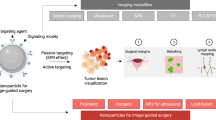
Abstract
Purpose
Negative surgical margins (NSMs) have favorable prognostic implications in breast tumor resection surgery. Fluorescence image-guided surgery (FIGS) has the ability to delineate surgical margins in real time, potentially improving the completeness of tumor resection. We have recently developed indocyanine green (ICG)-loaded self-assembled hyaluronic acid (HA) nanoparticles (NanoICG) for solid tumor imaging, which were shown to enhance intraoperative contrast.
Procedures
This study sought to assess the efficacy of NanoICG on completeness of breast tumor resection and post-surgical survival. BALB/c mice bearing iRFP+/luciferase+ 4T1 syngeneic breast tumors were administered NanoICG or ICG, underwent FIGS, and were compared to bright light surgery (BLS) and sham controls.
Results
NanoICG increased the number of complete resections and improved tumor-free survival. This was a product of improved intraoperative contrast enhancement and the identification of a greater number of small, occult lesions than ICG and BLS. Additionally, NanoICG identified chest wall invasion and predicted recurrence in a model of late-stage breast cancer.
Conclusions
NanoICG is an efficacious intraoperative contrast agent and could potentially improve surgical outcomes in breast cancer.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Surgery is the primary treatment modality for stage I-III breast cancer [1, 2]. Complete removal of the primary tumor, or a negative surgical margin (NSM), is critical in decreasing local recurrence and increasing disease-specific survival [3,4,5,6]. However, roughly 1/3 of women undergoing breast conservation surgery (BCS) experience positive surgical margins (PSMs), defined as tumor cells present at the edge of the resection margin [7, 8]. PSMs in breast cancer carry an approximately two-fold higher risk of local recurrence and have negative prognostic implications [7,8,9,10,11]. In the occurrence of a PSM, the National Comprehensive Cancer Network (NCCN) breast cancer treatment guidelines recommend re-excision to achieve negative margins, mastectomy, or radiotherapy boost, subjecting patients to additional treatment toxicities and economic burden [6, 12,13,14].
Imaging modalities such as computed tomography, magnetic resonance imaging, single-photon emission computed tomography, and positron emission tomography are useful for diagnosis, staging, and preoperative planning. However, translation to intraoperative use is limited due to prohibitive costs and complex infrastructure [15]. Intraoperative frozen section analysis (IFSA) of resected tissues can provide information to surgeons on margin status and has been shown to reduce local recurrence rates [16]. Nevertheless, IFSA can require up to 24–27 min to obtain results, and definitive margin status is not known until after surgery. Also, IFSA can be technically challenging as benign breast has prominent adipose tissue, which does not freeze well [17,18,19]. Therefore, surgeons primarily rely on visual and tactile cues to distinguish between healthy and malignant tissue, and can unintentionally leave behind residual disease in the tumor bed [20].
Fluorescence image-guided surgery (FIGS) has the ability to provide surgeons with real-time feedback on primary tumor, residual disease, and metastatic lymph nodes, thus increasing confidence in achieving NSMs. [21, 22]. FIGS offers advantages over other imaging modalities due to high spatial resolution and the absence of ionizing radiation [20, 23]. Furthermore, in vivo imaging in the near-infrared (NIR) range (700–1000 nm) is superior to the visible spectrum due to low scattering, negligible tissue autofluorescence, and relatively high tissue penetration [24]. Currently, there are three FDA-approved fluorophores that are utilized for FIGS, with several other non-targeted and targeted fluorescent probes in clinical development [25,26,27,28]. Of the three approved fluorophores, indocyanine green (ICG) optimally emits light in the NIR range. Though currently under investigation for sentinel lymph node (SLN) and tumor detection, ICG suffers from poor aqueous stability, poor photostability, non-specific binding to proteins, concentration-dependent aggregation, and reliance on the enhanced permeability and retention (EPR) effect for accumulation in solid tumors, resulting in relatively low intraoperative tumor signal and contrast [28,29,30,31].
Recently, we reported the development of NanoICG, an ICG-loaded self-assembled HA nanoparticle. NanoICG is composed of aminopropyl-1-pyrenebutanamide (PBA) conjugated to 10 kDa HA, which drives self-assembly into HA-PBA nanoparticles through hydrophobic interactions. PBA interactions within the HA nanoparticle form hydrophobic domains [32, 33], which allow for the incorporation of hydrophobic or amphiphilic molecules, such as ICG [34], through π-π stacking [23]. NanoICG improves the intraoperative detection of breast [35, 36], pancreas [37] and prostate tumors [38] by enhancing the tumor signal-to-noise ratio (SNR) and tumor-to-muscle contrast-to-noise ratio (CNR), both of which are essential for visual guidance of tumor removal. Due to the composition, size, and serum protein interactions of NanoICG and its components, it is likely that tumor uptake of NanoICG accumulates in tumor through a combination of CD44-mediated endocytosis [39, 40], the EPR effect [41], and macropinocytosis [42]. Additionally, in vitro and in vivo safety profiles indicated that NanoICG exhibits negligible toxicity [35, 37]. While NanoICG provides strong tumor contrast, the surgical efficacy of NanoICG has not been evaluated. In order to deem suitability for effective clinical translation, we sought to assess the efficacy of NanoICG to improve the complete resection of orthotopic, syngeneic murine triple-negative breast cancer (TNBC) tumors in comparison to ICG and bright light surgery (BLS) (standard of care). Our findings ultimately revealed a marked improvement in surgical efficacy with NanoICG, demonstrating the utility of using NanoICG for FIGS of TNBC.
Materials and Methods
Preparation and Characterization of NanoICG
Indocyanine green (ICG)-loaded hyaluronic acid (HA) nanoparticles (NPs) were synthesized and characterized as previously described [35,36,37,38]. HA was conjugated to aminopropyl-1-pyrenebutanamide (PBA), and ICG was entrapped in the HA-PBA nanoparticle during self-assembly. Following formulation, the mean hydrophobic diameter, ICG concentration, and fluorescence intensity were determined with a Zetasizer Nano instrument (Malvern, Worcestershire, UK), an Evolution 220 spectrophotometer (Thermo Fischer Scientific, Madison, WI), and a FluoroMax-4 Spectrofluorometer (Horiba, Edison, NJ), respectively.
Cell Lines and Cell Culture
4T1 murine breast cancer cells were obtained from American Type Culture Collection (Manassas, VA). Both wild-type and iRFP+/luciferace (luc)+ 4T1 cells (see Electronic Supplementary Material (ESM)) were cultured in RPMI 1640 medium (Corning; Corning, NY) supplemented with 10 % fetal bovine serum (FBS) (GE Healthcare) and 100 I.U. penicillin/100 μg/ml streptomycin (P/S) (Corning). Cells were incubated at 37 °C in a humidified incubator with 5 % CO2.
Animal Models
All animal work was performed under a protocol approved by the UNMC Institutional Animal Care and Use Committee (IACUC). Tumor models were established by implanting 2.5 × 104 (early-stage breast tumor study) or 2.0 × 105 (late stage breast tumor) iRFP+/luc+ 4T1 cells suspended in 100 μl 1:1 growth media:Matrigel (Corning) into the right mammary fat pads of 8 week-old female BALB/c mice (Jackson Laboratory; Bar Harbor, ME). Tumors were allowed to grow four (early-stage breast tumor study) or six (late-stage breast tumor study) days prior to survival surgery.
Bioluminescence Imaging
Bioluminescence imaging (BLI) was utilized to track tumor development and monitor mice for residual disease, local recurrence, and secondary tumor metastases after tumor resection surgery. One day prior to imaging, fur was removed with clippers and depilatory cream. Mice bearing orthotopic iRFP+/luc+ 4T1 breast tumors were injected with 150 mg/kg D-Luciferin (Gold Biotechnology; St. Louis, MO) via intraperitoneal administration. Approximately 10 min after injection, mice were placed under anesthesia via inhalation of isoflurane (1–3 % in O2) (Piramel Group; Mumbai, India). Anesthetized animals were transferred to an IVIS Spectrum in vivo imaging system (PerkinElmer Inc.; Waltham, MA) where bioluminescent (BL) signal and brightfield images were captured. After imaging, mice were returned to cages where they were monitored until regaining full consciousness. The area of the BL signal was measured using the ROI tool in ImageJ software (NIH; Bethesda, MD).
Survival Surgery and Study Design
Tumor resection surgery was performed on female BALB/c mice bearing orthotopic iRFP+/luc+ tumors. Mice were randomly distributed into four experimental groups: (1) bright light surgery (BLS), (2) sham + no contrast (phosphate-buffered saline, PBS), (3) ICG (20 nmol), and (4) NanoICG (20 nmol). Contrast agents (ICG or NanoICG; prepared in ultrapure water) or PBS (sham) were administered via tail vein 24 h prior to surgery in a volume of 100 μl. Previous studies indicated that intraoperative imaging 24 h post intravenous injection was optimal for ICG and NanoICG [36, 43,44,45]. The operating table was sterilized and covered with sterile drapes prior to surgery. A TP700 TPump Professional Core Warming and Cooling System (Stryker Corp.; Kalamazoo, MI) was used to maintain the operating table at 37 °C. Mice were anesthetized via inhalation of isoflurane (1–3 % in O2). Once unconscious, the animals were moved to the surgical stage where the incision area was sterilized by alternating swabbing with 70 % EtOH and 10 % povidone iodine solution by an assistant.
Surgery was conducted under white light (BLS) or the Lab-FLARE RP1 FIGS system (FIGSS) (Curadel; Marlborough, MA) (sham, ICG, and NanoICG groups). The color, 800 nm, and merge (cyan pseudo color) channels were utilized for image-guidance. The surgical assistant was responsible for the operation of the surgery system, acquisition of intraoperative images, and adjustment of exposure, brightness, and contrast parameters. Resection with BLS was determined by visual and tactile cues of malignant tissue, while only contrast-enhanced tissue was removed in the sham, ICG, and NanoICG groups. Surgery was deemed complete when identifiable malignant tissue (BLS) or contrast-enhanced tissue (sham, ICG, and NanoICG) no longer remained in the surgical cavity. Resected tissues were imaged with the color, 700 nm, and 800 nm channels of a Pearl Trilogy Small Animal Imaging System and images were analyzed with Image Studio Ver. 5.0 software (LI-COR Biosciences; Lincoln, NE).
The skin incision was closed with Ethilon® 5–0 nylon sutures (Ethicon, Inc.; Somerville, NJ). Mice were removed from anesthesia, and observed until full consciousness was regained. Mice were injected with 0.1 mg/kg buprenorphine (Reckitt Benckiser Healthcare; Hull, UK) via intraperitoneal injection twice daily, up to 3 days post-surgery. After a 3-day recovery period, mice were monitored for the presence of residual tumor and tumor growth with BLI and measurement of tumor size with calipers. Mice were removed from the study if they reached one of the following endpoint criteria: (1) 28-day tumor-free survival, (2) tumor volume ≥ 1000 mm3, (3) tumor size ≥1 cm in any direction, or (4) skin ulceration (Fig. 1).
Calculation of Tumor-to-Background, Signal-to-Noise Ratios
Intraoperative tumor-to-background ratio (TBR) and SNR were calculated with intraoperative images from the Lab-FLARE RP1 system (Curadel) or post-surgical images from the Pearl Trilogy Small Animal Imaging System, respectively. Mean fluorescence intensity (MFI) values of the tumor and tumor bed were calculated using an intraoperative image obtained with the 800 nm channel from the Lab-FLARE RP1 system on ImageJ software. TBR was calculated as the ratio of MFITumor/MFITumor Bed. SNR was calculated by (Average Intensity Per PixelTumor)/(SDBackground) [36]. A region of interest (ROI) was manually generated around each of the suspected tumors (800 nm channel) with Image Studio software.
Histological Analysis
Resected tissues were stained with black tissue marking dye (Cancer Diagnostics; Durham, NC), placed in containers prefilled with buffered 10 % formalin (Azer Scientific; Morgantown, PA), and fixed for 24 h. Formalin was replaced with 70 % EtOH following fixation. Samples were paraffin-embedded and blocks were cut into 4 μm-thick sections and mounted onto slides. H&E-stained slides were analyzed by a board-certified pathologist (SRL) blinded to the experimental groups and results of surgical resection. The pathologist was responsible for determining presence of tumor, margin status, and general characteristics of resected tissues.
Statistical Analysis
Statistical analysis was performed with GraphPad Prism 8 software (Version 8.0.1) (GraphPad Software; San Diego, CA). Chi-square analysis was used to assess rates of residual disease after surgery. The Kaplan-Meier method was used to create survival plots and log-rank (Mantel-Cox) tests were used to assess statistical significance. A two-tailed unpaired T-test was used to analyze differences in TBR and SNR.
Results
NanoICG Characterization and In Vitro Imaging
Physicochemcial properties of NanoICG characterization were consistent with past studies [35,36,37,38] and are reported in Suppl. Fig. 1 (see ESM). In the absence of the HA nanoparticle, free ICG exhibits poor tumor uptake and retention. This was illustrated with 2D monolayer culture and 3D spheroid culture of 4T1 cells, where fluorescence microscopy revealed minimal uptake of free ICG. However, when treated with NanoICG, 4T1 cells exhibited a strong NIR signal in both monolayer and 3D spheroid culture, indicating that the HA nanoparticle improved the efficiency of ICG uptake into tumor cells (Suppl. Fig. 2, see ESM).
Overall surgical efficacy study results. Waterfall plots depict percent change in BL signal area at a 3 days and b 28 days post-surgery. “#” indicates mouse with known chest wall invasion. This subject was removed from study. c Residual disease status after resection. d Kaplan-Meier curve. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
Study Design and Overall Results
The surgical efficacy of NanoICG compared to ICG, BLS (procedure control), and a sham control, which included NIR fluorescence (NIRF) imaging but no contrast agent to control for device bias, were evaluated in an orthotopic, syngeneic murine model of TNBC. There were no observed abnormalities with tumor growth. Female Balb/c mice bearing orthotopic iRFP+/luc+ 4 T1 tumors were used for each group (N = 9), except for NanoICG (N = 8). One mouse was excluded from the NanoICG group due to the presence of chest wall invasion, a clinicopathologic feature that is uncharacteristic of early-stage breast cancer [12]. There were no adverse events attributed to the administration of contrast agent, nor were there complications during surgery.
Effects of FIGS on Resection and Post-Surgical Survival
BLI was utilized to track tumor growth before surgery and residual disease after surgery. The percent change in baseline after tumor resection surgery was calculated at 3 and 28 days post-surgery using pre-surgery BLI as baseline measurements. A 100 % reduction in BL signal area indicated a complete resection, while reductions in BL signal <100 % indicated the presence of residual disease (Fig. 2a and b). Complete resections were observed in 7/8 mice treated with NanoICG. In contrast, 0/9 mice from the sham group, 2/9 mice from the BLS group and 3/9 mice in the ICG group experienced complete resections. Additionally, there were 4 instances (2 BLS, 2 ICG) where tumor was not detectible with contrast-enhancement (ICG) or with visualization and palpation (BLS). In these cases, little-to-no tissue was removed, resulting in an increase in tumor size after surgery. All mice with residual disease reached endpoint criteria prior to 28 days, thus failing to reach 28-day survival (Fig. 2a and b and Fig. 3). NanoICG improved complete resection compared to sham (P = 0.0003), BLS (P = 0.0071), and ICG (P = 0.0235) (Fig. 3c). Furthermore, it was found that NanoICG prolonged survival versus sham (P < 0.0001), BLS (P = 0.0041), and ICG (P = 0.0173), with 87.5 % (7/8) of mice reaching 28-day tumor-free survival. Although no significant differences were observed between ICG versus BLS (P = 0.2246) and BLS versus sham (P = 0.0992), ICG significantly prolonged survival versus sham (P = 0.0036) (Fig. 2d, Tables 1-2).
Assessment of Intraoperative NIRF Signal
NanoICG improved the completeness of tumor resection and, as a direct result, prolonged survival versus ICG, sham and BLS. Partially contributing to this result was the improved NIRF signal provided by NanoICG. NanoICG had a mean intraoperative TBR of 3.92 ± 2.25, which was significantly higher than ICG (1.96 ± 1.53) (P = 0.0466). This was consistent with visual observations during FIGS: NanoICG provided strong contrast enhancement and more readily identified tumor in the merge and 800 nm channels. On the contrary, ICG provided poor intraoperative contrast enhancement and thus, tumors were only identified in the 800 nm channel with weak fluorescence, making it difficult to fully resect the tumors (Fig. 4a). The improved intraoperative TBR of NanoICG allowed for the consistent identification of malignant tissue in the tumor bed and differentiation from normal healthy tissue.
NIRF signal of tumors resected with FIGS. Intraoperative images were obtained with the lab-FLARE RP1. a Intraoperative TBR of primary tumor was quantified using the adjacent tumor bed as reference tissue. b All resected tissues were imaged with the pearl trilogy small animal imaging system and were utilized to quantify SNR values of the 800 nm channel. Color bar values are reported in AU. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
In addition to identifying a greater number of lesions with contrast-enhancement than ICG (NNanoICG = 38; NICG = 19), it was found that NanoICG had an average SNR of 10.43 ± 6.20, significantly greater than the average SNR (800 nm channel) of tissues resected from the ICG group (3.73 ± 3.12) (P < 0.0001) (Fig. 4b). The superior intraoperative contrast-enhancement provided by NanoICG directly influenced the extent of resection and afforded the opportunity to identify and excise primary tumors and small, occult lesions. As a result, a greater number of complete resections were observed after surgery in the NanoICG group versus ICG and BLS (Fig. 2c).
Identification of Small, Occult Lesions
Although both NanoICG and ICG were effective at identifying the primary tumor with NIRF contrast-enhancement in the majority of cases, NanoICG exhibited a superior ability for the detection of small, occult lesions beyond the primary tumor. This is evident in Fig. 5a (red arrows) where a small, contrast-enhanced lesion was removed and placed on the skin adjacent to the tumor bed. Additionally, at the 12 o’clock position of the tumor bed (red boxes), small, occult lesions can be visualized with contrast-enhancement in the 800 nm channel. These lesions were otherwise non-palpable and non-visually-identifiable. ICG did not provide sufficient contrast-enhancement to identify additional lesions in the tumor bed (Fig. 5b). These findings were reflected in observations of the total number of resected specimens that were suspicious for tumor in each group: NNanoICG = 38, NICG = 19, NBLS = 17 (Fig. 5c).
Identification of small, occult lesions with FIGS. Color and 800 nm images were obtained during FIGS with the lab-FLARE RP1 FIGSS. Resected tissues were imaged with the pearl trilogy small animal imaging system to confirm presence of tumor with iRFP and 800 nm fluorescence. a, b Red boxes and arrows indicate tissues resected with NanoICG guidance. c Imaging was analyzed with image studio software with which tumor areas were calculated. H&E-stained sections of resected tissues were analyzed by a board-certified pathologist. Primary tumor from. d The NanoICG group was recognized as tumor (main image: 400×, scale bar = 20 μm; inset image: 40×, scale bar = 200 μm; green arrow indicates area of 400× image). Representative examples of e small, occult lesions that were classified as “tumor-positive” (main image: 400×, scale bar = 20 μm; inset image: 100×, scale bar = 100 μm; green arrow indicates area of 400× image) or f “Tumor-negative” (100×, scale bar = 100 μm). Color bar values are reported in AU.
To further investigate the detection of small lesions, resected tissues were imaged with the 700 nm (iRFP) and 800 nm (NanoICG or ICG) channels of the Pearl Trilogy Small Animal Imaging System. While 4T1 cells expressed iRFP, it was noted that there was autofluorescence from normal tissue specimens, e.g., adipose tissue and skin. Additionally, consistent with Fig. 5, resected tissues from the NanoICG group exhibited greater 800 nm NIRF signal than resected tissues from the ICG group. These images were further analyzed by calculating the area of tissues identified by NIRF. As expected, no significant differences in the average areas of resected tissues were observed among the groups (P > 0.05). However, of note, the average size of tissues identified with NanoICG (4.39 ± 4.42 mm2; size range = 0.17–14.70 mm2) encompassed a much wider range, particularly smaller masses, than ICG (3.61 ± 2.87 mm2; size range = 0.56–9.18 mm2) or BLS (4.33 ± 3.43; size range = 0.82–10.36 mm2). This data suggests that NanoICG has a greater versatility at detecting a large size range of lesions with NIRF than ICG and BLS.
Finally, NanoICG has a higher sensitivity to detect small, occult tumors compared to ICG or BLS. As depicted in Fig. 5c, NanoICG detected a greater number of submillimeter lesions than ICG or BLS (NNanoICG = 12, NICG = 2, NBLS = 2). These lesions were positive for iRFP fluorescence and retained ICG fluorescence after resection. Interestingly, several small lesions detected with fluorescence were as small as the 4T1 tumor spheroids (Suppl. Fig. 3, see ESM). After post-resection imaging, H&E-stained sections of resected tissues were assessed by a board-certified pathologist. Histological analysis confirmed the presence of cancer in all resected primary tumors (Fig. 5d). The majority of resected submillimeter lesions contained tumor (Fig. 5e). Taken together, these lesions were non-palpable, non-visually obvious, and would have otherwise gone unnoticed with traditional means of identification, potentially leading to a residual disease-positive post-surgical outcome. All larger sections (size >1 mm) enhanced by NanoICG that were assessed with histopathology were deemed tumor positive. In 4/10 sections (from 3 mice) with submillimeter resected specimens, NanoICG indicated the presence of tumor, while pathology was inconclusive as to the presence of tumor. An example of a potential false-positive is shown in Fig. 5f, which is suggestive of normal adipose tissue. Future studies will investigate true/false positive and true/false negative rates on a larger population. Ultimately, the combination of (1) improved contrast-enhancement and (2) the identification of small, occult lesions, was critical in NanoICG improving complete tumor resection and prolonging survival versus sham, ICG, and BLS.
Prediction of Residual Disease with NIRF in a Model of Locally-Advanced Breast Cancer
The aforementioned surgical efficacy study was performed in an orthotopic, syngeneic model of early stage breast cancer. The area of primary tumor was relatively contained to the mammary fat pad (except for small, occult lesions that occasionally grew in chest wall tissue). Therefore, this was representative of stage I or II breast cancer [12]. However, surgery is still utilized in stage III breast cancer [1]. A hallmark feature of stage IIIB breast cancer is a T4 tumor status, namely, extension of the tumor into the chest wall and/or the skin [12]. We sought to establish a model of locally-advanced breast cancer with chest wall invasion by increasing the amount and volume of tumor cells injected into the mammary fat pad: 2 × 105 iRFP+/luc+ 4T1 cells in 100 μl 1:1 media:Matrigel. As a result of this modification, tumors rapidly progressed, were large in size, and consistently invaded into the chest wall.
Consistent with the previous surgical efficacy study, it was observed that the primary tumor was identified intraoperatively with fluorescence or visualization/palpation, and subsequently resected. In the case of BLS and ICG, almost all resections resulted in the absence of residual tumor or fluorescence in the surgical cavity, respectively. However, after resection of the primary tumor, NanoICG exhibited the ability to detect remaining invasive disease with fluorescence. Complete resection of locally invasive cancer in this mouse model was difficult due the close proximity of tumors to the subclavian vessels and jugular vein and high blood perfusion in the muscle, which led to hemorrhaging and placing mice at risk for death from exsanguination. Therefore, residual invasive disease that was detected with fluorescence was not resected and left in the surgical cavity. In every case (6/6) that NanoICG identified unresectible invasive disease during surgery, presence of residual tumor was detected with BLI after surgery. Interestingly, the fluorescence location from NanoICG was consistent with the location of recurrence (Fig. 6c). Thus, NanoICG could identify unresectable invasive tumors and also predict the extent and location of residual disease after surgery. On the contrary, almost all cases of residual disease-positive outcomes after surgery in the BLS (4/4 mice) and ICG (6/7 mice) groups did not show signs of residual disease presence during surgery (Fig. 6a and b).
Discussion
Although past studies of NanoICG have demonstrated the ability to improve tumor contrast, none have explored the effects on complete resection and survival after NanoICG administration [35,36,37,38]. The present study sought to investigate the translatability of NanoICG by assessing surgical efficacy in a clinically-relevant study. An early-stage model of TNBC was chosen due to its prominence in oncological surgery [1]. In this model, the NanoICG group experienced a greater number of complete tumor resections compared to ICG, BLS, or sham, which directly led to prolonged survival. On the contrary, the majority of mice in the ICG and BLS groups did not reach 28-day tumor-free survival (Fig. 2, Tables 1-2). Although ICG was ineffective at identifying residual tumor in the surgical cavity, it was still effective at identifying the primary tumor with weak contrast-enhancement. Therefore, ICG significantly-prolonged survival versus sham. Nevertheless, these findings ultimately demonstrated that NanoICG is efficacious as an optical contrast agent for the FIGS of breast cancer.
Directly contributing to improved resection status and prolonged survival was superior intraoperative contrast provided by NanoICG. In vitro studies demonstrated that addition of NanoICG to 4T1 cells in 2D monolayer or 3D tumor spheroid culture led to uptake in almost all cells, resulting in high NIR signal. In contrast, cells treated with free ICG, showed minimal fluorophore uptake, and thus minimal NIR fluorescent signal (Suppl. Fig. 1, see ESM). Consistent with in vitro findings, suspected tumors resected from the NanoICG group presented significantly greater NIR contrast enhancement with open-field TBR and closed-field SNR versus ICG (Fig. 4a-b). These measurements are especially important in assessing the ability of a contrast agent to provide tumor-specific contrast enhancement. Having a TBR sufficient in aiding the surgeon to differentiate tumor from healthy tissue is critical in preclinical and clinical testing [46, 47]. It is generally accepted that a TBR >3.0 is a sufficient to provide adequate intraoperative contrast in preclinical FIGS studies and is essential for successful clinical translation [22]. In the current study, it was found that NanoICG provided a TBR >3.0 in 7/9 cases while ICG only exceed this threshold in 1/9 cases, regardless of whether resected tissue originated from the primary tumor or small, occult lesions (Figs. 4 and 5). Though intraoperative TBR was only calculated for the primary tumors, SNRs of all resected tissues were calculated and demonstrated that NanoICG provides superior contrast enhancement of tumor (Fig. 4b). By these standards, NanoICG can potentially be a candidate for clinical investigation.
Novel fluorescent probes currently under clinical investigation have been found to intraoperatively detect submillimeter, microscopic tumors. The heightened sensitivity to detect lesions containing a small number of tumor cells plays a major role in identifying difficult-to-detect lesions [48,49,50,51]. Similar to the aforementioned probes, NanoICG improved the detection of small, occult, submillimeter lesions beyond the primary tumor. These lesions were invisible to the naked eye, would have otherwise gone unnoticed by traditional means of detection, and left unresected in the tumor bed (Fig. 5). This finding is clinically relevant because nonpalpable breast cancer lesions pose as a significant hurdle to surgeons. The management of these lesions often requires surgeons to rely on assistance from radiologists and wire-guided localization (WGL), where re-excision rates fall between 10 and 43 % [52]. Furthermore, though detectible with mammographic abnormalities, microinvasive breast cancer, defined as invasive carcinoma measuring less than 1 mm (T1mic tumor status), is challenging to detect intraoperatively [12, 53, 54].
In a few specimens, NanoICG falsely identified tissue that was ultimately tumor cell-negative. Though initial ex vivo fluorescence imaging suggested that these lesions contained tumor due to iRFP fluorescence (Fig. 5a), histological analysis by a board-certified pathologist indicated that some resected submillimeter lesions were actually normal adipose or skin tissue (Fig. 5f). There are two possible explanations for this observation. (1) Although unlikely, it is possible that tumor cells were not captured in the histological section that was assessed by the pathologist. Tumor cells may have been present in another plane of tissue and, thus the cells responsible for the iRFP fluorescence observed with ex vivo imaging were not present during histological analysis. Additionally, some samples that were deemed “tumor-negative” were still questionable and could still have contained a small number of tumor cells. (2) NanoICG could detect small lesions with high sensitivity. However, because these lesions were not always found to be tumor, the specificity of NanoICG decreases at submillimeter sizes. This could have been a product of tissue autofluorescence [55, 56] or retention in the skin/adipose tissue due to the lipophilic nature of ICG (logP = 6.05) [57, 58]. Nevertheless, the ability to detect submillimeter lesions with high sensitivity was a major contributing factor to the improved complete resection rate and prolonged post-operative survival afforded by NanoICG when compared to ICG or BLS. From a clinical standpoint, false-positives are more acceptable than false-negatives because there is a lesser possibility of leaving residual tumor in the surgical bed [25, 59]. Ultimately, it is not the primary tumor that goes undetected by the surgeon, but it is the detection of non-palpable and small, occult lesions that is critical in obtaining clear surgical margins [25, 48, 60,61,62]. Therefore, by intraoperatively delineating difficult-to-detect lesions, NanoICG has the potential to improve NSM rates after BCS.
Although surgical resection is primarily associated with early-stage breast cancer, it is also utilized in 86 % (21 % BCS, 65 % mastectomy) of stage III breast cancer [1]. Therefore, in order to assess the efficacy of NanoICG in stage III breast cancer, we established a model of locally-invasive breast cancer. It was found that NanoICG consistently identified chest wall invasion in the surgical cavity while BLS and ICG failed to do so (Fig. 6a-c). In the clinical scenario of late-stage breast cancer, NCCN guidelines recommend irradiation (to the chest wall, infraclavicular region, supraclavicular area, internal mammary nodes, and/or any part of the axillary bed at risk) after BCS or mastectomy [12]. Given the observations of this study, NanoICG could identify the presence, extent, and location of invasive disease during surgery and serve as an aid to inform post-operative treatment decisions. Therefore, in addition to serving as an optical contrast for the fluorescence-guided resection of early-stage breast cancer, NanoICG could potentially be of value for the treatment of late-stage invasive breast cancer.
Currently, there are several contrast agents that are undergoing clinical investigation for the intraoperative detection of breast cancer, including activatable probes, peptides, and antibody-dye conjugates [63]. NanoICG offers comparable contrast and survival benefit to these clinical candidates. With the exception of the nanoparticle, ONM-100 [48], all other probes under clinical investigation use dyes other than ICG. Although these dyes may offer superior optical performance and versatility (i.e., conjugatability), they are not FDA-approved and most FIGSS are optimized for ICG detection [64]. Additionally, due to its composition and size, NanoICG can be potentially used in a variety of cancers with active (CD44) and passive (EPR effect) targeting. Nevertheless, in order to advance into the clinic, further investigation on the sensitivity and specificity of NanoICG is necessary to determine where it falls in comparison with clinical-stage contrast agents.
There were several limitations in the current study. Although an orthotopic model of TNBC was used, it was difficult to establish an anatomically-accurate model of breast cancer. While mice undergo mammary development, they lack the anatomical and histological complexity of a human breast [65]. By this regard, it was difficult to accurately represent breast tumor growth and perform tumor resection surgery with a surgical margin. In reality, the murine model of TNBC displayed tumor growth that was either very small and localized to the mammary fat pad or extended beyond the mammary fat pad and, at times, invaded into the skin and/or chest wall. This made it difficult to obtain a histopathological PSM even though the result after surgery was “residual disease-negative” (Fig. 5d-e, tumor cells abut the inked margin). Therefore, future studies that focus on more anatomically relevant models of breast cancer could be beneficial. Additionally, the 4 T1 model is a syngeneic model of TNBC, which only comprises approximately 15 % of all human breast cancer cases [66]. Investigating the use of NanoICG as a contrast agent for other molecular subtypes of breast cancer (HER2, luminal A, and luminal B) would increase its clinical applicability.
Conclusions
NanoICG is an ICG-loaded self-assembled HA nanoparticle that improves the delivery of the NIR dye, ICG, to solid tumors. The current study built these previous findings and assessed the efficacy of NanoICG as a contrast agent for FIGS in a syngeneic, orthotopic model of breast cancer. It was demonstrated that NanoICG was efficacious as a contrast agent for FIGS by not only improving complete resections of breast tumors, but also improving post-surgical survival versus ICG, BLS (standard of care), and sham (control). These results are supported by the superior intraoperative contrast provided by NanoICG and the ability to identify small, occult lesions. This, in combination with its ability to detect locally-invasive breast cancer and predict recurrence, demonstrates the clinical utility of NanoICG.
References
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A (2016) Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 66:271–289
Nayyar A, Gallagher KK, McGuire KP (2018) Definition and Management of Positive Margins for invasive breast Cancer. Surg Clin North Am 98:761–771
Meric F, Mirza NQ, Vlastos G, Buchholz TA, Kuerer HM, Babiera GV, Singletary SE, Ross MI, Ames FC, Feig BW, Krishnamurthy S, Perkins GH, McNeese M, Strom EA, Valero V, Hunt KK (2003) Positive surgical margins and ipsilateral breast tumor recurrence predict disease-specific survival after breast-conserving therapy. Cancer 97:926–933
Houssami N, Macaskill P, Luke Marinovich M, Morrow M (2014) The association of surgical margins and local recurrence in women with early-stage invasive breast cancer treated with breast-conserving therapy: a meta-analysis. Ann Surg Oncol 21:717–730
Morrow M, Van Zee KJ, Solin LJ et al (2016) Society of Surgical Oncology–American Society for Radiation Oncology–American Society of Clinical Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in ductal carcinoma in situ. Pract Radiat Oncol 6:287–295
Orosco RK, Tapia VJ, Califano JA, Clary B, Cohen EEW, Kane C, Lippman SM, Messer K, Molinolo A, Murphy JD, Pang J, Sacco A, Tringale KR, Wallace A, Nguyen QT (2018) Positive surgical margins in the 10 Most common solid cancers. Sci Rep 8:5686. https://doi.org/10.1038/s41598-018-23403-5
Morrow M, Jagsi R, Alderman AK, Griggs JJ, Hawley ST, Hamilton AS, Graff JJ, Katz SJ (2009) Surgeon recommendations and receipt of mastectomy for treatment of breast cancer. JAMA 302:1551–1556
Marinovich ML, Azizi L, Macaskill P et al (2016) The Association of Surgical Margins and Local Recurrence in women with ductal carcinoma in situ treated with breast-conserving therapy: a meta-analysis. Ann Surg Oncol 23:3811–3821
Davidson N, Gelber R, Piccart M et al (2010) Overview of the randomized trials of radiotherapy in ductal carcinoma in situ of the breast. J Natl Cancer Inst - Monogr 41:162–177
Donker M, Litière S, Werutsky G, Julien JP, Fentiman IS, Agresti R, Rouanet P, de Lara CT, Bartelink H, Duez N, Rutgers EJ, Bijker N (2013) Breast-conserving treatment with or without radiotherapy in ductal carcinoma in situ: 15-year recurrence rates and outcome after a recurrence, from the EORTC 10853 randomized phase III trial. J Clin Oncol 31:4054–4059
Moran MS, Schnitt SJ, Giuliano AE, Harris JR, Khan SA, Horton J, Klimberg S, Chavez-MacGregor M, Freedman G, Houssami N, Johnson PL, Morrow M, Society of Surgical Oncology., American Society for Radiation Oncology. (2014) Society of Surgical Oncology-American Society for Radiation Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. J Clin Oncol 32:1507–1515
(2018) NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines® ): Breast Cancer
Smith BD, Jiang J, Shih Y-C et al (2017) Cost and complications of local therapies for early-stage breast Cancer. J Natl Cancer Inst 109:djw178. https://doi.org/10.1093/jnci/djw178
Greenup RA, Camp MS, Taghian AG, Buckley J, Coopey SB, Gadd M, Hughes K, Specht M, Smith BL (2012) Cost comparison of radiation treatment options after lumpectomy for breast Cancer. Ann Surg Oncol 19:3275–3281
Frangioni JV (2008) New technologies for human cancer imaging. J Clin Oncol 26:4012–4021
Olson TP, Harter J, Muñoz A, Mahvi DM, Breslin T (2007) Frozen section analysis for intraoperative margin assessment during breast-conserving surgery results in low rates of re-excision and local recurrence. Ann Surg Oncol 14:2953–2960
Jorns JM, Visscher D, Sabel M, Breslin T, Healy P, Daignaut S, Myers JL, Wu AJ (2012) Intraoperative frozen section analysis of margins in breast conserving surgery significantly decreases reoperative rates: one-year experience at an ambulatory surgical center. Am J Clin Pathol 138:657–669
Chiappa C, Rovera F, Corben AD et al (2013) Surgical margins in breast conservation. Int J Surg 11:S69–S72
Kennedy GT, Okusanya OT, Keating JJ, Heitjan DF, Deshpande C, Litzky LA, Albelda SM, Drebin JA, Nie S, Low PS, Singhal S (2015) The optical biopsy: a novel technique for rapid intraoperative diagnosis of primary pulmonary adenocarcinomas. Ann Surg 262:602–609
Vahrmeijer AL, Hutteman M, van der Vorst JR, van de Velde C, Frangioni JV (2013) Image-guided cancer surgery using near-infrared fluorescence. Nat Rev Clin Oncol 10:507–518
Maloney BW, McClatchy DM, Pogue BW, Paulsen KD, Wells WA, Barth RJ (2018) Review of methods for intraoperative margin detection for breast conserving surgery. J Biomed Opt 23:1–19. https://doi.org/10.1117/1.JBO.23.10.100901
Tummers WS, Warram JM, van den Berg NS, Miller SE, Swijnenburg RJ, Vahrmeijer AL, Rosenthal EL (2018) Recommendations for reporting on emerging optical imaging agents to promote clinical approval. Theranostics 8:5336–5347
Hill TK, Mohs AM (2016) Image-guided tumor surgery: will there be a role for fluorescent nanoparticles? Wiley Interdiscip Rev Nanomedicine Nanobiotechnology 8:498–511
Hong G, Antaris AL, Dai H (2017) Near-infrared fluorophores for biomedical imaging. Nat Biomed Eng 1:0010. https://doi.org/10.1038/s41551-016-0010
Tipirneni KE, Warram JM, Moore LS, Prince AC, de Boer E, Jani AH, Wapnir IL, Liao JC, Bouvet M, Behnke NK, Hawn MT, Poultsides GA, Vahrmeijer AL, Carroll WR, Zinn KR, Rosenthal E (2017) Oncologic procedures amenable to fluorescence-guided surgery. Ann Surg 266:36–47
Tringale KR, Pang J, Nguyen QT (2018) Image-guided surgery in cancer: a strategy to reduce incidence of positive surgical margins. Wiley Interdiscip rev Syst biol med 1–18
Zhang RR, Schroeder AB, Grudzinski JJ, Rosenthal EL, Warram JM, Pinchuk AN, Eliceiri KW, Kuo JS, Weichert JP (2017) Beyond the margins: real-time detection of cancer using targeted fluorophores. Nat Rev Clin Oncol 14:347–364
Olson MT, Ly QP, Mohs AM (2019) Fluorescence guidance in surgical oncology: challenges, opportunities, and translation. Mol Imaging Biol 21:200–218
Schaafsma BE, Mieog JSD, Hutteman M, van der Vorst J, Kuppen PJ, Löwik CW, Frangioni JV, van de Velde C, Vahrmeijer AL (2011) The clinical use of indocyanine green as a near-infrared fluorescent contrast agent for image-guided oncologic surgery. J Surg Oncol 104:323–332
Alander JT, Kaartinen I, Laakso A et al (2012) A review of Indocyanine green fluorescent imaging in surgery. Int J Biomed Imaging 2012:1–26
Wang H, Li X, Tse BW-C et al (2018) Indocyanine green-incorporating nanoparticles for cancer theranostics. Theranostics 8:1227–1242
Payne WM, Svechkarev D, Kyrychenko A, Mohs AM (2018) The role of hydrophobic modification on hyaluronic acid dynamics and self-assembly. Carbohydr Polym 182:132–141
Svechkarev D, Kyrychenko A, Payne WM, Mohs AM (2018) Probing the self-assembly dynamics and internal structure of amphiphilic hyaluronic acid conjugates by fluorescence spectroscopy and molecular dynamics simulations. Soft Matter 14:4762–4771
Mishra A, Behera RK, Behera PK, Mishra BK, Behera GB (2000) Cyanines during the 1990s: a review. Chem Rev 100:1973–2011. https://doi.org/10.1021/cr990402t
Hill TK, Abdulahad A, Kelkar SS, Marini FC, Long TE, Provenzale JM, Mohs AM (2015) Indocyanine green-loaded nanoparticles for image-guided tumor surgery. Bioconjug Chem 26:294–303
Hill TK, Kelkar SS, Wojtynek NE et al (2016) Near infrared fluorescent nanoparticles derived from hyaluronic acid improve tumor contrast for image-guided surgery. Theranostics 6:2314–2328
Qi B, Crawford AJ, Wojtynek NE et al (2018) Indocyanine green loaded hyaluronan-derived nanoparticles for fluorescence-enhanced surgical imaging of pancreatic cancer. Nanomedicine Nanotechnology, Biol Med 14:769–780
Souchek JJ, Wojtynek NE, Holmes MB et al (2018) Optimized hyaluronic acid formulation of near infrared fluorophores for surgical detection of a prostate tumor xenograft. Acta Biomater 75:1–28
Kelkar SS, Hill TK, Marini FC, Mohs AM (2016) Near infrared fluorescent nanoparticles based on hyaluronic acid: self-assembly, optical properties, and cell interaction. Acta Biomater 36:112–121
Bhattacharya DS, Svechkarev D, Souchek JJ, Hill TK, Taylor MA, Natarajan A, Mohs AM (2017) Impact of structurally modifying hyaluronic acid on CD44 interaction. J Mater Chem B 5:8183–8192
Yashuhiro M, Maeda H (1986) Tumor-selective delivery of macromolecular drugs via the EPR effect: background and future prospects. Cancer Res 46:6387–6392
Park JH, Oh N (2014) Endocytosis and exocytosis of nanoparticles in mammalian cells. Int J Nanomedicine 9:51–63
Newton AD, Predina JD, Corbett CJ, Frenzel-Sulyok LG, Xia L, Petersson EJ, Tsourkas A, Nie S, Delikatny EJ, Singhal S (2019) Optimization of second window Indocyanine green for intraoperative near-infrared imaging of thoracic malignancy. J Am Coll Surg 228:188–197
Keating J, Tchou J, Okusanya O, Fisher C, Batiste R, Jiang J, Kennedy G, Nie S, Singhal S (2016) Identification of breast cancer margins using intraoperative near-infrared imaging. J Surg Oncol 113:508–514
Zeh R, Sheikh S, Xia L, Pierce J, Newton A, Predina J, Cho S, Nasrallah M, Singhal S, Dorsey J, Lee JYK (2017) The second window ICG technique demonstrates a broad plateau period for near infrared fluorescence tumor contrast in glioblastoma. PLoS One 12:e0182034. https://doi.org/10.1371/journal.pone.0182034
Koller M, Qiu S-Q, Linssen MD et al (2018) Implementation and benchmarking of a novel analytical framework to clinically evaluate tumor-specific fluorescent tracers. Nat Commun 9:3739
Hoogstins C, Burggraaf JJ, Koller M, Handgraaf H, Boogerd L, van Dam G, Vahrmeijer A, Burggraaf J (2019) Setting standards for reporting and quantification in fluorescence-guided surgery. Mol Imaging Biol 21:11–18
Zhao T, Huang G, Li Y et al (2017) A transistor-like pH nanoprobe for tumour detection and image-guided surgery. Nat Biomed Eng 1:1–8
Prince AC, Jani A, Korb M, Tipirneni KE, Kasten BB, Rosenthal EL, Warram JM (2017) Characterizing the detection threshold for optical imaging in surgical oncology. J Surg Oncol 116:898–906
Prince AC, McGee AS, Siegel H, Rosenthal EL, Behnke NK, Warram JM (2018) Evaluation of fluorescence-guided surgery agents in a murine model of soft tissue fibrosarcoma. J Surg Oncol 117:1179–1187
Yano S, Takehara K, Miwa S, Kishimoto H, Tazawa H, Urata Y, Kagawa S, Bouvet M, Fujiwara T, Hoffman RM (2016) Fluorescence-guided surgery of a highly-metastatic variant of human triple-negative breast cancer targeted with a cancer-specific GFP adenovirus prevents recurrence. Oncotarget 7:75635–75647
Ahmed M, Rubio IT, Klaase JM, Douek M (2015) Surgical treatment of nonpalpable primary invasive and in situ breast cancer. Nat Rev Clin Oncol 12:645–663
Margalit DN, Sreedhara M, Chen Y-H et al (2013) Microinvasive breast cancer: ER, PR, and HER-2/neu status and clinical outcomes after breast-conserving therapy or mastectomy. Ann Surg Oncol 20:811–818
Vieira CC, Mercado CL, Cangiarella JF, Moy L, Toth HK, Guth AA (2010) Microinvasive ductal carcinoma in situ: clinical presentation, imaging features, pathologic findings, and outcome. Eur J Radiol 73:102–107
Tummers QRJG, Hoogstins CES, Gaarenstroom KN et al (2016) Intraoperative imaging of folate receptor alpha positive ovarian and breast cancer using the tumor specific agent EC17. Oncotarget 7:32144–32155
Andreou C, Neuschmelting V, Tschaharganeh DF, Huang CH, Oseledchyk A, Iacono P, Karabeber H, Colen RR, Mannelli L, Lowe SW, Kircher MF (2016) Imaging of liver tumors using surface-enhanced Raman scattering nanoparticles. ACS Nano 10:5015–5026
Kraft JC, Ho RJY (2014) Interactions of Indocyanine green and lipid in enhancing near-infrared fluorescence properties: the basis for near-infrared imaging in vivo. Biochemistry 53:1275–1283
Yanina IY, Tuchin VV, Navolokin NA, Matveeva OV, Bucharskaya AB, Maslyakova GN, Altshuler GB (2012) Fat tissue histological study at indocyanine green-mediated photothermal/photodynamic treatment of the skin in vivo. J Biomed Opt 17:058002. https://doi.org/10.1117/1.JBO.17.5.058002
van Keulen S, van den Berg NS, Nishio N, Birkeland A, Zhou Q, Lu G, Wang HW, Middendorf L, Forouzanfar T, Martin BA, Colevas AD, Rosenthal EL (2019) Rapid, non-invasive fluorescence margin assessment: Optical specimen mapping in oral squamous cell carcinoma. Oral Oncol 88:58–65
Madajewski B, Judy BF, Mouchli A, Kapoor V, Holt D, Wang MD, Nie S, Singhal S (2012) Intraoperative near-infrared imaging of surgical wounds after tumor resections can detect residual disease. Clin Cancer Res 18:5741–5751
Nguyen QT, Tsien RY (2013) Fluorescence-guided surgery with live molecular navigation-a new cutting edge. Nat Rev Cancer 13:653–662
Predina JD, Fedor D, Newton AD et al (2017) Intraoperative molecular imaging: the surgical Oncologist’s north star. Ann Surg 266:e42–e44
Hernot S, van Manen L, Debie P, Mieog JSD, Vahrmeijer AL (2019) Latest developments in molecular tracers for fluorescence image-guided cancer surgery. Lancet Oncol 20:e354–e367
DSouza AV, Lin H, Henderson ER et al (2016) Review of fluorescence guided surgery systems: identification of key performance capabilities beyond indocyanine green imaging. J Biomed Opt 21:080901. https://doi.org/10.1117/1.JBO.21.8.080901
Dontu G, Ince TA (2015) Of mice and women: a comparative tissue biology perspective of breast stem cells and differentiation. J Mammary Gland Biol Neoplasia 20:51–62
Waks AG, Winer EP (2019) Breast Cancer treatment. JAMA 321:288–300. https://doi.org/10.1001/jama.2018.19323
Acknowledgments
This work was supported in part by the National Institutes of Health grants, R01EB019449, P50CA127297, P20GM103480, 1S10RR17846, 1S10RR027940, and P30CA036727 (Fred and Pamela Buffett Cancer Center at UNMC), Department of Defense Breast Cancer Research Program grant (W81XWH-14-1-0567), and the Nebraska Research Initiative. NEW would like to thank the UNMC Graduate Studies Assistantship/Fellowship and the John Borrlson Memorial Scholarship for support. We would also like to thank Megan Holmes, Freshta Baher, Xiaoxiao Qi, and Melissa Malone for technical assistance. Finally, we would like to thank the UNMC Flow Cytometry Core Facilities for assistance with flow sorting analysis, UNMC Small Animal Imaging Core Facility for access to IVIS, UNMC Tissue Sciences Core Facility for sectioning and staining of breast cancer tissue, and UNMC Comparative Medicine Personnel (Kristin Leland and Wendy Schwendeman) for assistance with animal protocol development.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflicts of interest to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 2048 kb)
Rights and permissions
About this article
Cite this article
Wojtynek, N.E., Olson, M.T., Bielecki, T.A. et al. Nanoparticle Formulation of Indocyanine Green Improves Image-Guided Surgery in a Murine Model of Breast Cancer. Mol Imaging Biol 22, 891–903 (2020). https://doi.org/10.1007/s11307-019-01462-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-019-01462-y