Abstract
Study objectives
This study aims to explore the association between osteoporosis and nightly sleep duration among adult outpatients.
Design
This is a large-scale, retrospective cross-sectional study.
Setting
The setting was in a preventive medicine health center at a general community hospital in Tokyo, Japan.
Participants
There were 19,321 healthy individuals (≥50 years old) who underwent annual general health checkup between January and December 2008.
Measurements
The participants were divided into four groups according to their self-reported average nightly sleep duration (<6, 6–6.9, 7–7.9, and >8 h). Radial bone mineral density was measured using dual-energy X-ray absorptiometry, and T-score was compared to young adult mean to diagnose osteoporosis. Multivariate logistic regression was used to explore the association between sleep duration and osteoporosis.
Results
The mean age of the participants was 60.9 years (standard deviation [SD], 7.9) and 48.0% were female. The prevalence of osteoporosis was 8.0% (95% confidence interval [CI] = 7.6–8.4%). Those with sleep duration of >8 h were more likely to have osteoporosis (odds ratio [OR] = 1.35; 95% CI = 1.06–1.73) than those with short sleep duration (<6 h).
Conclusion
Those individuals with self-reported sleep duration of more than 8 h (long sleepers) appear to have higher odds of osteoporosis compared to the progressively shorter sleepers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
An estimated 200 million people suffer from osteoporosis worldwide [1], with approximately 10 million patients, most frequently postmenopausal women, in Japan [2]. Due to Japan’s rapidly aging population, the prevalence of this disease is increasing [3] and has garnered more attention to research and prevention.
Bone mineral density (BMD) reaches a peak at approximately 40 years of age and then gradually decreases [4] with a sharper drop-off after menopause. Once BMD begins to decline, recovery is difficult. Osteoporosis may be associated with significant morbidity, with sequelae including not only fractures of the vertebrae and hip [5] but also decreases in patients’ activities of daily living (ADL) and quality of life (QOL) [6]. Although both pharmacological and non-pharmacological therapies are available, osteoporosis treatment is often prolonged, during which time the patients continue to be at risk [7]. Thus, the prevention of osteoporosis is important in terms of QOL and ADL as well as cost of medical care.
A previous study by Stone et al. reported an association between sleep duration, falls, and fractures in the elderly [8]. Although the results were not statistically significant, they found that those who slept longer than 10 h tended to have more falls (OR = 1.23; 95%, CI = 0.86–1.75) and fractures (OR = 1.43 95%CI = 0.95–2.15) than the 8–9-h sleepers. However, it remains unclear whether long sleep duration is an independent risk factor for low BMD and osteoporosis given that many other factors may also be related to fractures. We hypothesize that long sleep duration may be an independent risk factor for osteoporosis after adjustment for comorbidities, lifestyle, and physiologic factors, likely through multiple mechanisms involving weight loading and sleep-associated hormonal changes.
In this study, we explored the relationship between BMD, osteoporosis, and sleep duration using a large sample of Japanese outpatients. Recognition of any salient association may facilitate both early disease identification and osteoporosis prevention.
Methods
Study participants
All participants who underwent annual health checkup at the Center for Preventive Medicine at St. Luke’s International Hospital in Tokyo, Japan, from January through December 2008 were included in this study. Attracting a large number of apparently healthy individuals, the purpose of the health checkup program is to promote health through the early detection of chronic diseases and their risk factors. In Japan, the Industrial Safety and Health Law requires all workers and their families to undergo an annual, employer-sponsored health checkup. Around 80% of those who participate in our program are employees of various companies and local governmental organizations in Tokyo and their dependents; 20% are independently registered participants.
Data collection
Data were collected from adults (>20 years old) undergoing annual health checkup at our center in 2008. Patients aged 50 years and older were enrolled in our osteoporosis analysis according to previously published screening recommendations [9].
Two investigators independently extracted and recorded information using a structured data form. A consensus was reached by discussing any points of disagreement. St. Luke’s International Hospital Ethical Committee Institutional Review Board approved all aspects of this study. To preserve patient confidentiality, direct patient identifiers were not collected as part of the dataset.
Measurements
The annual checkup program consisted of self-reported demographic and lifestyle information (pre-exam questionnaire), medical history, initial evaluation (vital signs and laboratory data), information on comorbidity (diabetes mellitus, hypertension, dyslipidemia), past medical history (myocardial infarction, cerebral infarction), current medications, and treatments. Regular alcohol consumption was defined as drinking any amount of alcohol once or more per week.
In the questionnaire, the participants reported their average duration of sleep per night, which was classified into four categories (<6, 6–6.9, 7–7.9, and ≧8 h) based on a previous study [10]. The frequency of exercise per week was also classified into four categories (none per week, 1–2 days per week, 3–5 days per week, and almost daily).
Radial BMD was measured using dual-energy X-ray absorptiometry (DEXA) DCS-6EX-3 by ALOKA (Tokyo Japan), and the T-score was compared to the young adult mean (YAM). Diagnosis of osteoporosis was made if T-score was less than 70% of YAM per Japanese criteria [11] and is comparable to the World Health Organization (WHO) criteria of T-score = −2.5 SD [12]. The participants with a prior diagnosis of osteoporosis or those whose BMD T-score was less than 70% at baseline were excluded.
Statistical methods
All analyses were conducted using SPSS 15.0J statistical software (SPSS Japan, Tokyo, Japan). The responses were analyzed using descriptive statistics, including mean, variance, SD, and percents. The chi-square or Fisher’s exact tests were used for cross-tabulated data, and t-test was used to compare the means of continuous data. The 95% confidence intervals (95% CI) were calculated using normal approximation methods. We subsequently constructed multivariate logistic regression models to evaluate the adjusted association between osteoporosis and sleep duration, reported as adjusted odds ratios (ORs) with 95% CI.
Results
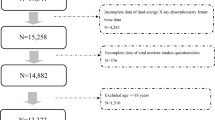
A total of 19,321 individuals (>50 years old) were included in this study. The mean age of the participants was 60.9 years old (SD 7.9), 48.0% of them were female, and the mean body mass index (BMI) was 22.6 kg/m2. Of the participants, 20.3% had hypertension, 6.5% diabetes, 14.6% dyslipidemia, 55.8% drank alcohol regularly, and 11.6% were current smokers. Other relevant characteristics are summarized in Table 1.
A total of 1,545 people (8.0%; 95% CI = 7.6–8.4%) were diagnosed with osteoporosis. There were 18.4% of participants who slept for less than 6 h per night while 12.3% slept for 8 h and more.
In total, 12,589 participants were included in the multivariate logistic regression as shown in Table 2. Surprisingly, no significant association regarding gender in relation to osteoporosis and sleep duration was found (P = 0.27). The multivariate logistic regression model indicated that the subjects who slept more than 8 h per night, on average, were significantly more likely to have osteoporosis compared to those sleeping less than 6 h (OR = 1.35; 95% CI = 1.06–1.73). Sleep duration of 6–6.9 and 7–7.9 h per night were similarly associated with progressively higher odds of osteoperosis, though these did not reach statistical significance. Moreover, while older age (>60 years old compared with 50–59 years old) was associated with osteoporosis (OR = 7.78; 95% CI = 6.44–9.34), as well as were women (OR = 8.78; 95%CI = 7.03–11.0), BMI was not (OR = 0.86; 95%CI = 0.84–0.89). Regular alcohol consumption was also negatively associated with osteoporosis (OR = 0.61; 95%CI = 0.52–0.72). Among comorbidities, hypertension was associated with osteoporosis (OR = 1.45; 95%CI = 1.22–1.72) while diabetes was not. People with healthier exercise habits tended to have osteoporosis less frequently. No association was found between history of myocardial infarction and osteoporosis.
Discussion
In our study, those with more than 8 h of sleep per night had osteoporosis more frequently compared to those with less than 6 h of sleep after adjusting for all other variables.
A previous study showed that longer sleep duration was associated with increased BMD in college women [13]. However, our study showed that middle-aged and elderly subjects, ostensibly a more relevant target population for discussions of osteoporosis, tended to have more osteoporosis with longer nightly sleep duration. This difference between populations in terms of the effect of sleep on osteoporosis may be age-related. For younger individuals, growth hormone is important for increasing BMD [4], with shorter sleep duration being associated with less growth hormone production [14]. Adequate sleep is vital for young people to maintain an appropriately high BMD.
In contrast, integrated growth hormone secretion has been demonstrated to be negatively associated with BMD in the elderly [15]. Once peak BMD is reached at approximately 40 years old, BMD gradually begins to decrease [4]. Therefore, substantial differences in endocrine physiology between age groups warrant a separate interpretation of the association between sleep duration and BMD.
We propose two hypotheses regarding how longer sleep durations may lead to osteoporosis. First, increased activity prevents osteoporosis, with previous studies reporting that exercise had beneficial effects on BMD [16, 17]. Specifically, mechanical load weight is an important factor in the prevention of osteoporosis [18]. Lengthened periods of sleep, by definition, shorten wake time and may effectively lessen daily mechanical loading, thus negatively affecting BMD. Although we adjusted for frequency of exercise using multivariate logistic regression, non-exercise activity time was not taken into consideration.
Second, short sleep duration may be associated with higher serum estrogen levels. Estrogen has been reported to be a significant, inversely associated factor for osteoporosis [19], even in men [20]. The physiology of bone loss in aging women and men has been largely explained by the effects of gonadal sex steroid deficiency [21]. In other words, maintaining estrogen levels is important for preventing osteoporosis. A previous study by Verkasalo et al. reported that exposure to light at night increased estrogen levels [22]; therefore, shortened sleep duration might raise estrogen levels through increased light-induced estrogen production.
Other results in our study were consistent with those in previous studies. For example, osteoporosis [23] was unsurprisingly associated with increased age, while increased exercise and weight were associated with reduced osteoporosis [17, 24]. Alcohol drinking was also related to greater BMD. While previous studies have shown that excessive alcohol consumption is related to osteoporosis [25], alternate data have demonstrated that alcohol intake of less than 75 g/week may beneficially increase BMD [26]. Although our data did not quantify alcohol consumption, it is reasonable to assume that the amount of alcohol consumed is an important factor in our findings.
There are some limitations in our study. First, only 12,589 of the 19,321 participants were included in the logistic regression analysis due to incomplete data. Second, we measured radial BMD using DEXA though WHO guidelines recommend BMD measurement at the spine or hip [27]. There may be differences between radial BMD and spine or hip BMD [28] which might influence these results. Third, all data were self-reported by participants at the time of annual checkup and therefore subject to memory bias. Verifying sleep duration through direct observation or a daily sleep diary may increase the reliability of the results, though direct observation risks a substantially smaller sample size. Fourth, this is a cross-sectional study; therefore, temporal relationships cannot be ascertained. Because most of our participants are employees of large companies in Tokyo, they may not be wholly representative of the Japanese general population in terms of lifestyle factors such as working duration, insolation, exercise habits, and tobacco use. The inclusion of dependents of employees and independent registries, however, mitigates this limitation. Finally, there are many confounders for low bone mineral density, including diet, absorption and metabolism, duration of smoking, and sleep quality and sleep pathology including obstructive sleep apnea syndrome. Our study may not include all possible confounders for low bone mineral density, and there are opportunities for further studies into these multiple areas.
Conclusion
Those individuals with self-reported sleep duration of more than 8 h (long sleepers) have higher odds of osteoporosis compared to those who are short sleepers. These data add to the understanding of the significant impact that sleep has on health and metabolism, and clinicians should consider adding education on appropriate sleep duration to the current counseling that patients receive on osteoporosis prevention.
References
Cooper C, Campion G, Melton LJ (1992) Hip fractures in the elderly: a world-wide projection. Osteoporos Int 2:285–289
Yamamoto I (1999) Presumption of osteoporosis population. Osteoporo Jpn 1:10–11
Reginster JY, Burlet N (2006) Osteoporosis: a still increasing prevalence. Bone 38:S4–S9
Mora S, Gilsanz V (2003) Establishment of peak bone mass. Endocrinol Metab Clin North Am 32:39–63
Melton LJ 3rd, Atkinson EJ, Cooper C, O’Fallon WM, Riggs BL (1999) Vertebral fractures predict subsequent fractures. Osteoporos Int 10:214–221
Suzuki N, Ogikubo O, Hansson T (2010) Previous vertebral compression fractures add to the deterioration of the disability and quality of life after an acute compression fracture. Eur Spine J 19:567–574
MacLean C, Newberry S, Maglione M, McMahon M, Ranganath V, Suttorp M, Mojica W, Timmer M, Alexander A, McNamara M, Desai SB, Zhou A, Chen S, Carter J, Tringale C, Valentine D, Johnsen B, Grossman J (2008) Systematic review: comparative effectiveness of treatments to prevent fractures in men and women with low bone density or osteoporosis. Ann Intern Med 148:197–213
Stone KL, Ewing SK, Lui LY, Ensrud KE, Ancoli-Israel S, Bauer DC, Cauley JA, Hillier TA, Cummings SR (2006) Self-reported sleep and nap habits and risk of falls and fractures in older women: the study of osteoporotic fractures. J Am Geriatr Soc 54:1177–1183
Qaseem A, Snow V, Shekelle P, Hopkins R Jr, Forciea MA, Owens DK (2008) Screening for osteoporosis in men: a clinical practice guideline from the American College of Physicians. Ann Intern Med 148:680–684
Hall MH, Muldoon MF, Jennings JR, Buysse DJ, Flory JD, Manuck SB (2008) Self-reported sleep duration is associated with the metabolic syndrome in midlife adults. Sleep 31:635–643
Orimo H, Hayashi Y, Fukunaga M, Sone T, Fujiwara S, Shiraki M, Kushida K, Miyamoto S, Soen S, Nishimura J, Oh-Hashi Y, Hosoi T, Gorai I, Tanaka H, Igai T, Kishimoto H (2001) Diagnostic criteria for primary osteoporosis: year 2000 revision. J Bone Miner Metab 19:331–337
Orimo H (2006) Guideline for prevention and treatment of osteoporosis. Life Science, Tokyo
Elgan C, Dykes AK, Samsioe G (2004) Bone mineral density changes in young women: a two year study. Gynecol Endocrinol 19:169–177
Rasmussen MH, Wildschiodtz G, Juul A, Hilsted J (2008) Polysomnographic sleep, growth hormone insulin-like growth factor-I axis, leptin, and weight loss. Obesity (Silver Spring) 16:1516–1521
Fall C, Hindmarsh P, Dennison E, Kellingray S, Barker D, Cooper C (1998) Programming of growth hormone secretion and bone mineral density in elderly men: a hypothesis. J Clin Endocrinol Metab 83:135–139
Friedlander AL, Genant HK, Sadowsky S, Byl NN, Gluer CC (1995) A two-year program of aerobics and weight training enhances bone mineral density of young women. J Bone Miner Res 10:574–585
Bonaiuti D, Shea B, Iovine R, Negrini S, Robinson V, Kemper HC, Wells G, Tugwell P, Cranney A (2002) Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database Syst Rev 3:CD000333
Slemenda CW (1995) Body composition and skeletal density—mechanical loading or something more? J Clin Endocrinol Metab 80:1761–1763
Cummings SR, Browner WS, Bauer D, Stone K, Ensrud K, Jamal S, Ettinger B (1998) Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med 339:733–738
Slemenda CW, Longcope C, Zhou L, Hui SL, Peacock M, Johnston CC (1997) Sex steroids and bone mass in older men. Positive associations with serum estrogens and negative associations with androgens. J Clin Invest 100:1755–1759
Clarke BL, Khosla S (2010) Physiology of bone loss. Radiol Clin North Am 48:483–495
Verkasalo PK, Lillberg K, Stevens RG, Hublin C, Partinen M, Koskenvuo M, Kaprio J (2005) Sleep duration and breast cancer: a prospective cohort study. Cancer Res 65:9595–9600
Hopper JL, Seeman E (1994) The bone density of female twins discordant for tobacco use. N Engl J Med 330:387–392
Reid IR (2002) Relationships among body mass, its components, and bone. Bone 31:547–555
Seeman E, Melton LJ 3rd, O’Fallon WM, Riggs BL (1983) Risk factors for spinal osteoporosis in men. Am J Med 75(6):977–983
Feskanich D, Korrick SA, Greenspan SL, Rosen HN, Colditz GA (1999) Moderate alcohol consumption and bone density among postmenopausal women. J Womens Health 8(1):65–73
WHO Scientific Group (2008) Prevention and management of osteoporosis, 2000. Available at: http://whqlibdoc.who.int/trs/WHO_TRS_921.pdf Accessed July 28
Fujiwara S, Nakamura T, Orimo H, Hosoi T, Gorai I, Oden A, Johansson H, Kanis JA (2008) Development and application of a Japanese model of the WHO fracture risk assessment tool (FRAX). Osteoporos Int 19:429–435
Acknowledgements
We gratefully acknowledge Dr. Toshiko Kawakita for contributing data of the Center for Preventive Medicine at St. Luke’s International Hospital and thank Mina Shapiro, RN for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kobayashi, D., Takahashi, O., Deshpande, G.A. et al. Association between osteoporosis and sleep duration in healthy middle-aged and elderly adults: a large-scale, cross-sectional study in Japan. Sleep Breath 16, 579–583 (2012). https://doi.org/10.1007/s11325-011-0545-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-011-0545-6