Abstract
The current study was undertaken to determine the ability of different carrier materials for sustaining the viability of microbial consortium during storage. Different bioformulations consisting of carrier material and microbial consortium were prepared and examined for viability and stability for one year stored at 4 °C and ambient temperature. Total 8 bio-formulations were prepared consisting five economically viable carriers (gluten, talc, charcoal, bentonite, broth medium) and a microbial consortium. In present study, maximum enhanced shelf-life of consortium based on colony forming unit count were recorded for talc + gluten based (B4) bioformulation (9.03 log10 cfu/g) over other bio-formulations stored for 360 days. Furthermore, the pot experiments was conducted to evaluate the efficacy of B4 formulation on growth of spinach in comparison with recommended dose of chemical fertilizer, uninoculated and no amendment control. The results depicted that B4 formulation increased biomass (176–666%), leaf area (33–123%), chlorophyll content (131–789%) and protein content (68.4–94.4%) of spinach over controls. Further B4 application significantly increased the nutrients like available nitrogen (131–475%), phosphorus (75–178%) and potassium (31–191%) of pot soil along with noteworthy improvement in root colonization as evident from scanning electron microscope analysis in comparison to controls at 60 days after sowing. Therefore, exploiting B4 formulation can serve as the environmentally sound approach to enhance the productivity, biomass and nutritional value of spinach. Thus, Plant growth promoting microbes-based formulation can be the novel paradigm to improve the soil health and eventually the crop productivity in economical and sustainable manner.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
To meet the food needs of growing population there is overuse of chemical fertilizer that hardly support the plant growth by leaching into surface water. The most possible alternatives to chemical fertilizers are the use of plant growth promoting microorganisms (PGPM) that can promote nutrient uptake by plants and protect them from adverse environmental conditions (Szopa et al. 2022). Inoculation of microbes for crop protection, growth and productivity is not a new concept (Bashan et al. 2014), but inoculation of monocultures under field conditions are not showing promising results due to the poor adaptability of microbial inoculants to changing environmental conditions (Mishra et al. 2021). The consortial formulations have multiple benefits to plant growth as it increases the population size and function of the inocula with many compatible microbes (Amalraj et al. 2015). Some other studies have showed that adaptability of the microbial inoculation could improve in the consortia based microbial communities (Vassilev et al. 2015; Khan et al. 2017). Microbial consortia proved more competent with a broad spectrum of action without genetic engineering towards crop productivity and plant health (Zahir et al. 2018; Ma 2019; Mishra and Sing 2022). Microbial consortia can perform better in different soils, as they can adapt in diverse range of climatic conditions with multiple inputs of root colonization, disease resistance, metabolite production, nutrient solubilization and antibiotic productions etc. (Elkoca et al. 2010; Upadhyay et al. 2012). The low microbial viability of the bioformulations during storage, marketing and application are the major concerns of its adoptability at large scale in the biofertilizer market. Due to ever changing soil environment, the population of PGPM starts declining after its application in the rhizospheric niche (Roy et al. 2010; Szopa et al. 2022) as their density in the rhizosphere depends on various factors like soil moisture, soil temperature and carriers etc. (Shilpa and Brahmaprakash 2016).
Carrier materials supports the growth and delivery of microbes to the rhizosphere and have potential to protect its competitive strength with other endemic microbes, soil pH and availability of organic carbon in soil (Brahmaprakash and Sahu 2012). Carrier materials may be organic (e.g., compost, peat, biogas slurry, crushed corn cob) or inorganic (e.g., zeolite, talc, perlite) in nature (Gunjal et al. 2012). For the preparation of good biofertilizer, carrier suitability is mainly studied to check the ability of carriers for offering a desirable micro-environment that can enhance the shelf life and effectivity of microbial strains introduced into the soil. The performance of carrier material varies according to strains, so there is no universal carrier identified for microbe-based formulations. Abd El Fattah et al. (2013) revealed that carrier-based microbial formulations are highly efficient due to their ease of handling and long-term preservation. Few carrier materials such as plant by-products, coal, talc, agricultural waste, organic waste and bentonite have been previously used to assess microbial shelf life but could not get proper recognition for its market value (Brockwell and Bottomley 1995; Stephens and Rask, 2000). Likewise, rhizospheric application of liquid microbial inoculum fails to increase the productivity and growth of crops due to certain factors. Short shelf life and contaminations are major bottlenecks in the commercialization of microbial bioformulations. In order to commercialize the technology, inoculums viability in an appropriate formulation for longer duration is very important. Satisfactory cfu of microbes for a carrier material is 107 cfug−1 of the carrier as recommended by Sethi and Adhikary (2012). Therefore, the selection of an appropriate carrier material for biofertilizer market is one amongst the important strategies towards sustainable agriculture. Thus, the present study was attempted to exploit the locally available carrier materials and potent microbial consortia that not only assist in improving the soil fertility and nutritional value of plants but also the viability and stability of bio-formulations for longer duration.
Methodology
Collection of microbial strains and compatibility test
In the present study seven pre-isolated and good plant growth promoting characterized microbial strains (Bacillus filamentosus RS3B, B. pseudomycoides RS6B, B. paramycoides RPB3, Alcaligenes faecalis RS10B, Aspergillus luchuensis RS6F, A. tamarii RS8F, and Trichoderma lixxi TvR1) (Sachdev and Singh 2018; Maddhesiya et al. 2021) were obtained from departmental laboratory (research group of Prof. Rana Pratap Singh). The compatibility of microbial strains with each other were tested based on the formation of inhibition zone or overlapping growth between the paired cultures. The bacterial and fungal compatibility were tested on nutrient agar (NA) and potato dextrose agar (PDA) media by spot inoculation and disc placement (James and Mathew 2017) respectively while a modified agar media (Hi-Media) was used to assess the compatibility between bacterial- fungal strains following the methods described by Mishra et al. (2021). Based on PGP traits (ammonia production, indole acetic acid (IAA) production, phosphate solubilization, nitrogen solubilization, potassium solubilization, hydrogen cyanide (HCN) production and siderophore production), and compatibility test, a consortium of seven microbial strains was used for the preparation of carrier-based formulation.
Selection and Physico-chemical characterization of carrier material
Four different carrier materials like gluten, talc charcoal, bentonite and their different combination were used [Gluten (C1), Talc (C2), charcoal (C3), Talc + gluten (C4), charcoal + gluten (C5), talc + charcoal + gluten (C6), bentonite (C7) and broth medium (C8)] to support the growth and nutrition of microbial inoculate during storage and application. All the selected carrier materials were purchased from G-1082 basement Sushant Lok-2 sector 57 Gurugram Haryana, 122011 IN and HiMedia Lab Pvt. Ltd, Mumbai, India. The pH and water holding capacity (WHC) of each carrier material were determined by following the methods of Maheshwari et al. (2015). Available nitrogen (AN), available phosphorus (AP), available potassium (AK) and organic matter (OM) percentage were estimated following the method described by Sohaib et al. (2020).
Preparation of carrier-based granular formulations
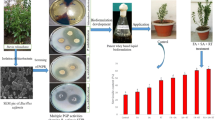
Initially, all the carrier materials and their combination were processed (dried, ground, sieved and sterilized) by following method of Tripathi et al. (2015). The granular bioformulations were prepared with the help of processed carrier materials and selected PGPM consortium following protocol given by Abd El Fattah et al. (2013). Carrier materials like talc + gluten (C4) and charcoal + gluten (C5) were mixed in the ratio of 3:1, while talc + charcoal + gluten (C6) were mixed in 1:1:1 ratio under asceptic conditions. Freshly cultured microbial consortium (1 × 109 CFU/ml) were used for the development of carrier based granular bioformulation (2:1 ratio; 100 g carrier and 50 ml inoculum) by following the methods of Kumar et al. (2014). Subsequently, the mixtures were kneaded between the fingers, granulated (using Dolphins granular maker) and were spread in trays covered with thin plastic films for a period of 48–72 h for curing, at 25 ± 1 °C. After curing, the formulations were sealed in low density, flexible, sterilized polythene bags leaving two-third vacant space for proper aeration (Maheshwari et al. 2015). Prepared bioformulations were stored at two different temperatures (4 °C and ambient temperature (AT) for 1 year to evaluate microbial viability in carrier materials (Tamreihao et al 2016).
Shelf-life assessment of microbial consortium
The microbial population in all the bioformulations was estimated by the standard plate count method. 1 g(g) samples from each bio-formulations at different time intervals were suspended in 9 mL of sterile distilled water and serially diluted upto 10–9. The number of colonies forming units (cfu) of microbial strains were recorded by following the method of Shilpa and Brahmaprakash 2016.
Inoculation of plants with selected bioformulations
Dose optimization
Based on viability of microbes in formulations, the formulation containing maximum number of viable cells was selected and evaluated for dose optimization with spinach in plastic pots (10 × 4x4 cm). Each pot was filled with 1000 g of non-autoclave soil. Different doses of bioformulation i.e., 5 g, 10 g, 15 g and 20 g/pot was added to the soil in the pot while one pot was kept as non-treated soil respectively. All dosage were layered following the ‘‘sandwich model’’ (Varma and Scheupp 1994). Spinach seeds were surface sterilized by following methos of Bakhsh et al. (2016). Sterilized seeds were germinated on water agar plates (1% agar in distilled water) and germinated seedlings were transferred in to the pots (5 seedlings/pot). The pots were irrigated with tap water on alternate days.
Growth parameters viz., shoot and root length (cm), shoot and root fresh and dry biomass(g) were recorded at 30 days after transplantation (See Section"Assessment of bioformulations"). Based on highest plant growth and biomass of spinach over control, 5 g/pot of bioformulation was selected for further application.
Assessment of bioformulations
Experimental design
Experiment was conducted in triplicates in earthen pots, during the months of October -December for two consecutive years (2020 and 2021). and pots were arranged randomly in three blocks under open greenhouse conditions (Inostroza et al. 2016) at Environmental Science Research Station, Babasaheb Bhimrao Ambedkar University, Lucknow, India (26° 72 E, 80° 85 N). Sandy loam soil (pH 7.8, EC 5.4 ds/m, organic matter (OM) 0.12%, available nitrogen (AN) 16.67 mg/kg, available potassium (AK) 70.61 mg/kg, available phosphorus (AP) 4.7 mg/kg, alkaline phosphatase (ALkP)12.10 µg/g, soil microbial biomass carbon (SMBC) 108 µg/g) was collected from the non- agricultural land from the premises of BBA University, Lucknow, Uttar Pradesh. The soil was mixed thoroughly before the experiment and 7 kg of non-autoclaved soil was filled in earthen pots (45 × 15x15cm). The maximum to minimum atmospheric temperature range during the period was 25 °C and 18 °C. There was total six different treatments included in the study i.e., freshly prepared granulated bioformulation (T1), one-year stored bioformulation (T2), Freshly prepared inoculated broth medium (T3), non-inoculated carrier material (T4), recommended dose of chemical fertilizer (T5) and non- treated soil only(T6). Approximately, 34 g (0.5% based on optimization) of one year stored and freshly prepared granulated bioformulations were added to each pot. The bioformulation was added once into the soil (at the time of sowing). The surface-sterilized seeds using 3% hydrogen peroxide solution (Bakhsh et al. 2016) were sowed in pots (10 seeds per pot) and 6 saplings per pots were maintained by thinning after germination for homogeneity. The pots were irrigated on alternate days, and weeding removal as per requirement was done.
The crop was harvested after 60 days after sowing (DAS) and their root and shoot length were measured by using meter scale. Freshly harvested plants were washed by running tap water to remove soil and dust particles then roots, stems and leaves were separated. Uniform leaf area was collected from each treatment by Image J software (Wayne Rasband NIH, http://imagej.nih.gov/ij/index.html) Vitale et al. (2020) and their fresh weight (g) and dry weight was taken by following the method described by Seymen (2021). Estimation of biochemical parameters i.e., chlorophyll, carotenoids and protein content were done by following the methods described by Mastan et al. (2019).
The study of microbial colonization on roots was carried out with a scanning electron microscope (SEM) (JEOL, JSM 6490 LV). For colonization assays, roots were gently washed three times with sterile distilled water to remove unbound microbes from the root surface before microscopic observation. (Gomez et al. 2018). Roots were cut into transverse section to observe colonies on root surface. Small sections of root samples were glued to a double-sided conductive carbon tab stuck on a standard vacuum-clean stub.
Soil samples were collected from each earthen pot at the depth of 10 cm before the plantation and after harvesting of seedlings. Soil samples were homogenized manually and sieved (2 mm sieve) to remove concrete and other debris. Soil was oven-dried at 65 °C temperature (for 24 h), for physico-chemical analysis while fresh and moist soil was used to analyze microbial and enzymatic activities. The physico-chemical properties including pH, electrical conductivity, available nitrogen (AN), phosphorus (AP) and potassium (AK), organic carbon, alkaline phosphatase (ALkP), soil microbial biomass carbon (SMBC) of the soil were estimated by following standard methods of Jackson (1973). Increased physio-chemical and enzymatic parameters of the post-harvest soil with respect to pre-sowing soil (control) were used to determine the fertility of the cultivated soil.
Statistical analysis
Statistical analysis of the data was done using MS Excel and IBM SPSS statistics 20. Data were analysed by one-way analysis of variance, and mean values were compared using Duncan’s multiple range test (P < 0.05). Significance of the data was calculated by Duncan multivariate test (DMRT) in which data indicated by the same letters in a column is not significantly different.
Results
Based on PGP traits and compatibility, seven PGPM as a consortium were mixed in different carrier materials for the preparation of a promising biofertilizer to the existing soil condition as well as crop growth and production. The findings of the study are as follows.
Physico-chemical properties of carrier materials
The pH values of all the carrier materials were recorded in between 6.2 to 7.3 which is reported optimal and suitable for PGP microbial species. The results depicted that highest pH was observed in case of C3 (7.3) and lowest pH in case of C1 and C7 (6.2). The average EC of carrier materials ranged from ~ 2.34 ± 0.22 to 4.25 ± 0.34 dSm−1. The EC of carrier materials shows the amount of soluble salts present, and these salts can undoubtedly affect the activities and longevity of inoculants. Most of the carrier materials were rich in carbon, nitrogen, potassium and phosphorus. The results of the current study shows that Organic matter (OM) content in selected carriers ranged between 0.73 and 4.2% and maximum OM was reported in C4. While available nitrogen (AN) varied from 58.3 to 125 mg/kg and highest AN was shown by C4. Similarly available phosphorus (AP) ranged from 1.46 to 5.3 mg/kg with highest in C5, whereas, available potassium (AK) was ranged between 16.5 to 197 mg/kg with highest in C1 (Table 1). The water holding capacity (WHC) of the carrier materials varied from 27 to 76% and C4 shows the maximum WHC (76%).
Shelf life of consortium in carrier-based bioformulations at different temperatures
In the present study, viability of the consortium was maintained in granular formulation containing 8 economically viable carriers for 360 days. Among all, B4 proved to be the most suitable carrier material in supporting the viability of consortium (Figs. 1, 2). For B4 bioformulation, the population density of 9.09, 9.09, 9.08, 9.06, 9.05 and 9.03 log10 cfu/ g of bioformulation was observed to sustain at 60, 120, 180, 240, 300 and 360 days after inoculation (DAI) respectively at 4 °C. At ambient temperature it was 9.09, 9.10, 9.10, 9.05, 9.05 and 9.03 log10 cfu/g of bioformulation at 60, 120, 180, 240, 300 and 360 DAI respectively followed by B1 bioformulation with maximum population of 8.68 log10 cfu at 60 DAI and minimum population of 5.55 log10 cfu at 360 DAI. The cfu count of consortium in other carrier-based granular bio-formulations was constant for 2 months. However, it was found to decline significantly after 240 days of storage at both temperatures (Figs. 1, 2).
Enumeration of the total viable count of microbial consortium in carrier-based granular formulation stored at 4 °C, log10 cfu/g of granular bioformulation. B bioformulation; b bentonite, C charcoal, D days, G gluten, T talc, B1 G + MC, B2 T + MC, B3 C + MC, B4 T + G + MC, B5 C + G + MC, B6 C + T + G + MC, B7 b + MC, B8 broth medium + MC, CFU colony forming unit, MC microbial consortium of seven microbes (Bacillus filamentosus + B. pseudomycoides + Alcaligenes faecalis + B. paramycoides + Aspergillus tamarii + A. luchuensis + Trichoderma lixxi); Data are mean of three replicas, in duplicate determination (n = 6) ± standard error of means. Means, followed by the same letter in a column are not significantly different by Duncan’s multivariate test (DMRT)
Enumeration of the total viable count of microbial consortium in carrier-based granular formulation stored at ambient temperature log10 cfu/g of granular bioformulation. B bioformulation, b bentonite, C charcoal, D days, G gluten, T talc, B1 G + MC, B2 T + MC, B3 C + MC, B4 T + G + MC, B5 C + G + MC, B6 C + T + G + MC, B7 b + MC, B8 broth medium + MC, AT ambient temperature, CFU colony forming unit, MC microbial consortium of seven microbes (Bacillus filamentosus + B. pseudomycoides + Alcaligenes faecalis + B. paramycoides + Aspergillus tamarii + A. luchuensis + Trichoderma lixxi); Data are mean of three replicas, in duplicate determination (n = 6) ± standard error of means. Means, followed by the same letter in a column are not significantly different by Duncan’s multivariate test (DMRT)
Effect of selected bioformulation on growth, root colonization and biochemical aspects of spinach
Influence of treatments on vegetative growth and biochemical properties of spinach
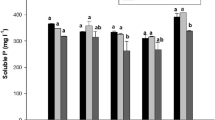
To evaluate the influence of selected bioformulation, 6 different (T1-T6) treatments were given to the plants (Fig. 3). To ascertain the effect of different treatments on yield, various plant growth parameters like shoot and root length, leaf area and biomass of spinach were recorded (Fig. 3). It was evident from the results that there was a clear enhancement in the vegetative growth of spinach upon the addition of carrier-based bioformulation as compared to non-treated plants (Fig. 4.). Plants treated with T1 and T2 bioformulations showed maximum enhancement in plant length and biomass as compared to other treatments. In present study, the T1 showed the maximum percentage increase of 113.07%, 128.05%, 222.1%, 184.6% and 120.1% for shoot length, root length, shoot biomass, root biomass and leaf area respectively at 60 DAS as compared to T6, and the minimum percentage increase of 10.3%, 6.8%, 25.6%, 37.0% and 24.7% for shoot length, root length, shoot biomass, root biomass and leaf area respectively as compared to T3 at 60 DAS. Similarly, T2 showed maximum increase for shoot length, shoot biomass, leaf area, root length and biomass by113.3%, 680.7%, 112.7%, 118.5% and 142.5% respectively at as compared to T6 at 60 DAS. The biochemical properties of spinach plants also showed drastic effect when treated with bioformulations (Fig. 3). The chlorophyll content of spinach leaves was enhanced by 714% and 603% after treatment with T1 and T2 over T6. Similarly, carotenoid content of leaves was also enhanced by1725% and 2825% with T1 and T2 bioformulations as compared to T6. The results further showed that protein content was increased by (62.9–91.3%) and (56.01–80.4%) in case of spinach leaves treated with T1 and T2 as compared to other treatments included in the study.
Effect of different treatments on vegetative growth and biochemical characters of Spinach (Spinacia oleracea L.). A Plant height, B plant biomass, C leaf area Index, D chlorophyll-a; E Chlorophyll b; F total chlorophyll, G carotenoids, H Protein. T1 freshly prepared inoculated carrier (t + G + MC) based bioformulation, T2 one year stored inoculated carrier-based bioformulation, T3 freshly prepared inoculated liquid bioformulation, T4 uninoculated carrier material, T5 chemical fertilizer, T6 untreated soil only, mg/g milligrams per grams, µmol/g micromole per grams, SL shoot length, RL root length, SFW shoot fresh weight, SDW shoot dry weight, RFW root fresh weight, RDW root dry weight. Data are mean of three replicas in two sets of experiments (n = 6) ± standard error of means. Means, followed by the same letter in a column are not significantly different by Duncan’s multivariate test (DMRT)
Plants of spinach grown with the application of bioformulations for 60 days. T1 freshly prepared inoculated carrier (t + G + MC) based bioformulation, T2 one year stored inoculated carrier-based bioformulation, T3 freshly prepared inoculated liquid bioformulation, T4 uninoculated carrier material, T5 chemical fertilizer, T6 untreated soil only
Root colonization
SEM analysis was conducted to assess the microbial colonization on root surface with and without effect of bioformulation. The results showed the clear difference between colonization of microbes on root surface of different treatments (Fig. 5). Spinach roots treated with carrier based bioformulations (T1 and T2) were fully colonized by microbial population, while roots treated with broth based bioformulation were less colonized, and there was very least or no colonization on the surface of roots treated with T4, T5 and T6 (Fig. 5).
SEM images of spinach roots showing microbial colonization on root surface. T1 freshly prepared inoculated carrier (t + G + MC) based bioformulation, T2 one year stored inoculated carrier-based bioformulation, T3 freshly prepared inoculated liquid bioformulation, T4 uninoculated carrier material, T5 chemical fertilizer, T6 untreated soil only
Effect of treatments on physico-chemical and microbial properties of soil
Effect of carrier-based bioformulation on microbial biomass and soil physico-chemical properties are presented in Table 2. All the studied parameters showed a remarkable difference between carrier-based bioformulation and non-inoculation. However, soil treated with T2 bioformulation showed maximum percentage increase of 750%, 178%, 191%, 475%, 166.6% and 278% for OM, AP, AK, AN, ALkP and SMBC respectively at harvesting as compared to control (pre-sowing control soil). Whereas minimum percentage increase of 16.6% of OM, 49.04% of AP, 131.1% AN and 33.1% of ALkP was observed for T4. The results further showed that microbial population in the rhizospheric soil was increased by the addition of different bioformulation (Fig. 6). Maximum increase in log10 cfu/g of rhizospheric soil for bacteria was observed for T1 (23.9%) followed by T2 (23.6%) as compared to pre-sowing soil. Furthermore, increased cfu/g of soil for fungi was 37% in case of T1 and 36.2% in T2 as compared to pre-sowing data (Fig. 6). While minimum increased microbial population after crop harvesting was shown by T6 (15.5%) for bacteria and T4 (25.2%) for Fungi respectively.
Microbial species richness in pot soil cultivated with Spinach. T1 freshly prepared inoculated carrier (t + G + MC) based bioformulation; T2 one year stored inoculated carrier-based bioformulation, T3 freshly prepared inoculated liquid bioformulation, T4 uninoculated carrier material, T5 chemical fertilizer, T6 untreated soil only. Data are mean of three replicas, in duplicate determination (n = 6) ± standard error of means; Means, followed by the same letter in a column are not significantly different by Duncan’s multivariate test (DMRT)
Discussion
Physico-chemical properties of carrier materials
PGP microbial cultures need an appropriate medium for their transport to the target site, and carrier materials may act as that medium. Viability of microbial inoculants in different carrier material might be different due to their different physicochemical properties (Ma 2019, Shoiab et al. 2020). Material having good WHC, non-toxic to inoculants, pH buffering capacity, cation exchange capacity, biodegradable and provide nutrients to the microbes are considered as a good inoculant’s carrier (Sohaib et al. 2020). In present study talc + gluten (C4) carrier was having highest WHC, organic carbon and available nitrogen content as compared to others selected carriers. Hence, it could be one amongst the selected carrier to support beneficial microbial cultures to target their sites as bio-inoculation. The results of this study are in agreement with Brahmaprakash and Sahu (2012) and Shahzad et al. (2017). Singh et al. (2020), also reported that, talc possesses high water holding capacity and near neutral pH that could help in sustaining microbial population for longer duration under different storage condition.
Shelf life of consortia in carrier-based bioformulation at different temperatures
The prime research finding of the current study was the use of different carrier material inoculated with consortium (of bacterial and fungal strains) instead of single inoculants to sustain the viability of microbes and better plant growth promotion. Several studies have been conducted on survivability of microbes in consortium as well as their single application (Rastegari et al. 2020; Kour et al. 2020). In our study, we used different carrier material for sustaining viability of microbial consortium at different temperature at storage. The results showed that, B4 proved to be the most suitable carrier material in supporting the viability of consortia at two different temperature (Figs. 1, 2). The reason might be the key characteristics of carrier material such as high WHC and near neutral pH which allow PGP inoculants to sustain longer shelf life, which is in lineation with the study of Aloo et al. (2022).
Tamreihao et al. (2016), also reported that talc-based bioformulation show better shelf life than corn starch formulation up to 6 months of storage at 4 °C and ambient temperature and further moisture-free environments and low-temperature conditions were reported to minimize the metabolic rate of microbial propagules. For boosting the property of talc, it was mixed with gluten for sustaining better shelf life of microbes in this study, as gluten form matrix, which immobilized the microbial cells (Sivakumar et al. 2014). Additionally, gluten is readily available, bio-degradable and environment-friendly (Cho and Lee 1999). PGP microbial cells when encapsulated with gluten remain viable for a longer time as reported by Fravel et al. (1985) and Park and Chang (2000) which supports our study. The main aim of microbial encapsulation in this study was to increase colonization on the host plant, protection from the harsh climatic conditions and to lower the competition by natural inhabitants in the soil. Enriched carrier material was found to retain the shelf life of microbial inoculants and increases the effectivity of bioformulation. Solanki and Shah (2016) also found better shelf life of microbial inoculants in carrier material enriched with nutrient rich medium like glycerol, calcium alginate, molasses, trehalose, maltose and carboxymethyl cellulose. In the present study, the results exhibited that liquid inoculants were ineffective than solid carrier-based bioformulation. Khandare et al. (2019) also reported that the low survival of microbial inoculants in broth based bioformulation might be the reason for ineffectiveness of liquid bioformulation over carrier based bioformulation. Thus, the present study suggests that the manipulation of carrier material talc with gluten (B4) would be a better option for maintaining stability and enhancing the shelf-life of the microbial consortium.
Influence of treatments on vegetative growth, and biochemical characteristics of spinach in pot condition
Effect of selected bioformulation on spinach growth was summarised in Fig. 3. Our results showed that maximum growth promotion was in plants treated with carrier based bioformulation rather than other treatments. Plant growth enhanced by application of bioformulation, might be facilitated due to better nutrient mobilization by microbes in the bioformulation (Awasthi et al. 2019; Maddhesiya et al. 2021; Mishra et al 2021). Furthermore, phytohormones like IAA production assist in cell division and cell differentiation, which in turn helps in accelerating overall plant growth and development (Gupta and Vandana 2019). In the present study, IAA production and phosphate solubilization by the microbial strains in the consortium might be the reason for increased plant growth and production. The study is in lineation with our previous study, Parveen et al. (2022a, b), which reported enhanced growth of tomato plant, root elongation and development, facilitated by IAA producing similar group of PGPM consortium, which thereby improved water and nutrient acquisition from the soil. The results are also supported by the findings of Petrillo et al. (2022) who reported increased seed germination, shoot and root length and overall growth of Spinach upon application of bioformulation containing consortia of B. amylolique faciens RHF6, B. amylolique faciens LMG9814, and B. sp. AGS84.
The results also showed significant increase in biochemical characteristics like chlorophyll, carotenoids and protein content of treated plants (with carrier based bioformulation) as compared to uninoculated plants. The improvement in biochemical characteristics may be attributed to enhancement in nitrogen fixation, nutrient solubilization, mineralization, production of phytohormones, secondary metabolites and siderophore stimulated by microbial bioformulations in spinach. The results of this study are in accordance with Shilev et al. (2019), Sohaib et al. (2020), Asif et al. (2020) and Parveen et al. (2022b).
Roots colonization by microbial colonies
Roots colonization by microbial strains is an important step required for plant growth. Accordingly, and considering the importance of probiotic microbes that promote the growth of spinach, we have analysed a bioformulation for root colonization in spinach. The results showed that roots treated with bioformulation (T1 and T2) were fully colonized by microbial colonies. Higher root colonization in these treatments might be due to higher microbial population present in the rhizospheric soil (Fig. 6). These results confirm the strong positive effect of bioformulation on spinach plant growth, which may be due to higher root colonization over other treatments and suggesting that this bioformulation may have probiotic potential, at least on spinach. Results were supported with the findings of Gómez et al. (2018), who found rhizobium as good colonizer for spinach seedling. These results were also supported by our previous study having the similar microbial species showing colonization with tomato roots (Parveen et al. 2022a).
Effect of treatments on pot soil’s physico-chemical and microbial properties
To assess the effect of bioformulation on pot soil cultivated with spinach, soil parameters like pH, EC, OM, AN, AP, AK, SMBC were analyzed. Electrical conductivity was highly reduced by the application of bioformulation. The reduction in EC was possibly due to accelerated growth of the plant and the higher solubility of the salt in presence of these inoculated microbes (Babalola 2010; Schoebitz and Vidal 2016 and Kapadia et al. 2021). From the results, it was found that T1 and T2 has maximum increase of SMBC, OM, AN, AK and AP. This might be due ability of microbial strains for solubilization of phosphorus, nitrogen and potassium in the soil. In the present study, the increment in AN, AP and AK in the soil may increase the soil fertility status and plant growth after introduction of bioformulation to the soil. High nitrogen and phosphorus content in soils treated with carrier-based bioformulation can be directly related to the presence of viable nitrogen fixers and phosphate solubilizing microbes in the consortium. Maddhesiya et al. (2021) and Mishra and Singh (2022) also reported that microbial communities have a significant role in improving fertility of the soil and productivity of plants. Furthermore, application of bioformulation to the soil containing phosphate solubilizing microbial strains may help in the solubilization of insoluble phosphate by secretion of organic acids, siderophore and enzyme production that makes phosphate available to the plants. Nutrients like phosphorus play indispensable role as a primary nutrient in better root development, plant growth and yield (Kushwaha et al. 2001). Further, the results showed that there was a remarkable change in the organic matter content of the post-harvested soil. The bio-formulations application improved the organic matter of the soil. Increased OM might be accelerated by the action of microbial consortium, that help in the decomposition of root exudates which in turn increases the OM content of the soil. It has also been concluded in previous studies that microbial consortium increases the level of organic matter (Monica et al. 2011; Xu et al. 2019). Likewise, in this study, the significant variations were observed in microbial population (cfu) and microbial biomass carbon (MBC) in the soil. Our results showed increase in rhizospheric microbial population upon addition of bioformulation which agrees with Tripti et al. (2017).
Conclusion
It can be concluded from the results of the present study that multistrain microbial consortium inoculated in carrier material talc + gluten is an effective bioformulation for enhancing shelf life of microbial strains under storage and plant growth in pot condition. Furthermore, this bio-formulation also improved the soil health, plant growth, helps in root colonization, and improved the biochemical characteristics of the spinach. Overall, bioformulation consisting of talc + gluten + microbial consortium best suited to enhance the efficacy of microbial consortium to promote plant growth, soil fertility and microbial richness in soil, root colonization providing the best micro-environment for the survival of PGP microbial strains. Therefore, application of microbial consortia along with talc + gluten can be the competent bioformulation that not only improve the soil health but enhance the yield and nutrient status of the crop in economical and eco-friendly manner.
References
Abd El Fattah DA, Eweda WE, Zayed MS, Hassanein MK (2013) Effect of carrier materials, sterilization method, and storage temperature on survival and biological activities of Azotobacter chroococcum inoculant. Annual Agri Sci 58(2):111–118
Aloo BN, Mbega ER, Makumba BA, Tumuhairwe JB (2022) Effects of carrier materials and storage temperatures on the viability and stability of three biofertilizer inoculants obtained from potato (Solanum tuberosum L.) rhizosphere. Agriculture 12:140
Amalraj ELD, Mohanty D, Kumar GP, Desai S, Ahmed SKMH, Pradhan R, Khan SS (2015) Potential microbial consortium for plant growth promotion of sunflower (Helianthus annuus L.). Proc Natl Acad Sci 85(2):635–642
Asif M, Pervez A, Irshad U, Mehmood Q, Ahmad R (2020) Melatonin and plant growth-promoting rhizobacteria alleviate the cadmium and arsenic stresses and increase the growth of Spinacia oleracea L. Plant Soil Environ 66(5):234–241
Awasthi A (2019) Field-specific microbial consortium are feasible: a response to Kaminsky et al. Trends Biotechnol 37(6):569–572
Babalola OO (2010) Beneficial bacteria of agricultural importance. Biotech Lett 32(11):1559–1570
Bakhsh K, Akram W, Jahanzeb A, Khan M (2016) Estimating productivity of Bt cotton and its impact on pesticide use in Punjab. Pakistan Pak Econ Soc Rev 54(1):15–24
Bashan Y, de-Bashan LE, Prabhu S, Hernandez JP (2014) Advances in plant growth-promoting bacterial inoculant technology: formulations and practical perspectives (1998–2013). Plant Soil 378:1–33. https://doi.org/10.1007/s11104-013-1956-x
Brahmaprakash G, Sahu PK (2012) Biofertilizers for sustainability. J Ind Inst Sci 92:37–62
Brockwell J, Bottomley PJ (1995) Recent advances in inoculant technology and prospects for the future. Soil Biol Biochem 27:683–687
Cho CF, Lee WC (1999) Formulation of a biocontrol agent by entrapping biomass of Trichoderma viride in Gluten matrix. J Biosci Bioeng 87(6):822–824
Elkoca E, Turan M, Donmez MF (2010) Effects of single, dual and triple inoculations with Bacillus subtilis, Bacillus megaterium and Rhizobium leguminosarum by Phaseoli on nodulation, nutrient uptake, yield and yield parameters of common bean (Phaseolus vulgaris L. cv. ‘Elkoca-05’). J Plant Nutr 33:2104–2119
Fravel DR, Marois JJ, Lumsden RD, Connick WJJ (1985) Encapsulation of potential biocontrol agents in an alginate-clay matrix. Phytopathology 75:774–777
Gomez AJ, Felix JDF, Fraile PG, Mateos PF, Menendez E, Velazquez E, Rivas R (2018) Probiotic activities of Rhizobium laguerreae on growth and quality of spinach. Sci Rep 8:295. https://doi.org/10.1038/s41598-017-18632
Gunjal A, Kapadnis B, Pawar N (2012) Agroindustry by-products as a carrier resource for plant-growth-promoting rhizobacterium, Bacillus subtilis. J Mater Cycles Waste Manag 14:274–280
Gupta, Vandana (2019) Effect of PGPR isolates on plant growth promotion in relation to salinity stress. Bull Environ Pharmacol Life Sci 8(12):18–26.
Inostroza NG, Barra PJ, Wick LY, Mora ML, Jorquera MA (2016) Effect of rhizobacterial consortia from undisturbed arid- and agroecosystems on wheat growth under differing conditions. Lett Appl Microbiol 64:158–163
Jackson MI (1973) Soil and chemical analysis prentice hall of India private limited, New Delhi.
James D, Mathew SK (2017) Compatibility studies on different endophytic microbes of tomato antagonistic to bacterial wilt pathogen. IJABR 7(1):190–194
Kapadia C, Sayyed RZ, El Enshasy HA, Vaidya H, Sharma D, Patel N, et al (2021) Halotolerant microbial consortia for sustainable mitigation of salinity stress, growth promotion, and mineral uptake in tomato plants and soil nutrient enrichment. Sustainability 13(15):8369
Khan MY, Zahir AZ, Asghar HN, Waraich EA (2017) Preliminary investigations on selection of synergistic halotolerant plant growth promoting rhizobacteria for inducing salinity tolerance in wheat. Pak J Bot 49(4):1541–1551
Khandare RN, Chandra R, Pareek N, Raverkar KP (2019) Carrier-based and liquid bioinoculants of Azotobacter and PSB saved chemical fertilizers in wheat (Triticum aestivum L.) and enhanced soil biological properties in Molli sols. J Plant Nutr 1–15.
Kour D, Rana K, Kaur T, Yadav N, Yadav AN, Rastegari AA, Saxena AK (2020) Microbial biofilms: functional annotation and potential applications in agriculture and allied sectors. In: Yadav MK, Singh BP (eds) New and future developments in microbial biotechnology and bioengineering: microbial biofilms. Elsevier, Cambridge, pp 283–301
Kumar S, Thakur M, Rani A (2014) Trichoderma: mass production, formulation, quality control, delivery and its scope in commercialization in India for the management of plant diseases. Afr J Agric Res 9(53):3838–3852. https://doi.org/10.5897/AJAR2014.9061
Kushwaha CP, Tripathi SK, Singh KP (2001) Soil organic matter and water-stable aggregates under different tillage and residue conditions in a tropical dryland agroecosystem. Appl Soil Ecol 16:229–241
Ma Y (2019) Seed coating with beneficial microorganisms for precision agriculture. Biotechnol Adv 107423.
Maddhesiya PK, Singh K, Singh RP (2021) Effect of perennial aromatic grass species richness and microbial consortium on soil properties of marginal lands and biomass production. Land Degrad Dev 32(2):1008–1021
Maheshwari DK, Dubey RC, Agarwal M, Dheeman S, Aeron A, Bajpai VK (2015) Carrier based formulations of biocoenotic consortia of disease suppressive Pseudomonas aeruginosa KRP1 and Bacillus licheniformis KRB1. Ecol Eng 81:272–277
Mastan A, Rane D, Dastager SG, Babu CSV (2019) Development of low-cost plant probiotic formulations of functional endophytes for sustainable cultivation of Coleus forskohlii; Microbiol Res 227:126310. https://doi.org/10.1016/j.micres.2019.126310
Mishra R, Singh RP (2022) Effect of species diversity levels and microbial consortium on biomass production, net economic gain, and fertility of marginal land. Land Degrad Dev 1–12. https://doi.org/10.1002/ldr.4195
Mishra R, Dubey P and Singh RP (2021) Assessing the efficacy of climate resilient microbial inoculants for enhanced phytochemical production from Indian licorice (Abrus precatorius L.). Medicinal Plants 13(2):330–338.
Monica S, Karthik L, Mythili S, Sathiavelu A (2011) Formulation of effective microbial consortia and its application for sewage treatment. J Microbial Biochem Technol 3:051–055
Park JK, Chang HN (2000) Microencapsulation of microbial cells. Biotechnol Adv 18:303–319
Parveen N, Singh DV, Singh RP (2022a) Developing an effective microbial community as bioinoculant for enhanced productivity of tomato (Lycopersicon esculantum Mill.) with improved soil fertility. IJFANS 11(3):2419–2430.
Parveen N, Mishra R, Singh RP (2022b) Assessing the efficacy of microbial bioformulations in enhancing biomass and defence related compounds of spinach (Spinacia Oleracea L.). IJFANS 11(3):3482–3497.
Petrillo C, Vitale E, Ambrosino P, Arena C, Isticato R (2022) Plant growth promoting bacterial consortia as a strategy to alleviate drought stress in Spinacia oleracea. Microorganism 10:1798.
Rastegari AA, Yadav AN, Yadav N (2020) Trends of microbial biotechnology for sustainable agriculture and biomedicine systems: diversity and functional perspectives. Elsevier, Amsterdam
Roy BD, Deb B, Sharma GD (2010) Evaluation of carrier-based inoculants of Azotobacter Chroococcun strain SDSA-112/2 in improving growth and yield of summer rice. IR-36. Biofrent 1:36–40
Sachdev S, Singh RP (2018) Isolation, characterisation and screening of native Aguirre environ. Sustan 6(1):46–58
Schoebitz M, Vidal G (2016) Microbial consortium and pig slurry to improve chemical properties of degraded soil and nutrient plant uptake (Schoebitz and Vidal, 2016). J Soil Sci Plant Nutr 16(1):226–236
Sethi SK, Adhikary SP (2012) Cost effective pilot scale production of biofertilizer using Rhizobium and Azotobacter. Afr J Biotechnol 11:13490–13493
Seymen M (2021) How does the flooding stress occurring in different harvest times affect the morpho-physiological and biochemical characteristics of spinach? Sci Hortic 275:109713
Shahzad S, Khan MY, Zahir AZ, Asghar HN, Chaudhry UK (2017) Comparative effectiveness of different carriers to improve the efficacy of bacterial consortium for enhancing wheat production under salt affected field conditions. Pak J Bot 49(4):1523–1530
Shilev ST, Babrikova I, Babrikov T (2019) Consortium of plant growth-promoting bacteria improves spinach (Spinacea oleracea L.) growth under heavy metal stress conditions. J Chem Technol Biotechnol. https://doi.org/10.1002/jctb.6077
Shilpa ME and Brahmaprakash GP (2016) Amendment of carrier with organic material for enhancing shelf life of microbial consortium. J Pure Appl Microbiol 10(4):2835–2842. https://doi.org/10.22207/JPAM.10.4.44
Singh J, Singh AV, Upadhayay VK, Amir K (2020) Comparative evaluation of developed carrier based bioformulations bearing multifarious PGP properties and their effect on shelf life under different storage conditions. Environ Ecol 38(1):96–103
Sivakumar PK, Parthasarthi R, Lakshmipriya VP (2014) Encapsulation of plant growth-promoting inoculant in bacterial alginate beads enriched with humid acid. Int J Curr Microbiol Appl Sci 3:415–422
Sohaib M, Zahir AZ, Khan MY, Ans M, Asghar HNSY, Al-Barakah FNI (2020) Comparative evaluation of different carrier-based multi-strain bacterial formulations to mitigate the salt stress in wheat. Saudi J Biol Sci 27:777–787. https://doi.org/10.1016/j.sjbs.2019.12.034
Solanki HK, Shah DA (2016) Formulation optimization and evaluation of probiotic Lactobacillus sporogenes-loaded sodium alginate with carboxymethyl cellulose mucoadhesive beads using design expert software. J Food Proc 1–1:4
Szopa D, Mielczarek M, Skrzypczak D, Izydorczyk G, Katarzyna M, Katarzyna C, Krowiak AW (2022) Encapsulation efficiency and survival of plant growth-promoting microorganisms in an aginate- based natrix—a systematic review and protocol for a practical approach. Ind Crops Prod 181:114846
Tamreihao K, Ningthoujam DS, Nimaichand S, Singh ES, Reena P, Singh SH, Nongthomb U (2016) Biocontrol and plant growth promoting activities of a Streptomyces corchorusii strain UCR3-16 and preparation of powder formulation for application as biofertilizer agents for rice plant. Microbiol Res 192:260–270. https://doi.org/10.1016/j.micres.2016.08.005
Tripathi S, Chandra ADA, Varma A (2015) Development of carrier-based formulation of root endophyte Piriformospora indica and its evaluation on Phaseolus vulgaris L. World J Microbiol Biotechnol 31:337–344
Tripti KA, Usmani Z, Kumar V, Anshumali (2017) Biochar and flyash inoculated with plant growth promoting rhizobacteria act as biofertilizer for luxuriant growth and yield of tomato plant. J Environ Manag 190:20–27. https://doi.org/10.1016/j.jenvman.2016.11.060
Upadhyay SK, Singh JS, Saxena AK, Singh DP (2012) Impact of PGPR inoculation on growth and antioxidant status of wheat under saline conditions. Plant Biol 14:605–611
Varma A, Schuepp H (1994) Positive influence of arbuscular mycorrhizal fungus on in vitro raised hortensia plantlets. Angew Bot 68:108–113
Vassilev N, Vassileva M, Lopez A, Martos V, Reyes A, Maksimovic I, Eichler LB, Malusa E (2015) Unexploited potential of some biotechnological techniques for biofertilizer production and formulation. Appl Microbiol Biotechnol 99:4983–4996
Vitale L, Vitale E, Guercia G, Turano M, Arena C (2020) Effects of different light quality and biofertilizers on structural and physiological traits of spinach plants. Photosynthetica 58(4):932–943. https://doi.org/10.32615/ps.2020.039
Xu J, Jiang Z, Li M, Li Q (2019) A compost-derived thermophilic microbial consortium enhances the humification process and alters the microbial diversity during composting. J Environ Manag 243:240–249
Zahir ZA, Ahmad M, Hilger TH, Dar A, Malik SR, Abbas G, Rasche F (2018) Field evaluation of multistrain biofertilizer for improving the productivity of different mungbean genotypes. Soil Environ 37:45–52
Acknowledgements
Authors are thankful to the university for providing lab facility. NP is grateful to the ministry of tribal affairs for providing financial assistance as NFST—JRF [Ref No—201920-NFST-JAM-02504).
Funding
Funding was provided by Ministry of Tribal Affairs, Govt of India (Ref No—201920-NFST-JAM-02504).
Author information
Authors and Affiliations
Contributions
RPS conceptualized the idea. NP performed the experiments. NP wrote the manuscript, NP, RM, DVS, PK and RPS reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no declarations of interest.
Ethical approval
This article does not contain any studies with human or animal subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Parveen, N., Mishra, R., Singh, D.V. et al. Assessment of different carrier materials for the preparation of microbial formulations to enhance the shelf life and its efficacy on the growth of spinach (Spinacia oleracea L.). World J Microbiol Biotechnol 39, 180 (2023). https://doi.org/10.1007/s11274-023-03594-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-023-03594-4