Abstract
Studied is the effect of the non-ionic surfactant Tween 80 on the microbial transformation of 4-androstene-3,17-dione into its 9α-hydroxy-derivative by resting Rhodococcus sp. cells. The surfactant was applied in the cultivation medium as an additional source of carbon, in the transformation reaction medium as a mediator of the steroid substrate solubility or was used for permeabilization of the glucose grown Rhodococcus sp. cells. Special attention is paid to the fact that Tween 80 accelerates the 9α-steroid hydroxylation reaction carried out by glucose-grown cells. When the surfactant was applied as a supplementary source of carbon, the rate of the steroid hydroxylation reaction was significantly lower. In addition, the kinetics of the transformation process changed into a linear one thus indicating a very slow, if any, product degradation. The fatty acid profile, cell surface hydrophobicity as well as cell ultrastructure observed by scanning and transmission electron microscopy in the Tween 80- and glucose-grown Rhodococcus sp. cells are compared and related with their 9α-hydroxylating activity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The manufacture of steroid drugs includes several important microbial hydroxylation steps which are essential links of the production chain due to their exclusive stereospecificity (Holland 1999; Fernandes et al. 2003). The interest in studying the reaction of 9α-steroid hydroxylation is determined by the key role of the 9α-hydroxy-derivatives in the manufacturing of highly effective halogenated corticosteroids (Kieslich 1984). The effectiveness of microbial steroid transformation reactions, however, is limited by the low water solubility of the steroid compounds.
In most of the cases steroid substrates are added to the bioconversion systems dissolved in water-miscible organic solvents. Some transformation processes are carried out in the presence of organic compounds enhancing steroid compounds solubilization like surfactants and cyclodextrins (Smith et al. 1993; Roglič et al. 2005). Aqueous-organic solvent two-phase systems where the water-immiscible organic solvent contained the steroid substrate are also applied (Angelova et al. 2005). Only a few reports, however, associate the effect of the compounds applied to enhance the steroid substrates solubilization (such as lecitin, cyclodextrin and bis(2-ethylhexyl)phthalate) with their impact on the cell viability and morphology, microbial cell wall and cellular membranes (de Carvalho et al. 2004a, b; Donova et al. 2007; Rumijowska et al. 1997).
In previous studies we reported about the transformation of 4-androstene-3,17-dione (androstenedione, AD) into 9α-hydroxy-4-androstene-3,17-dione (9α-OH-AD) by resting Rhodococcus sp. where the steroid substrate was added into the transformation reaction medium as water suspension (Angelova et al. 1996; Mutafov et al. 1997) or dissolved in the water immiscible bis(2-ethylhexyl)phthalate being a component of an aqueous-organic solvent two-phase system or comprising a single phase reaction medium (Angelova et al. 2005).
This study aimed at evaluation the influence of the non-ionic surfactant Tween 80 on the 9α-steroid hydroxylating activity of the resting Rhodococcus sp. using androstenedione as a model substrate. For this reason Tween 80 was applied in the cultivation medium as an additional source of carbon, in the transformation reaction medium as a mediator of the steroid substrate solubility or was used for permeabilization of the glucose grown Rhodococcus sp. cells. The fatty acid profiles, cell hydrophobicity and cell surface structures observed by scanning and transmission electron microscopy in the Tween 80- and glucose-grown Rhodococcus sp. cells were compared and related with their steroid hydroxylating activity.
Materials and methods
Reagents
Tween 80™ (polysorbate 80, polyoxyethylene (20) sorbitan monooleate) and Triton X-100™ (polyoxyethylene-octylphenyl ether) were from Sigma (St. Luis, MO). Span 80™ (sorbitan monooleate; sorbitan (Z)-mono-9-octadecenoate) and androstenedione were from Serva Feinbiochemica (Heidelberg, Germany). Durcopan™ resins for electron microscopy were from Fluka Chemie AG (Buchs, Switzerland).
The nutrient broth Standard I™ was from Merck AG (Darmstadt, Germany). The cultivation medium was prepared by enzymatically digested casein hydrolysate as described earlier (Angelova et al. 1996).
Phosphate buffer solution (0.04 M KH2PO4, pH 7.2) was used throughout the study.
Microorganism
The Rhodococcus sp. strain originates from the N.D. Zelinsky Institute of Organic Chemistry of the Russian Academy of Sciences. It is maintained on slants of agarised broth Standard I in the microbial collection of the Department of Microbial Biochemistry.
Growth conditions
The cells were grown in 500-ml Erlenmeyer flasks each containing 100 ml cultivation medium supplemented with either 2% (w/v) glucose or 0.5% (v/v) Tween 80. Each flask was inoculated with 1 ml of a 24-h culture obtained on the same medium. After cultivation for 48 h on a rotary shaker at 28°C the cells were harvested by centrifugation, washed with buffer solution and used for steroid transformation.
Permeabilization procedure
The pellet of glucose-grown of Rhodococcus sp. cells was re-suspended in buffer solution to optical density 1.0 at 540 nm. The cells were subjected for 30 min to permeabilization with 0.1% (v/v) Tween 80 as described by Galabova and co-authors (Galabova et al. 1996), harvested by centrifugation and washed with buffer solution.
Transformation procedure
The transformation of AD was carried out at 28°C on a rotary shaker in 100-ml iodine flasks each containing 10 ml phosphate buffer, 100 mg wet cells and 10 mg AD. The AD was added from a stock microcrystalline suspension obtained by ultrasonic disintegration. Surfactants were applied in a final concentration 0.1% (v/v).
Analytical procedures
The cell surface hydrophobicity (H%) was determined by microbial cell adhesion to hydrocarbons (Rosenberg et al. 1980).
The fatty acid composition was determined after extraction of the total lipids according to Cristie (2003). The extracted fatty acids were analyzed by GC as methyl esters. The analytical parameters were: 25-m capillary column Ca-Wax (Chromatography Associates, Inc., State College, PA); temperature range from 150° to 200°C; ascending temperature gradient 4°C min-1; injector temperature 260°C; nitrogen flow rate 10 mm min−1; flame ionization detector; detection temperature 260°C.
The degree of fatty acid saturation was determined as a ratio of saturated to unsaturated fatty acids. The relative acyl chain length was expressed as a ratio of the sum of C18-fatty acids to the sum of C16-fatty acids.
For assaying the steroid compounds samples of 0.1 ml of the reaction mixture were dissolved in 0.9 ml methanol and 10 μl were analyzed by reversed phase HPLC (Shimadzu LC-4A system, Shimadzu Corp., Kyoto, Japan). The analytical parameters were: column Zorbax ODS (5 μm, 150 × 4.6 mm), (Du Pont Co., Wilmington, DE); mobile phase methanol:water:acetic acid (80:20:0.05); flow rate 1 ml min−1; temperature 40°C; detection at 254 nm.
Quantifications
All numeric data presented are mean values of three independent experiments. For their graphical presentation the NURBS procedure (non-uniform, rational B-splines) was used (TableCurve 2D 5.01, SYSTAT Software, San Jose, CA).
Electron microscopy
For scanning electron microscopy (SEM) the cells were fixed in 2% glutaraldehyde for 2 h at 4°C. The fixed cells were washed with phosphate buffer and dehydrated in water–ethanol ascending series from 30 to 100% (v/v), followed by air-drying for 2–3 h. The samples were coated with 12–13 nm gold in argon medium using Edwards S 150 A sputter apparatus (Edwards High Vacuum International, Crawley, UK) operated at 0.2 mbar. The observations were carried out at accelerating voltage 20 kV and electron ray 5–6 nm on a scanning device attached to a Zeiss transmission electron microscope 10C (Carl Zeiss AG, Oberkochen, Germany).
For transmission electron microscopy (TEM) the cells were fixed in 1% glutaraldehyde for 15 min at room temperature and washed three times with phosphate buffer. After a post fixation with 1% osmium tetroxide in phosphate buffer for 18 h the samples were washed with buffer and dehydrated in ascending water–ethanol series. Samples were embedded in Durcopan resin, thin sections were cut, stained with 1% uranyl acetate and lead citrate as described by Reynolds (1963) and examined at 60 kV with Zeiss 10C transmission electron microscope.
Results
Effect of non-ionic surfactants, Tween 80, Triton X-100 and Span 80, on the 9α-hydroxylating activity
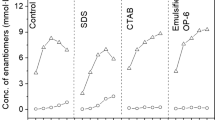
The results obtained (Fig. 1) from the transformation of AD in the presence of Triton X-100 showed a strong inhibition of the process due to the evident lysis of the bacterial cells. The presence of both, Tween 80 and Span 80 had a stimulating effect better expressed with Tween 80. The observed beneficial effect of Tween 80 on the accumulation of 9α-OH-AD was the reason for its selection for the experiments described below.
Effect of Tween 80 on the 9α-hydroxylating activity of intact and permeabilized cells
The dynamics of the 9α-hydroxylation of AD carried out by intact or permeabilized with Tween 80 cells in the presence and in the absence of the surfactant was studied (Fig. 2).
The effect of Tween 80 on the 9α-hydroxylation of AD carried out with intact and permeabilised cells: transformation of AD with intact cells in the presence of Tween 80 (filled triangle); transformation of AD with permeabilized cells in the presence of Tween 80 (inverted triangle); transformation of AD with permeabilized cells without Tween 80 (open square); transformation of AD with cells grown in the presence of Tween 80 (triangle). The control experiment (filled circle) was carried out with intact cells without Tween 80
The results obtained revealed a strong influence of the surfactant application mode on the transformation process. It is obvious that if the transformation medium does not contain Tween 80, the 9α-hydroxylation activity of the cells preliminary treated with the surfactant is practically the same as the activity of the intact cells. However, the presence of Tween 80 in the reaction medium causes acceleration of the 9α-OH-AD accumulation by permeabilized Rhodococcus sp. cells, which is much higher than the one observed with intact cells. This fact suggested that a prolonged contact of the cells with Tween 80 might enhance their 9α-hydroxylating activity.
The time course of the transformation reaction performed with cells harvested from a cultivation medium containing Tween 80 as a carbon source is presented on Fig. 2. Contrary to our expectations, the 9α-hydroxylating activity of cells grown in the presence of Tween 80 was lower and the transformation process was slower compared to glucose-grown cells regardless intact or permeabilized cells were used in the experiments (Fig. 2).
Fatty acid composition and cell surface hydrophobicity of Tween 80-grown cells
The analysis of the fatty acids content revealed several interesting differences between glucose- and Tween 80-grown cells. In particular, it was found that the glucose-grown cells contained mainly saturated fatty acids: tetradecanoic (myristic, 14:0); hexadecanoic (palmitic, 16:0); and octadecanoic (stearic, 18:0) and several unidentified fatty acids which sum did not exceed 5% of the total amount. The growth in the presence of Tween 80 resulted in the appearance of two unsaturated fatty acids with longer acyl chains: octadecenoic (oleic, 18:1) and octadecadienoic (linoleic, 18:2) (Fig. 3). The predomination of the longer chain unsaturated fatty acid decreased the fatty acid saturation and on other hand increased the relative acyl chain length up to 1.77 for Tween 80 grown cells, while for glucose grown Rhodococcus sp. cells the value is 0.27.
The total amount of lipids in the Tween 80-grown cells (14 mg g−1 wet weight) is slightly higher compared to the glucose-grown cells (11 mg g−1) while the cell surface hydrophobicity of glucose-grown cells was considerably higher compared to the hydrophobicity of the Tween 80-grown ones (72 vs. 57%).
Scanning and transmission electron microscopy of glucose- and Tween 80-grown cells
Both, the scanning and the transmission electron microscopy revealed considerable changes in cell surface structures (cell wall and cytoplasmic membrane) arising from the cultivation of the microorganism in the presence of the non-ionic surfactant Tween 80.
On the scanning electron micrographs the glucose-grown cells have dense surface with distinct even outlines (Fig. 4A, B). An amorphous electron dense material covers entirely the cells. Somewhere a separation of this outmost coating from the cells was observed. The TEM observations are in compliance with those carried out by SEM. The electron dense capsule-like material and its separation from certain cell surface zones are observed (Fig. 4C). The cell wall and the cytoplasmic membranes seen in the ultra thin sections revealed a good integrity and were found to fit tightly to the cytoplasm.
The cell surface of Tween 80-grown cells observed by SEM (Fig. 5A, B) looks rather rough with numerous concavities making the cell outline uneven. The cells have reduced turgor and reduced electron density. Cell disruption and lysis of cell membranes (Fig. 5A, B) are a good illustration of damages caused by the addition of Tween 80 to the cultivation medium.
The examination of the ultra thin sections of cells grown in the presence of Tween 80 (Fig. 5C, D) provided additional evidences for the deteriorating effect of the surfactant. The electron micrographs reveal a partial loss of the outmost electron dense layer with clearly recognizable zones of partial disruption of the underlying cell wall and membranes and a release of the cytoplasm (Fig. 5C). The visible decrease of the electron density in the area of the septal cell walls (Fig. 5D) is also a symptom of deep structural changes caused by the surfactant.
Discussion
The observed suppression of the steroid transformation process in the presence of Triton X-100 confirms results published earlier about its inhibiting effect on the activities of the Gram positive Rhodococcus erythropolis (Ahn et al. 2005) and Enterococcus sp. (Kang and Ryu 1999). We ascribe the observed fast cell lysis of the Rhodococcus sp. cells in the presence of Triton X-100 as a main reason for the suppression of the AD hydroxylation. According to Willumsen et al. (1998), the inhibiting effect of Triton X-100 might be due to the increased concentration of the toxic substrate in the aqueous phase or to the prevention of bacterial adhesion to the hydrophobic substrate.
It is largely accepted that the stimulating effect of Tween 80 on the microbial transformation/degradation processes of water-insoluble substrates is related to the increased solubility of the substrate in the aqueous medium (Willumsen et al. 1998; Kotterman et al. 1998; Lu et al. 2007). Our results are in a good agreement with this conception: in the presence of Tween 80 cells, preliminary treated with this surfactant, reveal much higher activity. However, this conception alone is not sufficient to explain why in the absence of the surfactant the Tween-treated cells have the same transformation activity as the non-treated ones. For that reason we assume that the presence of Tween 80 in the reaction medium gives rise to some modifications in the outmost layer that facilitate the absorption and the subsequent transport of the steroid substrate through the cell wall to the underlying membrane structures.
In this connection it is necessary to recall that the function of 9α-steroid hydroxylase is coupled with a well-organized membrane-bond electron-transport chain (Strijewski 1982). Recent investigations proved that ferredoxin reductase, a terminal oxygenase and NADH are essential for the 9α-hydroxylase activity with steroid substrates (Petrusma et al. 2009). In our experiments the observed by SEM and TEM disruptions of cell wall and membranes and the leakage of cytoplasm correlate with the decreased steroid hydroxylating activity of the cells cultured in medium containing Tween 80.
In view of the water insolubility and prominent hydrophobicity of the steroid substrate it could be suggest that the process of its microbial hydroxylation requires direct cell-substrate contact, so it seems reasonable any change of the cell surface hydrophobicity to influence the process effectiveness. We incline to explain the decreased transformation activity of the Tween 80-grown cells with the decreased hydrophobicity of these cells which possibly hinders the cell-substrate contact and as a consequence, the induction of the 9α-steroid hydroxylating activity. The 9α-hydroxylated product is accumulated slowly thus preventing the induction of the Δ1-steroid dehydrogenase and results in a very slow, if any, product degradation. As a consequence, the typical peak-like time course of the 9α-steroid hydroxylation process changes into a linear one (Fig. 2). In addition, the observed by SEM and TEM exfoliation of the electron dense capsule-like material and its partial disappearance from the Tween 80-grown possibly further contributes to lowering the cell surface hydrophobicity. Similar loss of hydrophobicity with parallel loss of surface lipids and mycolic acids were reported for the closely related mycobacteria and corynebacteria (Donova et al. 2007; Puech et al. 2001).
References
Ahn T-S, Lee G-H, Song H-G (2005) Biodegradation of phenanthrene by psychrotrophic bacteria from Lake Baikal. J Microbiol Biotechnol 15:1135–1139
Angelova B, Mutafov S, Avramova T, Dimova I, Boyadjieva L (1996) 9α-Hydroxylation of 4-androstene-3, 17-dione by resting Rhodococcus sp. cells. Process Biochem 31:179–184
Angelova B, Fernandes P, Cruz A, Pinheiro HM, Mutafov S, Cabral JMS (2005) Hydroxilation of androstenedione by resting Rhodococcus sp. cells in organic media. Enzyme Microb Technol 37:718–722
Cristie W (2003) Lipid analysis. Isolation, separation, identification and structural analysis of lipids, 3rd edn. The Oily Press, Bridgwater, England, pp 91–104
de Carvalho CCR, Cruz AARL, Pons MN, Pinheiro HMRV, Cabral JMS, da Fonseca MMR, Ferreira BS, Fernandes P (2004a) Mycobacterium sp., Rhodococcus erythropolis, and Pseudomonas putida behavior in the presence of organic solvents. Microsc Res Tech 64:215–222
de Carvalho CCR, Cruz A, Angelova B, Fernandes P, Pons MN, Pinheiro HM, Cabral JMS, da Fonseca MMR (2004b) Behaviour of Mycobacterium sp. NRRL B-3805 whole cells in aqueous, organic-aqueous and organic media studied by fluorescence microscopy. Appl. Microb. Biotech 64:695–701
Donova MV, Nikolayeva VM, Dovbnya DV, Gulevskaya SA, Suzina NE (2007) Methyl-β-cyclodextrin alters growth, activity and cell envelope features of sterol-transforming mycobacteria. Microbiology 153:1981–1992
Fernandes P, Cruz A, Angelova B, Pinheiro HM, Cabral JMS (2003) Microbial conversion of steroid compounds: recent developments. Enzyme Microb Technol 32:688–705
Galabova D, Tuleva B, Spasova D (1996) Permeabilization of Yarrowia lipolytica cells by Triton X-100. Enzyme Microb Technol 18:18–22
Holland HL (1999) Recent advances in applied and mechanistic aspects of the enzimatic hydroxylation of steroids by whole-cell biocatalists. Steroids 64:178–186
Kang K-H, Ryu H-W (1999) Enhancement of succinate production by organic solvents, detergents, and vegetable oils. J Microbiol Biotechnol 9:191–195
Kieslich K (1984) Biotransformations. In: Rehm HJ, Reed G (eds) Biotechnology. A comprehensive treatise in 8 volumes, vol 6A. Verlag, Weinheim, pp 1–78
Kotterman MJJ, Rietberg H-J, Hage A, Field JA (1998) Polycyclic aromatic hydrocarbon oxidation by the white-rot fungus Bjerkandera sp. strain BOS55 in the presence of nonionic surfactants. Biotechnol Bioeng 57:220–227
Lu W, Du L, Wang M, Guo Y, Lu F, Sun B, Wen J, Jia X (2007) A novel substrate addition method in the 11β-hydroxylation of steroids by Curvularia lunata. Food Bioprod Process 85(1C):63–72
Mutafov S, Angelova B, Avramova T, Boyadjieva L, Dimova I (1997) The inducibility of 9α-steroid hydroxylating activity in resting Rhodococcus sp. cells. Process Biochem 32:585–589
Petrusma M, Dijkhuizen L, van der Geize R (2009) Rhodococcus rhodochrous DSM 43269 3-ketosteroid 9a-hydroxylase, a two-component iron-sulfur-containing monooxygenase with subtle steroid substrate specificity. Appl. Environ. Microbiol. 75:5300–5307
Puech V, Chami M, Lemassu A, Lanéelle M-A, Schiffler B, Gounon P, Bayan N, Benz R, Daffé M (2001) Structure of the cell envelope of corynebacteria: importance of the non-covalently bound lipids in the formation of the cell wall permeability barrier and fracture plane. Microbiology 147:1365–1382
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–213
Roglič U, Žnidaršič-Plazl P, Plazl I (2005) The influence of β-cyclodextrin on the kinetics of progesterone transformation by Rhizopus nigricans. Acta Chim Slov 48:533–544
Rosenberg M, Gutnik D, Rosenberg E (1980) Adherence of bacteria to hydrocarbons: a simple method for measuring cell-surface hydrophobicity. FEMS Microbiol Lett 9:29–30
Rumijowska A, Lisowska K, Ziolkowski A, Sedlaczek L (1997) Transformation of sterols by Mycobacterium vaccae: effects of lecithin on the permeability of cell envelopes to sterols. World J Microbiol Biotechnol 13:89–95
Smith M, Zahnley J, Pfeifer D, Goff D (1993) Growth and cholesterol oxidation by Mycobacterium species in Tween 80 medium. Appl Environ Microbiol 59:1425–1429
Strijewski A (1982) The steroid-9α-hydroxylation system from Nocardia species. Eur J Biochem 128:125–135
Willumsen PA, Karlson U, Pritchard PH (1998) Response of fluoranthene-degrading bacteria to surfactants. Appl Microbiol Biotechnol 50:475–483
Acknowledgments
This work was partly supported by the Foundation for Scientific Investigations of the Bulgarian Ministry of Education, Science and Youth (contract HTC 01-161).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Avramova, T., Spassova, D., Mutafov, S. et al. Effect of Tween 80 on 9α-steroid hydroxylating activity and ultrastructural characteristics of Rhodococcus sp. cells. World J Microbiol Biotechnol 26, 1009–1014 (2010). https://doi.org/10.1007/s11274-009-0263-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-009-0263-4