Abstract
Purpose
The objective of this study was to evaluate endothelial function and carotid intima media thickness (CIMT) in moderate to severe obstructive sleep apnea (OSA) without comorbidities.
Methods
It is an observational case control study in which endothelial function was assessed using flow-mediated dilatation (FMD) and peripheral arterial tonometry (PAT), and carotid artery ultrasound was used to measure CIMT in study group subjects that included 20 normotensive, non-diabetic, treatment naïve, and moderate to severe OSA patients, and 20 normotensive, non-diabetic, and non-OSA subjects served as a control group. Study was conducted in Polysomnography Laboratory, Department of Internal Medicine, All India Institute of Medical Sciences (AIIMS) Hospital, New Delhi.
Results
FMD was significantly lower in the moderate to severe OSA group compared to non-OSA group (mean ± SD, 8.3 ± 2.8 vs. 13.4 ± 4.1 %; p = 0.0001). Reactive hyperemia index (RHI) was also significantly lower in the OSA group (1.55 ± 0.27 vs. 2.01 ± 0.48, p = 0.0007). CIMT was observed to be significantly higher in the OSA group compared to the non-OSA group (0.54 ± 0.09 vs. 0.48 ± 0.08 mm; p = 0.049). In the OSA group, FMD, RHI, and CIMT did not show a significant correlation with OSA disease severity indices [apnea hypopnea index (AHI), oxygen desaturation index (ODI), and minimum O2 saturation].
Conclusion
Endothelial function in macrovascular and microvascular circulation is significantly impaired in moderate to severe OSA patients without comorbidities. These patients also show evidence of subclinical atherosclerosis, in the form of increased CIMT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With increasing urbanization worldwide, particularly in developing countries where a large proportion of the world’s population resides, a new set of diseases are emerging. Obstructive sleep apnea (OSA) is one such disease. The incontrovertible data linking OSA to cardiovascular and cerebrovascular morbidity and mortality [1–3] has further put the spotlight on this condition in recent times. It has been hypothesized that impaired endothelial function may be an important mediator responsible for the increase in adverse cardiovascular and cerebrovascular consequences in OSA patients [1, 4]. Chronic intermittent hypoxia and sleep fragmentation are the two most important mechanisms which contribute to impaired endothelial function in OSA.
Endothelial function may be assessed in peripheral arteries (macrovascular) or in arterioles, capillaries, and venules (microvascular). Flow-mediated dilatation (FMD) is a useful non-invasive method for the assessment of macrovascular endothelial function [5]. Several studies in OSA patients have investigated endothelial function using FMD of brachial artery [6, 7]. A few studies have also attempted to evaluate microvascular endothelial function in OSA patients. These studies have utilized techniques such as venous occlusive plethysmography [8], laser Doppler flowmetry [9], and more recently, peripheral arterial tonometry [10]. Assessment of endothelial function in OSA patients allows us to address the larger problem of increased cardiovascular and cerebrovascular morbidity in this population at an incipient stage.
Carotid intima media thickness (CIMT) is a useful marker of subclinical atherosclerosis. Assessment of CIMT using carotid artery ultrasound offers a non-invasive method to detect atherosclerosis in its inchoative stages [11]. Previous studies have demonstrated an increased CIMT in OSA patients [12]. Additionally, an improvement in CIMT has been observed with continuous positive airway pressure (CPAP) therapy [13].
In the present study, we investigated endothelial function and CIMT in moderate to severe OSA patients without comorbidities.
Materials and methods
Study population
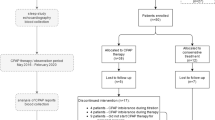
The present study was conducted in the Department of Internal Medicine at AIIMS Hospital, New Delhi, which is a tertiary care referral hospital located in North India. It was conducted between November 2013 and March 2015. Participants included subjects of either gender, older than 18 years of age. Study group subjects were selected from suspected OSA patients, who were initially evaluated at the Sleep Clinic of Department of Internal Medicine, AIIMS Hospital, New Delhi. The study group included 20 subjects with an apnea hypopnea index (AHI) ≥15 events/h and excessive daytime sleepiness (Epworth sleepiness scale (ESS) score >10), who were treatment naïve. Subjects with diabetes mellitus (FBS ≥126 mg/dL and/or HbA1c >6.5 %), hypertension (mean of three BP measurements taken at 1-min intervals >140/90 mmHg or those using antihypertensive medication), dyslipidemia (total serum cholesterol >200 mg/dL and/or low-density lipoprotein (LDL) cholesterol >130 mg/dL by immunocolorimetric assay), hypothyroidism (serum TSH >5.50 mIU/L by chemiluminescence), proteinuria [urine albumin creatinine ratio (ACR) >300 mg/g of creatinine], and smokers were excluded from the study. Control group subjects were drawn from hospital staff and relatives of patients admitted to the hospital. The control group consisted of 20 non-OSA subjects, with an AHI <5 events/h who met inclusion and exclusion criteria.
The study protocol was approved by the Institutional Ethics Committee, All India Institute of Medical Sciences, New Delhi. Written informed consent was obtained from all participants of the study.
Polysomnography
Prior to polysomnography (PSG), all subjects underwent a detailed anthropometry including a body composition analysis (TANITA TBF-410, TANITA Corp., Tokyo, Japan). All study participants underwent an overnight in-laboratory supervised polysomnography (SOMNOscreen plus, SOMNOmedics, Randersacker, Germany) at the Polysomnography Laboratory of the Department of Medicine at AIIMS. The study was supervised by a trained staff nurse. Polysomnography included electroencephalography, electrooculography, electrocardiography, surface electrodes for submental and tibialis anterior electromyography, pulse oximetry, pressure transducers for nasal and oral air flow, and piezoelectric strain gages for chest and abdominal movements [14]. Manual scoring of the sleep study was done the following morning by trained personnel using the scoring criteria of the American Academy of Sleep Medicine [15]. Apnea was scored in case of an air flow reduction of ≥90 % lasting for ≥10 s. Hypopnea was scored in case of an air flow reduction of ≥30 % lasting for ≥10 s and accompanied by either a ≥3 % arterial oxygen desaturation or an arousal. The AHI was determined as the total number of apneas and hypopneas per hour of sleep. The oxygen desaturation index (ODI) was determined as the number of arterial oxygen desaturations ≥3 % per hour of sleep.
Endothelial function
Macrovascular endothelial function was assessed by FMD of the brachial artery, performed by an experienced radiologist. Flow-mediated dilatation was performed in the morning after at least 8 h of sleep. Brachial artery FMD was measured using a 9 MHz transducer (LOGIQ e, GE Healthcare, Milwaukee, USA) with the patient lying comfortably in the supine position, 1 cm above the antecubital fossa in the right arm. Scan sections were obtained in the antecubital fossa in the longitudinal plane [16]. A baseline diameter was recorded initially. Subsequently, the brachial artery was occluded by inflating the sphygmomanometer cuff to 50 mmHg above the systolic blood pressure for 5 min. The cuff was then deflated rapidly, and the brachial artery diameter was recorded 1 min after the deflation of the cuff [17]. Brachial artery diameter, both pre- and post-cuff occlusion, was recorded at the peak systolic phase of cardiac cycle. FMD was calculated and expressed as the percentage change in the brachial artery diameter.
Microvascular endothelial function was assessed by using EndoPAT™ 2000 (Itamar Medical Ltd, Caesarea, Israel). Pneumatic probes were placed over the index fingers of both hands. One arm acted as the test arm and the other arm acted as the control arm. The test was conducted in three phases. In the initial phase, pulse wave tracings were obtained from both arms for 5 min. In the second phase, the sphygmomanometer cuff around the test arm was inflated to 50 mmHg above the systolic pressure for 5 min. In the last phase, pulse wave tracings were obtained from both the arms after rapid deflation of the sphygmomanometer cuff. Reactive hyperemia index (RHI) was calculated by the Endo-PAT™2000 software as the ratio of the post- to pre-occlusion peripheral arterial tonometry (PAT) amplitude of the test arm divided by the post- to pre-occlusion PAT amplitude of the control arm.
CIMT was measured by an experienced radiologist using a 7 MHz transducer (LOGIQ e, GE Healthcare, Milwaukee, USA), in the far wall of the common carotid artery just proximal to the carotid bulb [18]. Three readings each were taken from the right and left sides. Average of the six readings was taken as the mean CIMT. A carotid plaque was defined as a localized thickening >1.2 mm that did not uniformly involve the whole artery [19].
Statistical analysis
Data were expressed as mean ± SD in case of normally distributed data and median (interquartile range) in case of skewed data. Continuous variables were compared between the two groups using Student’s t test for independent samples in case of normal distribution. The Wilcoxon rank test was used to compare continuous variables in case of skewed data. The chi-square test or Fisher exact test was used to compare categorical data. Correlation between variables was expressed in terms of Spearman’s rank correlation coefficient. The comparison of endothelial function and CIMT between moderate OSA, severe OSA, and non-OSA groups was performed using one-way ANOVA followed by pairwise comparison using the Bonferroni correction. The p values <0.05 were considered statistically significant. Statistical analysis was performed using Stata 11.0 (College Station, TX, USA).
Results
A total of 468 subjects underwent polysomnography between November 2013 and March 2015. Three hundred and sixty two subjects were excluded because of associated comorbidities. Fifty-four subjects with mild OSA (AHI ≥5, but <15 events/h) were excluded. Twelve subjects declined to participate in the study. Twenty moderate to severe OSA subjects (AHI ≥15 events/h) constituted the study group. The study group was comprised of 10 subjects with moderate OSA (AHI ≥15, but <30 events/h) and 10 subjects with severe OSA (AHI >30 events/h). Twenty non-OSA subjects (AHI <5 events/h) constituted the control group. The baseline demographic, clinical, and peripheral blood biochemistry data are presented in Table 1. The two groups did not differ with regard to age, gender, body mass index (BMI), waist/hip ratio (WHR), total and LDL cholesterol, triglyceride, and fasting plasma glucose levels.
The polysomnographic data of two groups are presented in Table 2. AHI (p = 0.0001), ODI (p = 0.0001), and minimum O2 saturation (p = 0.0001) were significantly different in the two groups.
The baseline brachial artery diameter was not significantly different between the two groups (p = 0.15). FMD (p = 0.0001) and RHI (p = 0.0007) were significantly lower in the OSA group compared to non-OSA group (Table 3). In the OSA group, FMD and RHI did not show a significant correlation with OSA disease severity indices (AHI, ODI, and minimum O2 saturation). No correlation was observed between FMD and RHI (non-OSA group: Spearman’s rho = 0.05, p value = 0.82; OSA group: Spearman’s rho = −0.35, p value = 0.12).
Carotid intima media thickness (CIMT) was significantly higher in OSA group compared to non-OSA group, p = 0.049 (Table 3). None of the study participants demonstrated carotid plaques. No significant correlation was observed between CIMT and OSA disease severity indices (AHI, ODI, and minimum O2 saturation) in the OSA group.
Endothelial function and CIMT were also compared between severe OSA subjects (AHI >30 events/h), moderate OSA subjects (AHI ≥15, but <30 events/h), and non-OSA controls (AHI <5 events/h). FMD and RHI were significantly lower in both moderate and severe OSA groups compared to non-OSA controls (Figs. 1 and 2). However, CIMT did not differ significantly between the three groups (Table 4).
Discussion
Obstructive sleep apnea (OSA) is an increasingly prevalent condition, particularly in the developing world [20]. Several studies have demonstrated an association of OSA with cardiovascular and cerebrovascular morbidity [2]. Impairment of endothelial function represents one of the earliest steps in the development of vascular disease. The mechanisms underlying impairment of endothelial function in OSA include reduced nitric oxide (NO) availability, endothelial inflammation, and endothelial oxidative stress [21]. CIMT is an established marker of subclinical atherosclerosis [22]. The assessment of endothelial function and CIMT may best demonstrate the risk of future vascular disease in these patients. However, assessment of endothelial function and CIMT in OSA is frequently confounded by associated comorbidities. Present study evaluated endothelial function and CIMT in moderate to severe OSA subjects and non-OSA controls, after careful exclusion of diabetes mellitus, hypertension, dyslipidemia, hypothyroidism, and proteinuria.
Findings of the present study have demonstrated that endothelial function is significantly impaired in moderate to severe OSA patients without comorbidities. The impairment of endothelial function is observed in both macrovascular and microvascular beds. In a previous study, Bayram and colleagues [23] evaluated endothelial function in normotensive OSA patients. They demonstrated a significantly lower FMD in these patients. Although they excluded most cardiovascular risk factors, however, a significant number of study participants in their study were smokers. Smoking has been shown previously, to independently contribute to impaired endothelial function [24]. In contrast, all the participants of our study were non-smokers. They also observed an inverse correlation between FMD and AHI. An inverse correlation between FMD and AHI has been demonstrated in other studies as well [6, 25]. It is interesting to note that these studies pooled OSA and non-OSA subjects together while performing correlation analysis. In the present study, we observed a statistically significant correlation between FMD and OSA disease severity indices (AHI, ODI, and minimum O2 saturation) when we pooled OSA and non-OSA subjects together. However, a statistically significant correlation was not observed when we analyzed the OSA and non-OSA groups separately. We believe that correlation analysis should be performed separately in cases and controls, since combining the two may confound results. In a study by Chung and co-workers [25], it was observed that FMD was significantly lower in severe OSA patients compared to non-OSA controls. They however did not observe a significant difference in FMD between mild to moderate OSA and non-OSA subjects. We excluded mild OSA patients from our study. We observed a significantly lower FMD in both moderate OSA patients and severe OSA patients compared to non-OSA subjects. In a study from the UK [9], Butt and co-workers studied endothelial function in both macrovascular and microvascular beds in moderate to severe OSA patients. These investigators used FMD and laser Doppler flowmetry (LDF) to evaluate endothelial function. They demonstrated lower FMD as well as cutaneous perfusion response in moderate to severe OSA subjects. In the present study, we demonstrated similar results. However, we used a novel technology (PAT) to assess microvascular endothelial function. Peripheral arterial tonometry is an operator-independent method of endothelial function assessment and may be performed by trained nurses. Only a few studies have used PAT to assess microvascular endothelial function in OSA [10, 26]. In a study by Itzhaki and co-workers [10], RHI was observed to be significantly lower in subjects with moderate to severe OSA. The study subjects, however, were not free from cardiovascular risk factors such as hypertension, diabetes, or smoking.
Our study did not find a correlation between FMD and RHI. This lack of correlation has been demonstrated in previous studies of endothelial function in non-OSA population [27, 28]. It may be possible that different mechanisms of endothelial dysfunction may be acting in different vascular beds.
Present study has shown that CIMT is significantly higher in moderate to severe OSA patients. This finding suggests a greater risk of future clinical atherosclerotic disease in moderate to severe OSA patients. Similar findings were observed in a study by Tanriverdi and colleagues [29], who showed a significantly higher CIMT in moderate to severe OSA patients compared to non-OSA controls. In contrast to this study, none of the present study participants demonstrated carotid plaques. This difference may be attributed to the younger age of our study participants. Our study also confirms the findings of Drager and co-workers [30] who demonstrated a higher CIMT in severe OSA patients compared to mild to moderate OSA patients and non-OSA controls.
The present study has several unique features. This is the first study of endothelial function in OSA from the Indian subcontinent. To our knowledge, this is also the first study of endothelial function in OSA using both flow-mediated dilatation (FMD) and peripheral arterial tonometry (PAT) in the same cohort. The mean age of subjects in our cohort was nearly a decade lower compared to previous studies on endothelial function and CIMT in OSA [25, 29, 30]. Most major cardiovascular risk factors were carefully excluded in cases and controls. Endothelial function was evaluated in both macrovascular and microvascular beds in the present study. In addition, we used a novel non-invasive technology, peripheral arterial tonometry, which is relatively easier to perform and is operator independent.
Limitations of the present study include a small cohort. However, it is a unique cohort, in which diabetes mellitus, hypertension, dyslipidemia, hypothyroidism, proteinuria, and smoking were carefully excluded. A large number of patients (n = 468) had to be screened to exclude these risk factors in cases and controls. Second, we did not study endothelium-independent vasodilatation in our study. Third, it is a cross-sectional study and the impact of CPAP on endothelial function and CIMT was not evaluated. Fourth, the systolic blood pressure was significantly different between the OSA and non-OSA groups. However, none of the participants was hypertensive or being treated for hypertension. Whether this difference might have impacted the results remains debatable. Although the two groups were similar with regard to BMI and fat mass, they differed with regard to fat-free mass. Since fat-free mass is an important determinant of resting metabolism, this may have had an impact on the results. Lastly, given the significant prevalence of smoking among Indians, the confirmation of non-smoking status in the study participants using an objective technique such as breath analysis for carbon monoxide (CO) might have been more appropriate.
In conclusion, findings of the present study demonstrate an impaired endothelial function and CIMT in moderate to severe OSA patients. Impairment of endothelial function is observed in both macrovascular and microvascular beds. Given the increased incidence of adverse vascular events in OSA patients [1–3], our findings assume clinical significance. Non-invasive tests in the form of FMD, PAT, and CIMT may allow us to predict the risk of future vascular events in these patients.
References
Shamsuzzaman AM, Gersh BJ, Somers VK (2003) Obstructive sleep apnea: implications for cardiac and vascular disease. JAMA 290(14):1906–1914
Palomäki H (1991) Snoring and the risk of ischemic brain infarction. Stroke J Cereb Circ 22(8):1021–1025
Young T, Finn L, Peppard PE, Szklo-Coxe M, Austin D, Nieto FJ et al (2008) Sleep disordered breathing and mortality: eighteen-year follow-up of the Wisconsin sleep cohort. Sleep 31(8):1071–1078
Kunz AB, Kraus J, Young P, Reuss R, Wipfler P, Oschmann P et al (2012) Biomarkers of inflammation and endothelial dysfunction in stroke with and without sleep apnea. Cerebrovasc Dis 33(5):453–460
Celermajer DS, Sorensen KE, Gooch VM, Spiegelhalter DJ, Miller OI, Sullivan ID et al (1992) Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet 340(8828):1111–1115
Ip MSM, Tse H-F, Lam B, Tsang KWT, Lam W-K (2003) Endothelial function in obstructive sleep apnea and response to treatment. Am J Respir Crit Care Med 169(3):348–353
Oflaz H, Cuhadaroglu C, Pamukcu B, Meric M, Ece T, Kasikcioglu E et al (2006) Endothelial function in patients with obstructive sleep apnea syndrome but without hypertension. Respiration 73(6):751–756
Kato M, Roberts-Thomson P, Phillips BG, Haynes WG, Winnicki M, Accurso V et al (2000) Impairment of endothelium-dependent vasodilation of resistance vessels in patients with obstructive sleep apnea. Circulation 102(21):2607–2610
Butt M, Khair OA, Dwivedi G, Shantsila A, Shantsila E, Lip GYH (2011) Myocardial perfusion by myocardial contrast echocardiography and endothelial dysfunction in obstructive sleep apnea. Hypertension 58(3):417–424
Itzhaki S, Lavie L, Pillar G, Tal G, Lavie P (2005) Endothelial dysfunction in obstructive sleep apnea measured by peripheral arterial tone response in the finger to reactive hyperemia. Sleep 28(5):594–600
Cobble M, Bale B (2010) Carotid intima-media thickness: knowledge and application to everyday practice. Postgrad Med 122(1):10–18
Minoguchi K, Yokoe T, Tazaki T, Minoguchi H, Tanaka A, Oda N et al (2005) Increased carotid intima-media thickness and serum inflammatory markers in obstructive sleep apnea. Am J Respir Crit Care Med 172(5):625–630
Agha MA, Habib RM (2014) Assessment of carotid artery wall in patients with OSA syndrome and the effect of CPAP on its thickness. Egypt J Chest Dis Tuberc 63(1):155–160
Sharma SK, Nehra A, Sinha S, Soneja M, Sunesh K, Sreenivas V, et al. (2015) Sleep disorders in pregnancy and their association with pregnancy outcomes: a prospective observational study. Sleep Breath Schlaf Atm;1–7
Berry RB, Budhiraja R, Gottlieb DJ, Gozal D, Iber C, Kapur VK et al (2012) Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med JCSM Off Publ Am Acad Sleep Med 8(5):597–619
Corretti MC, Anderson TJ, Benjamin EJ, Celermajer D, Charbonneau F, Creager MA et al (2002) Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the International Brachial Artery Reactivity Task Force. J Am Coll Cardiol 39(2):257–265
Uehata A, Lieberman EH, Gerhard MD, Anderson TJ, Ganz P, Polak JF et al (1997) Noninvasive assessment of endothelium-dependent flow-mediated dilation of the brachial artery. Vasc Med Lond Engl 2(2):87–92
Sidhu PS, Desai SR (1997) A simple and reproducible method for assessing intimal-medial thickness of the common carotid artery. Br J Radiol 70:85–89
Nicolaides AN, Shifrin EG, Bradbury A, Dhanjil S, Griffin M, Belcaro G et al (1996) Angiographic and duplex grading of internal carotid stenosis: can we overcome the confusion? J Endovasc Surg Off J Int Soc Endovasc Surg 3(2):158–165
Sharma SK, Kumpawat S, Banga A, Goel A (2006) Prevalence and risk factors of obstructive sleep apnea syndrome in a population of Delhi, India. Chest 130(1):149–156
Atkeson A, Jelic S (2008) Mechanisms of endothelial dysfunction in obstructive sleep apnea. Vasc Health Risk Manag 4(6):1327–1335
Mancini GBJ, Dahlöf B, Díez J (2004) Surrogate markers for cardiovascular disease structural markers. Circulation 109(25 suppl 1):IV – 22–IV – 30
Bayram NA, Ciftci B, Keles T, Durmaz T, Turhan S, Bozkurt E et al (2009) Endothelial function in normotensive men with obstructive sleep apnea before and 6 months after CPAP treatment. Sleep 32(10):1257–1263
Ozaki K, Hori T, Ishibashi T, Nishio M, Aizawa Y (2010) Effects of chronic cigarette smoking on endothelial function in young men. J Cardiol 56(3):307–313
Chung S, Yoon I-Y, Shin Y-K, Lee CH, Kim J-W, Lee T et al (2007) Endothelial dysfunction and C-reactive protein in relation with the severity of obstructive sleep apnea syndrome. Sleep 30(8):997–1001
Cereda CW, Tamisier R, Manconi M, Andreotti J, Frangi J, Pifferini V et al (2013) Endothelial dysfunction and arterial stiffness in ischemic stroke the role of sleep-disordered breathing. Stroke 44(4):1175–1178
Gori T, Di Stolfo G, Sicuro S, Dragoni S, Lisi M, Parker JD et al (2006) Correlation analysis between different parameters of conduit artery and microvascular vasodilation. Clin Hemorheol Microcirc 35(4):509–515
Hamburg NM, Palmisano J, Larson MG, Sullivan LM, Lehman BT, Vasan RS et al (2011) Relation of brachial and digital measures of vascular function in the community: the Framingham heart study. Hypertension 57(3):390–396
Tanriverdi H, Evrengul H, Kara CO, Kuru O, Tanriverdi S, Ozkurt S et al (2006) Aortic stiffness, flow-mediated dilatation and carotid intima-media thickness in obstructive sleep apnea. Respiration 6:741–750
Drager LF, Bortolotto LA, Lorenzi MC, Figueiredo AC, Krieger EM, Lorenzi-Filho G (2005) Early signs of atherosclerosis in obstructive sleep apnea. Am J Respir Crit Care Med 172(5):613–618
Acknowledgments
The authors acknowledge the study subjects, highly trained staff nurses (Mr. Sreejith, Mr. Shashikant, Mr. Mohan Babu, Ms. Tsering, and Ms. Dhanya), medical social worker, Mr Ajeet, laboratory technician, Mr. Rohit Rai, and laboratory attendant, Mr. Dharmender Sharma, for their immense help in the recruitment, conduct, and completion of this study. The authors are thankful to AIIMS administration for the provision of EndoPAT™ probes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Support statement
Professor Surendra Kumar Sharma was supported by a J.C. Bose National Fellowship (SB/S2/JCB-04/2013), from the Ministry of Science and Technology, Government of India.
Funding
No funding was received for this research.
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This study was approved by the ethics committee of All India Institute of Medical Sciences (AIIMS). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Farooqui, F.A., Sharma, S.K., Kumar, A. et al. Endothelial function and carotid intima media thickness in obstructive sleep apnea without comorbidity. Sleep Breath 21, 69–76 (2017). https://doi.org/10.1007/s11325-016-1371-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-016-1371-7