Abstract
Introduction
Obstructive sleep apnea (OSA) is influenced by sleep architecture with rapid eye movement (REM) sleep having the most adverse influence, especially in women. There is little data defining the influence of slow-wave sleep (SWS) on OSA. We wished to study the influence of SWS on OSA and identify differences attributable to gender and/or age, if any.
Methods
Retrospective study of polysomnography (PSG) records of adult patients referred for diagnostic PSG. Records were excluded if they underwent split night or positive airway pressure titration studies, had <180 min of total sleep time (TST) and/or <40% sleep efficiency, or had SWS <5 min and/or <1% of TST. The apnea–hypopnea index (AHI) recorded during SWS was compared with that measured during other non-rapid eye movement (NREM) sleep and during REM sleep. The REM–SWS difference in AHI was measured, and compared between genders.
Results
Records from 239 patients were included. The mean AHI in all subjects was 17.7 ± 22.6. The SWS AHI was 6.8 ± 18.9, compared to the REM AHI of 24.9 ± 25.8, and NREM AHI of 15.8 ± 22.8. Females had significantly higher SWS by percentage, and lower NREM AHI (P < 0.0001) and SWS AHI (P = 0.03). Among patients with OSA (AHI ≥5), the difference between REM AHI and SWS AHI was greater in women than in men (34.2 ± 27.4 vs. 21.6 ± 26.0, P = 0.006).
Conclusions
The upper airway appears to be less susceptible to OSA during SWS than during REM and other NREM sleep. This may be related to phase-specific influences on both dynamic upper airway control as well as loop gain. Gender and age appear to modify this effect.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnea (OSA) is characterized by recurrent episodes of interruption of airflow during sleep due to partial or complete obstruction of the upper airways, often with concomitant oxygen desaturation. Relief of the apneas is associated with arousals or awakenings, leads to sleep fragmentation and prevents deep non-rapid eye movement (NREM) sleep (slow-wave sleep, SWS) [1, 2]. These qualitative and quantitative sleep abnormalities are responsible for daytime hypersomnolence in patients with OSA. Most obstructive apnea patients have an anatomically small upper airway with augmented pharyngeal dilator muscle activation maintaining airway patency awake, but not while asleep. However, OSA develops as the result of a variety of physiologic characteristics. These include: (1) upper airway anatomy, (2) the ability of upper airway dilator muscles to respond to rising intrapharyngeal negative pressure and increasing CO2 during sleep, (3) arousal threshold in response to respiratory stimulation, and (4) loop gain (ventilatory control instability) [3].
The occurrence of apneic events depends on the sleep stage, apneas being more commonly observed in stages I and II and in rapid eye movement (REM) sleep and less commonly in SWS [4, 5]. REM sleep seems to have the most adverse influence, especially in women. However, there is little data defining the influence of SWS on OSA.
In our study, we aimed to investigate the influence of SWS on OSA and identify gender differences if any. Because sleep-related breathing is known to worsen in females peri-and post-menopausally due to changes in estrogen and progesterone [6, 7], we also aimed to identify any influences of age on breathing during SWS.
Methods
Study design and participants
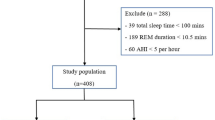
We retrospectively reviewed polysomnography (PSG) records of consecutive adult patients (>18 years) who were referred for diagnostic PSG between June 15, 2006 and July 22, 2007. Subjects were excluded who underwent a split night study, CPAP titration study or had less than 3 h of total sleep time (TST). We also excluded subjects with sleep efficiency <40%, or SWS <5 min or <1% of TST.
Polysomnographic evaluation
Overnight comprehensive PSG was performed in an accredited sleep laboratory (the Methodist Hospital in Houston, TX, USA), with multichannel recordings monitoring electroencephalogram, electrocardiogram, electrooculograms, submentalis electromyogram, airflow, respiratory effort, oxygen saturation, and anterior tibialis electromyogram. Data were scored manually by a single-blinded technologist in the context of routine clinical care, according to published guidelines [8, 9]. An obstructive apnea or hypopnea was scored when there was either a 50% reduction in oral–nasal airflow or a discernable reduction in flow associated with an arousal or oxyhemoglobin desaturation >3% for 10 s or longer; a single definition of apnea/hypopnea was used throughout each individual study. Obstructive sleep apnea was defined as an apnea–hypopnea index (AHI) ≥5 events/hour.
Statistics
Comparisons between the means of two normally distributed groups were performed with the unpaired t test. Comparisons between two non-normally distributed groups were done with the Mann–Whitney U test. Comparisons of the means of three or more non-normally distributed groups were done with the Kruskal–Wallis one-way analysis of variance and Dunn’s multiple comparisons post-test. A p value of <0.05 was considered statistically significant.
Results
Study participants
A total 327 records were evaluated for inclusion into the study, of which 239 met criteria and were included in the final analysis. The demographic and clinical characteristics of the participants are shown in Table 1. Participants primarily identified themselves as “white” and other races were distributed evenly. There were no significant differences between males and females in age, body mass index, or Epworth Sleepiness score. Males had more frequent respiratory events during sleep than females, as measured by the overall AHI (P < 0.0001). In the overall population, 146 of the subjects (93 males, 53 females) had polysomnographic OSA, as defined by AHI ≥5 events/hour.
Sleep-related breathing during SWS
The severity of OSA was found to differ by stage of sleep, shown in Fig. 1. The overall AHI during sleep was 17.7 ± 22.6 events/hour. The AHI during REM sleep was 24.9 ± 25.8, during NREM sleep (other than SWS) was 15.8 ± 22.8, and during SWS was 6.8 ± 18.9. In subjects with OSA, the mean AHI during sleep was 27.7 ± 24.0. Events were more frequent during REM sleep (36.9 ± 26.2 events/hour) than NREM sleep (25.1 ± 25.2 events/hour), or SWS (10.7 ± 23.3 events/hour) in these subjects. Forty-seven subjects had obstructive respiratory events during SWS ≥5 per hour. These subjects had more severe OSA on average (AHI = 45.4 ± 30.2 events/hour), with the mean SWS AHI 14.8 events/hour lower than the REM AHI (REM–SWS difference). This finding is similar to the overall group (REM–SWS difference = 18.2), but lower than seen in those with OSA (REM–SWS difference = 26.2).
Sleep-related respiratory events are attenuated during slow-wave sleep. Box plots (±1.5 times interquartile distance) are plotted for the apnea–hypopnea index (AHI) during rapid eye movement (REM) sleep, non-rapid eye movement sleep other than slow-wave sleep (NREM), and slow-wave sleep (SWS). Single asterisk indicates NREM AHI (16 ± 23 events/hour) significantly lower than REM AHI (25 ± 26 events/hour), P < 0.05. Double asterisk indicates SWS AHI (7 ± 19 events/hour) is significantly lower than the NREM AHI and REM AHI, P < 0.0001
Effects of gender on the attenuation of OSA during SWS
The polysomnographic sleep characteristics (sleep architecture and respiratory events) are shown in Table 2. The percent of sleep time in SWS was higher in females (17% vs. 14%, P = 0.02); the percent of time in REM sleep was similar in both groups. The SWS AHI in males was 10.1 ± 23.6 compared to 3.1 ± 11.0 in females (P = 0.03). The REM AHI was similar in males and females (27.2 ± 26.7 vs. 22.4 ± 24.6, P = 0.20), as was the REM–SWS difference (17.1 ± 23.8 vs. 19.3 ± 24.2, P = 0.49). The largest difference in respiratory events between gender was seen in the NREM AHI (22.1 ± 26.1 in males vs. 9.0 ± 16.2 in females, P < 0.0001), accounting for the majority of the difference in the overall mean AHI.
Gender effect on the attenuation of sleep-disordered breathing events during SWS is more appreciable in subjects with OSA (overall AHI ≥5 events/hour), shown in Fig. 2. The AHI during REM (P = 0.28) and SWS (P = 0.43) were similar between males and females. While both genders showed significantly lower AHI during SWS than REM sleep, the REM–SWS difference was significantly greater in females (34.2 ± 27.4) than in males (21.6 ± 26.0, P = 0.006).
The attenuation of sleep-disordered breathing events during slow-wave sleep is more pronounced in females. The mean + standard error of the mean of the apnea–hypopnea index (AHI) during rapid eye movement sleep (REM) and slow-wave sleep (SWS) and the difference between REM sleep and SWS (REM–SWS difference) is graphed for males (filled bars, N = 93) and females (open bars, N = 53). *P < 0.001
As shown in Fig. 3, there are notable differences in changes in breathing during sleep between males and females. Females demonstrate increasingly higher overall AHI and REM AHI with increasing age, whereas both values are high but stable over the age of 40 in males. SWS AHI remains low in females of all ages and, again, males ≥40 years have an elevated, stable SWS AHI. Because of these differences, male <50 years have larger REM–SWS difference than females of similar ages; this association is reversed in ages ≥50 years. The amount of SWS as a percent of TST (SWS%) remained relatively constant through all age groups. All female age groups, with means approximating 17%, were similar to each other and to the comparable male groups. Males <40 years of age had significantly higher mean SWS% (18.8 ± 6.5%) compared with males age 40–49 (12.2 ± 9.6%), 50–59 (14.6 ± 12.8%), and ≥60 (10.7 ± 7.0%) years.
Gender differences in the effects of age on breathing during deep sleep Top—The mean + standard error of the mean of the apnea–hypopnea index during the full night (overall, diagonal lines), rapid eye movement sleep (REM, solid black), and slow-wave sleep (SWS, dotted), as well as the difference between REM sleep and SWS (REM–SWS difference, solid white) in females (a, N = 114) and males (b, N = 125) by age group. Bottom—The amount of SWS, as a percent of the total sleep time (TST), is plotted by age group in females (filled circles, mean = solid line) and males (open circles, dashed line)
Discussion
Our results showed that OSA severity is lowest during SWS. This was the case in the majority of patients evaluated in this study. In each subset of patients studied (males, females, and those with OSA), the mean REM AHI and NREM AHI were both higher than the mean SWS AHI. We have demonstrated, for the first time to our knowledge, a possible influence of gender on the way in which respiration during sleep changes with changing sleep stages. The greatest decrease in AHI from REM sleep to SWS was in females with OSA. We observed a more complex interplay in terms of the influence of age. Older females consistently showed higher AHI and REM AHI than younger age groups, with low SWS AHI across age groups; on the other hand, males ≥40 years had a higher proportion of respiratory events occurring during SWS, though this proportion was stable across age groups.
Prior studies have also shown that sleep-disordered breathing is reduced during SWS. Most notably, a recent study found sharply reduced severity of OSA as NREM sleep progressed from stages 1 to 4, most pronounced in those with moderate or severe OSA [10]. Goh et al. showed that obstructive apneas were rare in SWS in children with OSA and that SWS appeared to be protective; only 5% of obstructive apneas occurred during SWS, even though SWS was more abundant than REM sleep [11]. The underlying mechanism was felt to be partly related to an increase in genioglossus muscle tone activity [12]. However, adult data demonstrated an exacerbation of sleep-disordered breathing when sleep quality improved with both REM and SWS rebound on the second night of sleep [13]. It is well known that sleep depth modifies the skeletal muscle tone including the upper airway muscles. Prior studies have suggested that collapsibility of the upper airways during light sleep and REM sleep is higher than during SWS even if skeletal muscle tone is lower during SWS [14, 15]. However, others have found no effect of sleep stage on upper airway collapsibility [16].
The mechanisms behind this change in breathing during SWS have been the subject of recent investigation. A relatively higher respiratory arousal threshold during SWS has been identified as a stabilizing influence for breathing during SWS [17, 18]. In a similar manner, eszopiclone, a benzodiazepine receptor agonist, was shown to increase the respiratory arousal threshold from sleep and decrease AHI in a group of patients with OSA and low arousal threshold [19]. Additional reports have suggested that ventilatory control is less stable in OSA and may have a substantial impact on apnea severity in certain patients with OSA [20]. The effect of ventilatory instability in producing upper airway obstruction is dependent on the underlying predisposition to pharyngeal collapse. If the fluctuation in ventilatory drive (loop gain) is elevated, breathing becomes unstable leading to cyclic upper airway obstruction. If the loop gain is low, as during SWS, stable breathing would then occur. Lastly, SWS is associated with numerous endocrine changes, such as release of growth hormone, renin, and prolactin, and inhibition of cortisol- and thyroid-stimulating hormone. In many cases, the effects of these hormones on upper airway resistance and sleep-disordered breathing are largely unknown.
Sex hormone regulation has been shown to play a role in breathing during sleep in females, as estrogen and progesterone are thought to protect against OSA, and estrogen replacement therapy is known to reduce sleep-disordered breathing in menopausal and post-menopausal women [6, 7]. The effects of these hormones on breathing during SWS are unclear: the SWS AHI remained relatively constant throughout all age groups in females, even though males showed an increase in SWS AHI with age; conversely, the REM AHI clearly showed an association with age in females, likely representing a delayed peak after menopause relative to males. Because the menstrual and menopausal statuses of our female participants are unknown, it is impossible to draw definitive conclusions about the impact of hormone regulation on the relationship between SWS and sleep-disordered breathing; this possibility certainly bears further study. Our overall population of women, with a mean age of 52 years, had a higher percentage of their sleep as SWS, compared with the population of men, consistent with earlier studies [21, 22]. Comparable age groups of males and females did not differ significantly with each other in SWS%. It is unclear if there is a causal relationship between a higher amount of SWS and attenuation of sleep-disordered breathing. Additionally, the differences in the severity of OSA between males and females keeps open the possibility that the effects seen are a reflection of disease severity rather than age and gender. This is an issue that will also impact future studies, given the relative protection female gender provides against OSA.
A recent study [23] showed that the prevalence of REM-related sleep-disordered breathing (REM AHI/NREM AHI ratio >2) was much higher in females (25%) than in males (8%). The findings from our data indirectly support this conclusion, as the difference between REM AHI and SWS AHI was higher in females (34 events per hour) than in males (22 events per hour). Additionally, the mean AHI during other (non-SWS) NREM sleep was lower in females (9 ± 16 events per hour) than in males (22 ± 26 events per hour), further supporting the idea that REM-related sleep-disordered breathing is comparatively more important in females. Our findings did not, however, support the prior conclusions that younger (<55 years) subjects had more REM-related sleep-disordered breathing than older subjects. In our population, the REM AHI was progressively higher in older age groups, whereas the SWS AHI remained fairly stable. Again, this effect was most pronounced in females. This discrepancy may be explained by the definition of REM-related sleep-disordered breathing. As opposed to an absolute cutoff (REM AHI/NREM AHI ratio >2), our analysis focused on trends with aging; while the cutoff may have clinical significance and allowed for prevalence analysis in the previous paper, we felt that analyzing the difference between REM AHI and SWS AHI allowed for better understanding of the relative contributions of each sleep stage in the overall severity of sleep-disordered breathing.
The high percent and stability of SWS% seen in our subjects may appear unusual. A report by Van Cauter et al. [24] showed that SWS% declines markedly from early adulthood to midlife, where it stabilized at a mean of 3.4% through later life. That study was done in healthy men while the current study, done in men and women referred for sleep studies, also showed stability in SWS% through midlife, albeit at a higher level (11–19%). This may be related to the presence of OSA in our population; with lower severity of disordered breathing, the SWS remains intact while the remainder of sleep becomes fragmented due to respiratory arousals and awakenings. However, Ratnavadivel et al. [10] showed the opposite effect: OSA patients demonstrated delayed and a reduced proportion of SWS compared with non-OSA patients (23.2% vs. 28.2%). The SWS% in our population falls between the values seen in these two other studies. Our findings of reduced SWS% with age in men and stability in women are consistent with the results from Ehlers and Kupfer [22], which showed that men in their 30s have a reduced SWS% and slow-wave activity compared with their 20s, but the same variables in women remain stable.
Our data showed a clear age and gender differences in breathing during SWS, especially when compared with breathing during REM sleep. When hypothesizing about causes of these effects, medications must be considered. Many medications are known to affect the quality and quantity of REM sleep; fewer are known to affect SWS. Frequency of prescription of some of these medications may differ, or the medication may demonstrate varying effects, among gender or age groups. Future investigations into sleep-disordered breathing during REM and SWS should consider medication use as a potential independent or confounding variable.
This population of patients was studied in a single sleep laboratory. This provides consistency in the application of scoring rules, but may impair the application of these results to the general population. The wide distribution of demographics, including age, gender, and ethnicity, improve generalizability. While there was a high prevalence of OSA in this population, that is to be expected at a referral sleep center, and identifies our population of interest. Identifying mechanisms that may lead to improvement in OSA in the most severely afflicted patients must be a priority.
Conclusions
SWS appears to render the upper airway less susceptible to occlusion, suggestive of a protective effect. This may be related to phase-specific influences on both dynamic upper airway control as well as loop gain. Gender may significantly influence this response. Increasing the SWS may theoretically improve OSA by increasing sleep time and improving sleep architecture.
References
Remmers JE, de Groot WJ, Sauerland EK, Anch AM (1978) Pathogenesis of upper airway occlusion during sleep. J Appl Physiol 44:931–938
Guilleminault C, Partinen M, Quera-Salva M, Hayes B, Dement WC, Nino-Murcia G (1988) Determinants of daytime sleepiness in obstructive sleep apnea. Chest 94:32–37
White DP (2005) Pathogenesis of obstructive and central sleep apnea. Am J Respir Crit Care Med 172:1363–1370
Krieger J, Kurtz D (1978) EEG changes before and after apnea. In: Guilleminault C, Dement WC (eds) Sleep apneas syndromes. Alan R. Liss, New York, pp 161–176
Guchu R, Findlay LJ, Woodson H, Fabrizio M, Suratt PM (1991) Upper airway stability is increased during slow wave sleep in obstructive sleep apnea [abstract]. Am Rev Respir Dis 143:A796
Pickett CK, Regensteiner JG, Woodard WD, Hagerman DD, Weil JV, Moore LG (1989) Progesterone and estrogen reduce sleep-disordered breathing in postmenopausal women. J Appl Physiol 66:1656–1661
Polo-Kantola P, Rauhala E, Helenius H, Erkkola R, Irjala K, Polo O (2003) Breathing during sleep in menopause: a randomized, controlled, crossover trial with estrogen therapy. Obstet Gynecol 102:68–75
Rechschaffen A, Kales A (1968) A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects. Washington Public Health Service, US Government Printing Office, Washington, DC
AASM Task Force (1999) Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. Sleep 22:667–689
Ratnavadivel R, Chau N, Stadler D, Yeo A, McEvoy RD, Catcheside PG (2009) Marked reduction in obstructive sleep apnea severity in slow wave sleep. J Clin Sleep Med 5:519–524
Goh DY, Galster P, Marcus CL (2000) Sleep architecture and respiratory disturbances in children with obstructive sleep apnea. Am J Respir Crit Care Med 162:682–686
Basner RC, Ringler J, Schwartzstein RM, Weinberger SE, Weiss JW (1991) Phasic electromyographic activity of the genioglossus increases in normals during slow-wave sleep. Respir Physiol 83:189–200
Le Bon O, Hoffmann G, Tecco J, Staner L, Noseda A, Pelc I, Linkowski P (2000) Mild to moderate sleep respiratory events: one negative night may not be enough. Chest 118:353–359
Boudewyns A, Van de Heyning PH, De Backer WA (1997) Site of upper airway obstruction in obstructive apnoea and influence of sleep stage. Eur Respir J 10:2566–2572
Horner RL (1996) Motor control of the pharyngeal musculature and implications for the pathogenesis of obstructive sleep apnea. Sleep 19:827–853
Penzel T, Möller M, Becker HF, Knaack L, Peter JH (2001) Effect of sleep position and sleep stage on the collapsibility of the upper airways in patients with sleep apnea. Sleep 24:90–95
Ratnavadivel R, Stadler D, Windler S, Bradley J, Paul D, McEvoy RD, Catcheside PG (2010) Upper airway function and arousability to ventilatory challenge in slow wave sleep versus stage 2 sleep in obstructive sleep apnoea. Thorax 65:107–112
Saboisky J, Eckert D, Malhotra A (2010) Stable breathing though deeper sleeping. Thorax 65:95–96
Eckert DJ, Owens RL, Kehlmann GB, Wellman A, Rahangdale S, Yim-Yeh S, White DP, Malhotra A (2011) Eszopiclone increases the respiratory arousal threshold and lowers the apnoea/hypopnea index in obstructive sleep apnea patients with a low arousal threshold. Clin Sci 120:505–514
Wellman A, Jordan AS, Malhotra A, Fogel RB, Katz ES, Schory K, Edwards JK, White DP (2004) Ventilatory control and airway anatomy in obstructive sleep apnea. Am J Respir Crit Care Med 170:1225–1232
Fukuda N, Honma H, Kohsaka M, Kobayashi R, Sakakibara S, Kohsaka S, Koyama T (1999) Gender differences of slow wave sleep in middle aged and elderly subjects. Psychiatry Clin Neurosci 53:151–153
Ehlers CL, Kupfer DJ (1997) Slow-wave sleep: do young adult men and women age differently? J Sleep Res 6:211–215
Koo BB, Dostal J, Ioachimescu O, Budur K (2008) The effects of gender and age on REM-related sleep-disordered breathing. Sleep Breath 12:259–264
Van Cauter E, Leproult R, Plat L (2000) Age-related changes in slow-wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA 284:861–868
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Subramanian, S., Hesselbacher, S., Mattewal, A. et al. Gender and age influence the effects of slow-wave sleep on respiration in patients with obstructive sleep apnea. Sleep Breath 17, 51–56 (2013). https://doi.org/10.1007/s11325-011-0644-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-011-0644-4