Abstract
Background
The purpose of this study was to investigate the effect of semi-rapid maxillary expansion (SRME) orthodontic treatment on biomarkers and respiratory parameters in children with obstructive sleep apnea syndrome (OSAS) and maxillary transverse deficiency.
Methods
Thirty children with OSAS were included in this study. Fifteen children were enrolled as control, and 15 children were subjected to SRME orthodontic treatment method for 5 months. Beside respiratory parameters, pharyngeal area, dental arch, and postero-anterior widths and the levels of OSAS biomarkers in serum and urine were measured.
Results
Pharyngeal airway space, dental arch, and postero-anterior widths were increased after SRME treatment. Sleep tests showed a decrease in the apnea-hypopnea index (AHI) after 5-month control/treatment duration. Serum kallikrein (KLK)1 levels decreased significantly in the treatment group. There was a significant increase in serum orosomucoid (ORM)2 levels and a decrease in urine perlecan levels in the control group after a 5-month follow-up. A significant negative correlation between serum ORM2, perlecan, gelsolin, and KLK1 levels and intercanin width, as well as between serum ORM2 and KLK1 levels and intermolar width, was observed.
Conclusions
SRME treatment can be considered as a useful approach in children with OSAS. A further investigation of OSAS-related biomarkers and their relationship with sleep and orthodontic parameters is needed for providing easier and reliable modulatory strategies in the treatment of OSAS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnea syndrome (OSAS) is a common disease in children, characterized by a habitual snoring associated with prolonged, repeated events of partial and/or complete upper airway obstruction and hypercapnia and hypoxemia during sleep [1]. The symptoms of pediatric OSAS such as snoring, pauses or absence of breathing or mouth breathing and night sweats could cause morning fatigue, daytime sleepiness, growth inadequacy, hyperactivity, and low school performance [2]. Untreated sleep disorders can also be the reason for cardiovascular, metabolic, and neurocognitive disorders [3]. Polysomnography (PSG) has been used as a reference standard for the diagnosis of OSAS as it measures the apnea-hypopnea index (AHI) [4]. Due to the disadvantages of PSG such as being time-consuming, expensive, and having difficult accessibility, portable devices can also be used as an alternative diagnostic test to confirm the diagnosis of OSAS [5].
Orthodontic treatment is carried out to correct dental and skeletal discrepancies, and this treatment may be effective on nasorespiratory problems of the growing children with OSAS [6]. Rapid maxillary expansion (RME) is an effective and valid orthodontic treatment approach for managing OSAS in children with maxillary constriction [6, 7]. RME can be used as an alternative orthopedic treatment method to expand the maxilla until providing appropriate and stable maxillary width increase, maxillary and mandibular dental arch coordination, nasal respiratory function, opening the mid-palatal suture, preventing posterior uni- or bilateral cross-bite, and dental crowding among children with OSAS [6, 8,9,10,11].
In order to find out cheaper and effective alternative methods for the diagnosis of OSAS, studies have focused on hypoxia and inflammation-related molecules linking OSAS with metabolic alterations [12]. OSAS is predictably associated with altered protein expression patterns such as orosomucoids (ORMs) which have been implicated in the modulation of a variety of immune responses, endothelial function, and permeability [13, 14]. The fatty acid binding proteins (FABPs) are also potential indicators of OSAS and metabolic alterations [12]. Other proteins which show different protein expression profiles in the urinary samples of children with OSAS are gelsolin, perlecan, and kallikrein (KLK). Gelsolin is a 93-kDa cytosolic protein and it is activated by Ca2+ [15, 16]. Perlecan is a heparan sulphate proteoglycan that belongs to a family of glycosaminoglycans [1]. KLK1 reduction in urine samples emerged as a potentially valuable tool in the prediction of OSAS [1]. Tissue hypoxia due to repeated sleep apneas leads to increased serum levels of uric acid in patients with OSAS [17].
In this study, we aimed to investigate the relationship between the effect of 5-month semi-rapid maxillary expansion (SRME) orthodontic treatment and the magnitude of the changes in serum and urine biomarkers and respiratory parameters in children with OSAS.
Materials and methods
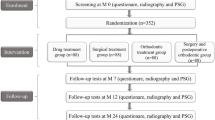
Thirty white children (16 male/14 female) attending the Department of Orthodontics of Ankara University School of Dentistry were enrolled in the study. We enrolled children who have clinical signs of maxillary transverse deficiency, malocclusion (high, narrow palate associated with deep bite, retrusive bite, or cross-bite), and signs and symptoms of OSAS (AHI > 1) including habitual snoring and oral respiration as identified by a parental questionnaire. This population was divided into two groups randomly as control (mean age, 11.46 ± 2.06) and SRME-treated group (mean age, 12.27 ± 1.93). This study was performed with the approval of Ankara University Health Sciences Institutional Review board (2012-37-4), and standard informed consent was obtained from the parents of each child.
Ear, nose, and throat assessment
At the beginning of the treatment, children underwent an ear, nose, and throat examination and every volunteer had an ear nose throat consultation before orthodontic assessment. According to the results of the consultation, patients who require adenotonsillectomy due to adenoid hypertrophy or tonsillar hypertrophy have been excluded from the study. Therefore, the etiological factors in OSAS except for mandibular retrognathia and maxillary construction were eliminated by ear, nose, and throat consultation in this study.
Semi-rapid maxillary expansion
A detailed personal and family history was obtained for all participants and a general clinical examination was performed. All investigations were carried out before orthodontic treatment (T0), and at the end of the treatment period (T1). Children with OSAS, as the control group, were followed up for 5.38 ± 1.36 months. SRME proceeded 5.01 ± 0.96 months in the treatment group. A modified McNamara RME device was cemented with an expansion screw connecting right and left maxillary dental acrylic segments to each other with a Hyrax type maxi screw (Forestadent) (Fig. 1a–c). The screw was turned twice a day for the first 7 days, then once a day until the palatal cusp of the upper molar came into contact with the buccal cusp of the lower molar [18]. After this initial treatment, when the maxillary arch was sufficiently over-expanded, the screw was fixed with a steel ligature wire.
The measurements of the study
Pharyngeal linear widths and pharyngeal area measurements were done by using Dolphin computer software and a digital planimeter device (Ushikata X-Plan 380dIII/460dIII, Tokyo, Japan) on lateral cephalograms (Fig. 2a, b). Each area was measured in triplicate in an average manner.
a Lateral cephalometric landmarks and planes. Eb base of epiglottis, Ba basion, P palate point, Ptm the most superior point of the pterygomaxillary fissure, PNS posterior nasal spine, PPW1 the point where the plane parallel to the palatal plane and passing through PNS intersects the posterior pharyngeal wall, PPW2 the pharyngeal wall point intersecting the plane parallel from the palatal plane and passing through the point P, PPW3 the point where the plane parallel to the palatal plane and passing through Eb point intersects the posterior pharyngeal wall. b Pharyngeal area measurements. Nasopharynx: the area confined within the anterior and posterior pharyngeal wall on the side, the PNS-PPW1 line below, and the region above where the line drawn from Ba point to the sphenoid bone intersects the perpendicular line drawn from Ptm. Superior oropharynx: the area confined within the anterior and posterior pharyngeal wall on the side, the P-PPW2 line below, and the PNS-PPW1 line above. Inferior oropharynx: the area confined within the anterior and posterior pharyngeal wall on the side, the Eb-PPW3 line below, and the P-PPW2 line above
Maxillary dental arch measurements (intercanin, interpremolar, intermolar distance) were performed on dental casts twice by using digital compasses (Absolute Digimatic Mitutoyo) before and after SRME treatment (Fig. 3).
Maxillary dental arch measurements. (1) Intercanine width: the distance between the cusp tips of the left and right upper canine tips. (2) Interpremolar width: the distance between the buccal cusp tips of the left and right upper first premolars. (3) Intermolar width: the distance between the mesio-buccal cusp tips of the left and right upper first molars
Postero-anterior cephalometric analyses (nasal and maxillary widths) were done and evaluated by using Dolphin computer software (Fig. 4).
Sleep analysis by using a multichannel device
The measurements for OSAS in control and SRME-treated groups were performed by a portable multichannel device at the baseline (C0 and T0) and after a 5-month follow-up (C1 and T1). The ApneaLink Plus (ResMed Corporation, Poway, CA) is a multichannel screening tool designed to screen for OSAS as shown in Fig. 1d. The device records air flow via a nasal cannula connected to a nasal pressure transducer. A belt placed around the chest records thoracic respiratory effort and electrocardiogram. Pulse oximetry is also recorded via the digital probe to measure hemoglobin saturation. The compact screening device was attached to the patient’s wrist and it provided information regarding the extent of sleep fragmentation.
Each ApneaLink study was scored manually by the same investigator based on pediatric criteria. AHI, flow limitation, snoring, blood oxygen saturation, and desaturation data were obtained by ApneaLink software. Respiratory events were scored in the presence of at least 3% of desaturation, when there is no movement and change causing snoring and pulse changes. All desaturations which are decreased more than 3% from baseline arterial oxygen saturation (SaO2) were quantified. OSAS was identified as an AHI > 1 [19].
ELISA for determination of OSAS-related parameters in serum and urine
The blood samples in serum separator tubes and first-morning urine samples were collected on the day after the sleep analysis. Serum and urine specimens were stored at − 80 °C until analysis. Serum and urine levels of ORM2, FABP4, perlecan, gelsolin, KLK1, and uric acid were assayed with commercially available ELISA kits used according to the manufacturer’s instructions; human ORM2, with a sensitivity of 0.05 ng/mL, and linearity between 0.05 and 15 ng/mL; human FABP4; human perlecan; human gelsolin; human KLK1 (Sunred Biological Technology Co., China), which has a sensitivity of 0.083 ng/mL, exhibits linearity between 0.1 and 30 ng/mL, and has inter- and intra-assay coefficients of variability 12 and 10%, respectively; and human uric acid (Cell Biolabs, Inc., San Diego, CA), which has a sensitivity of 0.5 μM.
Statistical analysis
Data are expressed as means ± SE (standard error). The changes before and after control/treatment were compared by Student’s t test and Mann-Whitney U test. The Pearson correlation test and Spearman’s rank correlation were applied to determine the relationship between parameters. p values less than 0.05 were considered as statistically significant.
Results
Study population
The orthodontic evaluation and parental questionnaire results demonstrated that all children had dental malocclusion with narrow maxilla and snoring. The characteristics of control and treatment groups were shown in Table 1. There were no significant differences in age, sex, and BMI distribution between groups. Half of the children (T0, n = 15) completed the 5-month SRME treatment (T1), and other 15 children (C0, n = 15) with OSAS were accepted as the control group and subjected to a follow-up for 5 months (C1). None of the volunteers experienced adverse effects when SRME was applied. The alterations in sleep parameters, pharyngeal area, dental arch, and posterio-anterior widths were measured before and after SRME treatment.
According to the sleep test results, AHI decreased significantly in the treatment group from T0 to T1 and the control group from C0 to C1 (Table 1, p < 0.05). Oxygen desaturation index (ODI) and SaO2 values were not altered significantly in groups before and after the 5-month control/treatment duration (Table 1). There were also no differences in sleep parameters between before and after treatment (T1-T0) and control (C1-C0) (Table 1).
As shown in Table 2, a significant increase in nasopharyngeal (p < 0.05) and total pharyngeal area (p < 0.01) was observed after SRME in children with OSAS. Oropharynx area did not change before and after treatment. In the dental model measurement, intercanine, interpremolar, and intermolar area showed an increase after SRME treatment (Table 2, p < 0.001). The nasal, maxillary, and intermolar widths were increased by treatment (Table 2, p < 0.001 and p < 0.05).
The change in protein levels associated with OSAS in serum and urine and its relationship between arch widths
KLK1 levels were decreased significantly in the serum samples of children with OSAS after SRME treatment (Table 1, p < 0.05). An increase in serum ORM2 (p < 0.05) and a decrease in urine KLK1 and perlecan levels (p < 0.05 and p < 0.01) were observed after 5 months of follow-up in the control group as shown in Table 1. Changes in serum levels of ORM2 before and after treatment (T1-T0) also showed a significant difference compared to changes in serum levels of ORM2 before and after the follow-up period in the control group (C1-C0) (Table 1, p < 0.05). Although urinary uric acid levels were increased slightly in the control group and decreased in the treatment group, the alteration was not statistically significant at the end of the control and treatment period (Table 1). Also, FABP4 and gelsolin levels were not altered in serum after control/treatment (Table 1). Besides a significant negative correlation found between serum ORM2, perlecan, gelsolin, KLK1 levels, and intercanin width, a negative correlation at the p < 0.01 level was observed between serum uric acid levels and interpremolar width (Table 3). We also found an inverse relationship between serum ORM2, KLK1, and uric acid levels and intermolar width (Table 3, p < 0.05).
Discussion
In this study, we evaluated the beneficial effect of SRME orthodontic treatment in children with OSAS by comparing the levels of biomarkers as well as sleep test data, pharyngeal area, maxillary dental arch, and postero-anterior widths before and after treatment. We observed that pharyngeal airway space and maxillary dental arch width (intercanine, interpremolar, and intermolar) and postero-anterior width (nasal and maxillary) increased after SRME. AHI, an indicator of sleep apnea severity, was decreased following the treatment. The SRME-treated group showed a statistically significant decrease in serum KLK1 levels. In the control group, we observed a decrease in perlecan levels of urine and an increase in serum ORM2 levels at the fifth month. There was also a significant negative correlation between serum levels of OSAS-associated markers, ORM2, KLK1, gelsolin and perlecan, and intercanin width. Serum uric acid levels and interpremolar/intermolar width also demonstrated an inverse relationship in the study.
According to the results, the significant increase in pharyngeal airway space, intercanine, interpremolar, intermolar, and postero-anterior width can be considered as a positive progress induced by SRME. A study by Ashok et al. showed that RME is a useful treatment option for widening of maxilla [20]. AHI measured by an ApneaLink portable device was significantly decreased after control/treatment duration, while one of the most important markers of intermittent hypoxia, ODI, and desaturation (SaO2 < 95%) did not show any alteration before/after control and treatment. Similarly, Villa et al. reported statistically significant decreases in AHI of children with OSAS by RME [7, 8, 21]. An improvement in sleep parameters after RME was shown in children with sleep-disordered breathing [20]. Also, a recent study showed that RME treatment reduced AHI in children with OSAS [22]. Previous studies reported that a high-sensitive and specific determination of AHI could be provided by ApneaLink Plus portable devices [23, 24]. We can suggest that the clinical symptoms of OSAS may be resolved by expanding the pharyngeal airway, dental arch, and posterio-anterior widths and decreasing AHI at the end of the treatment period.
As a potential OSAS biomarker, serum ORM2 levels showed an increase in the control group after 5 months from the first specimen collection. Serum ORM2 levels showed an inverse correlation with intercanin width. Similarly, a previous study by Gozal et al. showed that urine ORM2 levels were increased in children with OSAS [1]. Magid et al. showed that ORM2 levels were increased in urine in association with acute inflammation [25]. Increased expression of ORM2 in the serum samples of children with OSAS may also induce the propagation of inflammation-related markers like FABP4.
FABP4 is a cytosolic lipid chaperone, and our present data showed that serum levels of FABP4 were increased in OSAS and decreased at a clinical level following SRME treatment. As a supporting evidence by Bhushan, children with OSAS showed an increased morning plasma FABP4 levels [26]. Several studies also reported a higher serum FABP4 levels in adults with OSAS [27, 28]. Lam et al. have reported that plasma FABP4 levels were significantly associated with sleep hypoxemia parameters, including duration of oxygen desaturation and minimal oxygen saturation, independent of age and obesity [28]. FABP4 levels may be upregulated markedly in subjects with high AHI and severe degree of hypoxemia. In our study, the sleep apnea in children with OSAS may not be severe enough to cause tissue hypoxia. The different intensity of intermittent hypoxia in OSAS subjects results in different expression profiles of FABP4. We also suggested that increased FABP4 levels may also be associated with inflammation arisen from increased hypoxia during OSAS.
At present study, urine perlecan levels were decreased at the fifth month in the control group, while we did not observe any change in the treatment group. The inflammatory events can induce an increase in perlecan levels [29]. OSAS is a disease which has an inflammatory component [30]. No significant change of perlecan levels of the treatment group may be the result of the beneficial effect of RME on OSAS. In addition, the decreased urine perlecan levels of the control group may be related to the mild severity of OSAS. The urine perlecan level has been suggested as a potential clinical marker for many diseases. Serum contains low levels of perlecan relative to those found in urine [31, 32]. Krishna et al. [33] reported an increased expression of perlecan in the urinary proteome of children with OSAS. Considering a negative relationship between serum perlecan levels and intercanin width in this study, we can suggest that perlecan levels, as a hypoxia-related marker, can be affected by dental arch width. The marker of perlecan can be used to understand if the hypoxic condition is still present in children with OSAS or not.
In the present study, unchanged gelsolin levels before and after control/treatment may be related to the mild severity of OSAS. We suggest that the mild OSAS could not induce hypoxia and inflammation-related changes like protein permeability, catabolism, and excretion in children. The role of gelsolin in inflammation associated with OSAS is still unknown. A study by Zhang et al. [34] showed that treatment with gelsolin reduces brain inflammation in mice. Another study showed that gelsolin expression is necessary for the development of pulmonary inflammation [35]. A significant negative relationship between serum gelsolin levels and intercanin width may suggest that an increase in intercanin width may prevent hypoxia through decreasing gelsolin levels in patients with OSAS. Our data showed that KLK1 levels, another important marker for OSAS, were decreased in both the control and SRME-treated groups. Similarly, Gozal et al. [1] showed that KLK1 levels were significantly reduced in urine samples of children with OSAS. Previous studies showed that tissue KLK1 protected against retinal and neuronal ischemic damage in different animal models [36, 37]. We can suggest that a decrease in serum KLK1 levels can exert inflammatory effects in OSAS. SRME treatment may prevent hypoxia and inflammation related with OSAS by increasing KLK1 levels.
According to our results, a slightly lower uric acid level of urine in response to SRME is an important clinical finding, despite the low number of individuals in study groups. Several studies demonstrated a positive correlation between the severity of sleep apnea and increased levels of serum uric acid levels as a reflection of oxidative stress and hypoxia [38,39,40]. Oxidative stress is the result of a hypoxia-reoxygenation phenomenon and is associated with elevated serum uric acid levels [17, 40]. Van Hoorenbeeck et al. have reported that improvements in uric acid levels were correlated with improvements in sleep parameters such as ODI [39]. We can suggest that sleep parameters such as ODI and SaO2 values can show positive changes depending on uric acid levels. The findings of studies may vary because of different skeletal and dental expansion rates in different age groups. Our findings indicated that there was a decrease in uric acid excretion due to maxillar expansion and decreased airway blockage by SRME treatment.
Although this study is the first to evaluate the relation between SRME treatment and biochemical markers, it has some limitations. Therefore, further studies are needed to make certain interpretations about the role of relevant biochemical markers in patients with OSAS. A limitation of our study was the short follow-up, the small number of subjects, and the mild degree of OSAS. Our results pointed to the alteration of some biomarker levels (perlecan, KLK1, uric acid) in snoring children with SRME treatment. Our findings warrant further studies based on larger numbers of subjects and a population with severe OSAS. In conclusion, the 5-month personalized SRME treatment showed beneficial effects on children with OSAS and may be considered as a useful approach in children with OSAS to deal with abnormal breathing during sleep. Future studies which aim to characterize the protein maps in OSAS are required to provide reliable and convenient alternative clinical approaches in the treatment of patients with OSAS.
References
Gozal D, Jortani S, Snow AB, Kheirandish-Gozal L, Bhattacharjee R, Kim J, Capdevila OS (2009) Two-dimensional differential in-gel electrophoresis proteomic approaches reveal urine candidate biomarkers in pediatric obstructive sleep apnea. Am J Respir Crit Care Med 180(12):1253–1261. https://doi.org/10.1164/rccm.200905-0765OC
Guilleminault C, Eldridge FL, Simmons FB, Dement WC (1976) Sleep apnea in eight children. Pediatrics 58(1):23–30
Chhangani BS, Melgar T, Patel D (2010) Pediatric obstructive sleep apnea. Indian J Pediatr 77(1):81–85. https://doi.org/10.1007/s12098-009-0266-z
Health Quality O (2006) Polysomnography in patients with obstructive sleep apnea: an evidence-based analysis. Ont Health Technol Assess Ser 6(13):1–38
Bilgin C, Erkorkmaz U, Ucar MK, Akin N, Nalbant A, Annakkaya AN (2016) Use of a portable monitoring device (Somnocheck Micro) for the investigation and diagnosis of obstructive sleep apnoea in comparison with polysomnography. Pak J Med Sci 32(2):471–475. https://doi.org/10.12669/pjms.322.9561
Pirelli P, Saponara M, Guilleminault C (2004) Rapid maxillary expansion in children with obstructive sleep apnea syndrome. Sleep 27(4):761–766
Villa MP, Rizzoli A, Miano S, Malagola C (2011) Efficacy of rapid maxillary expansion in children with obstructive sleep apnea syndrome: 36 months of follow-up. Sleep Breath 15(2):179–184. https://doi.org/10.1007/s11325-011-0505-1
Villa MP, Malagola C, Pagani J, Montesano M, Rizzoli A, Guilleminault C, Ronchetti R (2007) Rapid maxillary expansion in children with obstructive sleep apnea syndrome: 12-month follow-up. Sleep Med 8(2):128–134. https://doi.org/10.1016/j.sleep.2006.06.009
Katyal V, Pamula Y, Daynes CN, Martin J, Dreyer CW, Kennedy D, Sampson WJ (2013) Craniofacial and upper airway morphology in pediatric sleep-disordered breathing and changes in quality of life with rapid maxillary expansion. Am J Orthod Dentofac Orthop 144(6):860–871. https://doi.org/10.1016/j.ajodo.2013.08.015
Baratieri C, Alves M Jr, de Souza MM, de Souza Araujo MT, Maia LC (2011) Does rapid maxillary expansion have long-term effects on airway dimensions and breathing? Am J Orthod Dentofac Orthop 140(2):146–156. https://doi.org/10.1016/j.ajodo.2011.02.019
McNamara JA Jr, Lione R, Franchi L, Angelieri F, Cevidanes LH, Darendeliler MA, Cozza P (2015) The role of rapid maxillary expansion in the promotion of oral and general health. Prog Orthod 16(1):33. https://doi.org/10.1186/s40510-015-0105-x
Catala R, Cabre A, Hernandez-Flix S, Ferre R, Sangenis S, Plana N, Texido A, Masana L (2013) Circulating FABP4 and FABP5 levels are differently linked to OSA severity and treatment. Sleep 36(12):1831–1837. https://doi.org/10.5665/sleep.3210
Costello MJ, Gewurz H, Siegel JN (1984) Inhibition of neutrophil activation by alpha1-acid glycoprotein. Clin Exp Immunol 55(2):465–472
Sorensson J, Matejka GL, Ohlson M, Haraldsson B (1999) Human endothelial cells produce orosomucoid, an important component of the capillary barrier. Am J Phys 276(2 Pt 2):H530–H534
Yin HL, Stossel TP (1979) Control of cytoplasmic actin gel-sol transformation by gelsolin, a calcium-dependent regulatory protein. Nature 281(5732):583–586. https://doi.org/10.1038/281583a0
Yin HL, Albrecht JH, Fattoum A (1981) Identification of gelsolin, a Ca2+-dependent regulatory protein of actin gel-sol transformation, and its intracellular distribution in a variety of cells and tissues. J Cell Biol 91(3 Pt 1):901–906. https://doi.org/10.1083/jcb.91.3.901
Hira HS, Shukla A, Kaur A, Kapoor S (2012) Serum uric acid and lactate levels among patients with obstructive sleep apnea syndrome: which is a better marker of hypoxemia? Ann Saudi Med 32(1):37–42. https://doi.org/10.5144/0256-4947.2012.37
Iseri H, Ozsoy S (2004) Semirapid maxillary expansion—a study of long-term transverse effects in older adolescents and adults. Angle Orthod 74(1):71–78. https://doi.org/10.1043/0003-3219(2004)074<0071:SMESOL>2.0.CO;2
Witmans MB, Keens TG, Davidson Ward SL, Marcus CL (2003) Obstructive hypopneas in children and adolescents: normal values. Am J Respir Crit Care Med 168(12):1540. https://doi.org/10.1164/ajrccm.168.12.954
Ashok N, Varma NK, Ajith VV, Gopinath S (2014) Effect of rapid maxillary expansion on sleep characteristics in children. Contemp Clin Dent 5(4):489–494. https://doi.org/10.4103/0976-237X.142817
Villa MP, Rizzoli A, Rabasco J, Vitelli O, Pietropaoli N, Cecili M, Marino A, Malagola C (2015) Rapid maxillary expansion outcomes in treatment of obstructive sleep apnea in children. Sleep Med 16(6):709–716. https://doi.org/10.1016/j.sleep.2014.11.019
Vale F, Albergaria M, Carrilho E, Francisco I, Guimaraes A, Caramelo F, Malo L (2017) Efficacy of rapid maxillary expansion in the treatment of obstructive sleep apnea syndrome: a systematic review with meta-analysis. J Evid Based Dent Pract 17(3):159–168. https://doi.org/10.1016/j.jebdp.2017.02.001
Erman MK, Stewart D, Einhorn D, Gordon N, Casal E (2007) Validation of the ApneaLink for the screening of sleep apnea: a novel and simple single-channel recording device. J Clin Sleep Med 3(4):387–392
Lesser DJ, Haddad GG, Bush RA, Pian MS (2012) The utility of a portable recording device for screening of obstructive sleep apnea in obese adolescents. J Clin Sleep Med 8(3):271–277. https://doi.org/10.5664/jcsm.1912
Magid E, Guldager H, Hesse D, Christiansen MS (2005) Monitoring urinary orosomucoid in acute inflammation: observations on urinary excretion of orosomucoid, albumin, alpha1-microglobulin, and IgG. Clin Chem 51(11):2052–2058. https://doi.org/10.1373/clinchem.2005.055442
Bhushan B, Khalyfa A, Spruyt K, Kheirandish-Gozal L, Capdevila OS, Bhattacharjee R, Kim J, Keating B, Hakonarson H, Gozal D (2011) Fatty-acid binding protein 4 gene polymorphisms and plasma levels in children with obstructive sleep apnea. Sleep Med 12(7):666–671. https://doi.org/10.1016/j.sleep.2010.12.014
Balci MM, Arslan U, Firat H, Kocaoglu I, Vural MG, Balci KG, Maden O, Gurbuz OA, Ardic S, Yeter E (2012) Serum levels of adipocyte fatty acid-binding protein are independently associated with left ventricular mass and myocardial performance index in obstructive sleep apnea syndrome. J Investig Med 60(7):1020–1026. https://doi.org/10.2310/JIM.0b013e31826868f2
Lam DC, Xu A, Lam KS, Lam B, Lam JC, Lui MM, Ip MS (2009) Serum adipocyte-fatty acid binding protein level is elevated in severe OSA and correlates with insulin resistance. Eur Respir J 33(2):346–351. https://doi.org/10.1183/09031936.50075408
Garcia de Yebenes E, Ho A, Damani T, Fillit H, Blum M (1999) Regulation of the heparan sulfate proteoglycan, perlecan, by injury and interleukin-1alpha. J Neurochem 73(2):812–820
Calvin AD, Albuquerque FN, Lopez-Jimenez F, Somers VK (2009) Obstructive sleep apnea, inflammation, and the metabolic syndrome. Metab Syndr Relat Disord 7(4):271–278. https://doi.org/10.1089/met.2008.0093
Lee EY, Kim SH, Whang SK, Hwang KY, Yang JO, Hong SY (2003) Isolation, identification, and quantitation of urinary glycosaminoglycans. Am J Nephrol 23(3):152–157
Mitsuhashi H, Tsukada Y, Ono K, Yano S, Naruse T (1993) Urine glycosaminoglycans and heparan sulfate excretions in adult patients with glomerular diseases. Clin Nephrol 39(5):231–238
Krishna J, Shah ZA, Merchant M, Klein JB, Gozal D (2006) Urinary protein expression patterns in children with sleep-disordered breathing: preliminary findings. Sleep Med 7(3):221–227. https://doi.org/10.1016/j.sleep.2005.09.010
Zhang QH, Chen Q, Kang JR, Liu C, Dong N, Zhu XM, Sheng ZY, Yao YM (2011) Treatment with gelsolin reduces brain inflammation and apoptotic signaling in mice following thermal injury. J Neuroinflammation 8(1):118. https://doi.org/10.1186/1742-2094-8-118
Oikonomou N, Thanasopoulou A, Tzouvelekis A, Harokopos V, Paparountas T, Nikitopoulou I, Witke W, Karameris A, Kotanidou A, Bouros D, Aidinis V (2009) Gelsolin expression is necessary for the development of modelled pulmonary inflammation and fibrosis. Thorax 64(6):467–475. https://doi.org/10.1136/thx.2008.107946
Masuda T, Shimazawa M, Ishizuka F, Nakamura S, Tsuruma K, Hara H (2014) Tissue kallikrein (kallidinogenase) protects against retinal ischemic damage in mice. Eur J Pharmacol 738:74–82. https://doi.org/10.1016/j.ejphar.2014.05.033
Wang Z, Han X, Cui M, Fang K, Lu Z, Dong Q (2014) Tissue kallikrein protects rat hippocampal CA1 neurons against cerebral ischemia/reperfusion-induced injury through the B2R-Raf-MEK1/2-ERK1/2 pathway. J Neurosci Res 92(5):651–657. https://doi.org/10.1002/jnr.23325
Hirotsu C, Tufik S, Guindalini C, Mazzotti DR, Bittencourt LR, Andersen ML (2013) Association between uric acid levels and obstructive sleep apnea syndrome in a large epidemiological sample. PLoS One 8(6):e66891. https://doi.org/10.1371/journal.pone.0066891
Van Hoorenbeeck K, Franckx H, Debode P, Aerts P, Wouters K, Ramet J, Van Gaal LF, Desager KN, De Backer WA, Verhulst SL (2012) Weight loss and sleep-disordered breathing in childhood obesity: effects on inflammation and uric acid. Obesity (Silver Spring) 20(1):172–177. https://doi.org/10.1038/oby.2011.282
Verhulst SL, Van Hoeck K, Schrauwen N, Haentjens D, Rooman R, Van Gaal L, De Backer WA, Desager KN (2007) Sleep-disordered breathing and uric acid in overweight and obese children and adolescents. Chest 132(1):76–80. https://doi.org/10.1378/chest.06-2930
Acknowledgments
We would like to thank Dr. Nazlı Karaca for scoring sleep analysis data obtained from the portable multichannel device and Professor Ensar Baspinar who have been of great help in the statistical analysis.
Funding
This study was supported by Ankara University Scientific Research Foundation with 13L3334002 project number. The sponsor had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (name the institution/committee) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Additional informed consent was obtained from all individual participants for whom identifying information is included in this article.
Rights and permissions
About this article
Cite this article
Hoxha, S., Kaya-Sezginer, E., Bakar-Ates, F. et al. Effect of semi-rapid maxillary expansion in children with obstructive sleep apnea syndrome: 5-month follow-up study. Sleep Breath 22, 1053–1061 (2018). https://doi.org/10.1007/s11325-018-1636-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-018-1636-4