Abstract
Purpose
The relationship between obstructive sleep apnea (OSA) and atherosclerosis-related inflammation has been poorly investigated, particularly focusing on functional responses of immune cells playing a key role in atherogenesis and in comparison with control groups with similar cardiovascular risk factors which are known to be themselves associated with inflammation. We sought to determine cellular tumor necrosis factor-alpha (TNF-α) production from peripheral blood mononuclear cells (PBMCs) and interleukin (IL)-8 release from neutrophils (PMNs) in patients studied for suspected OSA.
Methods
Thirty-six consecutive patients who underwent a nocturnal complete cardiorespiratory evaluation for suspected OSA were initially evaluated. Serum, PBMCs, and PMNs were isolated (at baseline and after 12 weeks) from patients with apnea–ipopnea index (AHI) >20 (OSA group, n = 16) and from control patients with AHI <5 (nonOSA group, n = 11). All patients continued the same pharmacological therapy for 12 weeks; the OSA group was additionally treated with nocturnal continuous positive-airway-pressure ventilation (cPAP).
Results
The two groups had similar clinical characteristics (prevalence of hypertension, dyslipidemia, diabetes, and cardio-metabolic therapies) except for obesity. Resting and stimulated TNF-α production from PBMCs and IL-8 release from PMNs were similar in the two groups. Serum cytokines resulted within the normal range. In the OSA group, cPAP was not associated with changes in cellular responses.
Conclusions
In patients showing similar prevalence of major cardiovascular risk factors and cardio-metabolic therapies, differing for the presence or absence of OSA, cytokine productions from PBMC and PMN were similar and were not modified during cPAP therapy. Studies designed to investigate OSA-associated inflammation should carefully match the control group subjects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnea (OSA) is a clinical condition associated with increased cardiovascular morbidity and mortality [1, 2]. Some of OSA risk factors such as obesity, dyslipidemia, insulin resistance, impaired glucose tolerance, hypertension, hypercoagulability state, and male sex are well-known cardiovascular risk factors (CRF) too [2–5]. The increased prevalence of arterial hypertension observed in OSA patients has been related to various pathophysiological processes among which increased sympathetic drive and endothelial dysfunction are key elements [6]. Since most of the characteristic risk factors of metabolic syndrome are seen in OSA patients, not surprisingly, these subjects are prone to a nine time-increased probability to develop this pathological condition [7].
Continuous positive airway pressure ventilation (cPAP) therapy is known to be the gold standard treatment for OSA, able to improve symptoms and reduce cardiovascular mortality and morbidity [8]. Moreover, beside cPAP therapy, various treatments aimed at interfering with CRF are often used in patients with OSA.
Vascular inflammation and production of inflammatory mediators are well-studied pathways underlying atherosclerosis [9] and vascular lesion may result from an imbalance between pro-inflammatory and anti-inflammatory responses. In particular, tumor necrosis factor-α (TNF-α), one key cytokine of type 1 CD4+ T-helper cells, is known to be highly pro-inflammatory and to exacerbate the atherosclerotic disease [10]. The polymorphonuclear leukocytes (PMNs) are the first cellular type involved in early stages of vascular atheromata and interleukin (IL)-8, the major pro-inflammatory cytokine released by these cells, is well established as a proatherogenic factor, being able to attract PMNs, to activate monocytes and to direct their recruitment towards vascular lesions [11, 12].
A pro-inflammatory state has been proposed as one of the pathophysiological components of OSA associated with hypoxemia and sleep deprivation [13], and although a few published data reported a relationship between OSA and inflammation, both the evaluation at a cellular level of cytokine production and the comparison between OSA and nonOSA patients similarly burdened by CRF has been poorly investigated. Moreover, anti-hypertensive and/or metabolic therapies are associated with modulation of systemic and cellular parameters of inflammation [14–24]. For the presence of those potential confounding factors (i.e. CRF and therapies), the relationship between OSA and inflammation is doubtful and the association between OSA and inflammation could have been magnified by the concomitant presence of CRF in OSA subjects in previous reports. Therefore, we sought to assess the cytokine production from cells playing a key role in atherogenesis such as peripheral blood mononuclear cells (PBMCs; TNF-α production) and PMNs (IL-8 release) in patients undergoing a cardiorespiratory examination for suspected OSA, thus comparing a control group derived from the same population of patients as OSA subjects and differing for the absence of the sleep disorder. Serum levels of TNF-α and IL-8 were also analyzed. As a secondary endpoint, we investigated whether cPAP was associated with changes in cytokine production in patients affected by OSA.
Patients and methods
The initial patient population consisted of 36 consecutive patients who underwent a nocturnal complete cardiorespiratory evaluation for suspected sleep apnea. All the patients had been evaluated at the Pneumology Division, Cuasso al Monte, Circolo Hospital, Varese, and then studied at our Research Center on Dyslipidemia, Department of Clinical Medicine, University of Insubria, Varese, to be included in the study aimed at investigating the leukocyte cytokine production (TNF-α from circulating PBMCs and IL-8 from PMNs) and serum levels.
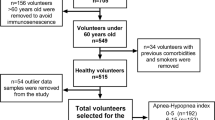
As for inclusion criteria we considered patients without ongoing infections (3 months before the study), any known immuno-inflammatory disease such as rheumatoid arthritis, chronic inflammatory bowel diseases, psoriasis, and without non-steroidal anti-inflammatory drugs or steroid therapy. The cardiorespiratory examination was performed to evaluate apnea–hypopnea index (AHI) using Embletta PDS (EMBLA, somnologica for EMBLA software); AHI was calculated as the total number of episodes of apnea (total airflow cessation for more than 10 s) and hypopnea (reduction in airflow ≥10 s with oxygen desaturation of ≥4%) per hour of sleep [13]. Patients showing an AHI >20 (n = 16) were included in the study and considered OSA group whereas patients who showed an AHI <5 were considered not affected and included in the study as control nonOSA group (n = 11). Exclusion criteria were the finding of an intermediate (range: 5–19) AHI value (n = 7) and/or any addition of novel drugs and/or changes in treatments during the study (n = 1, added chemotherapy) and/or the presence of infections throughout the study (n = 1, urinary infection).
All the patients underwent clinical assessment and testing for complete standard blood examination and blood sampling for isolation of PBMCs and PMNs from 8.00 and 9.00 am (blood sampling was evaluated for cell isolation within 1 h—see below). The following laboratory parameters were analyzed: total cholesterol, high-density lipoprotein cholesterol (HDL-c), triglycerides, fasting glycemia, whereas the low-density lipoprotein cholesterol (LDL-c) was calculated according to the Fridewall formula: LDL-c = total cholesterol − (HDL-c + triglycerides/5). High-sensitive C-reactive protein was also measured. The patients included in the study were studied twice: at baseline and after 12 weeks. After the first evaluation, in OSA group (average AHI, 39.6 ± 19.1; average nocturnal oxygen saturation, 90.7 ± 7%; oxygen desaturation index, 37.3 ± 16), the nocturnal cPAP therapy was added to the ongoing pharmacological treatments and subjects of nonOSA group continued their pharmacological treatment. No change in lifestyle was instituted, no significant change in weight was observed and adherence to pharmacological treatments and cPAP compliance was verified at the second evaluation, being compliance subjectively reported as good in all the patients. In particular, the body mass index did not change throughout the study in the two groups (OSA patients, 31.76 ± 4.39 kg/m2 at visit 1 vs. 31.71 ± 4.44 kg/m2 at visit 2, p = 0.87; nonOSA subjects, 27.68 ± 3.46 kg/m2 vs. 27.68 ± 3.36 kg/m2; p = 0.17). The mean age of the overall population studied (n = 36) was 58 ± 12 years. The mean age of the OSA patients (15 males, one female) and nonOSA subjects (seven males, four females) did not differ (61 ± 10 years vs. 55 ± 14 years, respectively, p = 0.33). Table 1 shows the clinical features of the two groups of patients. All subjects included in the study were white Caucasians. In particular, there was a similar prevalence in the two groups as regards hypertension (systolic blood pressure ≥ 140 mmHg, diastolic blood pressure ≥ 90 mmHg) [25], diabetes (fasting plasma glucose ≥ 126 mg/dl or symptoms of hyperglycemia or casual plasma glucose ≥ 200 mg/dl) [26], metabolic syndrome (defined as the presence of any three of the following risk factors: waist circumference, man > 102 cm, woman > 88 cm; triglycerides ≥ 150 mg/dl; HDL-c < 40 mg/dl for men and <50 mg/dl for women; blood pressure ≥ 130/≥85 mmHg; fasting glucose ≥ 110 mg/dl) [27] whereas a greater prevalence of obesity (body mass index ≥ 30 kg/m2) [28] was observed in the OSA group. Moreover, the prevalence of anti-hypertensive, hypolipemic, and antidiabetic therapy was similar in the two groups. In particular, as regards anti-hypertensive treatment, six patients with OSA and four without sleep disorder assumed a combination treatment. Angiotensin-converting enzyme inhibitors were assumed in seven OSA and in four nonOSA group patients, type 1 angiotensin receptor blockers were assumed in two patients in OSA and four in nonOSA groups, calcium-channel blocker in two OSA subjects and in one nonOSA patient, diuretics were used in eight patients of OSA group and three of nonOSA patients, and beta-receptor blockers were assumed in two OSA patients and in three nonOSA, and alpha-receptor blockers in three patients in OSA group and in one nonOSA patient. Statin as a hypolipemic therapy was assumed in four subjects in OSA (in one patient associated with polyunsatured fatty acids) and four in nonOSA group. As regards diabetes treatment, among patients with OSA, one assumed metformin and one glibenclamid whereas one patient in the nonOSA group was treated with metformin plus glibenclamid plus rosiglitazone. Taken together, the treatments for diabetes, dyslipidemia, and hypertension are referred to as “cardio-metabolic therapy” throughout the text.
The work has been conducted in accordance with the Declaration of Helsinki and all the patients gave an informed consent to the study which was approved by the local Ethical Committee.
Cell isolation
PBMCs
Whole blood was allowed to sediment on dextran at 37°C for 30 min. Supernatant was recovered and PBMCs were separated by Ficoll-Paque Plus density-gradient centrifugation. A typical PBMC preparation contained about 80% lymphocytes and 16% monocytes, and cell viability was always >99% as assessed by flow cytometric analysis.
PMNs
Whole blood was allowed to sediment on dextran at 37°C for 30 min. Supernatant was recovered and PMNs were isolated by standard density-gradient centrifugation. Contaminating erythrocytes were eliminated by 10 min hypotonic lysis in distilled water with added NH4Cl 8.2 g/l, KHCO3 1.0 g/l, and ethylenediamine tetraacetic 37.0 mg/l. Cells were then washed three times in NaCl 0.15 mol/l. Purity and viability of PMNs preparations were always greater than 95% and no platelets or erythrocytes could be detected either by light microscopic examination or by flow cytometric analysis.
Cytokine production from leukocyte subsets
TNF-α production from isolated PBMCs
TNF-α production by PBMCs was measured in resting and stimulated conditions. Cells were resuspended at the concentration of 1 × 107 cells/ml in RPMI culture medium for 24 h and incubated alone or in the presence of lypopolisaccharide (LPS; 1 μg/ml). Finally cells were harvested and centrifuged, cells supernatant were recovered and stored until assay at −80°C.
IL-8 production from isolated PMNs
PMNs, separated as above described, were resuspended at the concentration of 1 × 107 cells/ml in RPMI culture medium and incubated alone (resting IL-8 production) or in the presence of 0.1 μM of N-formyl-Met-Leu-Phe (fMLP-stimulated IL-8 production) at 37°C for 3 h. After incubation, the cells were centrifuged (600×g, 5 min, 20°C) and the supernatant was harvested and stored at −80°C until assay.
Serum cytokines
Serum obtained with standard procedures from venous blood of patients and controls were stored at −80°C until cytokine assay (TNF-α; IL-8). The normal values for the investigated serum cytokines are: 0 pg/ml for TNF-α and 1.2–16.7 pg/ml for IL-8, respectively.
Cytokine assay
Cytokines (TNF-α, IL-8) levels cell supernatants and serum were quantified using a sandwich-type enzyme-linked immunosorbent assay (ELISA kit; Amersham Biosciences, Little Chalfont, UK). All steps were performed at room temperature. The optical density was determined using a spectrophotometer set to 450 nm. The detection limit of the assay was 1 pg/ml for all the cytokines investigated.
Statistical analysis
Descriptive statistics were computed as mean and standard deviation for continuous variables. For skewed distributions median and interquartile range (IQR) were used.
The Chi square test was used to compare the prevalence of hypertension, dyslipidemia, metabolic syndrome, diabetes, and obesity between the two groups of OSA and nonOSA patients and to compare the prevalence of pharmacological treatments for CRF.
Parametric continuous variables were compared with the Student’s t test (age, hemodynamic parameters, lipid and metabolic profile, serum C-reactive protein, and body mass index) between the two groups of OSA and nonOSA patients.
The Mann–Whitney U test was used for independent variable comparison of cytokine production and the Wilcoxon test was performed to compare data obtained at baseline and after 12-week follow-up. Calculations were performed using a commercial software (GraphPad Prism version 5.00 for Windows, GraphPad Software, San Diego, CA, USA, www.graphpad.com) and a two-sided P < 0.05 was retained for statistical significance.
Results
Clinical and laboratory characteristics
Among our population undergoing a cardiorespiratory examination for suspected OSA, the subjects with and without OSA had similar prevalence of hypertension, diabetes, metabolic syndrome, and cardio-metabolic therapy (Table 1). Systolic blood pressure and heart rate were similar between the two groups whereas diastolic blood pressure was slightly higher in OSA patients (Table 2). A significantly higher body mass index was observed in OSA group. Total cholesterol, HDL-c, triglycerides, and fasting glycemia were similar in OSA and nonOSA groups; LDL-c was higher in nonOSA group (Table 2).
Cytokine production in OSA and nonOSA groups
No difference in cytokine production was observed between OSA and nonOSA groups. In particular, in PBMCs, the TNF-α production was similar between groups both in resting condition and after LPS stimulation (Fig. 1, upper panels); in neutrophils, IL-8 release was not statistically different in OSA and nonOSA patients either in a resting state or after fMLP stimulus (Fig. 1, bottom panels).
Resting (upper left panel) and LPS-induced (upper right panel) TNF-α production from PBMCs and resting (bottom left panel) and fMLP-stimulated (bottom right panel) IL-8 release from PMNs isolated from OSA patients and nonOSA control group. Comparison between the two groups: TNF-α: P = 0.61 for resting and P = 0.71 for stimulated production; IL-8: P = 0.43 for resting and P = 0.30 for stimulated production. Horizontal line median value, box 25th to 75th percentile, vertical bars range
Serum cytokines in OSA and nonOSA groups
As regards serum cytokines, serum levels were always found within the normal range. Therefore we report here a descriptive statistics: TNF-α: median (IQR): 0 pg/ml (0–0.87) vs. 0 pg/ml (0–0); IL-8: median (IQR): 0 pg/ml (0–0) vs. 0 pg/ml (0–0).
Longitudinal study
No change was evidenced at the second visit as regards lifestyle. Adherence to pharmacological treatments was verified in all the subjects and compliance towards cPAP was reported good for all the OSA patients.
The 12-week cPAP treatment in OSA group, when added to the pharmacological ongoing treatment, did not modify cytokine functional responses in both PBMCs (resting and stimulated TNF-α production) and neutrophils (resting IL-8 and IL-8 release after stimulus; Table 3).
In the nonOSA group, the resting cellular cytokine release from PBMCs was similar at baseline and after 12-weeks whereas the stimulated TNF-α was increased at the follow-up evaluation (Table 3).
The serum levels of TNF-α and IL-8 remained within the normal range both in the OSA group and in the nonOSA group.
Discussion
In the last few years OSA condition has been related to novel risk factors leading to atherogenesis such as systemic inflammation and oxidative stress [29]. Moreover, it is well known that isolated traditional CRF and clustered high cardiovascular risk conditions are associated with inflammation both expressed as increased systemic markers and pro-inflammatory cellular responses [19–24, 30–32].
Atherosclerosis is nowadays considered an immuno-inflammatory pathological process, being ongoing inflammatory responses accelerating and exacerbating molecular events associated with LDL-c oxidative modification occurring within the vessel walls and different types of cells are known to play pivotal roles trough the involvement of cytokine production [9]. Most of the previous studies investigating sleep disorders were performed using serum levels of inflammatory markers and comparing groups with various potential confounding factors such as a nonhomogeneous distribution of CRF and/or pathological conditions, including metabolic syndrome, and drug treatments for diabetes, hypertension, and dyslipidemia [33–35]. Furthermore a few published studies on inflammation and OSA do not provide a satisfactorily patient description as regards concomitant CRF and cardio-metabolic treatments [36–38].
In our study, the cellular production of key functional cytokines, such as TNF-α from PBMCs and IL-8 from PMNs (baseline cellular release and stimulated production), and their serum levels were found to be similar between subjects presenting a OSA condition and nonOSA patients. Moreover, although stimulated TNF-α was higher in the controls after 3 months, in the OSA group cytokine production did not change after cPAP therapy. All the patients and controls were recruited after a nocturnal cardiopulmonary examination for suspected OSA and divided according to AHI. These enrolling criteria resulted into two groups with similar clinical characteristics except for a higher prevalence of obesity and body mass index in the OSA group, together with a slight, though significant higher diastolic blood pressure and lower LDL-c levels. No further patient selection was used except for the exclusion of patients with systemic diseases associated with chronic inflammation, ongoing infections, or ongoing anti-inflammatory drug treatments.
As regards cellular functional responses of circulating leukocytes, to date, literature does not provide conclusive data matching OSA and pro-inflammatory cytokine production. Previously, only a few studies focused on cellular functional responses of lymphocytes and monocytes in OSA, analyzing results in selected patients, using healthy volunteers as controls or excluding confounding co-morbidities such as diabetes, hypertension, and ischemic heart disease [33, 39, 40]. In monocytes, an increased TNF-α was found in severe OSA when compared to healthy controls, obese controls, and mild OSA patients. However, no difference in TNF-α was observed between monocytes from healthy controls and obese controls [41].
A positive association of both subcutaneous and visceral fat with circulating markers of inflammation was reported in Framingham Heart Study participants [42]. In sleep apnoeic patients, plasma TNF-α and IL-6 were higher than the values found in the lean control group whereas did not differ from levels of obese controls [4]. Moreover, serum levels of TNF-α, IL-6, and leukotriene were found similar when OSA patients were compared with obese healthy subjects [43].
In our patients burdened by major CRF and poly-treated, the greater prevalence of obesity in OSA group did not result in increased cytokine cellular production from both PBMCs and PMNs or in increased serum levels of the investigated cytokines. Since it has been reported that pro-inflammatory changes in various cell types are affected by the risk profile of the subjects [16–24] it is likely that these CRF may constitute major confounding issues in studies associating OSA with inflammation. Moreover, it has been reported that drugs used in counteracting major CRF such as statins, calcium-channel blockers, and angiotensin-converting enzyme inhibitors can reduce the upregulated pro-inflammatory cellular responses [16–18, 21–24].
Arterial hypertension is more prevalent in OSA patients [3, 5]; in our study, OSA and nonOSA groups were similar for the prevalence of hypertension. Previously, when comparing serum cytokines, increased levels of TNF-α and IL-6 [35] were observed in a group of OSA hypertensive patients compared with nonOSA normotensive subjects. Similarly, the obese apneic subjects who showed increased plasma concentrations of IL-6 and TNF-α compared with non-apneic lean subjects in the previously cited study by Vgontzas and coworkers had systolic, diastolic, and mean arterial pressure levels significantly higher [4]. Moreover, the same authors observed a significant effect of anti-hypertensive medications in OSA patients, being the serum levels of TNF-α and IL-6 higher in the group not taking any drug [4].
As regards the association between metabolic cardiovascular risk factors and sleep-disordered breathing, in patients of the Sleep Heart Health Study cohort the degree of the disorder was related in men to total cholesterol, triglycerides, and to low HDL-c levels [2]. In previous studies on inflammation in OSA, since the lipid pattern and the metabolic syndrome prevalence were seldom investigated and considered in the analyses, another confounding factor could be the metabolic profile. In a study reporting the prevailing activation of T-helper 1-pattern cytokine in patients affected by OSA, 50% of individuals presented hyperlipidemia as a possible pro-inflammatory confounding factor [35]. In our data OSA and nonOSA control group showed similar prevalence of diabetes and metabolic syndrome and similar values of total cholesterol, HDL-c, and glycemia.
The similar pro-inflammatory cellular pattern found in our study comparing the OSA and nonOSA groups seems therefore to indicate that the contribution to inflammation exerted by the CRF and/or the modulation by cardio-metabolic therapy (assumed in 62.5% of our OSA patients and in 72% of nonOSA individuals) could predominantly affect the inflammatory response beyond the sleep disorder itself. However, altered cellular protein expression could pre-date cytokine production by leukocytes, since neutrophil kappaB binding activity determined by electrophoretic mobility shift assay was found to be increased in OSA subjects compared with controls [44]. Moreover, impaired endothelial function expressed by eNOS and phosphorylated eNOS was reported in patients with OSA together with a reduced capacity of endothelial repair, as shown by a reduced number of circulating endothelial progenitor cells found in OSA patients free of any other known CRF [45, 46].
A few previous studies examined the impact of cPAP therapy on inflammatory markers with controversial results. Reductions in serum C-reactive protein, TNF-α, IL-6, IL-8, and markers of oxidative stress were demonstrated after cPAP therapy in OSA [34, 40, 47]. However, in most of the studies, the recruited OSA patients did not have any concomitant diseases nor ongoing pharmacological treatments. Only 10% [33] and 17% [41] of the OSA patients who respectively showed a reduced monocyte production of IL-6 and TNF-α when treated with cPAP had concomitant hypertension treated with calcium channel blockers. Phillips and colleagues [38] could demonstrate that 7 days withdrawal of cPAP in OSA patients resulted in an immediate return of OSA clinical condition but interestingly it was not possible to observe a change in measured cytokines and systemic markers of inflammation. Recently serum inflammatory markers did not change after 4 weeks of either therapeutic or sub-therapeutic cPAP treatment in a comprehensive study in men with moderate–severe OSA [48].
In our OSA patients, most of whom were treated with cardio-metabolic therapy, cPAP was not able to modify the cellular cytokine production by PBMCs and PMNs. Therefore, the contribution of cPAP in treated patients seems to be less relevant than observed in OSA subjects presenting with milder CRF and less intensely treated. However, our controls, who did not undergo any change in treatment, showed increased stimulated TNF-α after 12 weeks, possibly reflecting a progression in atherosclerosis-related micro-inflammation. Therefore, it cannot be excluded that our OSA patient population presenting with high prevalence of CRF and poly-pharmacological treatment could have been worsened during follow-up without adding cPAP to the current drug therapy. It has to be acknowledged that we studied a relatively small patient population which was not randomized for cPAP to avoid a delay of ventilatory treatment when clinically indicated and that compliance to cPAP was verified according to subjective reports. Moreover our findings obtained in moderate to severe OSA may not apply to higher degrees of the sleep disorder and suggest a lack of primary role of inflammation in OSA patients when compared with subjects presenting with similar clinic characteristics.
In conclusion, our study indicates that distribution and treatments of CRF act as potent confounders when the relationship between sleep disorders and inflammation is focused on. Moreover, our results suggest that anti-hypertensive and hypolypemic-metabolic treatments seem to prevail over the potential pro-inflammatory effect of OSA and that, in patients treated for CRF, the role of cPAP in reducing cellular cytokine production is not a major one.
In our opinion, our study prompts the need for future studies to take into account CRF and cardiovascular treatments when drafting a study protocol designed to disentangle the role of OSA in determining cytokine imbalance and the response to cPAP in terms of modulation of the pro-inflammatory state.
References
Stradling JR, Crosby JH (1991) Predictor and prevalence of OSA and snoring in 1001 middle aged men. Thorax 46:85–90
Newman AB, Nieto FJ, Guidry U, Lind BK, Redline S, Pickering TG, Quan SF, Sleep Heart Health Study Research Group (2001) Relation of sleep-disordered breathing to cardiovascular disease risk factors: the sleep heart health study. Am J of Epidemiol 154:50–59
Peppard PE, Young T, Palta M, Skatrud J (2000) Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 342:1378–1384
Vgontzas AN, Papanicolaou DA, Bixler EO, Hopper K, Lotsikas A, Lin HM, Kales A, Chrousos GP (2000) Sleep apnea and daytime sleepiness and fatigue: relation to visceral obesity, insulin resistance, and hypercytokinemia. J Clin Endocrinol Metab 85:1151–1158
Quan SF, Gersh BJ, National Center on Sleep Disorders Research, National Heart, Lung, and Blood Institute (2004) Cardiovascular consequences of sleep-disordered breathing: past, present and future: report of a workshop from the national center on sleep disorders research and the national heart, lung, and blood institute. Circulation 109:951–957. doi:10.1161/01.CIR.0000118216.84358.22
Wolk R, Somers VK, Virend K (2007) Sleep and the metabolic syndrome. Exp Physiol 92:67–78. doi:10.1113/expphysiol.2006.033787
Coughlin SR, Mawdsley L, Mugarza JA, Calverley PM, Wilding JP (2004) Obstructive sleep apnoea is independently associated with an increased prevalence of metabolic syndrome. Eur Heart J 25:735–741. doi:10.1016/j.ehj.2004.02.021
Bazzano LA, Khan Z, Reynolds K, He J (2007) Effect of nocturnal nasal continuous positive airway pressure on blood pressure in obstructive sleep apnea. Hypertension 50:417–423. doi:10.1161/HYPERTENSIONAHA.106.085175
Hansson GK (2005) Inflammation, Atherosclerosis and coronary artery disease. N Engl J Med 352:1685–1695
Kleemann R, Zadelaar S, Kooistra T (2008) Cytokines and atherosclerosis: a comprehensive review of studies in mice. Cardiovasc Res 79:360–376. doi:10.1093/cvr/cvn120
Baggiolini M, Dewald B, Moser B (1994) Interleukin-8 and related chemotactic cytokines—CXC and CC chemokines. Adv Immunol 55:97–179
Gerszten RE, Garcia-Zepeda EA, Lim Y-C, Yoshidak M, Ding HA, Gimbrone MA Jr, Luster AD, Luscinskas FW, Rosenzweig A (1999) MCP-1 and IL-8 trigger firm adhesion of monocytes to vascular endothelium under flow conditions. Nature 398:718–723. doi:10.1038/19546
Somers VK, White DP, Amin R, Abraham WT, Costa F, Culebras A, Daniels S, Floras JS, Hunt CE, Olson LJ, Pickering TG, Russell R, Woo M, Young T, American Heart Association Council for High Blood Pressure Research Professional Education Committee, Council on Clinical Cardiology, American Heart Association Stroke Council; American Heart Association Council on Cardiovascular Nursing, American College of Cardiology Foundation (2008) Sleep apnea and cardiovascular disease. An american heart association/american college of cardiology foundation scientific statement from the American heart association council for high blood pressure research professional education committee, council on clinical cardiology, stroke council, and council on cardiovascular nursing council. Circulation 118:1080–1111. doi:10.1161/CIRCULATIONAHA.107.189420
Ridker PM, Danielson E, Fonseca FA, Genest J, Gotto AM Jr, Kastelein JJ, Koenig W, Libby P, Lorenzatti AJ, MacFadyen JG, Nordestgaard BG, Shepherd J, Willerson JT, Glynn RJ, JUPITER Study Group (2008) Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med 359:2195–2207. doi:10.1056/NEJMoa0807646
Kinlay S, Schwartz GG, Olsson AG, Rifai N, Leslie SJ, Sasiela WJ, Szarek M, Libby P, Ganz P, Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering Study Investigators (2003) High-dose atorvastatin enhances the decline in inflammatory markers in patients with acute coronary syndromes in the MIRACL study. Circulation 108:1560–1566. doi:10.1161/01.CIR.0000091404.09558.AF
Yasunari K, Maeda K, Nakamura M, Watanabe T, Yoshikawa J, Asada A (2004) Effects of carvedilol on oxidative stress in polymorphonuclear and mononuclear cells in patients with essential hypertension. Am J Med 116:460–465. doi:10.1016/j.amjmed.2003.10.029
Yasunari K, Maeda K, Nakamura M, Watanabe T, Yoshikawa J (2005) Benidipine, a long-acting calcium channel blocker, inhibits oxidative stress in polymorphonuclear cells in patients with essential hypertension. Hypertens Res 28:107–112. doi:10.1291/hypres.28.107
Seres I, Fóris G, Páll D, Kosztáczky B, Paragh G Jr, Varga Z, Paragh G (2005) Angiotensin II-induced oxidative burst is fluvastatin sensitive in neutrophils of patients with hypercholesterolemia. Metabolism 54:1147–1154. doi:10.1016/j.metabol.2005.03.021
Sela S, Shurtz-Swirski R, Cohen-Mazor M, Mazor R, Chezar J, Shapiro G, Hassan K, Shkolnik G, Geron R, Kristal B (2005) Primed peripheral polymorphonuclear leukocytes: a culprit underlying chronic low-grade inflammation and systemic oxidative stress in chronic kidney disease. J Am Soc Nephrol 16:2431–2438. doi:10.1681/ASN.2004110929
Shurtz-Swirski R, Sela S, Shapiro G, Nasser L, Shasha SM, Kristal B (2001) Involvement of polymorphonuclear leukocytes in oxidative stress and inflammation in type 2 diabetes. Diabetes Care 24:104–110. doi:10.2337/diacare.24.1.104
Sugano R, Matsuoka H, Haramaki N, Umei H, Murase E, Fukami K, Iida S, Ikeda H, Imaizumi T (2005) Polymorphonuclear leukocytes may impair endothelial function. Results of crossover randomized study of lipid-lowering therapies. Arterioscler Thromb Vasc Biol 25:1262–1267. doi:10.1161/01.ATV.0000163842.91226.ba
Guasti L, Marino F, Cosentino M, Cimpanelli M, Maio RC, Klersy C, Crespi C, Restelli D, Simoni C, Franzetti I, Gaudio G, Marnini P, Grandi AM, Lecchini S, Venco A (2006) Simvastatin treatment modifies polymorphonuclear leukocyte function in high-risk individuals: a longitudinal study. J Hypertens 24:2423–2430. doi:10.1097/01.hjh.0000251903.62804.77
Marino F, Guasti L, Cosentino M, Rasini E, Ferrari M, Maio RC, Loraschi A, Cimpanelli MG, Schembri L, Legnaro M, Molteni E, Crespi C, Crema F, Venco A, Lecchini S (2008) Simvastatin treatment in subjects at high cardiovascular risk modulates AT1R expression on circulating monocytes and T lymphocytes. J Hypertens 26:1147–1155. doi:10.1097/HJH.0b013e3282f97dde
Guasti L, Marino F, Cosentino M, Rasini E, Ferrari M, Castiglioni L, Klersy C, Gaudio G, Grandi AM, Lecchini S, Venco A (2008) Prolonged statin-associated reduction in neutrophil reactive oxygen species and angiotensin II type 1 receptor expression: 1-year follow-up. Eur Heart J 29:1118–1126. doi:10.1093/eurheartj/ehn138
Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, Jones DW, Materson BJ, Oparil S, Wright JT Jr, Roccella EJ, National Heart, Lung, and Blood Institute Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure, National High Blood Pressure Education Program Coordinating Committee (2003) The seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure. JAMA 289:2560–2572. doi:10.1001/jama.289.19.2560
American Diabetes Association (2008) Diagnosis and classification of diabetes mellitus. Diabetes Care 31(Suppl 1):S55–S60. doi:10.2337/dc08-S055
Grundy SM, Brewer HB Jr, Cleeman JI, Smith SC Jr, Lenfant C (2004) American Heart Association; National Heart, Lung, and Blood Institute. Definition of metabolic syndrome. Report of the National Heart, Lung, and Blood Institute/American Heart Association Conference on Scientific Issues Related to Definition. Circulation 109:433–438. doi:10.1161/01.CIR.0000111245.75752.C6
Poirier P, Giles TD, Bray GA, Hong Y, Stern JS, Pi-Sunyer FX, Eckel RH, American Heart Association; Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism (2006) Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss: an update of the 1997 American heart association scientific statement on obesity and heart disease from the obesity committee of the council on nutrition, physical activity, and metabolism. Circulation 113:898–918. doi:10.1161/CIRCULATIONAHA.106.171016
Alam I, Lewis K, Stephens JW, Baxter JN (2006) Obesity, metabolic syndrome and sleep apnea: all pro-inflammatory states. Obes Rev 8:119–127. doi:10.1111/j.1467-789X.2006.00269.x
Jacobi J, Sela S, Cohen HI, Chezar J, Kristal B (2006) Priming of polymorphonuclear leukocytes: a culprit in initiation of endothelial cell injury. Am J Physiol Heart Circ Physiol 290:H2051–H2058. doi:10.1152/ajpheart.01040.2005
Libby P, Ridker PM, Maseri A (2002) Inflammation and atherosclerosis. Circulation 105:1135–1143. doi:10.1161/hc0902.104353
Tripepi G, Mallamaci F, Zoccali C (2005) Inflammation markers, adhesion molecules, and all-cause and cardiovascular mortality in patients with ESRD: searching for the best risk marker by multivariate modelling. J Am Soc Nephrol 16(Suppl 1):S83–S88. doi:10.1681/ASN.2004110972
Yokoe T, Minoguchi K, Matsuo H, Oda N, Minoguchi H, Yoshino G, Hirano T, Adachi M (2003) Elevated levels of c-reactive protein and interleukin-6 in patients with obstructive sleep apnoea syndrome are decreased by nasal continuous positive airway pressure. Circulation 107:1129–1134. doi:10.1161/01.CIR.0000052627.99976.18
Ryan S, Taylor CT, McNicholas WT (2006) Predictors of elevated nuclear factor-KB-dependent genes in obstructive sleep apnea syndrome. Am J Respir Crit Care Med 174:824–830. doi:10.1164/rccm.200601-066OC
Alberti A, Sarchielli P, Gallinella E, Floridi A, Floridi A, Mazzotta G, Gallai V (2003) Plasma cytokine levels in patients with obstructive sleep apnea syndrome: a preliminary study. J Sleep Res 12:305–311. doi:10.1111/j.1365-2869.2003.00361.x
Ciftci TU, Kokturk O, Bukan N, Bilgihan A (2004) The relationship between serum cytokine levels with obesity and obstructive sleep apnea syndrome. Cytokine 28:87–91. doi:10.1016/j.cyto.2004.07.003
Vgontzas AN, Papanicolaou DA, Bixler EO, Kales A, Tyson K, Chrousos GP (1997) Elevation of plasma cytokines in disorders of excessive daytime sleepiness: role of sleep disturbance and obesity. J Clin Endocrinol Metab 82:1313–1316. doi:0021-972X/97/$03.00/0
Phillips CL, Yang Q, Williams A, Roth M, Yee BJ, Hedner JA, Berend N, Grunstein RR (2007) The effect of short-term withdrawal from continuous positive airway pressure therapy on sympathetic activity and markers of vascular inflammation in subjects with obstructive sleep apnoea. J Sleep Res 16:217–225. doi:10.1111/j.1365-2869.2007.00589.x
Liu H, Liu J, Xiong S, Shen G, Zhang Z, Xu Y (2000) The change of interleukin-6 and tumor necrosis factor in patients with obstructive sleep apnea syndrome. J Tongji Med Univ 20:200–202
Dorkova Z, Petrasova D, Molcanyiova A, Popovnakova M, Tkacova R (2008) Effects of continuous positive airway pressure on cardiovascular risk profile in patients with severe obstructive sleep apnea and metabolic syndrome. Chest 134:686–692. doi:10.1378/chest.08-0556
Minoguchi K, Tazaki T, Yokoe T, Minoguchi H, Watanabe Y, Yamamoto M, Adachi M (2004) Elevated production of tumor necrosis factor α by monocytes in patients with obstructive sleep apnea syndrome. Chest 126:1473–1479. doi:10.1378/chest.126.5.1473
Pou KM, Massaro JM, Hoffmann U, Vasan RS, Maurovich-Horvat P, Larson MG, Keaney JF Jr, Meigs JB, Lipinska I, Kathiresan S, Murabito JM, O'Donnell CJ, Benjamin EJ, Fox CS (2007) Visceral and subcutaneous adipose tissue volumes are cross-sectionally related to markers of inflammation and oxidative stress: the Framingham Heart Study. Circulation 116:1234–1241. doi:10.1161/CIRCULATIONAHA.107.710509
Arias MA, García-Río F, Alonso-Fernández A, Hernanz A, Hidalgo R, Martínez-Mateo V, Bartolomé S, Rodríguez-Padial L (2008) CPAP decreases plasma levels of soluble tumour necrosis factor-alpha receptor 1 in obstructive sleep apnoea. Eur Respir J 32:1009–1015. doi:10.1183/09031936.00007008
Htoo AK, Greenberg H, Tongia S, Chen G, Henderson T, Wilson D (2006) Liu SF (2006) Activation of nuclear factor kappaB in obstructive sleep apnea: a pathway leading to systemic inflammation. Sleep Breath 10:43–50. doi:10.1007/s11325-005-0046-6
Jelic S, Padeletti M, Kawut SM, Higgins C, Canfield SM, Onat D, Colombo PC, Basner RC, Factor P, LeJemtel TH (2008) Inflammation, oxidative stress, and repair capacity of the vascular endothelium in obstructive sleep apnea. Circulation 117:2270–2278. doi:10.1161/CIRCULATIONAHA.107.741512
de la Peña M, Barceló A, Barbe F, Piérola J, Pons J, Rimbau E, Ayllón O, Agustí AG (2008) Endothelial function and circulating endothelial progenitor cells in patients with sleep apnea syndrome. Respiration 76:28–32. doi:10.1159/000109643
Ohga E, Tomita T, Wada H, Yamamoto H, Nagase T, Ouchi Y (2003) Effects of obstructive sleep apnea on circulating ICAM-1, IL-8, and MCP-1. J Appl Physiol 94:179–184. doi:10.1152/japplphysiol.00177.2002
Kohler M, Ayers L, Pepperell JC, Packwood KL, Ferry B, Crosthwaite N, Craig S, Siccoli MM, Davies RJ, Stradling JR (2009) Effects of continuous positive airway pressure on systemic inflammation in patients with moderate to severe obstructive sleep apnoea: a randomised controlled trial. Thorax 64:67–73. doi:10.1136/thx.2008.097931
Conflicts of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guasti, L., Marino, F., Cosentino, M. et al. Cytokine production from peripheral blood mononuclear cells and polymorphonuclear leukocytes in patients studied for suspected obstructive sleep apnea. Sleep Breath 15, 3–11 (2011). https://doi.org/10.1007/s11325-009-0315-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-009-0315-x