Abstract
Variation in pollen formation and its cytological mechanism in an allotriploid white poplar were investigated by the squashed technique and indirect immunofluorescence. Besides 0.5% stuck pollen grains, this allotriploid produced regularly spherical pollen grains. It was estimated that 90.3% of pollen grains were viable. Diameters of the viable spherical pollen grains ranged from 23.2 to 72.9 μm, with a bimodal frequency distribution. Numerous meiotic abnormalities were found, including highly irregular chromosome pairing, lagging chromosomes and chromosome bridges, micronuclei, and multiple spindles, which indicate highly genetic imbalance of this allotriploid. Some micronuclei triggered minispindle formation in metaphase II and participated in cytokinesis to form microcytes in sporads. Abnormal orientation of metaphase II spindles contributed to production of dyads and triads, which produced unreduced microspores. However, parallel orientation of spindles was not necessary for dyad formation, because an organelle band positioned in the equatorial region prevented the spindles from coalescing. Some microsporocytes exhibited a complete or partial absence of cytokinesis, which resulted in the formation of monads and the increased frequency of dyads and triads. The perspective of this triploid in the polyploid breeding program of white poplar is discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Polyploidization plays an important role in plant speciation and evolution. It has been estimated that more than 70% of the flowering plants are polyploids (Masterson 1994). Triploids are considered part of the evolutionary dynamics of natural populations (Husband 2004) and act as a transition in the formation of new polyploids (Jackson 1976). Moreover, triploids can function in gene flow within populations (Den Nijs and Peloquin 1977).
In general, triploids are expected to be sterile because of their genetic imbalance. The idea of the “triploid block effect” suggests that triploids may be evolutionary dead ends (Den Nijs and Peloquin 1977); however, the block is not sufficiently strong to completely prevent the production of offspring. A triploid bridge hypothesis has been proposed, given that some fertile gametes can usually be produced (Jackson 1976). The potato, for example, exhibits some complex interploidy relationships, as established by Den Nijs and Peloquin (1977). Conspicuously, triploids act as an important intermediate cytotype for production of tetraploids, pentaploids, and hexaploids. Aneuploid progeny can be produced by crossing triploids with diploids in melon (Ezura et al. 1994), blueberry (Dweikat and Lyrene 1988), elm (Santamour 1971), and Populus (Nilsson-Ehle 1938), suggesting that some aneuploid gametes may be fertile.
The abundant variation of pollen size in triploids is usually attributed to irregular chromosome pairing and abnormal meiotic division. Irregular pairing of chromosomes can result in unequal chromosome segregation and chromosome elimination, giving rise to an asymmetrical cytokinesis after meiosis (Mendes-Bonato et al. 2006; Tel-Zur et al. 2005). Abnormal orientation of spindles in the second meiotic division and aberrant cytokinesis, which are responsible for unreduced gamete production, have been reported in polyploid plants of Brachiaria ruziziensis (Risso-Pascotto et al. 2004, 2005), rose (Crespel et al. 2006), potato (Carputo et al. 1995; Ramanna 1974), and Citrus (Del Bosco et al. 1999).
Polyploid breeding plays an important role in improvement of the genus Populus. Triploid Populus are widely used for production of fuel, fiber, and lumber, owing to their desirable properties in growth and pulp characteristics compared to diploids (Van Buijtenen et al. 1958; Zhu et al. 1995). Furthermore, triploids, which can produce fertile gametes, have been used in polyploid breeding of Populus. Nilsson-Ehle (1938) obtained some triploids, tetraploids, and aneuploids by crossing diploid with triploid plants of Populus tremula L. Johnsson (1940, 1942) undertook crossing experiments including triploid × triploid and diploid × triploid in P. tremula, giving rise to offspring with chromosome numbers varying between 38 (the normal diploid chromosome number of Populus) and 76. Additionally, tetraploid aspens have been produced by pollination of unreduced pollen from triploid males to diploids (Harder et al. 1976). In a natural triploid P. tremula, irregular chromosome pairing and segregation in meiosis have been observed by Müntzing (1936). He found that a fusion of metaphase II plates led to the formation of dyads. These abnormalities were responsible for wide arrays of pollen sizes and ploidy levels.
In natural diploid populations of Populus tomentosa Carr., unreduced pollen can occur spontaneously. Unreduced pollen was used to induce the formation of triploid hybrids with P. tomentosa × Populus bolleana (Zhu et al. 1995) and with Populus alba × Populus glandulosa (Zhu, unpublished). These induced triploid hybrids exhibit good performances in growth and wood properties and have been applied in forestry production in China. Kang et al. (1999) investigated meiotic chromosome behavior of the triploids from the (P. tomentosa × P. bolleana) × P. tomentosa cross, showing abnormal chromosome synapsis, highly irregular chromosome pairing, lagging chromosomes, and micronulei. Two such triploid hybrids, B302 and B312, could produce fertile pollen, and some offspring were obtained by crosses of the two male triploid hybrids with two diploid female hybrids, P. tomentosa × P. bolleana and P. alba × P. glandulosa, respectively (Kang and Mao 2001; Kang et al. 1999). However, the cytological characteristics and variation of pollen of the triploid from (P. alba × P. glandulosa) × P. tomentosa is still not clear, which has hindered its use in improvement efforts targeting the white poplar.
In this study, to further contribute to the polyploid breeding programs of white poplar, variation in pollen size and shape and viability of pollen in the allotriploid white poplar from (P. alba × P. glandulosa) × P. tomentosa were analyzed. Furthermore, chromosome behavior and microtubule changes during microsporogenesis were recorded using the squashed technique and indirect immunofluorescence to reveal the cytological mechanism of pollen variation.
Materials and methods
Plant material
A male allotriploid (2n = 3x = 57) of white poplar was investigated. This triploid hybrid was synthesized by professor Zhu of Beijing Forestry University (unpublished) through a cross between a diploid hybrid P. alba × P. glandulosa and a 2n pollen producer of diploid P. tomentosa in 1985. Male floral branches of this triploid were collected from the Bajia nursery garden in Beijing and water-cultured in a greenhouse (15–25°C). After the branches were cultured in the greenhouse, four male flower buds or catkins were collected from the branches every 2–3 h until pollen maturity. The collected buds were immediately fixed in Carnoy solution (3:1 ethanol/acetic acid) at 4°C for 24 h. A total of 156 buds and catkins were collected and fixed. A pollen sample was collected from freshly dehiscent anthers on male catkins.
Analysis of pollen size and viability
The fresh pollen sample was mounted immediately in a drop of 2% aceto-carmine on a microscope slide. Pollen viability was estimated based on its stainability and shape. Unstained and withered pollen grains were scored as sterile. The diameter of stained spherical pollen grains was measured using an ocular micrometer; a total of 1,000 viable spherical pollen grains were measured. Some stained pollen grains with abnormal shape were recorded.
Cytological observations of microsporogenesis
Microsporocytes or microspores were pressed out of anthers and squashed in 2% aceto-carmine on a microscope slide. Some buds under meiosis were selected for meiotic analysis. The preparations with a glass cover were heated slightly over a flame to increase chromosome stainability. All preparations were observed using a microscope (Olympus BX51) and photos taken with an attached charge-coupled device video camera (Olympus DP70). The frequency of laggard chromosomes or bridge formation in anaphase I, the presence of micronucleus in telophase I and II, and the number of parallel, fused, tripolar, and normal crossed spindles were analyzed together with the variation of sporad type and shape.
Indirect immunofluorescence of microtubules
For indirect immunofluorescence of microtubules, anthers were fixed in 4% paraformaldehyde in a PEM buffer (50 mM Pipes, 5 mM EGTA, 2 mM MgSO4, pH 6.9) for 45 min. After being rinsed in the PEM buffer three times for 5 min, the anthers were dipped in 10% dimethylsulfoxide for 15 min and then extracted with 1% Triton X-100 for 30 min. They were then rinsed, respectively, in the PEM buffer and a PBS buffer (137 mM NaCl, 2.7 mM KCl, 7 mM Na2HPO4, 1.5 mM KH2PO4, pH 7.3) again three times for 5 min. After that, microsporocytes were squeezed out from the anthers and transferred to a slide coated with 0.1% poly-l-lysine (Sigma p1274). The cells were incubated with a monoclonal anti-α-tubulin antibody (Sigma T9026) diluted 1:100 with the PBS buffer for 2 h at 37°C in a moist chamber. Following further washing in the PBS buffer, the microsporocytes were incubated in a FITC-conjugated anti-mouse IgG (Sigma F0257) diluted 1:50 with the PBS buffer for 2 h at 37°C in a dark chamber. After a final wash in the PBS buffer, the microsporocytes were mounted with a mounting medium with 4′,6-diamidino-2-phenylindole (DAPI; Vectashield H-1200). The preparations were observed and photographed with a Leica TCS-SP2 confocal laser scanning microscope.
Results
Stainability and size variation of pollen
In the allotriploid analyzed here, most of the pollen grains (90.3%) were viable and only 9.7% were shrunken and unstainable (Fig. 1a). In addition to regularly spherical pollen grains, a few (0.5%) stuck pollen grains were found, having resulted from partial failure of microspore separation (Fig. 1b). Size of spherical pollen grains varied considerably, suggesting a wide range of ploidy level. The diameter of stainable spherical pollen grains ranged from 23.2 to 72.9 μm, with an average of 36.95 μm (±6.78). The frequency of pollen diameter followed a bimodal distribution with a trough at 48 μm (Fig. 2). The frequency of pollen grains with diameters from 23 to 48 μm was 92.2%, while it was 7.6% for grains with diameters from 49 to 65 μm. In addition, some giant pollen grains with diameters of more than 68 μm were found, which might share very high ploidy level.
Meiotic abnormalities
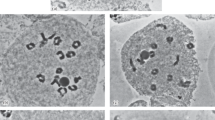
In this allotriploid, a number of meiotic abnormalities were recorded (Table 1), indicating its complex chromosome pairing and unbalanced chromosome segregation. Univalents, bivalents, trivalents, and multivalents were observed at diakinesis (Fig. 3a). At metaphase I, some chromosomes precociously migrated to the poles (Fig. 3b). Abnormal chromosome disposition was also observed, in which chromosomes were arranged on three plates. As a result, multiple spindles formed in metaphase I (Fig. 3c). In anaphase I, lagging chromosomes and chromosome bridges were found (Fig. 3d, e) with a frequency of 71.26%. The lagging chromosomes did not always reach the poles but instead formed micronuclei at telophase I (Fig. 3f), with a frequency of 62.76%. During the second meiotic division, micronuclei still were observed (Fig. 3g). Moreover, irregular metaphase II microsporocytes with three independent spindles were found (Fig. 3h), which developed into telophase II cells with more than four primary nuclei (Fig. 3i), indicating the property of high ploidy level of this hybrid. In telophase II, the frequency of microsporocytes with micronuclei decreased to 49.69%.
Meiotic abnormalities of the allotriploid. a Diakinesis with univalents and multivalents (arrows). b Metaphase I showing precocious migration of chromosomes to the poles. c Metaphase I with triple spindles. d Anaphase I with lagging chromosomes. e Late anaphase I with a chromosome bridge. f Telophase I with micronuclei. g Telophase II with micronuclei. h Metaphase II showing three independent spindles. i Telophase II with five primary nuclei and three micronuclei. Bars are equal to 10 μm
During microsporogenesis, some micronuclei were scattered in the cytoplasm without any action by microtubules; the others were positioned in equatorial region by microtubules (Fig. 4a, b). The scattered micronuclei were either incorporated into primary nuclei or remained in the cytoplasm until allocated to microspores together with primary nuclei (Fig. 4e–h). The latter formed minispindles in metaphase II (Fig. 4c, d) and underwent cytokinesis to develop into microcytes in meiotic products (Fig. 4f–l).
The fate of micronuclei. a Telophase I with two micronuclei stained by DAPI, one of which (arrow) was positioned in the equatorial region and the other (arrowhead) close to a primary nucleus. b Microtubular arrays of the same microsporocyte in (a) showing that microtubules emanated from the micronucleus located in the equatorial region and that no microtubule was connected to the other one. c Metaphase II showing the eliminated chromosomes in cytoplasm. d Indirect immunofluorescence of microtubules of the same microsporocyte in (c) showing a minispindle (arrow) associated with the eliminated chromosomes. e–h Dyad (e–g) and tetrad (h) showing primary nuclei together with micronuclei in same microspores. f–l Dyad (f, g, k, and l), triad (j), and tetrad (h and i) with one or more microcytes. Bars are equal to 5 μm in (a)–(d) and 10 μm in (e)–(l)
Abnormal orientation of spindles and cytokinesis
The orientation of spindles in the second meiotic division varied in this allotriploid. Apart from normal spindles with perpendicular alignment, parallel, fused, and tripolar spindles were observed, with frequencies of 16.40%, 10.88%, and 9.55%, respectively. As a result, dyads, triads, and tetrads were produced. Two patterns of tetrads, i.e., tetrahedral and tetragonal arrangements, were observed (Fig. 5a, b). In microsporocytes with normal and parallel spindles, staining with DAPI identified an organelle band in the equatorial region that separated the cytoplasm into dyad domains until meiotic division completed (Fig. 5c, d).
Patterns of tetrads and the organelle band in the second meiotic division. a A tetrahedral tetrad developed from perpendicular metaphase II spindles. b A tetragonal tetrad from parallel spindles. c, d An organelle band positioned in the equatorial region of the microsporocyte with parallel spindles at metaphase II (c) and anaphase II (d). Bars are equal to 10 μm in (a)–(b) and 5 μm in (c)–(d)
Indirect immunofluorescence of microtubular cytoskeletons clearly revealed aberrant cytokinesis in telophase II. Regularly, nuclear-based radial microtubule systems (RMS) formed entirely in telophase II (Fig. 6a, b). However, these RMS were abnormal in some microsporocytes (Fig. 6c–f), which resulted in the formation of monads (Fig. 6g) and an increased frequency of dyads and triads. The monads were likely to develop into the giant pollen grains. In addition, unequal sporads caused by aberrant cytokinesis were found (Fig. 6h).
Aspects of cytokinesis. a, b An intact nuclear-based radial microtubule systems (RMS, a) and nuclei (b) in a telophase II microsporocyte undergoing regular cytokinesis. c Partial absence of RMS in a microsporocyte. d Nuclei and organelles stained by DAPI in the same microsporocyte with (c), showing that a restituted nucleus was allocated to one microspore together with a reduced nucleus. e Defect of RMS in a microsporocyte. f Abnormal location of nuclei resulting from the complete absence of RMS in the microsporocyte of (e). g A monad with four nuclei. h A sporad caused by unequal cytokinesis. Bars are equal to 5 μm in (a)–(f) and 10 μm in (g)–(h)
Curiously, after sporads formed, cytoplasmic infurrowing tardily occurred in some monads (Fig. 7a, b) and dyads (Fig. 7c–e). In some cases, some cytoplasm lacking a nucleus was partitioned off. Triads with a weak cell wall (Fig. 7f) could be produced by the tardy infurrowing of dyads.
Tardy infurrowing of cytoplasm was observed in some monads and dyads. a, b Monads with tardy infurrowing, showing the whole nucleus staying on one side of the furrow. c, d, e Dyads with tardy infurrowing. The cell wall initiated from the location indicated by the arrows. f A triad with a weak cell wall (arrow) from tardy infurrowing of a dyad. Bars are equal to 10 μm
Discussion
Meiosis of allo-polyploids is characterized by complex chromosome pairing. Irregularities of chromosome pairing contribute to the production of lagging chromosomes and micronuclei (Del Bosco et al. 1999; Tel-Zur et al. 2005). As mentioned, Kang et al. (1999) found multiple meiotic abnormalities in triploids from a cross between diploid hybrid P. tomentosa × P. bolleana and a 2n pollen producer of diploid P. tomentosa. Similarly, various kinds of meiotic abnormalities, including irregular chromosome pairing, lagging chromosomes, multiple spindles, and micronuclei, were also found in the allotriploid investigated here.
Multiple spindles have been reported in several plant species (Caetano-Pereira and Pagliarini 2001; Risso-Pascotto et al. 2005; Tai 1970; Tilquin et al. 1984), and they are strongly correlated with the production of polyads in Fuchsia (Tilquin et al. 1984), B. ruziziensis (Risso-Pascotto et al. 2005), and Zea mays (Caetano-Pereira and Pagliarini 2001). Although triple spindles were observed in the second meiotic division of this allotriploid, no polyad with six equal microspores was found, which may be explained by the nuclear restitution and failure of cytokinesis. Additionally, according to investigations in Rubus (Thompson 1962) and Fuchsia (Tilquin et al. 1984), multiple spindles are probably a consequence of high ploidy level. Tai (1970) indicated that multiple spindles result in a multipolar cell division, which provides a mechanism of decreasing the ploidy level of polyploids.
The behavior and fate of micronuclei are associated with changes of microtubular cytoskeletons (Brown and Lemmon 1989, 1992). In our investigation, not all micronuclei were connected to microtubules. Some micronuclei not connected to microtubules were scattered in the cytoplasm and did not achieve cytokinesis. Consequently, they were allocated to a single microspore together with the primary nucleus. By contrast, the other micronuclei were connected with microtubules, which were positioned in equatorial region in telophase I and formed minispindles during the second division. Subsequently, this type of micronuclei took part in cytokinesis to develop into microcytes. Some investigations have led to a hypothesis that chromosomes can induce the organization of microtubules into a spindle (Steffen et al. 1986). Moreover, kinetochores seem to be necessary for generating a bipolar spindle (McKim and Hawley 1995). This hypothesis may explain the formation of minispindles in the allotriploid studied here. The lagging chromosomes with kinetochores, which were separated into two sister chromatids in metaphase II, formed a bipolar spindle, whereas the lagging chromosomes lacking kinetochores did not form a minispindle.
Stuck pollen grains were observed in this allotriploid, indicating the occurrence of abnormal microspore separation in some microsporocytes. On the one hand, microspore separation is accompanied by the initiation of the primexine wall and degradation of the callosic wall surrounding each microspore (Owen and Makaroff 1995; Rhee and Somerville 1998). This outcome suggests that the formation of the stuck pollen may result from the aborted degradation of the callosic wall in some sporads. On the other hand, a tardy infurrowing of the cytoplasm was observed in some monads and dyads, which might result in formation of a weak cell wall that is not strong enough to support the microspore separation.
It is known that cell size increases with increasing DNA content (Bretagnolle and Thompson 1995). Therefore, the ploidy level of pollen can be distinguished by analyzing pollen grain diameter. In general, the frequency distribution of pollen grain diameter is unimodal in normal diploid plants and is bimodal in unreduced pollen producers (Tondini et al. 1993). In this allotriploid poplar, the diameters of pollen grains followed a bimodal distribution, suggesting the production of unreduced pollen grains. Whereas some chromosomes tend to be eliminated, some unreduced pollen grains may not be exactly 3x in DNA content.
The meiosis of triploids is expected to give rise to aneuploid gametes with half the triploid chromosome number (n = 3x/2). It is usually not uniform, however, owing to irregular chromosome pairing and unbalanced chromosome segregation (Ramsey and Schemske 1998). In the allotriploid studied here, the wide range of pollen diameters may represent the various chromosome numbers of pollen grains owing to the unbalanced chromosome segregation. Similar results have been reported in triploid hybrids of potato (Carputo et al. 1995) and triploid and tetraploid vine cacti hybrids (Tel-Zur et al. 2003, 2005). Pollen, with a diameter above 37 μm, is considered as 2x pollen in the genus Populus (Mashkina et al. 1989). An incidental small saddle was present at 36.5 μm in the frequency distribution of pollen grain diameters in this allotriploid white poplar, and the frequency of pollen with a diameter under 36.5 μm was higher than that from 36.5 to 48 μm, indicating that some chromosomal combinations could be better tolerated to be fertile than others.
In general, the frequency of unreduced pollen can be estimated by the frequencies of dyads and triads. According to the observed frequencies of dyads and triads in this triploid, the expected frequency of unreduced pollen was 14.7%. However, the frequency of unreduced pollen was 7.6% from observation. One of the reasons is that some unreduced pollen from the dyads and triads might not be euploid owing to chromosome elimination and unbalanced chromosome segregation, and hence were not fully fertile. Those sterile pollen grains were out of statistics. The other is that the tardy infurrowing of cytoplasm in some sporads may be responsible for decreasing the frequencies of the dyads and triads.
The abnormal orientation of spindles may lead to meiotic nuclear restitution, resulting in the production of unreduced gametes (see Bretagnolle and Thompson 1995). Admittedly, fused spindles and tripolar spindles are identified respectively as reasons for dyad formation and triad formation (Ramanna 1979). As far as parallel orientation of spindles, however, Ramanna (1979) proposed that the occurrence of parallel spindles in the second meiotic division does not necessarily lead to dyad formation. Although parallel spindles are positively correlated with dyad production in alfalfa (Vorsa and Bingham 1979) and Ipomoea batatas (Becerra Lopez-Lavalle and Orjeda 2002), no correlation was found between the frequency of parallel spindles and the frequency of dyads in the investigations on triploid hybrids of potato (Carputo et al. 1995) and vine cacti (Tel-Zur et al. 2005). An organelle band, which separated the cytoplasm into dyad domains, was observed both in microsporocytes with normal and parallel spindle orientations. Bednara et al. (1986) indicated the organelle band served to prevent the coalescence of spindles. According to Brown and Lemmon (1992), the orientation of metaphase II spindles determines the pattern of tetrads, and the parallel spindles might result in the production of tetragonal tetrads. In this investigation, tetragonal tetrads were found in meiotic products, which indicated that some parallel spindles might not contribute to the formation of dyads.
Aberrant cytokinesis also can result in unreduced gamete formation (Pfeiffer and Bingham 1983; Ray and Tokach 1992; Tavoletti et al. 1991). In meiosis of higher plants, RMS are considered to play a primary role in the organization and apportionment of cytoplasm, the location of division planes, and the definition of nuclear cytoplasmic domains (Brown and Lemmon 1989, 1991, 1996). The cytokinesis is accomplished by centrifugal expansion of cell plates (Brown and Lemmon 1996; Traas et al. 1989). In the allotriploid poplar studied here, the RMSs were completely or partially lacking in some microsporocytes, which resulted in the production of monads and an increased frequency of dyads and triads.
Several cytokinetic mutants, which produce big and/or giant pollen, have been identified in potato (Ramanna 1974), alfalfa (Pfeiffer and Bingham 1983), and Arabidopsis (Spielman et al. 1997). In Arabidopsis, a stud (std) mutant (Hülskamp et al. 1997) and tetraspore (tes) mutant (Spielman et al. 1997) have been identified that failed in male meiotic cytokinesis, resulting in a single microspore with four nuclei. Furthermore, a TES protein associated with the tes mutant has been cloned and is likely to serve as a microtubule-associated motor, playing a role either in the formation of the RMS following meiosis or in maintaining their stability (Yang et al. 2003). In this study, both normal meiotic cytokinesis and defective meiotic cytokinesis were observed. The defective cytokinesis resulted in production of multinucleate microspores. This outcome suggests that the meiotic cytokinesis of this triploid may be involved in a similar mechanism at the molecular level.
In this triploid poplar, both the disoriented spindles and cytokinetic abnormalities are likely to be the major mechanism for 2n pollen production. However, these mechanisms are not typical for triploid plants, which generally contribute to irregular meiotic chromosome pairing and unbalanced chromosome segregation. In previous investigations on the 2n pollen donor of this triploid hybrid, P. tomentosa, Kang (2002) and Zhang et al. (2007) found that the disorientation of spindles at metaphase II and absence of cytokinesis at telophase II were responsible for the spontaneous formation of 2n pollen. Thus, it is suggested that the mutations of disoriented spindles and cytokinesis defect in this triploid hybrid are probably inherited from its male parent. Moreover, the 2n pollen formation in this triploid hybrid appeared to indicate that the genome of the P. alba × P. glandulosa was not strong enough to prevent the expression of the mutations from the P. tomentosa, or alternatively to indicate that the mutations were dominant to the P. alba × P. glandulosa genome.
Polyploid breeding is an important avenue in the improvement of the genus Populus, and using unreduced gametes is a routine and crucial approach for polyploid production in this genus (Kang et al. 2000; Mashkina et al. 1989; Zhu et al. 1995). Triploids usually act as a pollinator because of their ability to produce pollen with a high ploidy level. In aspen, pollen grains of triploid plants have been used to pollinate diploids, resulting in production of triploids, tetraploids, and aneuploids (Nilsson-Ehle 1938). This result suggested that chromosomally unbalanced pollen grains are probably fertile. Recently, Kang and Mao (2001) reported that the pollen grains of an allotriploid of white poplar, (P. tomentosa × P. bolleana) (haploid egg) × P. tomentosa (diploid pollen), could germinate on stigmas of P. tomentosa × P. bolleana and P. alba × P. glandulosa, with a number of offspring obtained. In the allotriploid poplar analyzed here, the viable pollen grains were estimated to make up 90.3% of the grains and showed a great diversity in ploidy level, suggesting that this allotriploid can be usefully applied to polyploid breeding programs of the genus Populus.
References
Becerra Lopez-Lavalle LA, Orjeda G (2002) Occurrence and cytological mechanism of 2n pollen formation in a tetraploid accession of Ipomoea batatas (sweet potato). J Heredity 93(3):185–192
Bednara J, Gielwanowska I, Rodkiewicz B (1986) Regular arrangements of mitochondria and plastids during sporogenesis in Equisetum. Protoplasma 130:145–152
Bretagnolle F, Thompson JD (1995) Gametes with the somatic chromosome number: mechanisms of their formation and role in the evolution of autopolyploid plants. New Phytol 129:1–22
Brown RC, Lemmon BE (1989) Minispindles and cytoplasmic domains in microsporogenesis of orchids. Protoplasma 148:26–32
Brown RC, Lemmon BE (1991) Pollen development in orchids. 1. Cytoskeleton and the control of division plane in irregular patterns of cytokinesis. Protoplasma 163:9–18
Brown RC, Lemmon BE (1992) Control of division plane in normal and griseofulvin-treated microsporocytes of Magnolia. J Cell Sci 103:1031–1038
Brown RC, Lemmon BE (1996) Nuclear cytoplasmic domains, microtubules and organelles in microsporocytes of the slipper orchid Cypripedium californicum A. Gray dividing by simultaneous cytokinesis. Sex Plant Reprod 9:145–152
Caetano-Pereira CM, Pagliarini MS (2001) A new meiotic abnormality in Zea mays: multiple spindles associated with abnormal cytokinesis in both divisions. Genome 44:865–871
Carputo D, Cardi T, Frusciante L, Peloquin SJ (1995) Male fertility and cytology of triploid hybrids between tetraploid Solanum commersonii (2n − 4x − 48, 2EBN) and Phureja–Tuberosum haploid hybrids (2n − 2x − 24, 2EBN). Euphytica 83:123–129
Crespel L, Ricci SC, Gudin S (2006) The production of 2n pollen in rose. Euphytica 151:155–164
Del Bosco SF, Tusa N, Conicella C (1999) Microsporogenesis in a Citrus interspecific tetraploid somatic hybrid and its fusion parents. Heredity 83:373–377
Den Nijs TPM, Peloquin SJ (1977) 2n gametes in potato species and their function in sexual polyploidization. Euphytica 26:585–600
Dweikat IM, Lyrene PM (1988) Production and viability of unreduced gametes in triploid interspecific blueberry hybrids. Theor Appl Genet 76:555–559
Ezura H, Kikuta I, Oosawa K (1994) Production of aneuploid melon plants following in vitro culture of seeds from a triploid × diploid cross. Plant Cell Tissue Organ 38:61–63
Harder ML, Verhagen S, Winton L, Einspahr DW (1976) Tetraploid aspen production using unreduced pollen from triploid males. For Sci 22:329–330
Hülskamp M, Parekh NS, Grini P, Schneitz K, Zimmermann I, Lolle SJ, Pruitt RE (1997) The STUD gene is required for male-specific cytokinesis after telophase II of meiosis in Arabidopsis thaliana. Dev Biol 187:114–124
Husband BC (2004) The role of triploid hybrids in the evolutionary dynamics of mixed-ploidy populations. Biol J Linn Soc 82:537–546
Jackson RC (1976) Evolution and systematic significance of polyploidy. Annu Rev Ecol Syst 7:209–234
Johnsson H (1940) Cytological studies of diploid and triploid Populus tremula and of crosses between them. Hereditas 26:321–352
Johnsson H (1942) Cytological studies of triploid progenies of Populus tremula. Hereditas 28:306–312
Kang XY (2002) Mechanism of 2n pollen occurring in Chinese white poplar. J Beijing For Univ 24(5/6):67–70
Kang XY, Mao JF (2001) Gamete fertility and morphological variations in offspring of triploid clones Populus tomentosa. J Beijing For Univ 23(4):20–23
Kang XY, Zhu ZT, Zhang ZY (1999) Morphology and meiosis of allotriploid Chinese white poplar. J Beijing For Univ 21(1):1–5
Kang XY, Zhu ZT, Lin HB (2000) Radiosensitivity of different ploidy pollen in poplars and its application. Acta Genet Sinica 27(1):78–82
Mashkina OS, Burdaeva LM, Belozerova MM, Vyunova LN (1989) Method of obtaining diploid pollen of woody species. Lesovedenie 1:19–25
Masterson J (1994) Stomatal size in fossil plants: evidence for polyploidy in majority of angiosperms. Science 264:421–424
McKim KS, Hawley RS (1995) Chromosomal control of meiotic cell division. Science 270:1595–1601
Mendes-Bonato AB, Risso-Pascotto C, Pagliarini MS, do Valle CB (2006) Cytogenetic evidence for genome elimination during microsporogenesis in interspecific hybrid between Brachiaria ruziziensis and B. brizantha (Poaceae). Genet Mol Biol 29(4):711–714
Müntzing A (1936) The chromosomes of a giant Populus tremula. Hereditas 21:383–393
Nilsson-Ehle H (1938) Production of forest trees with increased chromosome number and increased timber yield. Sven Papptidn 2:5
Owen HA, Makaroff CA (1995) Ultrastructure of microsporogenesis and microgametogenesis in Arabidopsis thaliana (L.) Heynh. Ecotype Wassilewskija (Brassicaceae). Protoplasma 185:7–21
Pfeiffer TW, Bingham ET (1983) Abnormal meiosis in alfalfa, Medicago sativa: cytology of 2N egg and 4N pollen formation. Can J Genet Cytol 25:107–112
Ramanna MS (1974) The origin of unreduced microspores due to aberrant cytokinesis in the meiocytes of potato and its genetic significance. Euphytica 23:20–30
Ramanna MS (1979) A re-examination of the mechanisms of 2n gametes formation in potato and its implications for breeding. Euphytica 28:537–561
Ramsey J, Schemske DW (1998) Pathways, mechanisms, and rates of polyploid formation in flowering plants. Annu Rev Ecol Syst 29:467–501
Ray M, Tokach MK (1992) Cytology of 2n pollen formation in diploid crested wheatgrass, Agropyron cristatum. Crop Sci 32:1361–1365
Rhee SY, Somerville CR (1998) Tetrad pollen formation in quartet mutants of Arabidopsis thaliana is associated with persistence of pectic polysaccharides of the pollen mother cell wall. Plant J 15(1):79–88
Risso-Pascotto C, Pagliarini MS, do Valle CB, Jank L (2004) Asynchronous meiotic rhythm as the cause of selective chromosome elimination in an interspecific Brachiaria hybrid. Plant Cell Rep 22:945–950
Risso-Pascotto C, Pagliarini MS, do Valle CB (2005) Multiple spindles and cellularization during microsporogenesis in an artificially induced tetraploid accession of Brachiaria ruziziensis (Gramineae). Plant Cell Rep 23:522–527
Santamour FS (1971) A triploid elm (Ulmus pumila × U. rubra) and its aneuploid progeny. Bull Torrey Bot Club 98(6):310–314
Spielman M, Preuss D, Li FL, Browne WE, Scott RJ, Dickinson HG (1997) TETRASPORE is required for male meiotic cytokinesis in Arabidopsis thaliana. Development 124:2645–2657
Steffen W, Fuge H, Dietz R, Bastmeyer M, Müller G (1986) Asterfree spindle poles in insect spermatocytes: evidence for chromosome-induced spindle formation? J Cell Biol 102:1679–1687
Tai W (1970) Multipolar meiosis in diploid crested wheat-grass Agropyrum cristatum. Am J Bot 57:1160–1169
Tavoletti S, Mariani A, Veronesi F (1991) Cytological analysis of macro- and microsporogenesis of a diploid alfalfa clone producing male and female 2n gametes. Crop Sci 31:1258–1263
Tel-Zur N, Abbo S, Bar-Zvi D, Mizrahi Y (2003) Chromosome doubling in vine cacti hybrids. J Heredity 94(4):329–333
Tel-Zur N, Abbo S, Mizrahi Y (2005) Cytogenetics of semi-fertile triploid and aneuploid intergeneric vine cacti hybrids. J Heredity 96(2):124–131
Thompson MM (1962) Cytogenetics of Rubus. III Meiotic instability in some higher polyploids. Am J Bot 49(6):575–582
Tilquin JP, de Brouwer K, Horvat F (1984) Unusual cytological patterns of microsporogenesis in a cultivar of Fuchsia. 1. Multiple spindles. Theor Appl Genet 67:413–417
Tondini F, Tawoletti S, Mariani A, Veronesi F (1993) A statistical approach to estimate the frequency of n, 2n and 4n pollen grains in diploid alfalfa. Euphytica 69:109–114
Traas JA, Burgain S, Dumas De Vaulx R (1989) The organization of the cytoskeleton during meiosis in eggplant (Solanum melongena (L.)): microtubules and F-actin are both necessary for coordinated meiotic division. J Cell Sci 92:541–550
Van Buijtenen JP, Joranson PN, Einspahr DW (1958) Diploid versus triploid aspen as pulpwood sources with reference to growth, chemical, physical and pulping differences. Tappi 41(4):170–175
Vorsa N, Bingham ET (1979) Cytology of 2n pollen formation in diploid alfalfa, Medicago sativa. Can J Genet Cytol 21:525–530
Yang CY, Spielman M, Coles JP, Li Y, Ghelani S, Bourdon V, Brown RC, Lemmon BE, Scott RJ, Dickinson HG (2003) TETRASPORE encodes a kinesin required for male meiotic cytokinesis in Arabidopsis. Plant J 34:229–240
Zhang ZH, Kang XY, Zhang PD, Li YH, Wang J (2007) Incidence and molecular markers of 2n pollen in Populus tomentosa Carr. Euphytica 154:145–152
Zhu ZT, Lin HB, Kang XY (1995) Studies on allotriploid breeding of Populus tomentosa B301 clones. Sci Silvae Sinicae 31(6):499–505
Acknowledgments
The authors would like to thank Dr. Jinfeng Zhang and Dr. Xiangming Pang from Beijing Forestry University and Dr. Paul Kretchmer at San Francisco Edit for revising the manuscript. This work was supported by the National Natural Science Foundation of China (Grant No. 30671708) and the National Key Technology R&D Programme of the 11th Five-Year Plan of China (Grant No. 2006BAD32B01).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by W. Boerjan
Rights and permissions
About this article
Cite this article
Wang, J., Kang, X. & Zhu, Q. Variation in pollen formation and its cytological mechanism in an allotriploid white poplar. Tree Genetics & Genomes 6, 281–290 (2010). https://doi.org/10.1007/s11295-009-0248-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11295-009-0248-3