Abstract
The diversity of easy-to-study organisms (e.g. vascular plants) is often used as a proxy for the diversity of other organisms whose investigation needs more effort, time and specialist knowledge. Some previous studies have found positive relationships between plant and macrofungal diversity and thus support this approach, while others question this practice. Our aim was to explore the possibility of using plant diversity as surrogate for macrofungal diversity in the forests of the Pannonian ecoregion. A total of 19 permanent plots in north-east Hungary were sampled for vascular plants and macrofungi. The effect on macrofungal abundance and diversity, as well as degradation level, of plant evenness and richness was tested using generalized linear models. Species richness of macrofungi assemblages proved to be independent of the diversity and naturalness of vascular plant communities; however, there was congruence in the composition of the two communities. In contrast to diversity, macrofungi abundance was significantly negatively correlated to plant species richness. There was a hump-backed relationship between the abundance of terricolous macrofungi and the degradation level estimated on the basis of the occurrence of vascular plants, although degradation did not influence the abundance of lignicolous macrofungi. Our results question the reliability of decisions on nature conservation actions based on a few groups of easy-to-observe organisms, and underline the necessity of studying as wide a range of taxonomic groups as possible.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human activities cause loss of biodiversity by degradation, reconstruction, fragmentation and over-utilisation of habitats, introduction of alien species as well as the spread of infectious diseases (Primack 1998). These negative effects are well-documented for some components of biodiversity (e.g. birds, reptiles, mammals, plants; Oleksyn and Reich 1994; Findlay and Houlahan 1997; Lenzen et al. 2009) while we have only scarce information on the influence of human activities on many other organisms. Macrofungi are an often neglected component of biodiversity; however, there is evidence that habitat degradation adversely influences the number of fruiting bodies of macrofungi and diminishes the diversity of the fungal community (Arnolds 1988, 1991; Ohenoja 1988; Zak 1992; Miller and Lodge 1997).
The main reasons that macrofungi are neglected when making decisions on nature protection activities (including selection of protected areas) is the lack of information on their abundance and distribution, and their unexplored taxonomic diversity. Researchers have attempted to solve these problems through the use of better-known surrogate taxa, such as vascular plants. Vascular plants are often used as a surrogate for estimating the diversity of other taxa because they are fundamental components of the structure of terrestrial ecosystems, reflecting environmental conditions and providing habitat for other organisms. Moreover, sampling of vascular plants is easier (Ryti 1992; Pharo et al. 2000; Chiarucci et al. 2005; Santi et al. 2010). The practice of using plant communities as a proxy for fungal communities is supported by the congruence in composition between them reported in several studies (e.g. Nantel and Neumann 1992; Packham et al. 2002; Saetersdal et al. 2003; Chiarucci et al. 2005; Brown et al. 2006; McMullan-Fisher et al. 2010). However, there is no clear evidence that higher plant diversity indicates higher fungal diversity: continental-scale meta-analysis (Schmit et al. 2005), and regional analyses (Villeneuve et al. 1989; Gabel and Gabel 2007; McMullan-Fisher 2008) have found significant positive correlation. On the other hand, studies by Chiarucci et al. (2005) and Santi et al. (2010) found that the species richness of the two communities were not correlated in spite of the positive correlation between their compositions in earlier studies. More recently, Gómez-Hernández and Williams-Linera (2011) reported a negative correlation between macrofungi and overstorey tree richness. Simulation studies (Virolainen et al. 2000; Chiarucci et al. 2005; McMullan-Fisher et al. 2010) showed that site-selection optimised based on plant communities cannot maximize fungal diversity, even if there is a statistical relationship between the compositions of the two groups. In studies by Virolainen et al. (2000) and McMullan-Fisher et al. (2010), random selection performs better than selection based on plant species.
Several factors may underlie these contradictory results: (1) the studies were performed at different scales (Flather et al. 1997) (from continental scale meta-analysis to comparing degradation stages of the same vegetation type); (2) different fungal groups may respond differently to environmental factors and may correlate with different proxies (cf. Villeneuve et al. 1989); (3) previous studies focussed on species composition and species richness and neglected other components of fungal diversity (e.g. evenness).
The aim of the present study was to overcome the limitations in our knowledge in relation to the last two points. Therefore, we investigated the relationship between abundance and diversity of macrofungi (and its two main subgroups) and diversity of vascular plants using different diversity measures (species richness, Shannon-diversity, evenness). Furthermore, we studied the consequences of habitat degradation on fungal communities using plant species as degradation indicators.
The study area is situated in the Carpathian Basin, whose biodiversity represents a very unique ecological entity in comparison with other European regions. For this reason, it is considered as a distinct ecological region (Pannonian ecoregion) in the Natura 2000 programme. Only some preliminary investigations of macrofungi–plant interactions have been published for the Pannonian ecoregion (Benedek et al. 2005; Pál-Fám et al. 2007).
Materials and methods
Study area
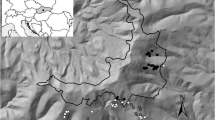
Our study was performed in the Cserehát—a hilly landscape in the north-east of Hungary (Fig. 1). The climate is rather cool, with an annual mean temperature 8–8.5 °C. The annual mean precipitation is about 600 mm, but about 400 mm falls during the growing season, which is important for fungal fructification. The most characteristic soil type is brown forest soil with clay illuviation on loess (Dorgai 1986).
The landscape is dominated by agricultural cultivation. Its forest vegetation has been changed significantly as a result of excessive forestry management, hunting and grazing. The area can be characterised by fragmented forest stands and alien tree plantations with invasive plant species in the herb layer. The study was carried out in five locations 50–80 km away from the city of Miskolc, at 200–250 m altitude above sea level near the villages Abaújlak (48°24′N, 20°57′E), Nyésta (48°23′N, 20°58′E), Irota (48°24′N, 20°52′E), Szendrőlád (48°20′N, 20°44′E) and Büdöskútpuszta (48°22′N, 20°44′E).
Semi-natural stands of beech (Melittio-Fagetum Soó 1964 emend. 1971), zonal Turkey oak (Quercetum petraeae-cerris Soó 1963), and extrazonal hornbeam-oak (Carici pilosae-Carpinetum Neuhäusl et Neuhäuslová-Novotná 1964 emend. Borhidi 1996), as well as oak wood-pastures, Scots pine (Pinetum sylvestris cultum) and Norway spruce plantations (Piceetum cultum) were investigated and characterised (Rudolf et al. 2008) (Table 1). The nomenclature of the plant associations follows the work of Borhidi (2003).
Sampling
Two sampling sites were selected in the most common forest type (i.e. zonal Turkey oak forest) and one site in other main forest types. In larger (1–3 ha) and more heterogeneous sites, five plots were selected, while in the smaller (<1 ha) and more homogeneous stands only 1 plot was sampled. Thus, in seven sites of the six vegetation types, 19 sample plots (1,200 m2/plot) were selected and marked. In the sites located at Nyésta and Abaújlak, the plant surveys were made in the spring and summer of 1995–1996; however, in the other locations (Irota, Szendrőlád and Büdöskútpuszta), sampling took place in 2004–2005. For each plot, all vascular plants (both woody and herbaceous) were recorded and their cover estimated visually in the herb, shrub, and canopy layers separately (Bullock 2006). The work of Simon (2000) was used for the identification of vascular plants. Fungi were sampled in the years 1995, 1996, 1998, 2002 in the sites at Nyésta and Abaújlak and in 2004–2006 at the other locations. Macrofungi species were recorded and fruit bodies were counted as measure of species abundance on six to eight occasions in each year between July and November. We investigated only epigeious macrofungi. These belong to following groups: soil saprotrophic, mycorrhizal, wood saprotrophic, necrotrophic parasites and saprotrophic on other plant remains. The literature used for determination of ‘semipileate’ species provides a measure of each species when they grow separately (Hansen and Knudsen 1997). Thus, we estimated abundance using these measures separately for each semipileate species; macrofungi were determined by K.R. and F.P.-F. Species names followed the work of Krieglsteiner (1991) and the Index Fungorum (CABI 2008). Many macrofungal species are known to fruit sporadically, with diverse patterns of occurrence from year to year (Watling 1995) due to different factors such as climatic and edaphic conditions. Accordingly, many years of intensive surveys are necessary to describe the macrofungal communities of a vegetation unit (Tofts and Orton 1998). On the other hand, vegetation composition dominated by long-lived perennials was recorded for only 1 year in each plot, thus the difference between years due to environmental fluctuations is negligible relative to differences between vegetation types.
The proportion of disturbance indicator (hereafter called degradation level) plant species was calculated using species categorization of Social Behaviour Types (Borhidi 1995). This measures the level of degradation caused by human activities or natural processes and indicated by changes in species composition (Morschhauser 1995). Social Behaviour Types categories are a refinement of Grime’s (1979) strategies adapted to Hungarian Flora. Plant species indicating degradation belong to Grime’s ‘ruderal’ category, except pioneer species of primary succession, which belong to the ‘natural pioneers’ category in Borhidi’s classification.
Applied diversity indices
There are a lot of diversity indices (Magurran 2004), and different indices may be sensitive to different aspects of abundance distribution, and thus may rank communities in different ways (Hurlbert 1971; Tóthmérész 1995). This problem can be solved by using parametric diversity families (Rényi 1961; Hill 1973; Aczél and Daróczy 1975; Patil and Taillie 1979; Keylock 2005) instead of a single diversity index (Tóthmérész 1995). Indices of these families show varying sensitivities to rare and abundant species as the scale parameter (α) changes. Jost (2006) suggested that, instead of entropy, “the effective number of species” or “numbers equals” (i.e. the number of equally abundant species that would result in the same entropy value) should be used as the unit of diversity. It can be proved that, if this suggestion is followed, all the above-mentioned diversity families lead to the same form (Jost 2006):
where p i is the relative abundance of the ith species, and α is the scale parameter.
Plotting diversity values (D α) as a function of scale parameter (α) is a useful tool with which to compare communities (Tóthmérész 1995), but lots of correlated diversity values related to similar α values cannot be used as independent variables in statistical modeling. Therefore, in this study, three scale parameter values were chosen: α = 0, which leads to species richness; α = 1, which corresponds to Shannon entropy; and α = 2, which corresponds to the Gini–Simpson index.
If α > 0, diversity can be split into two components: species richness and evenness of the species abundance values (Pielou 1977). Evenness was measured by the ratio of diversity and richness:
In this paper, we use the term diversity in general statements that are valid for any component of diversity. When referring to diversity values at α = 1 or α = 2, we use term ‘diversity value’.
Plant diversity was calculated from visually estimated cover data, while fungal diversity was calculated from abundance (number of sporocarps) data.
Large differences in total macrofungi abundance (which ranged from 119 to 8,961 for all species; from 47 to 1,131 for terricolous macrofungi; and from 24 to 8,460 for lignicolous macrofungi) might distort differences in diversity, resulting in high diversity in sample plots with high abundance (Gotelli and Colwell 2001). To eliminate this artifact, macrofungi richness, diversity and evenness values were calculated in 50 random subsamples and the averages were used in subsequent analyses. Random subsamples were generated by random selection of fruitbodies without replacement. The subsample size was set to slightly lower than the lowest abundance: 100 individuals for the whole macrofungi assemblage, 40 for terricolous macrofungi, and 20 for lignicolous macrofungi.
Statistical analysis
Species composition of plant and fungal communities was compared using the Mantel test based on Sorensen (binary data) and Bray-Curtis (abundance data) dissimilarities (Legendre and Legendre 1998) using 9,999 randomizations.
Diversity values calculated with different scale parameters (Hill 1973), and components of diversity (Stirling and Wilsey 2001) may be strongly correlated. Correlation among independent variables results in inaccurate estimation of regression coefficients, while correlation among dependent variables may lead to over-interpretation of the results. To avoid these problems, correlations (see Appendix) were calculated within variable sets (plants, macrofungi, terricolous macrofungi, lignicolous macrofungi), and uncorrelated variables (Table 2) were selected for the analyses.
The relationships between independent and dependent variables were studied by generalized linear models (Crawley 2007), which reveal non-monotonic (e.g. hump-backed) relationships. In each model, one dependent variable, and linear and quadratic terms of independent variables within one variable set, were entered. In the case of diversity, evenness and richness values, Gaussian distribution (according to the central limit theorem, the distribution of averages is approximately Gaussian) and identity link function were used; in the case of abundance data, quasi-Poisson distribution and log link function were used. The quasi-Poisson distribution allows over-dispersion (higher variance than mean), thus it is suitable if the spatial distribution of the individuals is aggregated. The effect of independent variables was tested by Type II F test (Fox 2009). All analyses were carried out in R version 2.9.2 (R Development Core Team 2009) using the vegan package (Oksanen et al. 2009) for rarefaction and calculating diversity and evenness values, and the car package (Fox 2009) for F tests.
Results
In all, 276 vascular plants and 266 macrofungi species were recorded in the 19 sample plots; 50 species of the vascular plants were trees or shrubs.
According to the Mantel tests, there was congruence between the composition of plant and fungal communities; however, the correlations were higher if dissimilarities were calculated from binary data. Composition of terricolous fungal communities was related more strongly to plant communities than to lignicolous fungal communities (Table 3).
Species richness of macrofungi assemblages proved to be independent of the diversity and degradation of vascular plant communities (Tables 4, 5). An exception was the quadratic relationship between evenness of plant communities and richness of lignicolous macrofungi (Table 4). However, even this relationship becomes non-significant after removing the plot with highest plant evenness from the analysis (Table 4). Improved fit of residuals to the normal distribution supports exclusion of this outlier point.
The total abundance of macrofungi seems to equal the quadratic function of evenness of plant communities (Table 4; Fig. 2). However, after excluding the plot with highest macrofungi abundance, which proved to be an outlier, the pattern changed: macrofungi abundance is negatively related to plant richness and is independent of plant evenness (Table 4; Fig. 3).
Relationship between evenness of plant communities and total abundance of macrofungi. Different forest types: open squares turkey oak forest, open circles scotch pine plantation, open triangles hornbeam-oak forest, crosses beech forest, asterisks Norway spruce plantation, filled triangles oak wood-pasture
Relationship between species richness of plant communities and total abundance of macrofungi. Different marks indicate different forest types: empty square turkey oak forest, empty circle scotch pine plantation, empty triangle hornbeam-oak forest, x beech forest, asterisk Norway spruce plantation, filled triangle oak wood-pasture
There is a hump-backed relationship between the abundance of terricolous macrofungi and the degradation level estimated by the occurring vascular plants (Table 5; Fig. 4), but degradation does neither influence abundance of lignicolous macrofungi nor species richness in any macrofungi group.
Relationship between degradation level % and abundance of terricolous macrofungi. Different forest types: open squares turkey oak forest, open circles scotch pine plantation, open triangles hornbeam-oak forest, crosses beech forest, asterisks Norway spruce plantation, filled triangles oak wood-pasture
Discussion
Composition of fungal and plant communities
Similar to previous studies (e.g. Nantel and Neumann 1992; Packham et al. 2002; Chiarucci et al. 2005; Saetersdal et al. 2003; Brown et al. 2006; McMullan-Fisher et al. 2010), we found that there is congruence between plant and fungal communities. This relationship is stronger for terricolous than for lignicolous fungal communities because, due to forest management, dead wood is almost absent from the studied plots except in the beech forest, which was richest in dead wood from both a qualitative and quantitative point of view.
Macrofungi diversity versus vegetation
Our results suggest that diversity of vascular plants (including richness and evenness) cannot be used as a proxy for macrofungi diversity; neither can diversity values or their components, i.e. species richness and evenness. These results coincide with those of Chiarucci et al. (2005) and Santi et al. (2010), while others (e.g. Schmit et al. 2005; Gabel and Gabel 2007; McMullan-Fisher 2008) have found significant positive correlations. In some previous studies, the lack of positive correlation may be attributed to methodological shortcomings: e.g. the sampling of Santi et al. (2010) covered only 1 year, which is not sufficient for macrofungi examinations. However, that is not the case in Chiarucci et al. (2005) and our study. In both these latter studies, small-sized habitats were sampled within a relatively small area.
There are different possible explanations for the positive correlation between diversity of different groups of organisms. To understand the lack of correlation in our case, we should explore why these possible mechanisms did not act. It could be that the expected diversity of both plants and macrofungi is influenced by habitat microheterogeneity, causing a positive correlation between these diversities. In European temperate forests, the species pool of woody species is small, thus the richness of vascular plants is determined mainly by the richness of the herb layer. Plant species richness was highest in the oak wood-pasture plots with the lowest canopy cover and a high variation of light availability, while it was lowest in Norway spruce plantations with evenly low light availability. This suggests that plant species richness (especially the richness of the herb layer) was influenced by the amount of, and variation in, light availability, which has a lower influence on heterotrophic macrofungi.
Another chain of events that could create a positive correlation between plant and macrofungi diversity is the following: higher plant diversity generates higher diversity of substrate for macrofungi, which generates higher macrofungi diversity. This is probably why the diversity of tree species is a promising surrogate for macrofungal diversity at the global scale (Schmit et al. 2005). The lack of positive correlation in our data is not evidence against this mechanism. Thus, it is possible that this mechanism operates in the Cserehát forests but that its effect is overwhelmed by other factors. Due to the pauperisation of European flora during the Ice Age, even the most diverse European forests are more species-poor than temperate forests in other continents. This narrower range of tree diversity values makes it hard to find a significant relationship. Gabel and Gabel (2007) showed a significant relationship between species diversity of macrofungi and plants in an area less influenced by glaciation.
Macrofungi abundance versus vegetation
Contrary to macrofungi diversity, macrofungi abundance is significantly correlated to plant diversity. Plots in Piceetum and Pinetum communities showed low plant evenness, while the abundance of macrofungi had high values. In both young plantations, the species pools of the canopy, shrub, and herb layers are small and fallen needles form a barely decayable layer of litter. The richness and productivity of terricolous saprotrophs (main litter decomposers) is known to depend on the quantity and quality of litter and is related significantly to the diversity of vascular plants (Villeneuve et al. 1989). In addition, the local abundance of a species is correlated with the size of its geographic range (Gaston 1994; Johnson 1998; Straatsma and Krisai-Greilhuber 2003). Therefore, in these fungal assemblages, a few less demanding, unspecific, higher-production species dominate. However, in the Norway spruce plots, the higher abundance values are caused mainly by the number of fruitbodies of lignicolous species. This fungal community is dominated by species living on Norway spruce deadwood (Trichaptum fuscoviolaceum, Marasmius androsaceus, Baeospora myosura) and widespread litter decomposers (Rhodocollybia butyracea, Lepista flaccida, Mycena filopes, Mycena pura). In macrofungal communities of artificial spruce forests examined by Kost (1992), the common litter decomposing and widespread lignicolous macrofungi can be found in abundance.
The old-growth beech forests, rich in arboreal species, showed greater evenness but low abundance of macrofungi. In most unmanaged beech forest plots, besides highly productive and frequent species such as the lignicolous Bisporella citrina, Trametes hirsuta, Micromphale foetidum, Schizophyllum commune, the more rare ectomycorrhizal fungi with low abundance such as Cortinarius alboviolaceus, C. terpsichores, Inocybe asterospora, I. furfurea, Lactarius flavidus, Russula fellea and Thelephora palmata can be found in greater proportion, presumably because of the host trees and forest structure (Bujakiewicz 1992; Pál-Fám et al. 2007) as well as the physical and chemical properties of the humus (Nantel and Neumann 1992).
The abandoned oak wood-pasture is the richest in vascular plants, but fungal abundance is low there. There are several possible explanations for low sporocarp abundance. This habitat is characterised by alternation of old trees (in small groups or standing alone) and patches of grass, thus it has an open canopy. This results in more extremes of temperature and lower humidity (Brown et al. 2006), which decreases the number of sporocarps. Due to the warmer and drier microclimatic conditions, the litter decomposes slowly, therefore, the abundance of the terricolous species may be reduced. The abandonment of grazing may be unfavourable for many macrofungi (Arnolds 1992). The total species richness is high but the species richness of trees and shrubs is low in this vegetation type. Low numbers of ectomycorrhizal partners and small quantity of fallen deadwood may also cause low sporocarp production.
The evenness of vegetation was greatest in the two Turkey oak forests. This results in a relatively high number of species and abundance of macrofungi due to the forest structure, quality of litter and edaphic conditions.
Effect of degradation
At low disturbance level (indicated by the degradation of vegetation) the number of sporocarps of terricolous macrofungi is low. The increase in degradation of vegetation is followed by increasing abundance of terricolous macrofungi up to a moderate disturbance level. In fungal communities living on soil of different vegetation types, the proportion of higher-productive species increases. However, further increase of disturbance causes a decrease in the number of sporocarps.
According to the intermediate disturbance hypothesis (Grime 1973; Connell 1978; Hobbs and Huenneke 1992), a moderate disturbance leads to highest diversity; diversity is decreased by too low or high a level of disturbance. This hypothesis has been proven for vascular plants (Kim et al. 2002; Rejmánek et al. 2004) and birds (Lepczyk et al. 2008). Sobek and Zak (2003), examining the functional diversity of soil fungal communities in a Georgia forest disturbed by human activity, have shown that a medium amount of disturbance (a greater complexity of carbon substrate of sites) results in the highest soil fungal functional diversity. Surprisingly, although we did find such a relationship between disturbance and abundance, we did not find species richness of macrofungi. Obviously fructification of macrofungi speeds up in case of increased disturbance, in response to this pressure. However, beyond a certain disturbance level, the fructification is blocked then ceases. This pattern appears only in terricolous macrofungi, because the occurrence of lignicolous macrofungi depends strongly on the presence, quantity and quality of dead wood (Kost and Haas 1989; Høiland and Bendiksen 1996; Siller 2004), which overwhelms the other factors (e.g. disturbance).
In conclusion, the vascular plant diversity cannot be used for proxy of macrofungi diversity in Pannonian forests and forest plantations. This result questions the reliability of decisions on nature conservation actions (including selection of protected areas) based on only a few groups of easy-to-observe organisms and underlines the necessity of studying as wide range of taxonomic groups as possible.
References
Aczél J, Daróczy Z (1975) On measures of information and their characterization. Academic, New York
Arnolds E (1988) The changing macromycete flora in the Netherlands. Trans Br Mycol Soc 90:391–406
Arnolds E (1991) Decline of ectomycorrhizal fungi in Europe. Agric Ecosyst Environ 35:209–244
Arnolds E (1992) Macrofungal communities outside forests. In: Winterhoff W (ed) Fungi in vegetation science. Kluwer, Dordrecht, pp 113–149
Benedek L, Pál-Fám F, Nagy J (2005) Comparison of macrofungi communities and examination of macrofungi–plant interactions in forest stands in North Hungary. Int J Horticult Sci 11(2):101–103
Borhidi A (1995) Social behaviour types, the naturalness and relative ecological indicator values of the higher plants in the Hungarian Flora. Acta Bot Hung 39:97–181
Borhidi A (2003) Plant associations of Hungary (in Hungarian). Academic, Budapest
Brown N, Bhagwat S, Watkinson S (2006) Macrofungal diversity in fragmented and disturbed forests of the Western Ghats of India. J Appl Ecol 43:11–17
Bujakiewicz A (1992) Macrofungi on soil in deciduous forests. In: Winterhoff W (ed) Fungi in vegetation science. Kluwer, Dordrecht, pp 49–78
Bullock JM (2006) Plants. In: Sutherland WJ (ed) Ecological census techniques: a handbook, 2nd edn. Cambridge University Press, Oxford, pp 186–190
CABI (2008) The index Fungorum. http://www.indexfungorum.org
Chiarucci A, D’Auria F, De Dominicis V, Laganá A, Perini C, Salerni E (2005) Using vascular plants as a surrogate taxon to maximize fungal species richness in reserve design. Conserv Biol 19:1644–1652
Connell JH (1978) Diversity in tropical rain forests and coral reefs. Science 199:1302–1310
Crawley MJ (2007) The R book. Wiley, New York
Dorgai L (1986) Cserehát (in Hungarian). BAZ Megyei Mezőgazdasági Szövetkezetek Szövetsége, MTA Miskolci Akadémiai Bizottság, Budapest
Findlay CS, Houlahan J (1997) Anthropogenic correlates of species richness in Southeastern Ontario wetlands. Conserv Biol 11:1000–1009
Flather CH, Wilson KR, Dean DJ, McComb WC (1997) Identifying gaps in conservation networks: of indicators and uncertainty in geographic-based analyses. Ecol Appl 7:531–542
Fox J (2009) Car: companion to applied regression. R package version 1.2-16. http://CRAN.R-project.org/package=car
Gabel AC, Gabel ML (2007) Comparison of diversity of macrofungi and vascular plants at seven sites in the Black Hills of South Dakota. Am Midl Nat 157:258–296
Gaston KJ (1994) Rarity. Chapman and Hall, London
Gómez-Hernández M, Williams-Linera G (2011) Diversity of macromycetes determined by tree species, vegetation structure, and microenvironment in tropical cloud forests in Veracruz, Mexico. Botany 89:203–216
Gotelli NJ, Colwell RK (2001) Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecol Lett 4:379–391
Grime JP (1973) Competitive exclusion in herbaceous vegetation. Nature 242:247–344
Grime JP (1979) Plant strategies and vegetation processes. Wiley, Chichester
Hansen L, Knudsen H (1997) Nordic macromycetes, vol 3. Nordsvamp, Copenhagen
Hill M (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54:427–432
Hobbs RJ, Huenneke LF (1992) Disturbance diversity and invasion: implications for conservation. Conserv Biol 6:324–337
Høiland K, Bendiksen E (1996) Biodiversity of wood-inhabiting fungi in a boreal coniferous forest in Sør-Trøndelag County, central Norway. Nord J Bot 16:643–659
Hurlbert SH (1971) The non-concept of species diversity: a critique and alternative parameters. Ecology 52:577–586
Johnson CN (1998) Species extinction and the relationship between distribution and abundance. Nature 394:272–274
Jost L (2006) Entropy and diversity. Oikos 113:363–375
Keylock C (2005) Simpson diversity and Shannon–Wiener index as special cases of a generalized entropy. Oikos 109:203–207
Kim YM, Zerbe S, Kowarik I (2002) Human impact on flora and habitats in Korean rural settlements. Preslia 74:409–419
Kost G (1992) Macrofungi on soil in coniferous forest. In: Winterhoff W (ed) Fungi in vegetation science. Kluwer, Dordrecht, pp 79–111
Kost G, Haas H (1989) Die Pilzflora von Bannwäldern in Baden–Württenberg Waldschutzgebiete im Rahmen der Mitteilungen der Forstlichen Versuchs- und Forschunganstalt. Band 4:9–182
Krieglsteiner GJ (1991, 1993) Verbreitungsatlas der Großpilze Deutschlands, Bander 1-2. Ulmer, Stuttgart
Legendre P, Legendre L (1998) Numerical ecology, 2nd English edn. Elsevier, Amsterdam
Lenzen M, Lane A, Cooper AW, Williams M (2009) Affects of land use on threatened species. Conserv Biol 23:294–306
Lepczyk CA, Flather CH, Radeloff VC, Pidgeon AM, Hammer RB, Liu J (2008) Human impacts on regional avian diversity and abundance. Conserv Biol 22:405–416
Magurran A (2004) Measuring biological diversity. Blackwell, New York
McMullan-Fisher SJM (2008) Surrogates for cryptogam conservation-associations between mosses, macrofungi, vascular plants and environmental variables. PhD thesis, University of Tasmania
McMullan-Fisher SJM, Kirkpatrick JB, May TW, Pharo EJ (2010) Surrogates for macrofungi and mosses in reservation planning. Conserv Biol 24:730–736
Miller RM, Lodge DJ (1997) Fungal response to disturbance: agriculture and forestry. In: Wicklow DT, Söderström BE (eds) The mycota IV environmental and microbial relationships. Springer, Berlin, pp 65–84
Morschhauser T (1995) Applications of isodegradation curves in nature conservation. Acta Bot Hung 39(3–4):393–405
Nantel P, Neumann P (1992) Ecology of ectomycorrhizal-basidiomycete communities on a local vegetation gradient. Ecology 73(1):99–117
Ohenoja E (1988) Effect of forest management procedures on fungal fruit body production in Finland. Acta Bot Fenn 136:81–84
Oksanen J, Kindt R, Legendre P, O’Hara B, Simpson GL, Solymos P, Stevens MHH, Wagner H (2009) Vegan: community ecology package. R package version 1. 15-4. http://CRAN.R-project.org/package=vegan
Oleksyn J, Reich PB (1994) Pollution, habitat destruction, and biodiversity in Poland. Conserv Biol 8:943–960
Packham JM, May TW, Brown MJ, Wardlaw TJ, Mills AK (2002) Macrofungal diversity and community ecology in mature and regrowth wet eucalypt forest in Tasmania: a multivariate study. Austral Ecol 27:149–161
Pál-Fám F, Siller I, Fodor L (2007) Mycological monitoring in the Hungarian biodiversity monitoring system. Acta Mycol 42(1):35–58
Patil GP, Taillie C (1979) An overview of diversity. In: Grassle JF, Patil GP, Smith W, Taille C (eds) Ecological diversity in theory and practice. Maryland, Fairland, pp 3–27
Pharo EJ, Beattie AJ, Pressey RL (2000) Effectiveness of using vascular plants to select reserves for bryophytes and lichens. Biol Conserv 96:371–378
Pielou EC (1977) Mathematical ecology. Wiley, New York
Primack RB (1998) Essentials of conservation biology, 2nd edn. Sinauer, Sunderland
R Development Core Team (2009) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna. ISBN 3-900051-07-0. http://www.R-project.org
Rejmánek M, Rejmánková E, Holzner W (2004) Species diversity of plant communities on calcareous screes: the role of intermediate disturbance. Preslia 76:207–222
Rényi A (1961) On measures of entropy and information. In: Neymann J (ed) Proceedings of the 4th Berkeley symposium on mathematical statistics and probability, vol. 1. University of California Press, CA, pp 547–561
Rudolf K, Pál-Fám F, Morschhauser T (2008) The macrofungi of Cserehát (in Hungarian). Clusiana 47(1):45–74
Ryti RT (1992) Effect of the focal taxon on the selection of nature reserves. Ecol Appl 2:404–410
Saetersdal M, Gjerde I, Blom HH, Ihlen PG, Myrseth EW, Pommeresche R et al (2003) Vascular plants as a surrogate species group in complementary site selection for bryophytes, macrolichens, spiders, carabids, staphylinids, snails, and wood living polypore fungi in a northern forest. Biol Conserv 115:21–31
Santi E, Maccherini S, Rocchini D, Bonini I, Brunialti G, Favilli L, Perini C et al (2010) Simple to sample: vascular plants as surrogate group in a nature reserve. J Nat Conserv 18:2–11
Schmit JP, Mueller GM, Leacock PR, Mata JL, Wu Q, Huang Y (2005) Assessment of tree species richness as a surrogate for macrofungal species richness. Biol Conserv 121:99–110
Siller I (2004) Macrofungi of montane beech forest reserves. Dissertation, Szent István University
Simon T (2000) Identification handbook of the Hungarian vascular plants (in Hungarian). Nemzeti Tankönyvkiadó RT, Budapest
Sobek EA, Zak JC (2003) The Soil FungiLog procedure: method and analytical approaches toward understanding fungal functional diversity. Mycologia 95(4):590–602
Stirling G, Wilsey B (2001) Empirical relationships between species richness, evenness, and proportional diversity. Am Nat 158:286–299
Straatsma G, Krisai-Greilhuber I (2003) Assemblage structure, species richness, abundance, and distribution of fungal fruit bodies in a seven year plot-based survey near Vienna. Mycol Resch 107(5):632–640
Tofts RJ, Orton PD (1998) The species accumulation curve for Agarics and Boleti from a Caledonian Pinewood. Mycologist 12:98–102
Tóthmérész B (1995) Comparison of different methods for diversity ordering. J Veg Sci 6:283–290
Villeneuve N, Grandtner MM, Fortin JA (1989) Frequency and diversity of ectomycorrhizal and saprophytic macrofungi in the Laurentide Mountains of Quebec. Can J Bot 67:2616–2629
Virolainen KM, Ahlroth P, Hyvarinen E, Korkeamaki E, Mattila J, Paiivinen J et al (2000) Hot spots, indicator taxa, complementarity and optimal networks of taiga. Proc R Soc London B 267:1143–1147
Watling R (1995) Assessment of fungal diversity: macromycetes, the problems. Can J Bot 73:15–24
Zak JC (1992) Response of soil fungal communities to disturbance. In: Carroll G, Wicklow DT (eds) The fungal community: its organization and role in the ecosystem. Dekker, New York, pp 403–425
Acknowledgments
We would like to thank the Department of Plant Taxonomy and Geobotany of University of Pécs for supporting the survey.
Author information
Authors and Affiliations
Corresponding author
Appendix
About this article
Cite this article
Rudolf, K., Morschhauser, T., Pál-Fám, F. et al. Exploring the relationship between macrofungi diversity, abundance, and vascular plant diversity in semi-natural and managed forests in north-east Hungary. Ecol Res 28, 543–552 (2013). https://doi.org/10.1007/s11284-013-1044-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-013-1044-y