore, the factor dividing groups I and III was not certain.
Quercus serrata seedlings were observed in all the treatment sites and they commonly germinated in lower plots. Given that Quercus species in oak forests can grow when there is stand-level dieback (Saito and Shibata 2012), we expected that the seedlings would germinate more in bright clear-cut sites. Quercus serrata showed relatively high scores on the axis 1 of the DCA, which indicates that it germinated at more shaded plots. However, several other studies also indicate that Q. serrata survive and grow at sites with sufficient light (Takahara 1986; Nishimura et al. 1990). Therefore, further monitoring is required to check whether the germinated seedlings will survive and grow further successfully.
Regardless of treatment, we observed a lower number of individuals and species for saplings and seedlings at the upper plots compared to the lower plots. This might be due to soil movement in the slope. Murashita et al. (unpublished) indicated that the average transport rates of fine earth at the three treatment sites were 0.621 g m−1 mm−1. This value was larger than those observed in other deciduous forests in Japan (0.051 g m−1 mm−1 at sites with 35°–40° of slope angle, Miura et al. 2002) and that observed at a deciduous forest after a year of clear-cutting (0.47 g m−1 mm−1 at site with average slope angle of 38.8°, Nakamori et al. 2012). Thus, soil erosion was observed at the upper slopes in each treatment site. Unstable soil condition might prevent the establishment of seedlings by loss of dispersed seeds, buried seeds and germinated seedlings (Yamase et al. 2009; Obora et al. 2013). It may also expose roots out of the soil (Chu et al. 2010), and thus reduce sapling recovery. On the contrary, at the lower plots, soil is deposited together with the seeds dispersed from the upper part of the slope (Yamase et al. 2009). This could increase the number of seedlings and saplings. However, it has been suggested that the vegetation recovery is delayed, when prone to deer browsing. For example, Yamase et al. (2014) showed that areas occupied by deer over 10 years had worse vegetation recovery than areas with fencing, even in lower slopes where soil is deposited more often. This indicates that the effect of deer-proof fencing should be stronger in a longer term. Therefore, the difference in vegetation recovery between sites A and site B might become clear by long-term monitoring.
Forty-two percent of the Q. serrata stump was sprouting and its DBH before cutting was around 25 cm. There were non-significant differences with the non-sprouted stumps. Only one stump of Q. variabilis sprouted, and its DBH was 27 cm before cutting. The age of Q. serrata trees with 20 cm diameter was around 65 years old, and that of Q. variabilis ranged from 57 to 71 years old at the Takaragaike hill (Kakimoto, unpublished). The average number of sprouted stems was 20 for Q. serrata and 68 for Q. variabilis. The sprouted stems of both species grew rapidly and reached more than 100 cm in height. Although it is difficult to discuss the relationship between sprouting possibility and DBH or stem age because the number of stumps is small, after a year of observation at this study site, it seems there is some recovery possibility for Q. serrata and Q. variabilis by sprouting, even for stands of around 60 years of age. However, Matsuura et al. (2002) reported that the ratio of stumps with survived sprouted stems decreased to 5–58% after 2–5 years past clear-cutting. Ito (2013) also suggested that the 11 sprouted stumps decreased to six 1 year after cutting, decreasing further to two stumps after 2 years of cutting. In the study of Itô (2013), the number of sprouted stems per stumps ranged from 10 to 50, but the stumps died even when sprouted stems reached 100 cm of height. Future monitoring is, therefore, necessary to predict the recovery probability by sprouting of Quercus species. Moreover, the main shrub species, R. reticulatum, L. ovalifolia, E. japonica and C. japonica showed relatively high stump sprouting ratios (80%). Rhododendron reticulatum is considered to sprout regardless of the stump age, thus shows stable recovery (Yamase 1999). Shade-tolerant and deer-unpalatable species, such as E. japonica and C. japonica (Takatsuki 1989), are expected to increase, but our study suggests that the sprouts of R. reticulatum should also grow faster and, therefore, the increase in E. japonica and C. japonica might not be so damaging. However, L. ovalifolia and I. pedunculosa might influence the recovery of Quercus species and R. reticulatum by becoming dominant in the future, because those species are known to increase their number of sprouted stems per stump, growing rapidly as the light conditions become brighter (Yamase 1999).
Ilex pedunculosa was also the dominant species in the new recruited saplings at the shrub layer and has a possibility to prevent Quercus species recovery by shading the lights at the shrub layer. Ilex pedunculosa is known as a species that regenerates in the gaps of evergreen broadleaved forests (Yamamoto 1994). The light condition improved by clear-cutting seemed to be a good environment for I. pedunculosa to regenerate. Moreover, I. pedunculosa is regarded as a deer-unpalatable species (Takatsuki 1989; Fujiki 2017) although deer can forage the species (Osaki 2006). This indicates the possibility of forest damaged both by oak wilt disease and deer browsing converting to forest dominated by I. pedunculosa at the sub-canopy and shrub layer, even at sites where clear-cutting was implemented. Careful monitoring is required to clarify the result of growth competition between I. pedunculosa and sprouts of Quercus species and R. reticulatum.
Conclusion
Clear-cut and deer-proof fencing contributed to the increase in the species number and the number of individuals of seedlings and saplings at the study site. Brighter environments also contributed in enhancing the germination of the seed bank, and thus pioneer species such as M. japonicus could become dominant at seedling stage. Although seedlings of Quercus species were not found profusely, sprouts of Q. serrata and Q. variabilis were observed and showed rapid growth. Rhododendron reticulatum, another target species of vegetation recovery, was also recorded sprouting. The dominance of I. pedunculosa at the shrub layer and active sprouting, and the fast growth of L. ovalifolia have both a possibility of preventing the regeneration of Quercus species and R. reticulatum, and converting the forest dominated by I. pedunculosa, the deer-unpalatable species. Further monitoring is required to understand the effect of clear-cutting and fencing on the resilience of Quercus forests until the result of competition between I. pedunculosa and Quercus species becomes clear. However, after a year of observation, clear-cutting and fencing also appear to be effective for the rehabilitation of depleted Quercus forests. Furthermore, comparing sites A and B, the effect of deer-proof fencing on the vegetation recovery of Quercus forests has not yet become apparent. It might become clear after long-term monitoring according to Yamase et al. (2014), which might also give insight into interpreting the appropriate length of observation and understanding the ecosystem resilience.
References
Chu L, Ishikawa Y, Shiraki K, Wakahara T, Uchiyama Y (2010) Relationship between forest floor cover percentage and soil erosion rate on the forest floor with an impoverished understory grazed by deer (Cervus Nippon) at Doudaira, Tanzawa mountains (in Japanese with English abstract). J Jpn For Soc 92:261–268
Fujiki D (2017) List of food plants and unpalatable plants of sika deer (Cervus nippon) in Hyogo Prefecture (in Japanese). Wildl Monogr Hyogo 9:118–126
Fujiki D, Kishimoto Y, Sakata H (2010) Assessing decline in physical structure of deciduous hardwood forest stands under sika deer grazing using shrub-layer vegetation cover. J For Res 15:140–144
Furusawa H, Miyanishi H, Kaneko S, Hino T (2003) Movement of soil and litter on the floor of a temperate mixed forest with an impoverished understory grazed by deer (Cervus nippon centralis Temminck) (in Japanese with English abstract). J Jpn For Soc 85:318–325
Hashimoto Y, Fujiki D (2014) List of food plants and unpalatable plants of sika deer (Cervus nippon) in Japan (in Japanese). Hum Nat 25:133–160
Hirayama K, Yamada K, Nishimura T, Kawamura S, Takahara H (2011) Changes in species composition and diversity with respect to the successional stage of urban forests in Kyoto City, western Japan (in Japanese with English abstract). J Jpn For Soc 93:21–28
Itô H (2013) A sprouting experiment of a Quercus serrata stand aged 64 years since last coppicing (in Japanese with English abstract). Bull FFPRI 427:105–109
Itô H (2015) Effects of Sika Deer (Cervus Nippon) on the dynamics of a broadleaved secondary forest after mass mortality of oak trees (in Japanese with English abstract). J Jpn For Soc 97:304–308
Itô H (2016) Changes in understory species occurrence of a secondary broadleaved forest after mass mortality of oak trees under deer foraging pressure. Peer J 4:e2816. https://doi.org/10.7717/PeerJ.2816
Itô H, Igarashi T, Kinuura H (2009) Changes in stand structure after the mass mortality of oak trees in Keihoku area, Kyoto city, Japan (in Japanese with English abstract). J Jpn For Soc 91:15–20
Itô H, Kinuura H, Oku H (2011) Stand structure of a broadleaved forest with Sasa-type floor after mass mortality by oak wilt disease (in Japanese with English abstract). J Jpn For Soc 93:84–87
Japan Meteorological Agency (2017) Search for the past meteorological data. http://www.data.jma.go.jp/obd/stas/etrn/index.php. Accessed 17 Jan 2018
Kinuura H, Kobayashi M (2006) Death of Quercus crispula by inoculation with adult Platypus quercivorus (Coleoptera: Platypodidae). Appl Entomol Zool 41:123–128
Kobayashi M, Ueda A (2005) Wilt disease of Fagaceae trees caused by Platypus quercivorus (Murayama) (Coleoptera: Platypodidae) and the associated fungus: aim is to clarify the damage factor (in Japanese with English abstract). J Jpn For Soc 87:435–450
Kuroda K, Yamada T (1996) Discoloration of sapwood and blockage of xylem sap ascent in trunks of wilting Quercus spp. following attack by Platypus quercivorus (in Japanese with English abstract). J Jpn For Soc 78:84–88
Kuroda K, Itô H, Osumi K, Oku H, Kinuura H, Takahara Y, Matsumoto K (2009) How to manage “Satoyama” forest: instruction for the volunteers and local governments (in Japanese). Forest and Forest Products Research Institute, Kyoto
Kuroda K, Osumi K, Oku H (2012) Reestablishing the health of secondary forests “Satoyama” endangered by Japanese oak wilt: a preliminary report. J Agric Ext Rural Dev 4(9):192–198
Kyoto City (2011) Guideline for conserving and restoring forest landscapes in Kyoto city, Kyoto City, Kyoto (in Japanese)
Matsuura M, Kobayashi T, Arita Y (2002) Limiting factors in the coppicing of the over-grown secondary forest of Quercus serrata Thunb (in Japanese with English abstract). J Jpn Soc Reveg Tech 28:115–120
Matsuura M, Azuma K, Kobayashi T (2004) Plant species composition of the herbaceous layer in relation to coppicing and bush cutting in secondary forest of Quercus serrata Thunb. In Sayama hill, central Japan (in Japanese with English abstract). J Jpn Soc Reveg Tech 30:121–126
McCune B, Mefford MJ (1999) PC-ORD: multivariate analysis of ecological data, ver 4. MJM Software Design, Gleneden Beach
Ministry of Agriculture Forestry and Fisheries of Japan (2018) Oak wilt disease damage by prefectures. http://www.rinya.maff.go.jp/j/press/hogo/attach/pdf/170927-1.pdf. Accessed 17 Jan 2018
Miura S, Hirai K, Yamada T (2002) Transport rates of surface materials on steep forested slopes induced by raindrop splash erosion. J For Res 7:201–211
Morishita M, Ando M (2002) Change in cover types of urban forests damaged by pine wilt disease in the northern part of Kyoto City (in Japanese with English abstract). For Res 74:35–45
Morisita M (1959) Measuring of interspecific association and similarity between communities. Bulletin of faculty of science, ser E3. Kyushu University, Fukuoka, pp 65–80
Nagashima K (2017) Seedling establishment and its factors in forests damaged by oak-wilt disease and deer browsing (in Japanese with English abstract). J Jpn Soc Reveg Tech 43(1):80–85
Nakajima H, Ishida M (2014) Decline of Quercus crispula in abandoned coppice forests caused by secondary succession and Japanese oak wilt disease: stand dynamics over twenty years. For Ecol Manag 334:18–27
Nakamori Y, Takii T, Miura S (2012) Variation in fine earth, sediment, and litter movement with different forest management practices on a steep slope in a Chamaecyparis obtusa plantation (in Japanese with English abstract). J Jpn For Soc 94:120–126
Nishimura N, Yamamoto S, Chiba K (1990) Structure and dynamics of a Quercus serrata stand near urban areas (I): stand structure and the population characteristics of Quercus serrata (in Japanese with English summary). J Jpn Soc Reveg Tech 16:8–17
Obora T, Watanabe H, Yokoi S (2013) The effect of sika deer browsing damage on regeneration in mass mortality area of Japanese oak trees (in Japanese). J Jpn Soc Reveg Tech 39(2):260–263
Osaki S (2006) Damages of forest vegetation by Sika deer in natural forests at Tonomine, Okawati-town. Hyogo Prefecture. Appl For Sci 15:41–46
Osawa T, Chiaki Igehara G, Ito C, Michimata S, Sugiyama D (2015) A method for the early detection of vegetation degradation caused by Sika deer. Jpn J Conserv Ecol 20:167–179
Saito S, Shibata M (2012) The forest structure and tree death rate of forest stands damaged by Japanese oak wilt in Yamagata prefecture (in Japanese with English abstract). J Jpn For Soc 94:223–228
Suzuki M (2013) Succession of abandoned coppice woodlands weakens tolerance of ground-layer vegetation to ungulate herbivory: a test involving a field experiment. For Ecol Manag 289:318–324
Suzuki M, Ito E (2014) Combined effects of gap creation and deer exclusion on restoration of belowground systems of secondary woodlands: a filed experiment in warm-temperate monsoon Asia. For Ecol Manag 329:227–236
Takahara H (1986) Effects of shading on the growth of some Quercus seedlings (in Japanese with English abstract). J Jpn For Soc 68:289–292
Takatsuki S (1989) Effects of deer on plants and plant communities. Jpn J Ecol 39:67–80
Takatsuki S (2009) Effects of sika deer on vegetation in Japan: a review. Biol Conserv 142:1922–1929
Takenaka, A. (2009) CanopOn 2. http://takenaka-akio.org/etc/canopon2/. Accessed 28 Oct 2016
Ward JS, Williams SC, Linske MA (2018) Influence of invasive shrubs and deer browsing on regeneration in temperate deciduous forests. Can J For Res 48:58–67
Yamamoto S (1994) Gap regeneration in primary evergreen broadleaved forests with or without a major canopy tree, Distylium racemosum, southwestern Japan: a comparative analysis. Ecol Res 9:295–302
Yamase K (1999) Stump sprouting of main woody species in secondary forest of pine mass-dieback under different thinning intensity (in Japanese with English summary). J JILA 63:465–468
Yamase K (2000) The growth environment and the seed dispersal form of the recruitment on different managements (in Japanese). J Jpn Soc Reveg Tech 25:547–550
Yamase K, Sekioka H, Tochimoto D, Todo C (2009) Seedbank potential in the movement soil (in Japanese). J Jpn Soc Reveg Tech 35:130–133
Yamase K, Todo C, Shibahara T (2014) Evaluation of topsoil seedbank potential during 10 years after felling in a site heavily used by sika deer (Cervus nippon Temminck) (in Japanese). J Jpn Soc Reveg Tech 40:265–268
Yoshii Y, Kobayashi M (2016) Characteristics of initial damage sites of Japanese oak wilt disease vectored by Platypus quercivorus (Murayama). Appl For Sci 25(1):7–14
Acknowledgements
This study was conducted as a part of JSPS Grant-in-Aid for Scientific Research C (no. 26450202).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagashima, K., Shimomura, T. & Tanaka, K. Early-stage vegetation recovery in forests damaged by oak wilt disease and deer browsing: effects of deer-proof fencing and clear-cutting. Landscape Ecol Eng 15, 155–166 (2019). https://doi.org/10.1007/s11355-019-00372-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11355-019-00372-z