Abstract
Vibrio vulnificus is a halophilic estuarine bacterium while it causes fatal septicemia or necrotizing wound infections in humans. This pathogen secretes the metalloprotease (V. vulnificus protease: VVP) and the cytolysin (V. vulnificus hemolysin: VVH) as protein toxins; however, their production was coordinated in response to the bacterial cell density. This regulation is termed quorum sensing (QS) and is mediated by the small diffusible molecule called autoinducer 2 (AI-2). In the present study, we investigated effects of disruption of luxO encoding a central response regulator of the QS circuit, as well as effects of temperature and growth phase, on the toxin production by V. vulnificus. Disruption of luxO was found to increase VVP production and expression of its gene vvpE. The expression of smcR, crp and rpoS, of which products positively regulate vvpE expression, and luxS encoding the AI-2 synthetase were also significantly increased. On the other hand, the luxO disruption resulted in reduction of VVH production and expression of its gene vvhA. Expression of other two genes affecting the QS circuit, luxT and rpoN, were also significantly decreased. The regulation systems of VVP production were found to exert their action during the stationary phase of the bacterial growth and to be operated strongly at 26 °C. By contrast, those of VVH production apparently started at the log phase and were operated more effectively at 37 °C.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In pathogenic bacteria, coordinated regulation of the virulence gene expression is critical to successful colonization, invasion, in vivo growth and/or in situ toxin production. The bacterial virulence is often regulated by temperature, and this regulation occurs at both transcription and translation level (Hurme and Rhen 1998). Transfer from a natural reservoir to an infectious host offers a number of cues, which induce the signal transmission to turn on the virulence potentials (Hurme and Rhen 1998).
Vibrio vulnificus is a gram-negative halophilic estuarine bacterium, while it is an opportunistic human pathogen causing rapidly progressing fatal septicemia and necrotizing wound infection (Jones and Oliver 2009). The infectious diseases are preferentially in susceptible patients with hepatic diseases, hemochromatosis, heavy alcohol drinking habits, and other immunocompromised conditions (Jones and Oliver 2009). Virulence of V. vulnificus is multifactorial (Milton 2006), and the bacterium produces various kinds of virulent or toxic factors including capsular polysaccharides, type IV pili, hemolytic/cytolytic toxin, and proteolytic enzymes (Linkous and Oliver 1999; Strom and Paranjpye 2000). However, the metalloprotease (V. vulnificus protease: VVP/VvpE) and the hemolysin (V. vulnificus hemolysin: VVH/VvhA) are the most important toxins (Milton 2006). VVP causes serious hemorrhagic skin damage through digestion of the vascular basement membrane, especially type IV collagen forming the framework of the membrane (Miyoshi et al. 2001). It also elicits edema formation through induction of exocytotic histamine release from mast cells (Miyoshi et al. 2003) and/or activation of the factor XII-plasma kallikrein-kinin cascade (Miyoshi et al. 2004). On the other hand, VVH exhibits powerful hemolytic and cytolytic activities (Gray and Kreger 1985), and it causes vasodilation and may play a significant role in hypotensive septic shock (Kook et al. 1996).
Vibrio vulnificus coordinates expression of virulence genes in response to the bacterial cell density. This regulation is termed quorum sensing (QS), which is mediated by the small diffusible signal molecule called autoinducer 2 (AI-2) (Federle and Bassler 2003; Henke and Bassler 2004). Indeed, V. vulnificus possesses LuxS (the AI-2 synthetase), LuxPQ (membrane bound sensor protein), LuxU-LuxO (the response regulators of QS circuit of V. vulnificus), and SmcR (the master transcriptional regulator for target genes controlled by the QS system) (McDougald et al. 2000, 2001; Shao and Hor 2001; Chen et al. 2003; Kim et al. 2003; Kawase et al. 2004). Moreover, five small RNAs (sRNAs) regulating SmcR were also predicted, as well as Vibrio cholerae and Vibrio harveyi (Lenz et al. 2004). Besides, Roh et al. (2006) identified LuxT as a negative regulator of SmcR. At low cell density, when the signal molecule AI-2 is absent, LuxPQ functions as kinase and it acts on LuxU and add phosphate group to the protein. The phosphorelay protein LuxU then transfers the phosphate group to LuxO. Therefore, at low cell density, LuxO remains phosphorylated which is the active form of the protein. Active LuxO, in association with sigma factor 54 RpoN, activates expression of sRNAs (small regulatory RNAs). The sRNAs along with sRNA binding protein Hfq represses the transcriptional regulator SmcR (Milton 2006). Also active LuxO activates the expression of LuxT, which is a negative regulator of SmcR (Roh et al. 2006). On the other hand at high cell density, when there is sufficient concentration of the signal molecule AI-2, it interacts with its specific sensor LuxPQ and converts its function from kinase to phosphatase. Subsequently the sensor protein dephosphorylates LuxO via LuxU. The dephosphorylated LuxO is inactive and it cannot activate the expression of sRNAs or LuxT. As a result SmcR is not inhibited any more. Therefore, at high cell density, SmcR functions actively and results in change of transcriptional status of the target genes (Milton 2006; Roh et al. 2006). In V. vulnificus, both AI-2 and SmcR positively regulate VVP production while negatively regulate VVH production (Shao and Hor 2001; Kim et al. 2003; Kawase et al. 2004). However, the primary target of V. vulnificus QS cascade may be VVP because VVP production is regulated more strongly (Milton 2006; Kim and Shin 2010, 2011). In addition to QS, two global regulators, namely RpoS (the stationary-phase sigma factor) and CRP (cAMP-receptor protein) responsible for catabolic repression, are known to control directly expression of vvpE encoding VVP (Kim and Shin 2011).
In the present study, we outlined effects of disruption of luxO on production of VVP and VVH, and on expression of their genes (vvpE and vvhA). In addition, we examined expression of genes consisting of the QS circuit (luxS, luxT and smcR) and those related to the QS cascade (rpoN that activates expression of sRNAs and hfq that acts together with sRNAs) (Milton 2006), rpoS and crp. The expression of rpoD, the house keeping sigma factor during log phase, was also examined. These experiments were carried out at two different temperatures, 26 °C (around estuarine temperature in the summer season) and 37 °C (around human intestinal temperature) and at different phases of the bacterial growth.
Materials and methods
Bacterial strains, plasmids and culture conditions
Bacterial strains and plasmids used in this study are listed in Table 1. Escherichia coli strains were grown on Luria–Bertani (LB) agar plate or in LB broth containing 0.5 % NaCl, and when required an appropriate antibiotic was added to the media as follows: chloramphenicol 10 μg/ml, streptomycin 50 μg/ml and kanamycin 50 μg/ml.
For cultivation of V. vulnificus strains, TYE broth (0.5 % tryptone, 0.25 % yeast extract, 2 % NaCl, 25 mM K2HPO4, pH 7.5) was used. Thiosulfate-citrate-bile-salts-sucrose (TCBS) agar plate containing chloramphenicol 10 μg/ml was used to select luxO mutants. In all experiments, V. vulnificus was cultivated in TYE broth (5 ml) at 37 °C with shaking overnight (strain YY0507, the luxO mutant, was cultivated in TYE broth containing chloramphenicol 10 μg/ml), and then, an aliquot of the first culture was taken and re-cultivated in fresh TYE broth at either 26 or 37 °C till reaching the desired growth phase.
For the AI-2 assay, autoinducer bioassay (AB) broth (1 mM l-arginine, 2 % glycerol, 10 ng/ml riboflavin, 1 μg/ml thiamin, 300 mM NaCl, 10 mM K2HPO4, 50 mM MgSO4, 0.2 % casamino acids; pH 7.5) was used.
Construction of the luxO mutant and its revertant
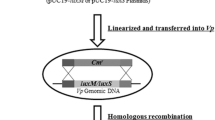
The luxO mutant was constructed by the single crossover homologous recombination as described previously (Nishibuchi et al. 1991; Funahashi et al. 2002). A 781 bp region of the luxO was amplified by PCR using a primer set luxO-1, a forward primer containing the recognition sequence for XbaI (TCTAGA) and a reverse primer containing the recognition sequence for EcoRI (GAATTC) (Table 2), and digested with XbaI and EcoRI. The XbaI-EcoRI digested PCR product was inserted into the suicide vector pKTN701 (Nishibuchi et al. 1991). The hybrid plasmid obtained was transformed into E. coli SY327λpir, then into E. coli SM10λpir. Thereafter, it was transferred to V. vulnificus L-180 by conjugation, and the conjugants were cultivated on TCBS agar plates containing chloramphenicol 10 μg/ml. One suitable luxO mutant named strain YY0507 was selected by 48 h cultivation at 37 °C, and disruption of the luxO gene was confirmed by PCR. The revertant strain named AAER12 was obtained by repeated sub-culturing of strain YY0507 in LB broth at 37 °C, and reversion of the gene was confirmed by PCR.
Measurement of bacterial growth
Vibrio vulnificus strains were grown at 37 °C under aeration in TYE broth (5 ml) overnight with shaking. Then, an aliquot of the first culture was taken and re-cultivated in fresh TYE broth at 26 or 37 °C, and the growth monitored by measuring the optical density at 600 nm (OD600) of the cultures every 1 h. Then, the growth curves were drawn. Thereafter, early log phase, late log phase, early stationary phase, and stationary phase were determined.
RT-PCR
Total RNA was extracted from the bacterial cells cultivated at 26 or 37 °C at early log, late log or early stationary phase, by using RNeasy Mini Kit (Qiagen GmbH, Hilden, Germany) according to the manufacturer’s manual. Total RNA thus obtained was added to the Ready-To-Go RT-PCR kit (GE Healthcare Bio-science, Buckinghamshire, UK) and incubated at 42 °C for 30 min for reverse transcription. Thereafter, the reverse transcriptase was inactivated by heating at 95 °C for 5 min, and PCR amplification with an appropriate primer set (Table 2) was performed as follows: 30 s denaturation at 95 °C, 30 s annealing at an appropriate temperature, and 60 s extension at 72 °C. The PCR products were electrophoresed on a 1.5 % agarose gel and visualized by staining with ethidium bromide and the intensity of bands were analyzed using imageJ program. The relative amount of each mRNA was estimated using the amount of mRNA of the house keeping gene 16s rRNA as 1.0.
Assay of VVP, VVH and AI-2 activity
Quantitative assays of VVP, VVH and AI-2 activity of the culture supernatants were performed. Cultures of V. vulnificus strains were grown in TYE medium until early log phase, late log phase, early stationary phase and stationary phase. Cell free culture supernatants were prepared from these cultures by centrifugation (at 12,000g for 5 min at 4 °C) and filtration (through 0.2 μm Millipore filter). Sterile TYE medium was used as negative control in the assays.
The proteolytic activity of VVP was assayed with azocasein (Sigma-Aldrich, St. Louis, MO, USA) as described by Miyoshi et al. (1987a, b). Briefly, the sample was allowed to act at 30 °C for an appropriate time on 1.0 mg of azocasein in 0.6 ml of 50 mM Tris–HCl buffer (pH 8.0). The reaction was stopped by the addition of 1.4 ml of 5 % trichloroacetic acid. After centrifugation at 5,000g for 5 min, an aliquot of the supernatant was withdrawn and mixed with the same volume of 0.5 M NaOH. Thereafter, the absorbance at 440 nm was measured. One protease unit (PU) was defined as the amount of VVP hydrolyzing 1 μg of the substrate in 1 min. In these experiments, purified VVP was used as a positive control.
The hemolytic activity of VVH was assayed with 1 % sheep erythrocytes as described by Shinoda et al. (1985). Briefly, the sample (0.6 ml) was allowed to act on the erythrocytes (0.6 ml) at 37 °C for 2 h in 20 mM Tris–HCl buffer containing 0.9 % NaCl (pH 7.5). Thereafter, the reaction mixtures were centrifuged at 1,000g for 5 min, and the amount of hemoglobin released from the disrupted erythrocytes was determined by measuring the absorbance of the supernatant at 540 nm. One hemolysin unit (HU) was defined as the amount of VVH eliciting 50 % hemoglobin release. In these experiments, purified VVH was used as a positive control.
The AI-2 activity was measured using the reporter strain V. harveyi BB170 as described by Bassler et al. (1993). Briefly, the reporter strain was cultured overnight in LB broth containing 3.0 % NaCl at 30 °C. The bacterial culture was diluted 1:5,000 with AB broth. An aliquot of the diluted culture (540 μl) was mixed with 60 μl of the sample, and the mixture was cultivated at 30 °C for 4 h with shaking. Thereafter, the intensity of bioluminescence (relative light unit: RLU) was measured with a luminometer K-210 (Kikkoman, Tokyo, Japan). In these experiments, the supernatant from V. harveyi BB152, a mutant producing only AI-2, was used as the 100 % control.
Western blot analysis
For Western blot analysis, proteins in the sample were precipitated by mixing with the same volume of 25 % tricloroacetic acid, and collected by centrifugation for 5 min at 15,000g at 4 °C. Thereafter, the protein collected was washed by 100 % ethanol, treated with 2 % sodium dodecyl sulfate (SDS) at 100 °C for 5 min and subjected to SDS-PAGE on the PhastSystem™ using a PhastGel™ Gradient 10–15 (GE Health Bio-Sciences). After SDS-PAGE, the proteins separated were transferred to a polyvinylidene difluoride (PVDF) membrane (GE Health Bio-Sciences). The membrane with the bound proteins was then incubated with rabbit IgG antibody against VVP or VVH, and the antigen–antibody complex was visualized using the antibody against rabbit IgG conjugated with horseradish peroxidase (Santa Cruz Biotechnology, Santa Cruz, CA, USA) and a chromogenic substrate 4-methoxy-1-naphthol and hydrogen peroxide. In these experiments, sterile TYE medium (the cultivation medium) was used as a negative control and purified VVP or VVH was used as a positive control.
Statistical analysis
To evaluate the significance of difference in the results, all experiments were repeated at least three times, the data were compared by student t test, and the P values less than 0.05 were considered significantly different.
Results
Construction of the luxO mutant and its revertant
To clarify inactivation of the luxO gene, the RNA preparations from the luxO mutant (strain YY0507, luxO::Cmr), the revertant (strain AAER12), as well as the wild type strain (strain L-180), were analyzed by RT-PCR. As shown in Fig. 1a, in the case of strain L-180 and AAER12, the significant transcription of luxO was observed; however, the luxO mRNA could not be detected in strain YY0507.
Expression of luxO, luxOU and luxU. In order to confirm inactivation of luxO in the luxO mutant strain YY0507 and its reversion in strain AAER12, total RNA was prepared from each strain, and RT-PCR to detect luxO (a), luxOU (b), or luxU mRNA (b) was carried out. The 16S rRNA was used as the positive control. Lane M 1-kb DNA ladder, lane 1 strain L-180, lane 2 strain YY0507 and lane 3 strain AAER12
The luxO gene makes an operon with a downstream gene luxU. Therefore, the RT-PCR experiments targeting the luxOU mRNA also showed that null expression of the operon in the luxO mutant (Fig. 1b). However, a comparative amount of luxU mRNA was detected in all strains (Fig. 1b), indicating the luxU gene has own promoter and it functions normally in the luxO mutant.
Growth of the luxO mutant and its revertant
Strain YY0507 was found to grow slightly faster than the wild type strain at both 26 and 37 °C; however, the growth speed of strain AAER12, the revertant from strain YY0507, was the same as that of the wild type strain (Fig. 2).
Growth of the bacterial strains at 26 °C (a) and 37 °C (b). Strain L-180 (grey diamond), YY0507 (black square) and AAER12 (white triangle) were cultivated in TYE broth at 26 or 37 °C, and the optical density at 600 nm (OD600) was measured every 1 h. Thereafter, early log phase (a), late log phase (b), early stationary phase (c), and stationary phase (d) were determined. Data represent the mean of three experiments
Expression of sigma factor genes (rpoD, rpoS and rpoN)
By disruption of luxO gene, the rpoD expression was significantly increased at log and early stationary phase, and the expression of rpoS was also increased at late log and early stationary phase (Fig. 3). On the other hand, disruption of luxO resulted in decrease in the expression of rpoN at late log and early stationary phase (Fig. 3). It should be noted that expression of rpoD and rpoS gene at 26 °C was higher than at 37 °C, but rpoN was more expressed at 37 °C (Fig. 3).
Effect of luxO disruption on expression of rpoD (a), rpoS (b) and rpoN (c). Strain L-180 (grey bar), YY0507 (black bar) and AAER12 (white bar) were cultivated in TYE broth at 26 or 37 °C, total RNA was extracted at early log phase (a), late log phase (b) and early stationary phase (c), and the level of mRNA was measured by RT-PCR. Thereafter, PCR products were electrophoresed on 1.5 % agarose gel, visualized by staining with ethidium bromide and analyzed by imageJ program. The amount of mRNA was represented using the amount of 16s rRNA as 1.00. The data is the mean + SD of three experiments. The asterisk indicates the significant difference (P < 0.05) between strain YY0507 and both strain L-180 and AAER12
Expression of the QS cascade genes (luxS, luxT and smcR), crp and hfq
As shown in Fig. 4, disruption of luxO resulted in significant increase in the expression of luxS, smcR and crp genes at late log and early stationary phase, and these genes were expressed more at 26 °C. By contrast, expression of luxT was decreased by disruption of luxO, and the transcription level of luxT was higher at 37 °C. On the other hand, no significant difference of hfq expression was observed by disruption of luxO gene (data not shown).
Effect of luxO disruption on expression of luxS (a), luxT (b) and smcR (c) and crp (d). Strain L-180 (grey bar), YY0507 (black bar) and AAER12 (white bar) were cultivated in TYE broth at 26 or 37 °C, total RNA was extracted at early log phase (a), late log phase (b) and early stationary phase (c), and the level of mRNA was measured by RT-PCR. Thereafter, PCR products were electrophoresed on 1.5 % agarose gel, visualized by staining with ethidium bromide and analyzed by imageJ program. The amount of mRNA was represented using the amount of 16s rRNA as 1.00. The data is the mean + SD of three experiments. The asterisk indicates the significant difference (P < 0.05) between strain YY0507 and both strain L-180 and AAER12
Expression of vvpE and vvhA gene
Expression of vvpE and vvhA was highly dependent on the bacterial growth and cultivation temperature (Fig. 5). Namely, vvpE expression was maximum at early stationary phase and was distinctly higher at 26 °C. Although transcription of vvpE gene was started at log phase, upon the entry in the stationary phase, its level was increased about 10 times. On the other hand, vvhA was most expressed at log phase at 37 °C.
Effect of luxO disruption on expression of vvpE (a) and vvhA (b). Strain L-180 (grey bar), YY0507 (black bar) and AAER12 (white bar) were cultivated in TYE broth at 26 or 37 °C, total RNA was extracted at early log phase (a), late log phase (b) and early stationary phase (c), and the level of mRNA was measured by RT-PCR. Thereafter, PCR products were electrophoresed on 1.5 % agarose gel, visualized by staining with ethidium bromide and analyzed by imageJ program. The amount of mRNA was represented using the amount of 16s rRNA as 1.00. The data is the mean SD of three experiments. The asterisk indicates the significant difference (P < 0.05) between strain YY0507 and both strain L-180 and AAER12
The disruption of luxO gene showed apparent increase in the vvpE expression. As shown in Fig. 5a, the transcription level of vvpE gene in the luxO-disrupted strain YY0507 was markedly higher than that of strain L-180 and AAER12. By contrast, the luxO disruption resulted in significant decrease in the expression of vvhA (Fig. 5b).
Production of VVP, VVH and AI-2
The VVP activity in the culture supernatant was highly dependent on the bacterial growth and cultivation temperature. Upon the entry in the stationary phase, the activity was increased about 10 times of that at log phase, and the activity was distinctly higher at 26 °C (Fig. 6a). By Western blot analysis, the VVP antigen could not be detected at early log phase; however, the antigen was steadily detected at late log and stationary phase (Fig. 6b). The disruption of luxO caused increase in production of VVP. The activities of VVP in the culture supernatants from strain YY0507 were significantly higher than those of strain L-180 and AAER12, and more steadily bands of the VVP antigen were detected when the culture supernatants of strain YY0507 were analyzed by western blotting (Fig. 6).
The activity of VVP in the culture supernatants (a) and Western blot analysis of the VVP antigen (b). a Culture supernatants were prepared from early log phase (a), late log phase (b), early stationary phase (c) and stationary phase (d) of growth of L-180 (grey bar), YY0507 (black bar) and AAER12 (white bar). The activities of VVP (PU/ml) in the culture supernatants were determined using azocasein as substrate, and the specific protease activity (PU/OD600) was calculated. The data is the mean + SD of three experiments. The asterisk indicates the significant difference (P < 0.05) between YY0507 and both strain L-180 and AAER12. In these experiments, purified VVP was used as a positive control, and the cultivation media used as a negative control showed no activity. b Western blot analysis of the VVP antigen was performed by precipitating the proteins in the collected culture supernatants by adding the same volume of 25 % tricloroacetic acid, the protein pellets was washed by 100 % ethanol, dissolved by suspending in SDS sample buffer and boiled for 5 min, and an aliquot of each preparation was subjected to SDS-PAGE. Thereafter, the proteins were transferred to a PVDF membrane, and VVP antigens were detected with the antibody against purified VVP. Purified VVP was used as a positive control and cultivation medium used as a negative control. Lane 1 strain L-180, lane 2 strain YY0507 and lane 3 strain AAER12
The VVH activity in the culture supernatant was also dependent on the bacterial growth and cultivation temperature. However, in contrast to the VVP activity, the VVH activity was apparently higher when cultivated at 37 °C, and the highest activity was detected in the culture supernatant at late log phase (Fig. 7). Additionally, disruption of luxO gene resulted in significant decrease in the activity caused by VVH at both cultivation temperatures (Fig. 7).
The activity of VVH in the culture supernatants (a) and Western blot analysis of the VVH antigen (b). a Culture supernatants were prepared from early log phase (a), late log phase (b), early stationary phase (c) and stationary phase (d) of growth of L-180 (grey bar), YY0507 (black bar) and AAER12 (white bar). The activities of VVH (HU/ml) in the culture supernatants were determined using 1 % sheep erythrocytes, and the specific hemolysin activity (HU/OD600) was calculated. The data is the mean + SD of three experiments. The asterisk indicates the significant difference (P < 0.05) between YY0507 and both L-180 and AAER12. In these experiments, purified VVH was used as a positive control, and the cultivation media used as a negative control showed no activity. b Western blot analysis of the VVH antigen was performed by precipitating the proteins in the collected culture supernatants by adding the same volume of 25 % tricloroacetic acid, the protein pellets was washed by 100 % ethanol, dissolved by suspending in SDS sample buffer and boiled for 5 min, and an aliquot of each preparation was subjected to SDS-PAGE. Thereafter, the proteins were transferred to a PVDF membrane, and VVH antigens were detected with the antibody against purified VVH. Purified VVH was used as a positive control and cultivation media used as a negative control. Lane 1 strain L-180, lane 2 strain YY0507 and lane 3 strain AAER12
As well as VVP and VVH, the activity of AI-2 was highly dependent on the bacterial growth and cultivation temperature. The activity reached a maximum level at early stationary phase of cultivation at 26 °C (Fig. 8). The luxO-disrupted mutant YY0507 showed the higher activity than the wild type strain and the revertant strain, indicating negative regulation of luxS by LuxO.
AI-2 activity in the culture supernatants. Culture supernatants were prepared from early log phase (a), late log phase (b), early stationary phase (c) and stationary (d) of growth of L-180 (grey bar), YY0507 (black bar) and AAER12 (white bar). Culture supernatants of test bacteria (60 μl) were added to the diluted reporter strain V. harveyi BB170 (540 μl) and incubated at 30 °C with shaking for 4 h. Bioluminescences of 600 μl aliqoutes of samples were measured with a luminometer, Lumitester K-210 (Kikkoman, Tokyo, Japan) that measured the amount of bioluminescence in relative luminescence unit (RLU). In these experiments, the supernatant from V. harveyi BB152 was used as a positive control (RLU 1.0), and the cultivation media used as a negative control showed no activity. The data is the mean + SD of three experiments. The asterisk indicates the significant difference (P < 0.05) between YY0507 and both L-180 and AAER12
Discussion
Vibrio vulnificus inhabits sea water or brackish water, but it can cause infection in human. During the infection process, V. vulnificus must sense and sustain changes in environmental factors. The most important environmental difference in many respects is the temperature (Lee et al. 2007). Signals from the changing environmental factors are relayed to specific genes by cognate signal transduction systems, resulting in the expression of genes including specific virulence factor genes. Virulence factors required for in vivo survival and growth are produced at an appropriate place and time in a tightly regulated fashion (Heithoff et al. 1997; Lee et al. 1999; Kim et al. 2007). In V. vulnificus, VVP is the best-known virulence factor regulated by various environmental signals (Jeong et al. 2003; Kim et al. 2003; Roh et al. 2006; Kim and Shin 2011), and three global regulators, RpoS, SmcR, and CRP, have been reported to control directly production of VVP (Chiang and Chuang 2003; Kim and Shin 2010, 2011). Here we studied the effects of disruption of luxO, which encodes the master regulator of QS cascade, on the expression of the genes of three global regulators at different temperatures, 26 and 37 °C at different phases of the bacterial growth. Also, the effects of luxO disruption on the expression of sigma factor genes, the QS cascade genes, vvpE and vvhA genes, and production of VVP, VVH and AI-2 were examined.
Our results demonstrated that V. vulnificus produced two protein toxins, VVP and VVH, at specific times of the bacterial growth in a tightly regulated fashion. As summarized in Table 3, disruption of luxO showed increased expression of rpoS, luxS, smcR, crp and vvpE at stationary phase. Production of AI-2 and VVP was also distinctly higher in the luxO mutant. However, at log phase, the vvpE expression and VVP production was not increased. These results verified the critical roles of LuxO in the regulation of V. vulnificus QS system. Two promoters, promoter L (PL) and promoter S (PS), are known to direct differentially transcription of the vvpE gene in a growth phase-dependent manner (Jeong et al. 2001, 2003, 2010). The PL activity is constitutive through the log and stationary phases, but is lower than the PS activity. The transcription from PS is induced only in the stationary phase and is dependent on RpoS, CRP and SmcR. These findings put the light on the exact time of SmcR, CRP and RpoS regulation of vvpE expression, namely, the regulation occurs only upon the entry to the stationary phase. Because transcription of vvpE from PL is starting from the log phase, RNA polymerase with RpoD, the log phase housekeeping sigma factor, would recognize the PL. However, in the present study, it was not confirmed that RpoD has significant role in VVP production during log phase. Although rpoD expression was significantly increased in the luxO mutant, vvpE transcription and VVP production were not significantly different from both the wild type strain and the revertant strain.
As summarized in Table 4, it is clear that both the cultivation temperature and growth phase are very critical determinants for regulation of expression vvpE and vvhA gene. The transcription level of vvpE and production of VVP were distinctly higher at 26 °C. It is also noteworthy that the expression of genes encoding positive regulators of vvpE (smcR, rpoS and crp) and AI-2 synthetase (luxS), and the AI-2 production were also higher at 26 °C. Taken together, it is concluded that the regulation systems for VVP production are operated strongly at 26 °C, but not at 37 °C. Therefore, as documented previously by Kawase et al. (2004), V. vulnificus produces a significant amount of VVP only in the interstitial tissue of limbs, in which temperature is lower than that in the small intestine and the blood-stream, and VVP produced causes serious hemorrhagic and edematous skin damage (Miyoshi et al. 2001, 2003, 2004).
In contrast to VVP, the production of VVH started during log phase and reached a maximum level at late log phase. Disruption of luxO resulted in decreased expression of vvhA and less production of VVH. It is also noteworthy that, at 37 °C, the expression of rpoN was higher at log phase. This may indicate that RNA polymerase with RpoN recognizes the promoter of vvhA gene. Taken together, it is concluded that the regulation systems for VVH production are operated effectively at 37 °C. Therefore, V. vulnificus produces an enough amount of VVH in the small intestine, which results in acceleration of the bacterial invasion to the blood-stream.
References
Bassler BL, Wright M, Showalter RE, Silverman MR (1993) Intercellular signaling in Vibrio harveyi: sequence and function of genes regulating expression of luminescence. Mol Microbiol 9:773–786
Chen CY, Wu KM, Chang YC, Chang CH, Tsai HC, Liao TL, Liu YM, Chen HJ, Shen AB, Li JC, Su TL, Shao CP, Lee CT, Hor LI, Tsai SF (2003) Comparative genome analysis of Vibrio vulnificus, a marine pathogen. Genome Res 13:2577–2587
Chiang SR, Chuang YC (2003) Vibrio vulnificus infection: clinical manifestations, pathogenesis, and antimicrobial therapy. J Microbiol Immunol Infect 36:81–88
Federle MJ, Bassler BL (2003) Interspecies communication in bacteria. J Clin Invest 112:1291–1299
Funahashi T, Moriya K, Uemura S, Miyoshi S, Shinoda S, Narimatsu S, Yamamoto S (2002) Identification and characterization of pvuA, a gene encoding the ferric vibrioferrin receptor protein in Vibrio parahaemolyticus. J Bacteriol 184:936–946
Gray LD, Kreger AS (1985) Purification of characterization of an extracellular cytolysin produced by Vibrio vulnificus. Infect Immun 48:62–72
Heithoff DM, Conner CP, Mahan MJ (1997) Dissecting the biology of a pathogen during infection. Trends Microbiol 5:509–513
Henke JM, Bassler BL (2004) Bacterial social engagements. Trends Cell Biol 14:648–656
Hurme R, Rhen M (1998) Temperature sensing in bacterial gene regulation—what it all boils down to. Mol Microbiol 30:1–6
Jeong HS, Jeong KC, Choi HK, Park KJ, Lee KH, Rhee JH, Choi SH (2001) Differential expression of Vibrio vulnificus elastase gene in a growth phase-dependent manner by two different types of promoters. J Biol Chem 276:13875–13880
Jeong HS, Lee MH, Lee KH, Park SJ, Choi SH (2003) SmcR and Cyclic AMP receptor protein coactivate Vibrio vulnificus vvpE encoding elastase through the RpoS-dependent promoter in a synergistic manner. J Biol Chem 278:45072–45081
Jeong HS, Kim SM, Lim MS, Kim KS, Choi SH (2010) Direct interaction between quorum-sensing regulator SmcR and RNA polymerase is mediated by integration host factor to activate vvpE encoding elastase in Vibrio vulnificus. J Biol Chem 285:9357–9366
Jones MK, Oliver JD (2009) Vibrio vulnificus: disease and pathogenesis. Infect Immun 77:1723–1733
Kawase T, Miyoshi S, Sultan Z, Shinoda S (2004) Regulation system for protease production in Vibrio vulnificus. FEMS Microbiol Lett 240:55–59
Kim CM, Shin SH (2010) Regulation of the Vibrio vulnificus vvpE expression by cyclic AMP-receptor protein and quorum-sensing regulator SmcR. Microb Pathog 49:348–353
Kim CM, Shin SH (2011) Change of Vibrio vulnificus metalloprotease VvpE production by temperature and salinity. J Bacteriol Virol 41:147–156
Kim SY, Lee SE, Kim YR, Kim CM, Ryu PY, Choy HE, Chung SS, Rhee JH (2003) Regulation of Vibrio vulnificus virulence by luxS quorum sensing system. Mol Microbiol 48:1647–1664
Kim CM, Park RY, Chun HJ, Kim SY, Rhee JH, Shin SH (2007) Vibrio vulnificus metalloprotease VvpE is essentially required for swarming. FEMS Microbiol Lett 269:170–179
Kook H, Lee SE, Baik YH, Chung SS, Rhee JH (1996) Vibrio vulnificus hemolysin dilates rat thoracic aorta by activating guanylate cyclase. Life Sci 59:41–47
Lee SH, Hava DL, Waldor MK, Camilli A (1999) Regulation and temporal expression patterns of Vibrio cholera virulence genes during infection. Cell 99:625–634
Lee SE, Kim SY, Kim CM, Kim MK, Kim YR, Jeong K, Ryu HJ, Lee YS, Chung SS, Choy HE, Rhee JH (2007) The pyrH gene of Vibrio vulnificus is an essential in vivo survival factor. Infect Immun 75:2795–2801
Lenz DH, Mok KC, Lilley BN, Kulkarni RV, Wingreen NS, Bassler BL (2004) The small RNA chaperone Hfq and multiple small RNAs control quorum sensing in Vibrio harveyi and Vibrio cholerae. Cell 118:69–82
Linkous DA, Oliver JD (1999) Pathogenesis of Vibrio vulnificus. FEMS Microbiol Lett 174:207–214
McDougald D, Rice SA, Kjelleberg S (2000) The marine pathogen Vibrio vulnificus encodes a putative homologue of the Vibrio harveyi regulatory gene, luxR: a genetic and phylogenetic comparison. Gene 248:213–221
McDougald D, Rice SA, Kjelleberg S (2001) SmcR dependent regulation of adaptive phenotypes in Vibrio vulnificus. J Bacteriol 183:758–762
Miller VL, Mekalanos JJ (1988) A novel suicide vector and its use in construction of insertion mutations: osmoregulation of outer membrane proteins and virulence determinants in Vibrio cholerae requires toxR. J Bacteriol 170:2575–2583
Milton DL (2006) Quorum sensing in vibrios: complexity for diversification. Int J Med Microbiol 296:61–71
Miyoshi N, Shimizu C, Miyoshi S, Shinoda S (1987a) Purification and characterization of Vibrio vulnificus protease. Microbiol Immunol 31:13–25
Miyoshi S, Sugiyama K, Suzuki Y, Furuta H, Miyoshi N, Shinoda S (1987b) Enhancement of vascular permeability due to histamine-releasing effect of Vibrio vulnificus protease. FEMS Microbiol Lett 40:95–98
Miyoshi S, Kawata K, Tomochika K, Shinoda S, Yamamoto S (2001) The C-terminal domain promotes the hemorrhagic damage caused by Vibrio vulnificus metalloprotease. Toxicon 39:1883–1886
Miyoshi S, Kawata K, Hosokawa M, Tomochika K, Shinoda S (2003) Histamine-releasing reaction by the N-terminal domain of Vibrio vulnificus metalloprotease. Life Sci 72:2235–2242
Miyoshi S, Watanabe H, Kawase T, Yamada H, Shinoda S (2004) Generation of active fragments from human zymogens in the bradykinin-generation cascade by extracellular proteases from Vibrio vulnificus and V. parahaemolyticus. Toxicon 44:887–893
Nishibuchi M, Kumagai K, Kaper JB (1991) Contribution of the tdh1 gene of Kanagawa phenomenon-positive Vibrio parahaemolyticus to production of extracellular thermostable direct hemolysin. Microb Pathog 11:453–460
Roh JB, Lee MA, Lee HJ, Kim SM, Cho Y, Kim YJ, Seok YJ, Park SJ, Lee KH (2006) Transcriptional regulatory cascade for elastase production in Vibrio vulnificus: LuxO activates luxT expression and LuxT represses smcR expression. J Biol Chem 281:34775–34784
Shao CP, Hor LI (2001) Regulation of metalloprotease gene expression in Vibrio vulnificus by V. harveyi luxR homologue. J Bacteriol 183:1369–1375
Shinoda S, Miyoshi S, Yamanaka H, Miyoshi N (1985) Some properties of Vibrio vulnificus hemolysin. Microbiol Immunol 29:583–590
Simon R, Priefer U, Puhler A (1983) A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in gram negative bacteria. Bio/Technology 1:784–791
Strom MS, Paranjpye RN (2000) Epidemiology and pathogenesis of Vibrio vulnificus. Microbes Infect 2:177–188
Acknowledgments
This study was supported by a grant from the Program of Japan Initiative for Global Research Network on Infectious Diseases (J-GRID), the Ministry of Education, Culture, Sports, Science and Technology in Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elgaml, A., Higaki, K. & Miyoshi, Si. Effects of temperature, growth phase and luxO-disruption on regulation systems of toxin production in Vibrio vulnificus strain L-180, a human clinical isolate. World J Microbiol Biotechnol 30, 681–691 (2014). https://doi.org/10.1007/s11274-013-1501-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1501-3