Abstract
Introduction
Technetium (Tc) methylene diphosphonate (MDP) has been the standard method for bone scintigraphy for three decades. 18F sodium fluoride (18F NaF) positron emission tomography (PET)/computed tomography (CT) has better resolution and is considered superior. The role of 2-deoxy-2-[18F]fluoro-D-glucose (18F FDG) PET/CT is proven in a variety of cancers, for which it has changed the practice of oncology. There are few prospective studies comparing these three methods of detection of skeletal metastases. Thus, we were prompted to initiate this prospective pilot trial.
Methods
This is a prospective study (Sep 2007–Dec 2010) of 52 patients with proven malignancy referred for evaluation of skeletal metastases. There were 37 men and 15 women, 19–84 years old (average, 55.6 ± 15.9). Technetium-99m (99mTc) MDP bone scintigraphy, 18F NaF PET/CT, and 18F FDG PET/CT were subsequently performed within 1 month.
Results
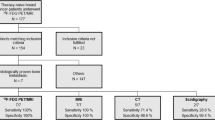
Skeletal lesions were detected by 99mTc MDP bone scintigraphy in 22 of 52 patients, by 18F NaF PET/CT in 24 of 52 patients, and by 18F FDG PET/CT in 16 of 52 patients. The image quality and evaluation of extent of disease were superior by 18F NaF PET/CT over 99mTc MDP scintigraphy in all 22 patients with skeletal lesions on both scans and over 18F FDG PET/CT in 11 of 16 patients with skeletal metastases on 18F FDG PET/CT. In two patients, 18F NaF PET/CT showed skeletal metastases not seen on either of the other two scans. Extraskeletal lesions were identified by 18F FDG PET/CT in 28 of 52 subjects.
Conclusion
Our prospective pilot-phase trial demonstrates superior image quality and evaluation of skeletal disease extent with 18F NaF PET/CT over 99mTc MDP scintigraphy and 18F FDG PET/CT. At the same time, 18F FDG PET detects extraskeletal disease that can significantly change disease management. As such, a combination of 18F FDG PET/CT and 18F NaF PET/CT may be necessary for cancer detection. Additional evaluation with larger cohorts is required to confirm these preliminary findings.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Imaging malignancies of bone, whether primary or metastatic, can be addressed using a variety of different modalities. These are broadly separated into anatomic-based modalities such as radiography, computed tomography (CT), and magnetic resonance imaging or functional modalities such as bone scintigraphy and positron emission tomography (PET).
Scintigraphic evaluation of bone cancer using Technetium-99m (99mTc) methylene diphosphonate (MDP) bone scintigraphy has been the standard functional modality for the evaluation of skeletal malignancy for many decades [1, 2]. Moreover, the use of single photon emission computed tomography (SPECT) alone or in combination with CT (SPECT/CT) has been shown to improve the sensitivity and specificity of bone scintigraphy over planar imaging alone [3, 4]. Since the advent of PET, 2-deoxy-2-[18F]fluoro-D-glucose (18F FDG) PET/CT has also been used for this indication. In particular, there is evidence that an MDP bone scan and an FDG PET scan may provide complementary information with regard to skeletal lesions, given the different mechanisms of radiotracer uptake in bone [5, 6]. That is, an MDP bone scan is better able to show sclerotic lesions while an FDG PET scan is better able to resolve lytic lesions [7]. Understandably then, comparison of these two modalities shows some discordant findings [5, 8, 9]. As such, in clinical practice, MDP bone scans and FDG PET scans are often used adjunctively for the complete staging of cancer patients with either primary or metastatic disease in the bone [10].
Another radiopharmaceutical that is of utility in bone imaging is 18F sodium fluoride (18F NaF). Historically, 18F NaF was initially used as a planar scintigraphy tracer and showed good quality results [11, 12]. However, it was abandoned with the introduction of 99mTc-based agents in the 1970s. In the 1990s, 18F NaF experienced resurgence with the advent of PET imaging [13–17]. Physiologically, 18F NaF behaves like MDP but, as a positron emitter, is more suitable for PET imaging [18, 19]. Theoretically, then, it may provide the sensitivity of a bone scan with the improved resolution and specificity of a PET scan. Thus, imaging skeletal lesions with 18F NaF PET/CT is a logical approach and has been shown to have clinical utility [20–23].
Since the advent of PET/CT, there have been a handful of studies showing the added value of 18F NaF PET/CT imaging over 18F NaF PET alone [20]. Comparisons have also been made between 18F NaF PET/CT and 99mTc MDP SPECT or 18F FDG PET/CT, again showing the improved sensitivity and specificity afforded by 18F NaF over the other tomographic, hybrid modalities [3, 24]. However, in non-small cell lung cancer, Kruger et al. [25] report that 18F FDG PET/CT is superior to bone scintigraphy in the detection of osteolytic bone metastases and may obviate the need to perform additional bone scans or 18F NaF PET for staging.
There are few prospective studies comparing all three of these methods of detecting skeletal metastases in a wide variety of malignancies, as might be seen in a routine clinical practice. Therefore, we were prompted to initiate this prospective study to compare the utility of 18F NaF PET/CT against 99mTc MDP bone scans and 18F FDG PET/CT in identifying skeletal lesions in various cancer types.
Methods
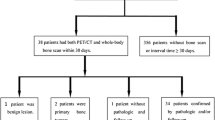
Stanford’s Institutional Review Board and Cancer Center Scientific Review Committee approved this study. The 52 consecutive patients in this study all had biopsy-proven recurrent malignancy and presented to the Nuclear Medicine Division between September 2007 and December 2010 prior to selection of therapy. There were 37 men and 15 women, ranging in age from 19 to 84 years (average ± standard deviation, 55.6 ± 15.9 years). These patients had a variety of malignancies. Nineteen had sarcoma, 18 had prostate cancer, 6 had breast cancer, 2 had colon cancer, 1 had bladder cancer, 1 had lung cancer, 1 had malignant paraganglioma, 1 had lymphoma, 1 had gastrointestinal stromal tumor, 1 had renal cancer, and 1 had salivary gland cancer. All were referred for evaluation of possible skeletal metastases with a 99mTc MDP bone scan. After obtaining a written informed consent, they were enrolled in the study and had the additional 18F NaF PET/CT and 18F FDG PET/CT scans done. All three scans were completed within 1 month.
Bone Scintigraphy Protocol
No patient preparation was required. Pregnancy was excluded by history. The intravenous dose of 99mTc MDP was 25 ± 2.5 mCi (925 ± 92.5 MBq). The patients were asked to return to the clinic in 3 h, during which time they were encouraged to hydrate and void. Upon returning, planar images of the whole body as well as spot views of the thorax and pelvis in the anterior and posterior views were acquired. Per routine clinical practice, additional spot (planar or oblique) views of the body were obtained if deemed necessary. The images were acquired on either of two dual-head gamma cameras (Infinia Hawkeye, GE Healthcare, Milwaukee, WI, USA or E-CAM, Siemens AG, Erlangen, Germany). All images were interpreted using a dedicated GE Xeleris workstation (GE Medical Systems, Haifa, Israel; version 2.0551). SPECT or SPECT/CT images were not acquired.
PET/CT Protocol
The standard protocol for an 18F FDG PET/CT scan was performed. Patients were asked to fast for 4–6 h if non-diabetic or 12 h if diabetic. Normal medications and water were allowed. Short-acting insulin was held for 4 h prior to the scan. The intravenous dose 18F FDG was 15 ± 1.5 mCi (555 ± 55.5 MBq). After a 1-h wait/circulation time, the patients were scanned on a GE Discovery LS scanner. No patient preparation was required for the 18F NaF scan. The intravenous dose 18F NaF was 10 ± 1.0 mCi (370 ± 37 MBq). After waiting for 45 min, the patients were scanned on the same GE Discovery LS scanner.
For both radiopharmaceuticals, total body (vertex to toes) PET/CT images were obtained in 2D mode, with the patients’ arms at their sides. The PET images were reconstructed with a standard iterative algorithm (OSEM, two iterative steps, 28 subsets). Images were the reformatted into axial, coronal, and sagittal views and reviewed using the same GE Xeleris workstation used for bone scans.
Data Analysis
Interpretation of all the 99mTc MDP bone scans, 18F NaF PET/CT, and 18F FDG PET/CT was performed by two board-certified nuclear medicine readers (AI, EM) blinded to the diagnosis and the results of the other imaging studies. Agreement was reached by consensus. Subsequently, a direct comparison of the detected lesions was performed among the three scans. Diagnostic accuracy was evaluated by comparing the bone scintigraphy and PET/CT results with final diagnoses (as confirmed by histological evaluation), clinical follow-up, or other imaging studies. The sensitivity and specificity of each imaging modality were calculated using a 2 × 2 contingency table. Confidence interval (CI) estimations were performed using the Wilson score method.
Results
A large number of the 52 patients demonstrated skeletal metastases by one or all of the modalities examined. Skeletal metastases were detected by 99mTc MDP scintigraphy in 22 of 52 patients, by 18F NaF PET/CT in 24 of 52 patients, and by 18F FDG PET/CT in 16 of 52 patients. The image quality and evaluation of extent of disease was superior by 18F NaF PET/CT over 99mTc MDP scintigraphy in all 22 patients with skeletal lesions on both scans and over 18F FDG PET/CT in 11 of 16 patients with skeletal metastases on 18F FDG PET/CT. In two patients (one with sarcoma and another with prostate cancer), 18F NaF PET/CT showed skeletal metastases not seen on either of the other two scans. Both these patients, however, had extensive metastatic disease. Extraskeletal metastases were identified by 18F FDG PET/CT in 28 of 52 subjects.
Using pathology reports (46% of the patients) or clinical follow-up (54% of the cases) as the gold standard, the per-patient sensitivity, specificity, positive predictive value, negative predictive value, and the accuracy of 99mTc MDP bone scintigraphy, 18F NaF PET/CT, and 18F FDG PET/CT for detecting malignant lesions were calculated (Table 1). As can be seen, 18F NaF PET/CT was equal to or better than 99mTc MDP bone scintigraphy on all these parameters. It also outperformed 18F FDG PET/CT in sensitivity, negative predictive value, and accuracy. However, 18F FDG PET/CT outperformed 18F NaF PET/CT in specificity and positive predictive value. The high efficacy of 18F FDG PET/CT is as expected and in accordance with the literature.
In addition, the sensitivity and specificity of 99mTc MDP bone scintigraphy, 18F NaF PET/CT, and 18F FDG PET/CT for detecting skeletal lesions was calculated in the subgroups of participants with sarcomas and prostate cancer. These were the most common malignancies included in the project; the findings are presented in Tables 2 and 3. Even in this subanalysis, 18F NaF PET/CT was better than or equal to 99mTc MDP bone scintigraphy and outperformed 18F FDG PET/CT in sensitivity for the detection of osseous metastases. However, 18F FDG PET/CT was more specific than 18F NaF PET/CT and 99mTc MDP bone scintigraphy in the prostate cancer subgroup. The statistical significance of this analysis is limited due to the small number of subjects in each category.
Cases
Figure 1 shows a 61-year-old woman with breast cancer and skeletal metastases seen on 99mTc MDP bone scan, 18F PET/CT, and 18F FDG PET/CT. In addition, 18F FDG PET/CT showed multiple pulmonary metastases. Figure 2 illustrates a biopsy-proven skeletal metastasis seen only on 18F NaF PET/CT, not on the 99mTc MDP bone scan or the 18F FDG PET/CT, in a 31-year-old man with history of L4 osteosarcoma. Figure 3 shows images from a 73-year-old man with prostate cancer: 99mTc MDP bone scintigraphy and 18F NaF PET/CT demonstrate extensive skeletal metastases, while 18F FDG PET/CT indicates liver and aorto-caval lymph node metastases. The osseous lesions are not seen on 18F FDG PET/CT.
Sixty-one-year-old woman with breast cancer. Skeletal lesions are seen on anterior whole-body planar 99mTc MDP bone scintigraphy (a), maximum intensity projection (MIP) image of 18F FDG PET/CT (b), and MIP image of 18F NaF PET/CT (c). Multiple bilateral pulmonary metastases are noted on the fused transaxial 18F FDG PET (d). Fused 18F NaF PET/CT demonstrates a spinal metastasis (e).
Discussion
99mTc MDP bone scintigraphy has been the method of choice for the evaluation of osseous metastases in various cancers since it allows a whole-body survey at a relatively reduced cost. Successful imaging of skeletal metastases is achieved for prostate, lung, breast, and other cancers. Applications of skeletal scintigraphy include initial staging, monitoring the response to therapy, and detection of areas at risk for pathological fracture. Although 99mTc MDP scintigraphy is sensitive for the detection of advanced skeletal metastatic lesions, early involvement may be missed because this technique relies on the identification of the osteoblastic reaction of the involved bone rather than the detection of the tumor itself. The technique relies significantly on the regional blood flow to bone as well. Limitations imposed by the spatial resolution of planar scintigraphy and SPECT also affect the sensitivity of bone scintigraphy in the detection of osseous metastases [3].
Thus, the transition to the better resolution of PET/CT for the detection of osseous metastases is appealing, with the use of the positron emitter 18F NaF as the radiotracer of choice. 18F NaF PET/CT appears superior in bone lesion detection over 99mTc MDP bone scan and SPECT [13]. 18F NaF bone scanning was performed prior to the introduction of 99mTc-based agents, achieving excellent quality studies. 18F NaF is an avid bone seeker, a property due to the fact that it is an analog of the hydroxyl group found in the hydroxyapatite bone crystals. 18F NaF has the desirable characteristics of high and rapid bone uptake accompanied by very rapid blood clearance, which results in a high bone-to-background ratio in a short time. High-quality images of the skeleton can be obtained less than an hour after the intravenous administration of 18F NaF. Therefore, imaging skeletal lesions with 18F PET/CT appears as a very promising approach for the acquisition of highly sensitive and specific images. Recent studies have compared 18F NaF PET with 99mTc MDP scintigraphy. These studies have demonstrated that 18F NaF PET is more accurate than planar imaging or SPECT with 99mTc MDP for localizing and characterizing both malignant and benign bone lesions. The higher quality imaging, increased clinical accuracy, greater convenience to the patient and referring physician, and more efficient use of nuclear medicine resources all indicate the need to reconsider the use of 18F NaF PET for imaging malignant diseases of the skeleton [18]. However, the lack of reimbursement for this study and the high cost associated with 18F NaF PET/CT imaging prevent its clinical utilization currently. The dramatic growth of 18F FDG imaging resulted in PET scanners in virtually all major medical institutions. PET centers located throughout the USA and other countries are able to provide positron-emitting agents. This, and the remarkable technological developments in positron imaging devices combined with co-registration with CT, has resulted in a renewed interest in 18F NaF.
18F FDG PET/CT contributes unique information regarding the metabolism of malignant lesions. By supplying a physiologic basis for more informed treatment and management, it influences prognosis and survival [26]. It is probable that for lung carcinoma, 18F FDG PET/CT has similar or better sensitivity, although poorer specificity, when compared with bone scintigraphy [25]. However, several researchers concluded that 99mTc MDP SPECT is superior to 18F FDG PET in detecting bone metastases in breast cancer and that the sensitivity for osteoblastic lesions is limited with 18F FDG PET/CT [27, 28]. It appears that surveillance of metastatic spread to the skeleton in breast cancer patients based on 18F FDG PET alone is not possible. There is convincing evidence that for prostate cancer, 18F FDG PET is less sensitive than bone scintigraphy [29]. While 18F FDG PET is limited in prostate cancer for the detection of osseous metastatic lesions, it may be useful in the detection of metastatic nodal and soft tissue disease [30]. There are limited data relating to the detection of osseous lesions in lymphoma, but 18F FDG PET seems to perform better than the bone scan [31]. There is an increasing body of evidence relating to the valuable role of 18F FDG PET in multiple myeloma where it is clearly better than bone scan, presumably because 18F FDG is identifying marrow-based disease at an early stage [32]. The morphology of the metastasis itself appears to be relevant for the ability of 18F FDG PET to detect disease. At least in breast cancer, different patterns of FDG uptake have been shown in sclerotic, lytic, or lesions with a mixed pattern. Furthermore, the precise localization of a metastasis in the skeleton may be important with regard to the extent of the metabolic response induced [33]. Functional imaging with PET and 18F FDG may also have an important role in the imaging evaluation of patients with bone and soft tissue sarcoma, including guiding biopsy, detecting local recurrence in amputation stumps, detecting metastatic disease, predicting and monitoring response to therapy, and assessing for prognosis [34]. Positron emission tomography has been shown to be superior to scintigraphy in the detection of metastases because it detects the presence of tumor directly by metabolic activity rather than indirectly by showing tumor involvement due to increased bone mineral turnover. This has allowed the detection of metastatic foci earlier with PET than with bone scintigraphy [35].
The results of our study are yet another indication that 18F NaF PET/CT is more sensitive than 99mTc MDP bone scintigraphy and 18F FDG PET/CT for the detection of skeletal metastases while maintaining the high specificity. In an era affected by frequent 99mTc shortages, the availability of an excellent agent for skeleton imaging such as 18F NaF should lead to its increased utilization in order to improve patient care.
Limitations of this study include the relatively small number of patients, the variety of cancers, as well as the selection bias toward patients with known cancers. In addition, the large number of subjects with prostate cancer may favor 18F NaF PET/CT over 18F FDG PET/CT for the detection of skeletal metastases. Therefore, larger prospective studies centered on specific malignancies are warranted.
Conclusion
Our prospective trial demonstrates superior image quality and evaluation of skeletal disease extent with 18F NaF PET/CT over 99mTc MDP scintigraphy. Whether the same is true about 18F NaF PET/CT and 18F FDG PET/CT remains to be investigated in larger projects enrolling a more homogenous patient cohort. However, 18F FDG PET detects extraskeletal disease, which can change disease management. As such, a combination of 18F FDG PET/CT and 18F NaF PET/CT may be necessary for cancer detection. Additional evaluation with larger cohorts is required to confirm these preliminary findings.
References
Bombardieri E, Aktolun C, Baum RP et al (2003) Bone scintigraphy: procedure guidelines for tumour imaging. Eur J Nucl Med Mol Imaging 30(12):BP99–BP106
Jacobson AF, Fogelman I (1998) Bone scanning in clinical oncology: does it have a future? Eur J Nucl Med 25(9):1219–1223
Even-Sapir E (2005) Imaging of malignant bone involvement by morphologic, scintigraphic, and hybrid modalities. J Nucl Med 46(8):1356–1367
Schirrmeister H, Glatting G, Hetzel J et al (2001) Prospective evaluation of the clinical value of planar bone scans, SPECT, and (18)F-labeled NaF PET in newly diagnosed lung cancer. J Nucl Med 42(12):1800–1804
Min JW, Um SW, Yim JJ et al (2009) The role of whole-body FDG PET/CT, Tc 99m MDP bone scintigraphy, and serum alkaline phosphatase in detecting bone metastasis in patients with newly diagnosed lung cancer. J Korean Med Sci 24(2):275–280
Chua S, Gnanasegaran G, Cook GJ (2009) Miscellaneous cancers (lung, thyroid, renal cancer, myeloma, and neuroendocrine tumors): role of SPECT and PET in imaging bone metastases. Semin Nucl Med 39(6):416–430
Hsu WK, Virk MS, Feeley BT, Stout DB, Chatziioannou AF, Lieberman JR (2008) Characterization of osteolytic, osteoblastic, and mixed lesions in a prostate cancer mouse model using 18F-FDG and 18F-fluoride PET/CT. J Nucl Med 49(3):414–421
Chen YW, Huang MY, Hsieh JS, Hou MF, Chou SH, Lin CL (2007) Discordant findings of skeletal metastasis between tc 99M MDP bone scans and F18 FDG PET/CT imaging for advanced breast and lung cancers—two case reports and literature review. Kaohsiung J Med Sci 23(12):639–646
Schirrmeister H, Guhlmann A, Elsner K et al (1999) Sensitivity in detecting osseous lesions depends on anatomic localization: planar bone scintigraphy versus 18F PET. J Nucl Med 40(10):1623–1629
Ben-Haim S, Israel O (2009) Breast cancer: role of SPECT and PET in imaging bone metastases. Semin Nucl Med 39(6):408–415
Bridges RL, Wiley CR, Christian JC, Strohm AP (2007) An introduction to Na(18)F bone scintigraphy: basic principles, advanced imaging concepts, and case examples. J Nucl Med Technol 35(2):64–76, quiz 78–69
Shirazi PH, Rayudu GV, Fordham EW (1974) 18F bone scanning: review of indications and results of 1,500 scans. Radiology 112(2):361–368
Grant FD, Fahey FH, Packard AB, Davis RT, Alavi A, Treves ST (2008) Skeletal PET with 18F-fluoride: applying new technology to an old tracer. J Nucl Med 49(1):68–78
Gamie S, El-Maghraby T (2008) The role of PET/CT in evaluation of Facet and Disc abnormalities in patients with low back pain using (18)F-fluoride. Nucl Med Rev Cent East Eur 11(1):17–21
Drubach LA, Sapp MV, Laffin S, Kleinman PK (2008) Fluorine-18 NaF PET imaging of child abuse. Pediatr Radiol 38(7):776–779
Bhargava P, Hanif M, Nash C (2008) Whole-body F-18 sodium fluoride PET-CT in a patient with renal cell carcinoma. Clin Nucl Med 33(12):894–895
Lim R, Fahey FH, Drubach LA, Connolly LP, Treves ST (2007) Early experience with fluorine-18 sodium fluoride bone PET in young patients with back pain. J Pediatr Orthop 27(3):277–282
Even-Sapir E, Metser U, Mishani E, Lievshitz G, Lerman H, Leibovitch I (2006) The detection of bone metastases in patients with high-risk prostate cancer: 99mTc-MDP planar bone scintigraphy, single- and multi-field-of-view SPECT, 18F-fluoride PET, and 18F-fluoride PET/CT. J Nucl Med 47(2):287–297
Even-Sapir E, Mishani E, Flusser G, Metser U (2007) 18F-fluoride positron emission tomography and positron emission tomography/computed tomography. Semin Nucl Med 37(6):462–469
Even-Sapir E, Metser U, Flusser G et al (2004) Assessment of malignant skeletal disease: initial experience with 18F-fluoride PET/CT and comparison between 18F-fluoride PET and 18F-fluoride PET/CT. J Nucl Med 45(2):272–278
Hetzel M, Arslandemir C, Konig HH et al (2003) F-18 NaF PET for detection of bone metastases in lung cancer: accuracy, cost-effectiveness, and impact on patient management. J Bone Miner Res 18(12):2206–2214
Schirrmeister H, Buck A, Guhlmann A, Reske SN (2001) Anatomical distribution and sclerotic activity of bone metastases from thyroid cancer assessed with F-18 sodium fluoride positron emission tomography. Thyroid 11(7):677–683
Cook GJ, Fogelman I (2001) Detection of bone metastases in cancer patients by 18F-fluoride and 18F-fluorodeoxyglucose positron emission tomography. Q J Nucl Med 45(1):47–52
Blake GM, Park-Holohan SJ, Cook GJ, Fogelman I (2001) Quantitative studies of bone with the use of 18F-fluoride and 99mTc-methylene diphosphonate. Semin Nucl Med 31(1):28–49
Kruger S, Buck A, Mottaghy F et al (2009) Detection of bone metastases in patients with lung cancer: 99mTc-MDP planar bone scintigraphy, 18F-fluoride PET or 18F-FDG PET/CT. Eur J Nucl Med Mol Imaging 36(11):1807–1812
Feldman F, van Heertum R, Manos C (2003) 18FDG PET scanning of benign and malignant musculoskeletal lesions. Skeletal Radiol 32(4):201–208
Uematsu T, Yuen S, Yukisawa S et al (2005) Comparison of FDG PET and SPECT for detection of bone metastases in breast cancer. Am J Roentgenol 184(4):1266–1273
Nakai T, Okuyama C, Kubota T et al (2005) Pitfalls of FDG-PET for the diagnosis of osteoblastic bone metastases in patients with breast cancer. Eur J Nucl Med Mol Imaging 32(11):1253–1258
Meirelles GSP, Schoder H, Ravizzini GC et al (2010) Prognostic value of baseline [18F] fluorodeoxyglucose positron emission tomography and 99mTc-MDP bone scan in progressing metastatic prostate cancer. Clin Cancer Res 16(24):6093–6099
Jadvar H, Pinski J, Conti P (2003) FDG PET in suspected recurrent and metastatic prostate cancer. Oncol Rep 10(5):1485–1488
Shulkin B, Goodin G, McCarville M et al (2009) Bone and [18F]fluorodeoxyglucose positron-emission tomography/computed tomography scanning for the assessment of osseous involvement in Hodgkin lymphoma in children and young adults. Leuk Lymphoma 50(11):1794–1802
Jadvar H, Conti P (2002) Diagnostic utility of FDG PET in multiple myeloma. Skeletal Radiol 31(12):690–694
Fogelman I, Cook G, Israel O, Van der Wall H (2005) Positron emission tomography and bone metastases. Semin Nucl Med 35(2):135–142
Jadvar H, Gamie S, Ramanna L, Conti P (2004) Musculoskeletal system. Semin Nucl Med 34(4):254–261
Peterson J, Kransdorf M, O’Connor M (2003) Diagnosis of occult bone metastases: positron emission tomography. Clin Orthop Relat Res 415(Suppl):S120–S128
Acknowledgments
This research was supported in part by NCI ICMIC CA114747 (SSG), and the clinical studies were supported in part by the Doris Duke Foundation and Canary Foundation (SSG). We would also like to thank Dr. Fred Chin in the Cyclotron Facility, Lindee Burton, and all the technologists in the Nuclear Medicine Clinic.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iagaru, A., Mittra, E., Dick, D.W. et al. Prospective Evaluation of 99mTc MDP Scintigraphy, 18F NaF PET/CT, and 18F FDG PET/CT for Detection of Skeletal Metastases. Mol Imaging Biol 14, 252–259 (2012). https://doi.org/10.1007/s11307-011-0486-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-011-0486-2