Abstract
The genus Warburgia (Canellaceae) contains four tree species that are of valuable medicinal importance and are all found in Africa. Genetic diversity present in wild populations of these species is under great threat due to unsustainable harvesting for medicines and indiscriminate felling for timber and agricultural expansion. There is therefore an urgent need for conservation of these species. Some authors disagree about the taxonomy of the genus and list different species as synonyms. Amplified fragment length polymorphism (AFLP) technique was used to determine the genetic relationships between three species to resolve the taxonomic confusion. The amount of genetic variation within and among populations was assessed to guide strategies for their conservation and sustainable utilization. Four AFLP primer pairs (EcoRI/MseI) generated a total of 185 amplification products. Analysis of molecular variance revealed most variation among individuals within populations (63%, P < 0.0001), but variation among populations (37%, P < 0.0001) was highly significant as well. Constrained analysis of principal coordinates based on the Jaccard distance confirmed the separation among populations (38.2%, P < 0.0001). A phenetic tree and ordination graphs showed a clear distinction of W. ugandensis from W. salutaris and W. stuhlmannii. W. ugandensis populations from Uganda and western Kenya formed a subgroup that clustered away from the rest of the W. ugandensis populations. W. salutaris and W. stuhlmannii populations showed little genetic differentiation. An implication of the data to genetic management and taxonomic clarification is discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Warburgia (Canellaceae) includes four species: W. salutaris Berto f. Chiov found in southern Africa (Lesotho, South Africa, Swaziland, Mozambique and Zimbabwe), W. elongata Verd. and W. stuhlmannii Engl. found along the East African coastline, and W. ugandensis Sprague found within eastern Africa highlands. All these species are highly valued within traditional health systems for curing several ailments such as stomachache, constipation, toothache, common cold, cough, fever, muscle pains, weak joints, measles, and malaria (Beentje 1994; Kokwaro 1976; Watt and Breyer-Brandwijk 1962). The curative efficacy of extracts is linked to their antibacterial and antifungal activities, which has been scientifically proven in different in vivo and in vitro trials of the isolated major bioactive ingredients (Jansen and Groot 1991; Taniguchi and Kubo 1993; Treurnicht 1997; Haraguchi 1998; Mashimbye et al. 1999; Rabe and van Staden 2000; Olila et al. 2001). However, commercial processing of extracts is currently only being carried out for W. salutaris (Botha et al. 2004).
The taxonomy of some of these species is a subject to controversy, as some authorities refer to some of the four distinct species as synonyms. For example, Palmer and Pitman (1972) and Noad and Birnie (1989) refer to W. ugandensis and W. salutaris as synonyms of the same species based on their morphological characteristics and localities. Dale and Greenway (1961) suggest that W. stuhlmannii and W. ugandensis are conspecific. W. salutaris was formerly described under the names of Chibaca salutaris Bertol.f. and W. breyeri Pott (Palmer and Pitman 1972; Verdcourt 1990), but taxonomists later agreed that these are both synonyms for W. salutaris whose description took preference according to taxonomic rules (Verdcourt 1990). So far, this classification has been based on morphological characteristics. According to Verdcourt (1954), the three east African species may be distinguished by the sizes of the flowers and fruits: W. elongata can immediately be distinguished by its elongate fruits; W. stuhlmannii has small flowers with a staminal tube about 3.7 mm long and 1.2 mm in diameter, ten ovules, anthers about 1.0 mm long, and small fruits about 1.5 cm in diameter; W. ugandensis has considerably larger flowers with a staminal tube about 5 mm long and 2–3 mm in diameter, 30 ovules, anthers of 2.0 mm, and much larger fruits.
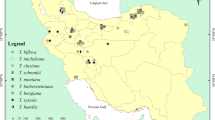
Deductions from several literature citations and herbaria specimen locations show that the species distribution falls into two main ecological categories: dry upland forests (including wooded riverines) and coastal grasslands (Fig. 1). The natural forests that these species inhabit are currently under great threat of destruction by clearing for farming and timber harvesting. Unsustainable exploitation of the species for medicinal purposes, mainly by harvesting the bark and roots, has also resulted in notable destruction in their natural habitats. As such, the species are reported as becoming rare in areas where they were once plentiful (Palmer and Pitman 1972; Beentje 1994). W. elongata and W. salutaris are listed as endangered and W. stuhlmannii as vulnerable in the ‘Red List’ of threatened species of the International Union for Conservation of Nature and Natural Resources (IUCN 2006). Indeed for this study, it was not possible to sample a W. elongata population. With the current trend in the global use of herbal medicine (Lange 2002) and the reliance of local communities worldwide on traditional medicines (estimated at about 80% by the World Health Organization; WHO 2002), continued exploitation of Warburgia species raises concerns regarding conservation and the sustainability of utilization. Thus, urgent measures to curb problems associated with overexploitation of both the trees and their habitats need to be addressed to ensure conservation of natural biodiversity and the traditional practices associated to its species.
Planting threatened medicinal tree species on farms and in plantations is one way of ensuring sustainability of production. This will reduce pressure exerted on natural forest stands while benefiting farmers with required products for their local use and generate income through commercial markets. There is great potential for commercial market development for extracts from Warburgia species. For example, in South Africa, Warburgia bark is highly valued, fetching more than 30 USD kg−1 in Zimbabwe (Cunningham 2003). Commercially processed W. salutaris in tablet form (to treat bronchitis, ulcers, chest infections, and thrush) are highly priced, ranging from 5.8 USD for 60 tablets (URL: http://www.vitalink.co.za) to 12.8 USD for 30 tablets (URL: http://www.biogenesis.co.za). A capsule form of the same species sells for US$ 60 per pack of ten tablets (URL: http://www.the-health-shop.net).
To develop sustainable management strategies for conservation and utilization, a clear taxonomic identification and a detailed understanding of the extent and distribution of genetic variation within a species are important. We employed the amplified fragment length polymorphism (AFLP; Vos et al. 1995) technique to evaluate the genetic structure of the morphologically defined species of the Warburgia genus to determine their phylogenetic relationships. We also assessed the amount of genetic variation within and among populations to guide genetic management. The AFLP technique is valuable for studying species where there is no prior sequence information and has been widely employed in taxa identification, determination of phylogenetic relationships as well as assessment of intraspecific diversity at molecular genetic level (Arens et al. 1998; Russell et al. 1999 and Muluvi et al. 1999 among others). This is the first study inclusive of all the Warburgia species where molecular markers have been used to assess genetic structuring and for validating their taxonomic relationships.
Materials and methods
Information from herbaria and literature citations on Warburgia species was used to outline their distribution to assist in identification of collection areas. Sampling was done for three species (W. salutaris, W. stuhlmannii, and W. ugandensis) from nine populations in Kenya (four), Mozambique (one), Tanzania (three), and Uganda (one), which were either natural or remnant on cultivated fields (Fig. 1 and Table 1). For the east African samples, identification of the specimens was based on the Flora of Tropical East Africa descriptions (Verdcourt 1954). A representative voucher specimen was also collected from each population and taken to herbarium in Nairobi (Kenya) and Lushoto (Tanzania) for confirmation of identity. Collection in Mozambique relied on local taxonomist for identification of the species. From each population, leaves were collected from 25 randomly sampled trees that were at least 100 m apart. The leaves were dried in snap-top plastic bags containing silica gel and stored at −20°C. Genomic DNA was extracted as described by Doyle and Doyle (1987). The AFLP method (Vos et al. 1995) from the plant mapping protocol of the Applied Biosystems (ABI), USA as designed for the AFLP Kit Module was used. Selective amplification was conducted using various combinations of two AFLP primers specific for MseI and EcoRI primer adaptors on a test panel of representative samples. The best primer combinations that gave reliable amplifications and polymorphisms in repeated trials were picked for the complete set of samples analyses. The capillary system (ABI PRISM 3730™) was used to resolve selective amplification products.
Data analysis
From the automated sequencers, the sample data were analyzed using GeneMapper™ software, which displays the sizing results as electrograms. The data were converted into a Microsoft Excel spreadsheet of allele frequencies in form of product presence (1) or absence (0). Genetic distance (D; Nei 1978) between population frequency data sets was generated with POPGENE 1.31 (Yeh et al. 1999). POPGENE also calculated genetic diversity levels for each population (H) according to Nei’s (1978) unbiased measure. Values were based on estimated allele frequency data assuming dominance and Hardy–Weinberg equilibrium. Analysis of molecular variance (AMOVA; Excoffier et al. 1992) based on Euclidean distances between individuals was undertaken with the ARLEQUIN 1.1 software package (Schneider et al. 2000).
A distance-based redundancy analysis (db-RDA; Legendre and Anderson 1999) was used to investigate the influence of geographical location (measured as population identity or as spatial coordinates of individuals) of the individual trees on differences in presence–absence of alleles measured by the Jaccard distance. db-RDA is a constrained ordination technique that combines ordination and regression approaches that allows for statistical investigation of explanatory variables on differences in distance among objects (Legendre and Legendre 1998; Legendre and Anderson 1999; Anderson and Willis 2003; Kindt and Coe 2005). The significance of ordination results was investigated by randomization test with 1,000 permutations. All the constrained analyses and ordination diagrams were obtained with the Biodiversity R software developed for the R statistical software (Kindt and Coe 2005; R Development Core Team 2005). The distribution of individuals of the same population within ordination diagrams was investigated by spiderplots (which connect each individual to the centroid or multivariate mean of their population), ordination ellipses (which calculate the average two-dimensional spread of positions of the same population), and by randomization tests (which randomly change population affiliation of individuals in a number of permutations).
Results
Of the 24 primer combinations tested, four combinations (EcoRI-ACA/MseI-CAA, EcoRI-ACT/MseI-CAT, EcoRI-AGC/MseI-CAT, and EcoRI-ACT/MseI-CAG) that showed good amplification and polymorphisms across all populations were selected for use on the full set of individuals. A total of 185 polymorphic makers were scored across the nine populations. Estimates of Nei’s unbiased genetic diversity (Table 1) showed two W. ugandensis populations (Laikipia from Kenya and Kibale from Uganda (H = 0.16 for both) and one W. stuhlmannii population (Pangani from Tanzania [H = 0.17]) being the most diverse, while one W. ugandensis population from (Lushoto from Tanzania [H = 0.12]) was the least diverse. Overall, the levels of genetic diversity within the sampled populations do not vary greatly. The percentages of polymorphic loci corresponded to the diversity estimates, as populations with higher diversity estimates also showed higher percentages of polymorphic loci (Table 1).
Summaries of AMOVAs based on all surveyed populations are shown in Table 2. Unstructured analyses based on all nine populations indicated that most variation was found among individuals within populations (63%, P < 0.0001) with significant variation partitioning among populations (37%, P < 0.0001). A similar case was observed in nested analyses where the populations were also partitioned by species. Only 10% of the total genetic variation partitioned among the species, with 29% of the variation among populations within species and 61% of the variation among individuals within populations, respectively. Further AMOVA based on W. ugandensis and W. stuhlmannii populations separately showed the same pattern. However, genetic differentiation among populations was higher in W. ugandensis (34%) than in W. stuhlmannii (14%). The phenogram (Fig. 2) based on Nei’s genetic distance showed three main groupings among the W. ugandensis populations, not corresponding to geographical distances. Two groups belonged to W. ugandensis, with one cluster of the two populations from the western side of the Rift Valley (Kitale from Kenya and Kibale from Uganda) and another cluster of populations from the eastern side of the Great Rift Valley. Interestingly, the Kibale/Kitale cluster of W. ugandensis formed an isolated out-group from all other species from this genus investigated in this study. W. stuhlmannii and W. salutaris populations clustered together, but formed two distinct subgroups: Kisarawe, a W. stuhlmannii population from the Tanzania, grouped together with Kwale, a W. stuhlmannii population from Kenya, while Pangani, a W. stuhlmannii population from Tanzania, grouped together with the geographically distant W. salutaris Changalane population from Mozambique.
Phenogram based on Nei’s (1978) genetic distance generated from 185 AFLP markers for 179 individuals sampled from nine populations of Warburgia species from Kenya (K), Mozambique (M), Tanzania (T), and Uganda (U)
The constrained ordination results for all individuals with population identities as explanatory variable show a clear differentiation between the various populations on the first two constrained ordination axes (Fig. 3; differentiation among populations explained 38.2% of squared Jaccard distance [P < 0.001]; differences between centroid positions of the populations explain 88.9% of variance of positions of the individuals in the two-dimensional ordination diagram of the first two ordination axes [P = 0.01]). Differentiation is quite clear between populations of W. ugandensis and those of W. stuhlmannii. The W. ugandensis populations are clearly split between a cluster of the Kitale (Kenya) and Kibale (Uganda) populations on the upper part of the ordination diagram and another cluster of the Laikipia (Kenya), Masai Mara (Kenya), and Lushoto (Tanzania) populations on the lower left-hand side of the diagram (Fig. 3). Although the Mozambican W. salutaris population has a centroid position within the same ordination quadrant as those for W. stuhlmannii populations, the overlap between positions of its individuals is less than for the latter species.
Ordination diagram for nine populations of Warburgia species generated with Jaccard db-RDA using population identity as explanatory variable with 185 AFLP markers. These first two ordination axes show 26.6% of squared Jaccard distance (P = 0.001 based on 1,000 permutations). Spiderplots connect each individual with the centroid of its population, whereas ellipses provide a summary of the distribution of individual positions for the same population
There was considerable overlap between the coastal W. stuhlmannii populations from Kisarawe (Tanzania) and Kwale (Kenya) as indicated by the overlap of ellipses and spiderplots of both populations (Fig. 3). For further insight, constrained ordination analysis with population identity as explanatory variable was therefore carried out for the three W. stuhlmannii populations (Kisarawe, Kwale, and Pangani). The ordination results show that the three populations are significantly different, although population differences explained a smaller amount of variation (Fig. 4; differentiation among populations explained 11.6% of squared Jaccard distance [P < 0.001]; differences between centroid positions of populations explained 40.8% of the variance of positions of the individuals in the two-dimensional ordination diagram of the first two ordination axes [P = 0.01]).
Ordination diagram for three W. stuhlmannii populations derived by Jaccard db-RDA using population identity as explanatory variable with 185 AFLP markers. These two constrained ordination axes explain 11.6% of squared Jaccard distance (P = 0.001 based on 1,000 permutations). Spiderplots and ellipses summarize the distribution of individuals of the same population
Discussion
Genetic structuring in relation to the taxonomic units of the genus Warburgia
The phenogram and ordination analyses revealed interesting information on the genetic relationships of the species in the genus Warburgia. The ordination results showed that there are significant differences between the investigated species and populations. W. ugandensis (which occurs within highlands) formed a major cluster away from other species, showing it is indeed a distinct taxonomic group. There is very little genetic differentiation between W. salutaris and W. stuhlmannii (D = 0.10; compared to D = 0.18 for the most genetically distant W. ugandensis populations). The two species occur at lower elevations than W. ugandensis. However, W. salutaris populations are also found at slightly higher elevations from W. stuhlmannii. Two W. stuhlmannii populations, Kwale (Kenya) and Kisarawe (Tanzania), were found to be genetically closer. Indeed their centroids in ordination diagrams occurred within very close range. However, the other W. stuhlmannii population (Pangani, Tanzania) clustered together with W. salutaris, which is geographically further away. This low genetic differentiation among W. salutaris and W. stuhlmannii raises questions on their taxonomic identities. The results imply that these two coastal species may be indeed one species and that the little genetic differentiation shown may be due to ecological adaptations resulting from random genetic drift (Heywood 1991). Isolated populations evolve separately as they adapt to new ecological habitats leading to changes in the allele frequencies hence the genetic differentiations (Epperson 1992). However, further analysis employing different phylogenetic markers, such as those slower involving evolving cpDNA, may provide more information in this respect.
Based on these results, inferences based on morphology only must be treated with reservation considering the uniformity in features across the coastal species and the close genetic relatedness revealed in the ordination analysis. There is a need for intensively sampling individuals from different species that are in close geographical distance, preferably where positions of the different populations overlap in the ellipse in ordination so that this genetic structuring can be discerned conclusively. Molecular studies have shown species previously classified as distinct taxa to belong to the same species. Within the eastern African flora, two such studies on Vitex species (Ahenda 1999; Lengeek et al. 2006) found little genetic differentiation between V. fischeri and V. keniensis and thus suggested that the two may be synonyms. Still within the coastal species group, the taxonomic status of W. elongata could not be concluded from this study. The W. stuhlmannii Kisarawe (Tanzania) population collection took place from the same locality indicated for W. elongata in the herbarium voucher specimens. However, none of the specimens was identified as having the morphological characters described in Verdcourt (1954) as the distinguishing characteristic for W. elongata (elongate fruits). Future collections narrowing down to the region where the species was earlier found could help to validate the taxonomic identity. Though only one specimen type of W. elongata is available at the East African Herbarium (Nairobi), it would be worthwhile to include the sample in future molecular analysis.
Considering the case of W. ugandensis populations alone, the results suggest that there may be two species among these populations that are quite genetically distinct from each other. Despite the geographical proximity of the Kitale (Kenya) population to other Kenyan W. ugandensis populations (Laikipia and Masai Mara, both <400 km away), it is genetically closer to Kibale from Uganda (>700 km away). This is the first time that such genetic distinction is being revealed for the species and suggests a possible case of speciation due to allopatry. However, a few other molecular works has been carried out on other trees species sampled across both central and western Kenya regions, which have shown similar genetic differentiation. While employing the random amplified polymorphic DNA (RAPD) analyses on an important timber species, V. fischeri (syn. V. keniensis), Lengkeek et al. (2006) found some limited differentiation in the species (19% of total variation among populations). Another RAPD study involving more populations across the two regions in Kenya on Prunus africana (Muchugi et al. 2006) and AFLP analysis of Lobelia giberroa (Mulugeta et al. 2007) showed a similar genetic disjunction between western and central highland Kenyan populations. These two species occur at slightly higher elevations than W. ugandensis. In the P. africana study, a substantially greater proportion of variation was partitioned among Kenyan populations (55%, P < 0.001), than in the current study, most of which was attributed to the split between central and western Kenyan regions.
Some authors (e.g. Hedberg 1951) have suggested that the East African mountain forest bridges are important for species dispersal, but this theory seems not to hold for the forests across the west and central Kenyan highlands. Muchugi et al. (2006) and Mulugeta et al. (2007) propose the distribution of many species across isolated African highland ‘islands’ to be a combination of long distance dispersal events and direct overland migrations facilitated by climate fluctuations. The major unique geological feature between the western and central Kenyan highlands is the Great Rift Valley. The climatic fluctuations of the Pleistocene are said to have contributed to the shaping of species (Hamilton 1982). Within Kenya, W. ugandensis populations are found in the ravine at the floor of the Rift Valley (e.g., the Masai Mara population included in this study), thereby providing connectivity of the western and central Kenyan highlands. However, the clear genetic differentiation of W. ugandensis Masai Mara (Kenya) population from the Kitale (Kenya) and Kibale (Uganda) from the AFLP data disputes chances of long distance dispersal model and instead supports migratory theory.
Very little genetic differentiation is seen among W. salutaris and W. stuhlmannii populations (D = 0.10 compared to D = 0.18 for the most genetically distant highland populations). Interestingly, the geographically proximate W. stulhmanii populations from Kenya and Tanzania do not cluster together as expected (Fig. 2). As W. salutaris and W. stuhlmannii show considerable genetic similarity, the seed dispersal hypothesis seems more likely to play the major role in the genetic exchange along the East African coast and southern region. According to the resolution of our AFLP markers, these two species seem not as genetically distinct as expected from their morphological characteristics that classified them into their individual taxonomic units.
It is important to note that morphological characters described in Verdcourt (1954) as distinguishing characteristic for W. elongata (elongate fruits) were not observed in the Kisarawe population, though collection took place from the same locality as the voucher specimens. The collector did not identify any specimen with distinguishing characteristic for W. elongata during the survey, concurring with the local community who claim not to have seen such specimen. Has the species therefore become extinct? This is quite an interesting question considering its 2006 IUCN classification as ‘endangered’. If W. elongata was found to be genetically close to the other two coastal species W. salutaris and W. stuhlmannii, this may suggest that the elongate fruits in W. elongata were an adaptive feature that could not be sustained. Therefore, inferences based on morphology only must be treated with reservation considering the uniformity in features across the coastal species and the results of the statistical analysis. There is a need for intensively sampling individuals of different species that are in close geographical distance, preferably where positions of the different populations overlap so that this genetic structuring can be discerned conclusively. Other approaches such as cytology can also be used to validate the species status in the genus.
Implications of these findings to genetic management of the species in utilization and conservation
AMOVA and db-RDA showed that most of the genetic variation is retained within individuals rather than among populations, as previously found in other tropical tree species (Hamrick et al. 1992; Nybom and Bartish 2000; Jamnadass et al. 2005). There is also significant genetic differentiation among populations resulting from differences observed between the coastal and highland species as well as the differences between eastern and western highlands populations. As genetic diversity of populations was high, intensive sampling of a few populations for each species will capture most of the genetic variation. Considering international and national genetic management programs, the Kenyan W. ugandensis populations occupying both the eastern and western highlands require different management approaches that take into account the diverse genetic resources in the region. If phytochemical studies would show that individuals from either the eastern or from the western highlands to be superior in efficacy of their extracts, planting programs should ensure that their genetic distinction is retained, as it may be related to ecological adaptation and transfers may erode genetic diversity. However, further studies can look at the possibilities of any existing genetic hybridization within the eastern and western populations and its implications to the conservation of the genetic resource.
Ecogeographical surveys during sample collection showed diminishing populations of Warburgia species. Population decline was especially critical along the coast. Based on these results, more emphasis should be placed on conservation strategies for the coastal species, despite the little genetic differentiation shown among them. As these species represent a diverse genetic resource from the highland groups, both cultivation and circa situ conservation should be encouraged in all countries. With respect to the cultural background of the local communities along the Kenyan and Tanzanian coasts, tree-planting practice is minimal unlike among communities living within the highlands. The few W. ugandensis trees that had been planted all occurred within farmer’s fields within the highlands. Individual governments or nongovernmental organizations should therefore take the initiative in the conservation of coastal regions. Maybe in situ conservation of the species in designated protected lands or ex situ in public lands (such as parks or schools) can be undertaken. However, there is still the risk of subdivision of these protected and public lands in the future; this may lead to indiscriminate tree felling. The extension of conservation sites along the coastline (e.g. the sacred Kayas of Kenya), a few kilometers into the mainland, can also help in in situ conservation of the Warburgia species.
References
Ahenda JO (1999) Taxonomy and the genetic structure of Meru oak populations, Vitex keniensis Turrill and Vitex fischeri Gurke, in East Africa. PhD Thesis. Wageningen University, The Netherlands
Anderson MJ, Willis TJ (2003) Canonical analysis of principal coordinates: a useful method of constrained ordination for ecology. Ecology 84:511–525
Arens P, Coops H, Jansen J, Vosman B (1998) Molecular genetic analysis of black poplar (Populus nigra L.) along Dutch rivers. Mol Ecol 7:11–18
Beentje HJ (1994) Kenya trees, shrubs and lianas. National Museums of Kenya, Nairobi, Kenya
Botha J, Witkowskie ETF, Shackleton CM (2004) The impact of commercial harvesting on Warburgia salutaris (‘pepper-bark tree’) in Mpumalanga, South Africa. Biodivers Conserv 13:1675–1698
Cunningham T (2003) Return of the pepper bark tree. Peoples and Plant Online. http://www.rbgkew.org.uk/peoplesplants/africa/warburgia/htm
Dale IR, Greenway PJ (1961) Kenya trees and shrubs. Buchanan’s Kenya Estates, Nairobi, Kenya
Doyle JJ, Doyle JL (1987) CTAB DNA extraction in plants. Phytocheml Bull 19:11–15
Epperson BK (1992) Spatial structure of genetic variation within populations of forest. New Forest 6:257–278
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: applications to human mitochondria DNA restriction data. Genetics 131:479–491
Grunwald J, Buttel K (1996) The European phytotherapeutics market: figures, trends and analyses. Drugs Made Ger 39:6–11
Hamilton AC (1982) Environmental history of East Africa. A study of the Quatenary. Academic, London
Hamrick JL, Godt MW, Sherman-Broyles SL (1992) Factors influencing genetic diversity in woody plants species. New For 6:95–124
Haraguchi H (1998) Mode of action of antimicrobial substances in plants. J Agri Food Chem 2:259–268
Hedberg O (1951) Vegetation belts of the east African Mountains. Sven Bot Tidskr 45:140–202
Heywood JS (1991) Spatial analysis of genetic variation in plant populations. Ann Rev Ecolog Syst 22:335–355
IUCN 2006. 2006 IUCN Red List of Threatened Species. http://www.iucnredlist.org. Downloaded on 20 October 2006
Jamnadass R, Hanson J, Poole J, Hanotte O, Simons AJ, Dawson IK (2005) High differentiation among populations of the woody legume Sesbania sesban in sub-Saharan Africa: implications for conservation and cultivation during germplasm introduction into agroforestry systems. For Ecol Manag 210:225–238
Jansen BMJ, de Groot A (1991) The occurrence and biological activity of drimane sesquiterpenoids. Nat Prod Rep 8:309–318
Kindt R, Coe R (2005) Tree diversity analysis. A manual and software for some common statistical methods for biodiversity and ecological analysis. World Agroforestry Centre (ICRAF), Nairobi. URL: http://www.worldagroforestry.org/treesandmarkets
Kokwaro JO (1976) Medicinal plants of East Africa. East African Literature Bureau. Nairobi, Kenya
Lange D (2002) The role of east and southeast Europe in the medicinal and aromatic plants’ trade. Med Plant Conserv 8:14–18
Legendre P, Anderson MJ (1999) Distance-based redundancy analysis: testing multi-species responses in multi-factorial ecological experiments. Ecol Monogr 69(1):1–24
Legendre P, Legendre L (1998) Numerical ecology. Elsevier Science BV, Amsterdam, p 853
Lengkeek AG, Mwangi AM, Agufa CAC, Ahenda JO, Dawson IK (2006) Comparing genetic diversity in agroforestry systems with natural forest: a case study of the important timber tree Vitex fischeri Gürke (syn. Vitex keniensis Turrill) in central Kenya. Agrofor Syst 67:293–300
Mashimbye MJ, Maumela MC, Drewes SE (1999) A drimane sesquiterpinoid lactone from Warburgia salutaris. Phytochemistry 51:435–438
Muchugi A, Lengkeek AG, Kadu CAC, Muluvi GM, Njagi ENM, Dawson IK (2006) Genetic variation in the threatened medicinal tree Prunus africana Cameroon and Kenya: implications for current management and evolutionary history. S Afr J Bot 72:498–506
Mulugeta K, Ehrich D, Taberlet P, Nemomissa S, Brochmann C (2007) Phylogeography and conservation genetics of giant lobelia (Loblia giberroa) in Ethiopia and Tropical East African mountains. Mol Ecol. DOI 10.1111/j1365-294X.2007.03232.x
Muluvi GM, Sprent JI, Soranzo N, Provan J, Odee D, Folkard G, Mcnicol JW, Powell W (1999) Amplified fragment length polymorphism (AFLP) analysis of genetic variation in Moringa oleifera Lam. Mol Ecol 8:463–470
Nei M (1978) Estimation of average heterozozygosity and genetic distance from a small number of individuals. Genetics 89:583–590
Noad T, Birnie A (1989) Trees of Kenya. General Printers, Nairobi, Kenya
Nybom H, Bartish IV (2000) Effects of life history traits and sampling strategies on genetic diversity estimates obtained with RAPD markers in plants. Perspect Plant Ecol Evol Syst 3:93–114
Olila D, Olwa-Odyek B, Opuda-Asibo J (2001) Antibacterial and antifungal activities of extracts of Zanthoxylum chalybeum and Warburgia ugandensis, Ugandan medicinal plants. Afr Health Sci 1(2):66–72
Palmer E, Pitman N (1972) Trees of Southern Africa Vol. 2. A.A. BalKema Cape Town, SA
Rabe T, van Staden J (2000) Isolation of an antibacterial sesquiterpenoid from Warburgia salutaris. J Ethnopharmacol 73:171–174
R Development Core Team (2005) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org
Russell JR, Weber JC, Booth A, Powell W, Sotelo-Montes C, Dawson IK (1999) Genetic variation of Calycophyllum spruceanum in the Peruvian Amazon Basin revealed by amplified length polymorphism (AFLP) analysis. Mol Ecol 8:199–204
Schneider S, Kueffer JM, Roessli D, Excoffier L (2000) Arlequin ver. 1.1: A software for population genetic data analysis. University of Geneva, Switzerland
Taniguchi M, Kubo I (1993) Ethnobotanical drug discovery based on medicine men’s trials in the African savannah: screening of East African plants for anti-microbial activity II. J Nat Prod 56:1539–1546
Treurnicht FT (1997) An evaluation of the toxic and potential antiviral effects of some plants used by South Africans for medicinal purposes. MSc thesis, University of Stellenbosch, South Africa
Verdcourt B (1954) Notes on the Tropical African Canellaceae (East African Herbarium, Nairobi). Kew Bulletin No 4, 1954
Verdcourt B (1990) Canellaceae. Flora of Zimbabwe. Volume 7, Part 4. http://www.zimbabweflora.co.zw/fams1.2/canellac.php. Accessed on 09/07/2007
Vos P, Hogers R, Bleecker M, Reijans M, van de Lee T, Hornes M, Freijters A, Pot J Peleman J, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acid Res 23:4407–4414
Watt JM, Breyer-Brandwijk MG (1962) The medicinal and poisonous plants of Southern and Eastern Africa. E & S. Livingstone, Edinburgh, UK
World Health Organisation (2002) Traditional medicine strategy 2002–2005. Department of Essential Drugs and Medicine Policy. WHO, Geneva, Switzerland
Yeh FC, Yang RC, Boyle T (1999) Popgene version 1.31. University of Alberta, Canada
Acknowledgments
This work formed part of Alice Muchugi’s Ph.D. thesis at Kenyatta University, Nairobi. We gratefully acknowledge the assistance of Levand Turyomurugyendo (Uganda), Evaline Sambane (Mozambique), and Nassoro Magogo (Tanzania) in the collection of plant material for this research. Funding was provided by the World Bank under the System-wide Genetic Resources Programme to World Agroforestry Centre (ICRAF).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Kremer
Rights and permissions
About this article
Cite this article
Muchugi, A., Muluvi, G.M., Kindt, R. et al. Genetic structuring of important medicinal species of genus Warburgia as revealed by AFLP analysis. Tree Genetics & Genomes 4, 787–795 (2008). https://doi.org/10.1007/s11295-008-0151-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11295-008-0151-3