Abstract
Objective
Supine body orientation plays an important role in precipitating upper airway collapse in a significant proportion of obstructive sleep apnea (OSA) patients known to have supine-predominant OSA (OSAsup). Traditionally, trunk position is used to assess OSAsup, but the role of the head position has not been established. We hypothesized that head position influences OSA independently of trunk position.
Methods
Head and trunk positions were determined from subjects undergoing overnight polysomnography. The apnea-hypopnea index (AHI), rapid eye movement (REM), and non-REM sleep time of all trunk and head positions (lateral and supine) were calculated and compared against the complete supine position, i.e., head and trunk supine.
Results
In 26 subjects, lateral rotation of the head to the right or left with the trunk supine resulted in a significant reduction in AHI from 36.0 ± 22.5 to 25.8 ± 16.6 (p = 0.008), and an AHI drop <10 in 27% of patients. The “trunk lateral–head lateral” position resulted in a more dramatic reduction in AHI from 31.6 ± 20.2 to 4.1 ± 4.1 (p < 0.0001). The distributions of REM and non-REM sleep were not different among positions. In the subgroup with a body mass index (BMI) <32 kg/m2 (15 subjects), the AHI reduction with lateral head rotation was significant (p = 0.005) but not in remaining 11 obese patient with a BMI ≥32 kg/m2 (p = 0.24).
Conclusion
OSA severity with the trunk in the supine position decreased significantly when the head rotated from supine to lateral, particularly in non-obese patients. These results demonstrate an important influence of head position on the AHI, independently of trunk position and sleep stage, in patients with OSA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnea (OSA) is a condition characterized by recurrent complete or partial collapse of the upper airway during sleep. This leads to complete (apnea) or partial (hypopnea) cessation of airflow, that is often accompanied by an oxygen desaturation and/or an arousal from sleep. This results in sleep disruption which, in turn, causes excessive daytime sleepiness and fatigue [1]. OSA is a common condition affecting approximately 7% of adults [2, 3] and is independently associated with increased risk of motor vehicle accidents [4], cardiovascular diseases [5], and impaired cognitive function [6]. The physiological presence and severity of OSA is assessed by the frequency of apneas and hypopneas per hour of sleep (apnea-hypopnea index, AHI).
Among several pathophysiological mechanisms of OSA is relapse of the tongue against the posterior pharyngeal wall due to gravity. This mechanism is particularly important in supine-predominant OSA (OSAsup), a condition characterized by markedly greater AHI in the supine than in the non-supine position [7, 8]. OSAsup constitutes a considerable proportion of OSA patients, ranging from to 25 to 70% [9,10,11,12]. Distinguishing OSAsup patients from the remaining OSA population is clinically important because it may influence treatment, since OSAsup patients can be treated by means of body position adjustment during sleep. In standard clinical practice, a patient’s position is characterized as supine or lateral depending on trunk orientation using overnight video recording or gyroscopic transducers on the trunk. On the other hand, the collapsible segment of the upper airway, the pharynx, is contained within the head and upper neck, and spans the segment between the nasal turbinates and the vocal cords. Therefore, it stands to reason that head position may play a role in influencing the AHI in patients with OSA.
However, the effect of lateral head rotation to the left and right on the AHI during sleep has not been thoroughly investigated in patients with OSA. The goal of this study was, therefore, to investigate the effects of head position, independently of trunk position, on OSA severity during sleep.
Methods
Subjects
We recruited subjects referred for overnight polysomnography (PSG) to the Sleep Research Laboratories of the Toronto Rehabilitation Institute and the Toronto General Hospital, University Health Network. Analysis of PSG data was approved by the institutional research ethics board and participants signed a consent form.
Polysomnography
Patients underwent overnight PSG using standard techniques and scoring criteria for sleep stages and arousals from sleep [13, 14]. Respiratory signals including thoracoabdominal movements by respiratory impedance plethysmography (RIP), nasal pressure via nasal pressure cannulae, and arterial oxyhemoglobin saturation (SaO2) via pulse oximetry were recorded on a computerized sleep scoring system as described previously [15]. Apnea was defined as a reduction in the sum channel of the RIP by ≥90% lasting ≥10 s and hypopnea as a reduction by ≥30 to 90% lasting ≥10 s and accompanied by a ≥3% desaturation or an arousal from sleep. Respiratory events were classified as obstructive if there was out-of-phase thoracoabdominal motion or flow limitation on the nasal pressure tracing, and central if there was absent thoracoabdominal or in-phase thoracoabdominal motion without evidence of airflow limitation, during apneas and hypopneas, respectively. The scoring technician was blinded to the purpose of this study. An overnight video recording using a Sony PTZ IP/Analog Hybrid Camera was used to determine body and trunk positions. Patients were given the choice to sleep on one or two pillows.
The AHI was calculated for total sleep time and for times spent in the various positions described below (Table 1). AHI for each position was calculated as the total number of events over the cumulative time slept in that position. Based on PSG data, subjects were included in the subsequent examination of the head and body posture if they had an overall AHI ≥5, spent ≥20 min supine, had adequate quality of video recordings, had a total sleep time of ≥3.5 h, and were predominantly obstructive, i.e., 50% or greater of their respiratory events were obstructive.
Determination of trunk and head positions
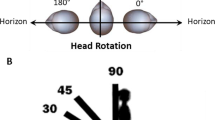
Trunk and head positions were identified by visual inspection of the PSG video recording, frame-by-frame by an examiner who was blinded to apnea and hypopneas scoring. Head and trunk positions were visually classified as supine if facing upwards or within approximately 45 degree from the sagittal (vertical) plane and non-supine otherwise, as shown in Fig. 1. Accordingly head and trunk positions were categorized into the classes listed in Table 1.
To detect a change in AHI related to a change in position, only subjects with an AHI ≥10 in the complete supine position (TSHS) who spent ≥20 min in TSHS and ≥20 min with a trunk supine–head lateral position were included. Subjects with low AHI in the TSHS position were excluded because they are not likely to have sleep apnea or to exhibit a change in AHI with a change in position.
Determination of sleep structure
In order to control for the effect of sleep structure on AHI, the durations of rapid eye movement (REM) sleep and non-REM sleep were quantified, and their proportions to the total sleep time were calculated for the postures of interest.
Effect of body mass index
To examine the effect of body mass index (BMI) on AHI, a subgroup analysis of patient with a BMI <32 and ≥32 kg/m2 was performed.
Statistical analysis
The changes in AHI between the TSHS and other sleep positions were calculated. The Shapiro–Wilk test was used to examine normality. The median and median absolute deviation (MAD) for non-normally distributed data, or mean and standard deviation for normally distributed data, were calculated for each group. The intra-subject percent change in AHI and the median percent change (med-Δ) for all subjects were calculated. Intra-subject differences in AHI were analyzed using the one-tailed Wilcoxon signed-rank sum test for paired comparisons of non-normally distributed data.
Results
Subjects
A total of 140 patients consented in the period between January 2011 and July 2013. Seventy-one subjects did not meet the PSG inclusion criteria (Section 2.2), mainly because of the absence of OSA or insufficient supine sleep time. Of the remaining 69 subjects, 43 were excluded based on criteria in Section 2.3, mainly short durations in TSHS or in head-lateral positions. The remaining 26 subjects were included, whose characteristics are displayed in Table 2 . The duration spent in each position is listed in Table 3.
Changes in AHI related to changes in trunk and head positions
AHIs in both TSHS and TLHL positions were available in 14 subjects. The intra-subject AHI decreased from the TSHS to the TLHL position with a med-Δ of −94% (p < 0.001). Similarly, AHIs in both TSHS and TRHR were available in 14 subjects in whom the med-Δ was −86% (p < 0.001).
Lateral head rotation, to either left of right, while maintaining the supine trunk orientation resulted in drops in the AHI. The change in head position from TSHS to TSHL led to a significant reduction in AHI with a med-Δ of −44% (p = 0.006) in 19 subjects in which those two positions were available. The change in head position from TSHS to TSHR resulted in a med-Δ of −40% (p = 0.055) in 22 subjects in which those positions were observed.
No significant difference was found between the TRHR and TLHL positions (p = 0.82), nor between the TSHR and TSHL (p = 0.47) positions. Therefore we combined lateral positions to form the “trunk lateral–head lateral” position (TRHR and TLHL positions combined). The combined AHI was obtained for the total time spent in those positions. Similarly, TSHR and TSHL positions were combined to form one group: “trunk supine–head lateral,” and the corresponding AHI was calculated.
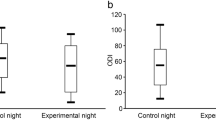
Both lateral head rotation and complete lateral position resulted in a significant drop in overall AHIs compared with TSHS, as shown in Fig. 2. The intra-subject drop in AHI upon change from TSHS to the “trunk supine–head lateral” position gave a med-Δ of −29% (p = 0.008) whereas in the “trunk lateral–head lateral” position med-Δ was −85% (p < 0.0001). It should be noted that overall AHI medians are not necessarily within the range of the single-side positions due to the non-linearity of the median operator.
In seven subjects (27%) with AHIs ranging from 10.5 to 38.5 in TSHS, OSA was essentially eliminated since the AHI fell to <10 with head rotation, while maintaining a supine trunk position. AHIs of those seven subjects are presented in Fig. 3.
Sleep stages and duration
As shown in Fig. 4, the proportion of REM and non-REM sleep to total sleep time did not differ significantly in any of the three positions (p > 0.13 for all comparisons).
Effect of BMI on AHI drop with lateral head position
Fifteen subjects had a BMI <32 (average = 27.0 ± 3.2 kg/m2) and the remaining 11 subjects had a BMI ≥32 (average = 39.9 ± 9.4 kg/m2). In subjects with a lower BMI, there was a significant reduction of AHI from TSHS to trunk supine–head lateral by a med-Δ of −48.7% (p = 0.005). On the other hand, in patients with a higher BMI there was a reduction by a med-Δ of −24.3% that was not significant (p = 0.24). In the between-group comparison, the reduction in the lower BMI group was larger than in the high BMI group (p = 0.04). Fig. 3 illustrates the change in AHI in both subgroups.
Discussion
In this study, we demonstrate that head position, independent from trunk position, has a significant impact on AHI in patients with OSA. OSA severity in the trunk supine position significantly decreased when the head was rotated from supine to lateral, and was essentially resolved in 27% of patients in whom the AHI fell to below 10. These results show that lateral rotation of the head can reduce the AHI even when the trunk continues to assume a supine posture and that distribution of REM to non-REM sleep does not play a role in this reduction. Our results confirm the findings of Safiruddin et al. who showed that the severity of upper airway collapse in OSA patients undergoing endoscopy during drug-induced sleep decreased, most prominently in patients with supine-predominant OSA, when the head was rotated to the left or right [16]. Our findings are also consistent with van Kesteren et al., who demonstrated in patients with supine–head related OSA, that lateral head rotation while the trunk is supine resulted in a significant reduction in the AHI [17]. Our study extends those findings to OSA patients in general whether or not their OSA is supine-related, during natural sleep and by ruling out differences in sleep structure as a cause of differences in AHI in relation to differing trunk and head positions.
The influence of trunk orientation on OSA is a well-known [18]. However, whether the cause of AHI fluctuation is due to a change in trunk position or head position or both has received little attention in the literature. In clinical practice, nonetheless, posture is generally judged on the basis of trunk position. It may be important to distinguish the effects of trunk and head position on OSA severity since OSA is supine-predominant in a considerable proportion of patients and since management of head versus trunk position may vary [9,10,11,12].
As expected, we found a significant decrease in AHI from the complete supine to the complete lateral position. Furthermore, the decrease in the AHI in the full lateral position was more pronounced than that during the “trunk supine–head lateral” position indicating an additive effect of the trunk lateral to the head lateral position on maintaining upper airway patency and reducing the AHI.
Since sleep stages can influence the AHI, such that REM sleep is generally associated with a higher AHI than non-REM sleep [19, 20], it is important to control for this when examining the effect of posture on the AHI. Unlike previous studies, we were able to control for potential effect of sleep stage distribution on AHI in relation to head position since we observed that the proportions of REM and non-REM sleep were evenly distributed among different trunk and head positions. Accordingly, the reduction in the AHI from the head supine to the head lateral position cannot be attributed to changes in sleep stage distribution.
The impact of changes in head and trunk on the AHI points to an anatomical or functional effect of such changes on the upper airway. The collapsible segment of the upper airway, the pharynx, is located within the head and upper neck, and extends from the nasal turbinates to the vocal cords [21]. Therefore, head position could play an important role in altering the size, shape, and collapsibility of the pharynx in relation to gravity and other factors. Generally, patients with OSA have a narrower pharynx than subjects without OSA [22]. When moving from the upright to the supine position, further narrowing of the pharynx can occur due to gravitational effects that cause oro-pharyngeal soft tissue, especially the tongue, to relapse towards the posterior pharyngeal wall rendering it more susceptible to collapse [23, 24]. Treatments aimed at preventing sleep in the supine posture, such as shirts with balls sewn on the back, have been shown to eliminate or attenuate supine-related OSA, presumably because they prevent relapse of the tongue against the posterior pharyngeal wall [25].
The reason why rotation of the head from supine to non-supine reduces the AHI in OSA patients is not clear. One likely possibility is that when the head is in the lateral, as opposed to the supine position, the tendency of the tongue to relapse posteriorly is mitigated, and since the lateral pharyngeal walls have relatively smaller tissue mass than the tongue, this results in a weaker effect of gravity and less tendency for them to collapse inwards. This potential mechanism is supported by magnetic resonance imagining during head rotation from supine to lateral with the trunk supine showing a marked increase in upper airway caliber, especially the retroglossal and retropalatal pharyngeal cross-sectional area and volume [26]. Collectively, the decreased gravitational effect on the tongue, increased upper airway caliber, and volume of the upper airway associated with head rotation from supine to lateral might explain the reduced collapsibility and decreased AHI seen in the “trunk supine–head lateral” position compared to the TSHS position in the present study.
In this study, we found that the reduction in AHI from head supine to head lateral was greater in those with a BMI <32 than in those with a BMI of ≥32. On the other hand, in those with a BMI ≥32 the reduction in AHI was not significant. The reason for this is not clear but may be related to deposition of excess parapharyngeal fat tissue in very obese patients that could prevent the favorable effects of lateral head rotation on OSA severity. This observation should be interpreted with caution because of the small number of patients in the high BMI group.
Our results suggest that manipulation of head position could be another type of postural therapy to treat selected patients with OSA in whom changing from the supine to lateral head position with the trunk supine reduces the AHI. For example, in qualified patients, optimization of head posture can be an alternative to the generic positional therapy tools that restrict trunk rotation, or can be used as an adjunct modality to mainstream treatments, such as CPAP or oral appliances, in order to achieve a better outcome and higher compliance rates. Inflatable–deflatable pillows, for example, could be used to optimize head position and ameliorate sleep apnea [27].
One limitation of our study is some subjectivity in visual position assessment of the overnight video recording. It was sometimes difficult to determine if head position was greater than or less than 45° from the vertical plane. Also it was sometimes difficult to determine head position if the head was covered by bedsheets. Such data had to be excluded from analysis.
In conclusion, our results demonstrate an important influence of head position, independently of trunk position, on the AHI in patients with OSA. We show that, in general, rotation of the head from the supine to lateral position causes a significant lowering of the AHI that, in some patients, is sufficient to eliminate OSA. This raises the possibility that manipulation of head position during sleep could be used in the management of OSA in selected patients. Further research will be required to shed light on mechanisms whereby head position influences severity of OSA.
Abbreviations
- AHI:
-
Apnea-hypopnea index
- CPAP:
-
Continuous positive airway pressure
- OSA:
-
Obstructive sleep apnea
- OSAsup:
-
Supine-predominant obstructive sleep apnea
- PSG:
-
Polysomnography
- RIP:
-
Respiratory impedance plethysmography
- REM:
-
Rapid eye movement
- SaO2 :
-
Arterial oxyhemoglobin saturation
- TSHS:
-
Trunk supine–head supine
- TSHL:
-
Trunk supine–head left
- TSHR:
-
Trunk supine–head right
- TLHL:
-
Trunk left–head left
- TRHR:
-
Trunk right–head right
- TLHS:
-
Trunk left–head supine
- TRHS:
-
Trunk right–head supine
References
Kimoff RJ, Cheong TH, Olha AE, Charbonneau M, Levy RD, Cosio MG, Gottfried SB (1994) Mechanisms of apnea termination in obstructive sleep apnea. Role of chemoreceptor and mechanoreceptor stimuli. Am J Respir Crit Care Med 149(3 Pt 1):707–714. doi:10.1164/ajrccm.149.3.8118640
Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S (1993) The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med 328(17):1230–1235. doi:10.1056/nejm199304293281704
Young T, Peppard PE, Gottlieb DJ (2002) Epidemiology of obstructive sleep apnea: a population health perspective. Am J Respir Crit Care Med 165(9):1217–1239
Young T, Blustein J, Finn L, Palta M (1997) Sleep-disordered breathing and motor vehicle accidents in a population-based sample of employed adults. Sleep 20(8):608–613
Kohli P, Balachandran JS, Malhotra A (2011) Obstructive sleep apnea and the risk for cardiovascular disease. Current atherosclerosis reports 13(2):138–146. doi:10.1007/s11883-011-0161-8
Kim HC, Young T, Matthews CG, Weber SM, Woodward AR, Palta M (1997) Sleep-disordered breathing and neuropsychological deficits. A population-based study. Am J Respir Crit Care Med 156(6):1813–1819. doi:10.1164/ajrccm.156.6.9610026
Cartwright RD (1984) Effect of sleep position on sleep apnea severity. Sleep 7(2):110–114
Ravesloot MJ, van Maanen JP, Dun L, de Vries N (2013) The undervalued potential of positional therapy in position-dependent snoring and obstructive sleep apnea—a review of the literature. Sleep & breathing = Schlaf & Atmung 17(1):39–49. doi:10.1007/s11325-012-0683-5
Oksenberg A, Silverberg DS, Arons E, Radwan H (1997) Positional vs nonpositional obstructive sleep apnea patients: anthropomorphic, nocturnal polysomnographic, and multiple sleep latency test data. Chest 112(3):629–639
Richard W, Kox D, den Herder C, Laman M, van Tinteren H, de Vries N (2006) The role of sleep position in obstructive sleep apnea syndrome. Eur Arch Otorhinolaryngol 263(10):946–950. doi:10.1007/s00405-006-0090-2
Oksenberg A, Khamaysi I, Silverberg DS, Tarasiuk A (2000) Association of body position with severity of apneic events in patients with severe nonpositional obstructive sleep apnea. Chest 118(4):1018–1024
Shetty M, Mador MJ (2015) Prevalence of Positional Obstructive Sleep Apnea in Patients Undergoing Polysomnography and the Effect of Sleep Stage. In: de Vries N, Ravesloot M, van Maanen JP (eds) Positional Therapy in Obstructive Sleep Apnea. Springer International Publishing, pp 53–64. doi:10.1007/978-3-319-09626-1_4
EEG arousals: scoring rules and examples: a preliminary report from the Sleep Disorders Atlas Task Force of the American Sleep Disorders Association (1992). Sleep 15 (2):173–184
Rechtschaffen A, Kales A (1968) A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects. UCLA Brain Information Service/Brain Research Institute, Los Angeles
Alshaer H, Fernie GR, Tseng WH, Bradley TD (2016) Comparison of in-laboratory and home diagnosis of sleep apnea using a cordless portable acoustic device. Sleep Med 22:91–96. doi:10.1016/j.sleep.2015.11.003
Safiruddin F, Koutsourelakis I, de Vries N (2014) Analysis of the influence of head rotation during drug-induced sleep endoscopy in obstructive sleep apnea. Laryngoscope 124(9):2195–2199. doi:10.1002/lary.24598
van Kesteren ER, van Maanen JP, Hilgevoord AA, Laman DM, de Vries N (2011) Quantitative effects of trunk and head position on the apnea hypopnea index in obstructive sleep apnea. Sleep 34(8):1075–1081. doi:10.5665/SLEEP.1164
Cartwright RD, Diaz F, Lloyd S (1991) The effects of sleep posture and sleep stage on apnea frequency. Sleep 14(4):351–353
Findley LJ, Wilhoit SC, Suratt PM (1985) Apnea duration and hypoxemia during REM sleep in patients with obstructive sleep apnea. Chest 87(4):432–436
Peregrim I, Gresova S, Pallayova M, Fulton BL, Stimmelova J, Bacova I, Mikulakova A, Tomori Z, Donic V (2013) Does obstructive sleep apnea worsen during REM sleep? Physiol Res 62(5):569–575
Patil SP, Schneider H, Schwartz AR, Smith PL (2007) Adult obstructive sleep apnea: pathophysiology and diagnosis. Chest 132(1):325–337. doi:10.1378/chest.07-0040
Fleetham JA (1992) Upper airway imaging in relation to obstructive sleep apnea. Clin Chest Med 13(3):399–416
Isono S, Tanaka A, Nishino T (2002) Lateral position decreases collapsibility of the passive pharynx in patients with obstructive sleep apnea. Anesthesiology 97(4):780–785
Walsh JH, Leigh MS, Paduch A, Maddison KJ, Armstrong JJ, Sampson DD, Hillman DR, Eastwood PR (2008) Effect of body posture on pharyngeal shape and size in adults with and without obstructive sleep apnea. Sleep 31(11):1543–1549
Brijbassi M, Kasai T, Montemurro LT, Bradley TD (2014) Effect of an Anti-Supine Shirt for Treatment of Supine-related Obstructive Sleep Apnea. Journal of Sleep Disorders & Therapy 2014
Ono T, Otsuka R, Kuroda T, Honda E, Sasaki T (2000) Effects of head and body position on two- and three-dimensional configurations of the upper airway. J Dent Res 79(11):1879–1884
Ishizaka S, Moromugi S, Kobayashi M, Kajihara H, Koga K, Sugahara H, Ishimatsu T, Kurata S, Kirkness JP, Oi K (2014) A remote-controlled airbag device can improve upper airway collapsibility by producing head elevation with jaw closure in normal subjects under Propofol anesthesia. IEEE journal of translational engineering in health and medicine 2:1–9
Acknowledgements
We would like to thank the technicians at the Toronto Rehab sleep laboratory for their support during data collection period; in particular, Fiona Rankin for her help in the interpretation of polysomnography data, and Wen-Hou Tseng for his aid in recruiting subjects for the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This project has been supported partially by these grants: MaRS Innovation has provided financial support, Ontario Centre of Excellence has provided financial support, Johnson and Johnson Inc. has provided financial support, and Ontario Brain Institute via FedDev has provided financial support. Dr. Alshaer received the NSERC PhD scholarship. Dr. Bradley is supported by the Clifford Nordal Chair in Sleep Apnea and Rehabilitation Research. Maryam Patel received the Youth Employment Grant from the NRC (National Research Council of Canada) Industrial Research Assistance program.
Conflicts of interest
H. Alshaer, T.D. Bradley, M Patel have received grant funding from the agencies listed in the Funding section above. The results of this study are not influenced by any of the aforementioned grants as the granting agencies had no role in the design or conduct of this research nor had a bias towards a certain outcome.
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Comment
Obstructive sleep apnea continues to be a significant problem affecting approximately 10% of the adult population in developed countries. Multiple therapeutic options exist today for treatment. Some of the least invasive include devices which maintain a lateral sleeping position at night. This article provides a new concept for patients who prefer to sleep supine - movement of the head alone to a lateral position.. While the study is small and should be reproduced in a larger cohort, the data supports that some patients with OSA may see a substantial benefit when their head is turned to a lateral position.
Robin Germany
Minnesota, USA
Rights and permissions
About this article
Cite this article
Zhu, K., Bradley, T., Patel, M. et al. Influence of head position on obstructive sleep apnea severity. Sleep Breath 21, 821–828 (2017). https://doi.org/10.1007/s11325-017-1525-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-017-1525-2