Abstract
US federal conservation programs, including the National Resource Conservation Service’s Wetland Reserve Program (WRP) and the US Fish and Wildlife Service’s Partners for Fish and Wildlife Program (PFWP), partner with private landowners to conserve and restore wetland habitats. Despite the success of these programs in terms of wetland area enrolled, uncertainties exist as to whether they are meeting their stated goals, including the restoration of wildlife habitat. In the St. Lawrence Valley of New York State, we investigated two questions related to WRP and PFWP wetland restorations. First, was whether restorations provide habitat for wetland-associated wildlife, including Species of Greatest Conservation Need (SGCN) prioritized by the New York State Wildlife Action Plan (SWAP). Second, was whether restorations support wildlife assemblages that are comparable to natural reference wetlands. Bird, anuran, turtle, snake, and fish species assemblages were surveyed at 47 WRP and PFWP restorations, and 18 natural reference wetlands. We detected 31 SGCN at restorations, including SGCN from each assemblage surveyed. Assemblage metrics, including species richness and relative abundance, did not differ between restored and reference wetlands for any of the assemblages surveyed. These results indicate that restorations provide habitat for SGCN and other wetland-associated wildlife, and that assemblages at restorations are similar to those at natural reference wetlands. We conclude that WRP and PFWP wetland restorations in this region are meeting federal program-level goals related to the restoration of wildlife habitat, and are contributing to the recovery of SGCN.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Globally, one of the most consequential changes in land-use is wetland loss due to degradation and drainage (Millennium Ecosystem Assessment 2005; Mitsch and Gosselink 2015). Wetlands ecosystems are valuable due to the ecosystem services they provide; not only do they have significant commercial and recreational value, wetlands also capture excess nutrients and pollutants, facilitate groundwater recharge, decrease erosion, mitigate flooding, increase water availability, regulate regional climate, and serve as attractive habitat for a variety of species (Costanza et al. 2014). Despite the value of these services, human-dominated landscapes are characterized by the loss of wetland habitats to agricultural, residential, or industrial development, as these services are external to existing markets (Dahl and Allord 1997; Zedler and Kercher 2005; Ghermandi et al. 2010). In the US, since the Clean Water Act of 1972 and the 1989 US federal policy objective of no net wetland loss, mitigation programs that conserve and restore wetland habitats have been instituted to compensate for past and present wetland losses. Most wetland restoration projects have been completed on public land, but the greatest need for the augmentation of wetland ecosystem services is on private land, as historically this is where the greatest losses have occurred (Dahl et al. 1991; Scodari 1997; Zedler 2003; Fairfax et al. 2005; Maresch et al. 2008).
Among the most important mechanisms for mitigating wetland losses in the US are voluntary programs that provide incentives to private landowners to conserve or restore wetland habitats, often within a conservation easement held by a public agency partner or land trust. Two public–private partnership programs in the US, the National Resource Conservation Service’s (NRCS) Wetlands Reserve Program (WRP) and the US Fish and Wildlife Service’s (USFWS) Partners for Fish and Wildlife Program (PFWP), have restored over one million hectares of wetlands, mostly within the last two decades (Filsinger and Milmoe 2012; USDA NRCS 2014a). WRP is focused on agricultural land and was created by the 1990 Farm Bill with the goal of “achieving the greatest [wetland] functions and values, along with optimum wildlife habitat, on every acre enrolled in the program” (USDA NRCS 2014a). Since the 2014 Farm Bill, WRP has been incorporated into the new Agricultural Conservation Easement Program (USDA NRCS 2014b). PFWP was created in 1987 with the objective of offering assistance to landowners with habitat restoration for the benefit of federal trust species (USFWS 2015a). Despite the success of these programs in terms of wetland area enrolled, uncertainties exist as to whether they are meeting their stated goals (Heard 2000; updated 2008; Rewa 2005; Gray and Teels 2006; Maresch et al. 2008; Brinson and Eckles 2011). Although agencies are obligated to conduct some post-restoration monitoring, there are few resources allocated to this task relative to those available for establishing new partnerships. More is known about the outcomes of wetlands restored via mandatory mitigation under the Clean Water Act, but it is unlikely that mitigation sites are comparable to wetlands restored via voluntary public–private partnership programs (National Research Council, NRC 2001; Kihslinger 2008). The United States Department of Agriculture (USDA) has recently conducted a series of cumulative effects studies on how ecosystem services, such as carbon-storage and nutrient management, are affected by NRCS programs, but these assessments focused on the regional program-level (aggregated-projects) rather than the project-level (Duriancik et al. 2008; Maresch et al. 2008; Euliss et al. 2010; Brinson and Eckles 2011). While results from program-level assessments indicate that the wetland restoration programs are generally meeting their goals, evaluative surveys at the project-level are also needed to compare different methods of restoration, to determine how restorations change over time, and to examine how they compare to natural wetlands on the landscape (Mitsch and Wilson 1996; Brinson and Rheinhardt 1996; Zedler 2000; Rewa 2005; Brinson and Eckles 2011).
One way to evaluate the success of WRP and PFWP is to assess the use of restorations by assemblages of biological indicator species (e.g., birds, amphibians, aquatic macroinvertebrates) or other target species [e.g., Species of Greatest Conservation Need (SGCN), federal trust species]. Biological indicator species provide information on the functionality of a particular ecosystem, as species within an assemblage vary in their habitat requirements and their sensitivity to disturbances. The number of studies describing the response of a single species assemblage to wetland restoration continues to grow (e.g., Ratti et al. 2001; Kaminski et al. 2006; Larkin et al. 2008; O’Neal et al. 2008), however, studies that include multiple species assemblages are much fewer (e.g., Juni and Berry 2001; Balcombe et al. 2005; Nedland et al. 2007). Assessing multiple species assemblages at restorations should be a priority because as with individual species, assemblages also vary in their habitat requirements and sensitivity to disturbance. It is also possible that a particular restoration might benefit members of one assemblage over another.
In 2000, the US Congress funded the State Wildlife Grant Program, which provides states with money to conserve SGCN. To participate in the program, each state is required to draft and periodically revise a State Wildlife Action Plan (SWAP) that identifies SGCN and proposes a prioritized plan for research, management, and species recovery (USFWS 2015b). New York State’s 2015 SWAP identifies many wetland-associated species as SGCN, and the state’s 2005 Comprehensive Wildlife Conservation Strategy identified the need to determine whether restored wetlands play a role in conserving SGCN (New York State Department of Environmental Conservation, NYSDEC 2005, 2015).
The St. Lawrence Valley of New York is a regional hotspot for wetland restoration, and is home to over 200 WRP and PFWP projects (USFWS 2006). As per program regulations, all restorations are completed on private property and most sites enrolled in WRP are protected under conservation easements. Restored wetlands are usually small, often under New York State’s regulatory threshold of 5 ha, and may consist of one several-hectare wetland, multiple small (under 1 ha) wetlands, or a combination of both large and small wetlands. A small number of PFWP wetland restoration projects in this region were surveyed for vegetation, macroinvertebrates, birds, and amphibians pre- and 2 years post-restoration in the early 1990s (Brown and Bedford 1997; Brown et al. 1997; Brown 1998, 1999; Brown and Smith 1998) and a sub-set of these sites were re-sampled several years later by Robinson (2000).
Our study had two objectives. First, was to determine whether WRP and PFWP wetland restorations in the St. Lawrence Valley provide habitat for wetland-associated wildlife, including SGCN. Second, was to determine whether WRP and PFWP wetland restorations support wildlife assemblages that are comparable to those found at natural reference wetlands in the region. To meet these objectives, we surveyed bird, anuran, turtle, snake, and fish assemblages at 47 WRP and PFWP restorations, and compared assemblage metrics, such as species richness and relative abundance between 18 restored–reference pairs.
Methods
Study area and site selection
The wetlands surveyed were located within a 6000 km2 expanse of northern Jefferson County (43.66° N, 75.81° W) and adjacent St. Lawrence County (43.92° N, 75.53° W), New York, and distributed across three ecological regions (Reschke 1990). The Eastern Ontario Plains and St. Lawrence Plains are rolling agricultural lowlands of hayfields, dairy farms, maize and other row crops (regions 42% and 31% agricultural, respectively), with natural vegetation predominately consisting of northern mixed hardwood and conifer forests. The Indian River Lakes region is comprised of numerous lakes, granite outcrops, as well as greater forest cover and less agricultural land (15%) than the other two regions (Fry et al. 2011). Human population densities outside of the scattered villages are low (around 10 persons/km2). The climate is cool (annual average temperature 6.6 °C, average minimum winter temperature −17.5 °C, and average maximum summer temperature 24.9 °C at Canton, NY) and seasonal, including long cold winters and short cool summers (growing season 125 days); precipitation is high (94 cm annually) and occurs throughout the year (National Oceanic Atmospheric Administration National Climatic Data Center, NOAA NCDC , NOAA NCDC 2010).
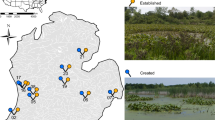
We surveyed 47 wetland restorations in 2009–2011 and 2014, including 28 WRP projects, 15 PFWP projects, and four projects that were shared between these programs (Fig. 1). Ducks Unlimited provided financial and/or technical assistance for at least 18 restorations. Thirty-one WRP restorations were protected under conservation easements, whereas PFWP projects were not. We selected 18 WRP and PFWP projects to pair with natural reference wetlands based on year surveyed, proximity, basin morphometry, and landscape context (c.f. Brinson and Rheinhardt 1996; Figs. 1, 2). As restorations were located on private land, we attempted to select reference sites that were also on private property. However, due to a scarcity of small natural wetlands and issues with obtaining landowner access, two reference wetlands were located on nearby state Wildlife Management Areas. Restored and reference sites were characterized as either scrub–shrub or emergent palustrine wetlands (Cowardin et al. 1979). Restoration techniques included excavating small depressions, constructing berms and water control structures, ditch-plugging, or tile drain removal. Land-uses bordering wetlands included hayfield, row crops, cattle pasture, shrub-successional stage fallow farm fields, forest, or landowner residences. Restorations (n = 47) ranged from 0.14 to 7.49 ha (mean ± SD = 5.32 ± 2.26 ha), while reference sites (n = 18) ranged from 0.17 to 10.73 ha (7.86 ± 2.69 ha).
Wildlife surveys
The bird assemblage was surveyed at each site between sunrise and 10:00 in May and June, via two 10 min, 100 m fixed-radius point counts and playback vocalization surveys, where species were identified visually or by their vocalizations (Ralph et al. 1995; Bibby et al. 2000; Rehm and Baldassarre 2007). We surveyed from a single vantage point located on the open water and emergent vegetation interface; one skilled observer (TAL) did all point counts. Playback vocalization surveys consisted of 30 s of vocalization and 30 s of silence, with three replicates per species. The speaker volume was standardized to a sound pressure level of 80–90 dB at a distance of 1 m (Conway 2009). Focal species, in order of broadcast, included the Least Bittern (Ixobrychus exilis), Sora (Porzana carolina), Virginia Rail (Rallus limicola), Common Gallinule (Gallinula galeata), American Coot (Fulica americana), Pied-billed Grebe (Podilymbus podiceps), King Rail (Rallus elegans), Yellow Rail (Coturnicops noveboracensis), American Bittern (Botaurus lentiginosus), Sedge Wren (Cistothorus platensis), Marsh Wren (Cistothorus palustris), and Golden-winged Warbler (Vermivora chrysoptera; Cornell Lab of Ornithology 2009); seven of these species are SGCN. Bird assemblage metrics, summed over the two surveys per site, included relative species richness, which was calculated by summing the species detected within the 100 m radius during point counts and all focal species detected during playback vocalizations. Additionally, we report the richness of SGCN and a weighted-average index reflecting the dominance of each assemblage by wetland-associated birds. To calculate the index, each species was assigned a rank based on its wetland indicator status (as given in Brooks and Croonquist 1990), and the rank values were summed for each site, such that a higher index value indicated greater dominance by wetland-associated species.
The anuran (frog and toad) assemblage was surveyed on a total of three nights, once in late April, once in late May, and once in early July. Surveys were done a minimum of 15 days apart at sites and coincided with three air temperature ranges (5–9, 10–16, and >16 °C). Each survey occurred between 0.5 h after sunset and 01:00. Surveys consisted of a 5 min, 50 m semi-circular fixed-radius point count located on the open water and emergent vegetation interface. Survey protocols were adapted from the North American Amphibian Monitoring Program, and species were identified by their vocalizations and chorus size was estimated using call codes (Weir and Mossman 2005). Anuran assemblage metrics measured included relative species richness and a relative abundance index calculated by summing the highest recorded call code per species per site. During anuran surveys, we also noted detections of the Whip-poor-will (Antrotomus vociferus), a nocturnal bird SGCN.
The turtle assemblage was surveyed via 10 baited hoop traps (30 cm aperture) checked daily for three consecutive days (30 trap-nights) in June (Steen and Gibbs 2005). Individuals captured were identified, sexed, measured (straight line carapace length), and marked by filing a notch on the pygal scute to detect recaptures (Ferner 2007). We surveyed the snake assemblage via two 1 h visual searches in May and June. Visual searches were conducted between 07:00 and 12:00 and consisted of an observer walking along the water’s edge searching for snakes (Heyer et al. 1994; McDairmid et al. 2012). Individuals captured were identified, measured (body length), and released. No attempt was made to mark individuals. Turtle and snake assemblage metrics included relative species richness and relative abundance, as indicated by the turtle catch-per-unit-effort (CPUE; individuals/trap-night) and the number of snake detections.
We surveyed the fish assemblage, including small species and the young of larger species, via five baited cylindrical funnel traps (42 cm long, 22 cm in diameter, 3 cm diameter entry opening, 0.48 cm2 mesh size, baited with 40 cat food pellets in 30 mL, perforated plastic containers) checked daily for three consecutive days (15 trap-nights). Three traps were placed amid submerged hydrophytes in the littoral zone along the perimeter of each wetland at 1 m depth. Individuals were identified and released. No attempt was made to mark individuals. Fish assemblage metrics included relative species richness and CPUE (individuals/trap-night).
Data analysis
To evaluate whether assemblage metrics differed between restored and reference wetlands, we conducted statistical hypothesis tests of the restored–reference pairs. Analogous analyses using full, unpaired dataset provided qualitatively identical results. The average mean difference in assemblage metrics between restored and reference wetland pairs was compared using paired t-tests or, in the case that parametric assumptions were violated, Wilcoxon signed-rank tests. We used the Anderson–Darling test to detect deviations from normality in the pair difference scores. All statistical tests were completed using the R Project for Statistical Computing (R Core Team 2012). When observed mean differences do not differ significantly from zero, some would advocate that a retrospective power analysis be reported (either an ‘observed power’ or ‘detectable effect size’ analysis), in order to evaluate the likelihood that the null hypothesis (average mean difference = 0.0) would have been rejected were it false. However, we agree with Hoenig and Heisey (2001) that the use of power analysis in this way is fallacious, and that confidence intervals (CIs) are more informative. An average mean difference near zero and tight CIs provide the most convincing evidence that there is little to no difference between treatment types. Thus, we report the 95% CI for all pair-wise comparisons. To maintain an experiment-wise Type I error rate of 5%, we applied a Bonferroni correction; given we included 11 metrics, the criterion for rejecting a null hypothesis was p < 0.0045.
We did not attempt to model species-by-species detectability via occupancy modeling, as our study represents a snapshot assessment of biodiversity. However, we assumed that detectability was similar per species across sites, and since surveys were done using the same methods at each site, our assemblage metrics should be positively correlated with true metric for each assemblage surveyed. We have no reason to suspect that detection probabilities of SGCN differed between restored and reference wetlands. All raw and summary data are available as Online Supplementary Materials.
Results
A total of 124 of 183 bird species known to breed in this region were detected, with 115 species at restorations (n = 47) and 100 species at reference wetlands (n = 18; McGowan and Corwin 2008). Both the American Goldfinch (Carduelis tristis) and the Common Yellowthroat (Geothlypis trichas) were the most commonly detected species, found at all restored and reference wetlands. A total of 22 and 18 obligate wetland species were detected at restored and reference wetlands, respectively. The Mallard (Anas platyrhynchos), Swamp Sparrow (Melospiza georgiana), Wood Duck (Aix sponsa), and Great Blue Heron (Ardea herodias) were the four most commonly detected obligate wetland species at both restored and reference wetlands. A total of 159 detections of 24 bird SGCN were made across sites, including 9 High Priority SGCN and 15 SGCN (Table 1). Twenty-four SGCN were detected at restorations and 17 SGCN were detected at reference wetlands. Both the Scarlet Tanager (Piranga olivacea) and Wood Thrush (Hylocichla mustelina) were among the most common SGCN at both restored and reference sites. Of the 24 SGCN detected, 7 were obligate wetland species. There was no difference in detected species richness, the richness of SGCN, or the wetland index (dominance by wetland-associated species) between restored and reference wetland pairs (Table 2).
All 10 anuran species native to the region were detected, including the Spring Peeper (Pseudacris crucifer), Boreal Chorus Frog (Pseudacris maculata), Grey Tree Frog (Hyla versicolor), American Toad (Anaxyrus americanus), Northern Leopard Frog (Lithobates pipiens), Green Frog (Lithobates clamitans), American Bullfrog (Lithobates catesbeianus), Pickerel Frog (Lithobates palustris), and Mink Frog (Lithobates septentrionalis; Gibbs et al. 2007). All species were detected at restorations, and all but the Wood Frog were detected at reference sites. The Spring Peeper, Green Frog, Grey Tree Frog, and American Bullfrog were the four most commonly detected species at both restored and reference wetlands. Both the Boreal Chorus Frog and the Mink Frog are classified as Potential SGCN (Table 1). Neither detected species richness nor relative abundance (indicated by the call-code index) differed between restored and reference wetlands (Table 2).
A total of 158 individuals of four of the seven turtle species found in the region were detected, including 106 individuals of four species at restorations and 52 individuals of three species at reference wetlands (Gibbs et al. 2007). The Snapping Turtle (Chelydra serpentina), a SGCN, was the most commonly detected species across sites (99 individuals). The Blanding’s Turtle, identified as a High Priority SGCN, was detected at one restored (one individual) and one reference (three individuals) wetland (Table 1). We also detected 56 Painted Turtles (Chrysemys picta) and one Red-eared Slider (Trachemys scripta elegans), a non-native species that was likely a release. There were no differences in detected species richness and relative abundance (indicated by CPUE) between restored and reference wetlands (Table 2).
We only completed snake surveys from 2009 to 2011, therefore results are restricted to surveys of 33 restorations and 14 restored–reference pairs. A total of 56 individuals of five of the nine snake species found in the region were detected, including the Common Gartersnake (Thamnophis sirtalis), Eastern Ribbonsnake (Thamnophis sauritus sauritus), Northern Watersnake (Nerodia sipedon), Eastern Milksnake (Lampropeltis triangulum), and Eastern Ratsnake (Pantherophis alleghaniensis; Gibbs et al. 2007). Captures at restorations included 31 individuals of five species and 25 individuals of four species were detected at reference wetlands. The Eastern Milksnake was only detected at a single restoration. The two SGCN snakes detected were the Eastern Ribbonsnake, the most commonly detected snake species, and the Eastern Ratsnake (Table 1). Neither detected species richness nor relative abundance (indicated by total detections) differed between restored and reference wetlands (Table 2).
A total of 2582 individuals of 20 of the estimated 35 fish species found in this region were detected, including 2105 individuals of 19 species at restorations and 477 individuals of 15 species at reference wetlands (Werner 2004). The Pumpkinseed (Lepomis gibbosus), Brook Stickleback (Culaea inconstans), Central Mudminnow (Umbra limi), and Fathead Minnow (Pimephales promelas) were the four most commonly detected species at both restored and reference sites. We detected one SGCN, the Black Bullhead (Ameiurus melas) at three restorations. We did not detect any fish at 11 restorations and 6 reference wetlands. There were no differences in detected species richness or relative abundance (indicated by CPUE) between restored and reference wetlands (Table 2).
Discussion
Our results indicate that WRP and PFWP restorations in the St. Lawrence Valley provide habitat for a variety of wetland-associated wildlife, including, but not limited to, 115 species of birds, 10 species of anurans, 4 species of turtles, 5 species of snakes, and 19 species of fish. A total of 31 SGCN were detected at restorations, including SGCN for each assemblage surveyed. Additionally, restorations were also found to provide habitat for several woodland and grassland-associated species, many of which are also SGCN.
Birds
Similar to other studies, we detected no difference in bird assemblage metrics between restored and reference wetlands; six of six studies comparing bird species richness between restored palustrine wetlands and reference wetlands also report no difference in species richness between sites (Brawley et al. 1998; Brown and Smith 1998; Juni and Berry 2001; Ratti et al. 2001; Balcombe et al. 2005; Kahara et al. 2012). Brown and Smith (1998), in the same region of New York as our study, compared the avian assemblages at PFWP restorations 3 years post-restoration with natural wetlands, and detected no differences in avian species richness or abundance. Kahara et al. (2012) compared WRP sites in California to reference wetlands and reported that waterbird use at WRP restorations was similar to or greater than similarly managed natural wetlands in the region.
Few studies have examined factors that affect bird use of WRP and PFWP wetland restorations. Among the four studies of which we are aware, three have focused on the effects of hydrological management on waterbirds. Fleming et al. (2015) and Kahara et al. (2012) concluded that active hydrological management increased the use of restorations by migrating and wintering waterbirds in Mississippi and California, respectively. Similarly, Kaminski et al. (2006) found that active management increased the number of breeding waterbirds at WRP sites in New York. Although we did not characterize the hydrologic regime at restorations, we know that 26 sites were restored without water control structures, and beaver dams had rendered water control structures inaccessible at four sites. Therefore, these sites could not be hydrologically manipulated. Of the 21 sites restored using water control structures, only 5 sites were actively managed by landowners.
A total of 24 bird SGCN were detected at restored wetlands, representing both wetland-associated (n = 12) and upland (n = 12) species. Given the small size of the wetlands and because we surveyed at the water’s edge, we detected both species found in wetlands and those inhabiting the adjacent upland habitat. Depending on the landscape context of the restoration, we detected not only wetland-associated SGCN, but also grassland SGCN [e.g., Bobolink (Dolichonyx oryzivorus), Eastern Meadowlark (Sturnella magna)], shrub-successional SGCN [e.g., Golden-winged Warbler, Brown Thrasher (Toxostoma rufum)], and forest SGCN (e.g., Wood Thrush, Scarlet Tanager). This is important from a management perspective because it indicates that WRP and PFWP wetland restorations have the potential to provide habitat for bird SGCN with diverse habitat requirements. Additionally, many WRP restorations are protected under conservation easements, which often include a substantial area of upland habitat. Therefore, the potential for conserving bird SGCN not traditionally associated with wetlands should be considered during project planning and easement delineation. For example, the Golden-winged Warbler, a SGCN that is currently under review for listing under the Endangered Species Act, was detected at 28% of restored wetlands surveyed, most commonly where the upland habitat surrounding the restoration was shrub-successional or early stage successional forest (Buehler et al. 2007).
Anurans
That we found no difference in the anuran assemblage metrics between restored and reference wetlands is consistent with many other studies, as Brown et al. (2012) reports that amphibian species richness and abundance at restored wetlands was either the same or greater than that at natural reference wetlands in 18 of 22 peer-reviewed studies. For example, Walls et al. (2014) and Beas and Smith (2014) both report that amphibian species richness at WRP and USFWS wetland restorations was higher than or did not differ from that of reference wetlands. Brown et al. (2012) further concludes that the colonization and persistence of amphibian populations at restorations is affected by connectivity to existing wetlands, hydroperiod, and the presence of emergent vegetation. Probably the most important driver of anuran colonization of restored wetlands is wetland connectivity. Restorations, especially those lacking connection to surface water flow, must be within the dispersal range of existing populations and connected by areas of land-uses that suitable for movement for colonization to occur (Semlitsch 2000a; Petranka and Holbrook 2006). Based on the US Fish and Wildlife Service, USFWS (1990) National Wetland Inventory, all restorations surveyed were located within 0.65 km from the nearest wetland, which is within the dispersal range of most pond-breeding amphibians (Semlitsch 2008).
Although we did not characterize the hydrologic regime at sites, we observed that larger, deeper restorations often had surface water year-round, while small depressional wetlands often dried during the late summer and filled again during the fall. Given this, we tended to detect highly aquatic species, such as the American Bullfrog, Green Frog, and Mink Frog (a Potential Species of Conservation Need) at larger restorations. Other studies of amphibian use of wetlands in agricultural landscapes highlight the importance of deep-water habitats. Mushet et al. (2012) found that deep-water overwintering habitat was an important predictor of the occurrence of four of five anuran species examined in wetlands in North Dakota. Similarly, Beas and Smith (2014) compared amphibian assemblages between playa wetlands located in grassland, cropland, and restored watersheds. They found no difference in amphibian species richness during the first survey year when rainfall was abundant, but reported higher species richness at restored playas during their second survey year when there was a drought. They attributed the difference between years to the presence of deep-water habitats at restorations that increase year-round water availability. While highly aquatic species were generally detected at large wetlands, smaller wetlands provided habitat for species that breed in wet meadows, such as the Spring Peeper and Boreal Chorus Frog (a Potential Species of Conservation Need); the three restorations that consisted solely of clusters of small depressional wetlands in a grassland matrix had exceptionally large choruses of these species.
Landowner actions that protect emergent and adjacent upland vegetation may be encouraging anuran occupancy during the breeding season at both restored and reference wetlands. Many landowners indicated that they intended to manage restored wetlands in a manner that enhanced wildlife habitat, which included not altering natural vegetation in and around wetlands. Additionally, the terms of most conservation easements prohibit mowing vegetation adjacent to wetlands, or specify a mowing schedule that is consistent with management goals (e.g., after grassland birds have fledged). Most restorations in our study had well-developed and relatively undisturbed zones of emergent and upland vegetation which are known to enhance anuran breeding habitat (Monello and Wright 1999; Semlitsch 2000b; Semlitsch and Bodie 2003).
Reptiles
Reptiles are not typically surveyed as biological indicator species, so few studies are available for comparison. However, a large number of reptiles native to this region, including several SGCN, are associated with wetlands. There is evidence that reptiles will colonize wetland restorations; Palis (2007) assessed turtle and snake colonization of 3 created wetlands and detected 18 species, including 2 species of conservation concern in Illinois. We detected four turtle species, including two SGCN: the Snapping Turtle and the Blanding’s Turtle. The Snapping Turtle was the most commonly detected species, likely due to the fact that it is a generalist that can be found in a variety of aquatic habitats, including wetlands, streams, rivers, and farm ponds (Paterson et al. 2012; Obbard and Brooks 1981). The Blanding’s Turtle was detected at one restored and one reference site in proximity to known populations that were located within larger wetland complexes, indicating that both restored and natural wetlands on private property have the potential to enhance existing populations of this High Priority SGCN (Stryszowska et al. 2016).
We believe that our study is the first to survey snakes at WRP or PFWP wetlands. Snake species richness and relative abundance were low across sites, due to low success of the visual-encounter, hand-capture sampling technique used. The Eastern Ribbonsnake, a SGCN, was the most commonly detected species at both restored and reference wetlands, likely because the surveys were done at the water’s edge, which is the preferred habitat of this species (Bell et al. 2007); moreover, when disturbed, ribbonsnakes fled into uplands and were more easily captured by observers. The other SGCN snake detected was the Eastern Ratsnake, which is not traditionally associated with wetlands, but instead is most often found in fields and deciduous forest edges (Weatherhead and Charland 1985; Blouin-Demers and Weatherhead 2001). At three restorations and one reference site, representing over half of all ratsnake detections, this species was observed preying on bird eggs (Wood Duck eggs in nest boxes and eggs of ground-nesting waterfowl). Overall, our survey was biased towards larger, more conspicuous snakes and did not yield many captures. We recommend that multiple survey techniques be used to more thoroughly sample snake assemblages in future studies (Ryan et al. 2002; McDairmid et al. 2012).
Fish
To the best of our knowledge, ours is the first study to examine fish assemblages at WRP or PFWP wetland restorations. While there are many studies of fish assemblages at marine and estuarine wetland restorations (e.g., Williams and Zedler 1999; Talley 2000; Gray et al. 2002), there is only one other study that has investigated fish assemblages at palustrine wetland restorations. Similar to our results, Juni and Berry (2001) found no difference in fish species richness between restored and reference wetlands in South Dakota. We detected one fish SGCN, the Black Bullhead, at three restored wetlands. This species is generally associated with low velocity riverine and lacustrine habitats characterized by medium to high cover of aquatic vegetation (Stuber 1982). Each of the restorations where the Black Bullhead was detected were located along larger streams, which likely provided the necessary habitat for this species. Similar to amphibians, we expected that surface water connectivity would be important in determining fish colonization at restorations (Bouvier et al. 2009). We expected isolated restorations to be absent of fish and were surprised to find diverse communities of fish at a number of restorations lacking in surface water connectivity. However, we believe this can be attributed to a number of sources. Many landowners indicated they stocked desirable species at both restored and reference wetlands, including sportfish, such as Brown Bullhead (Ameiurus nebulosus) or Smallmouth Bass (Micropterus dolomieu), and baitfish, such as shiners and minnows (family Cyprinidae). Many restorations were adjacent to streams and other natural wetlands without direct connectivity, and it is plausible that during high rainfall events small fish colonized these wetlands by swimming across flooded uplands (Snodgrass et al. 1996). To quantify inter-annual repeatability, we sampled several sites 2 years in a row, and at one restored site we detected no fish during 2010, but detected two species during 2011, after the region experienced 100-year flood conditions. It is also possible that fish reached isolated restorations via dispersal by waterfowl. Although this is a commonly invoked mechanism of dispersal that is plausible and has been directly observed in other groups of aquatic organisms, there is little quantitative evidence in support of this claim (Figuerola and Green 2002; van Leeuwen et al. 2012). Additionally, restored sites may have contained remnant ponds or drainage ditches with fish prior to restoration. Unfortunately, there is little information, particularly for older sites, on past land-uses or site characteristics prior to restoration. Restoration project managers should be encouraged to record detailed pre-restoration site information, as identifying site-specific characteristics that result in successful restoration should be a priority for future research.
Observations, limitations, and recommendations
The most challenging aspect of this study was locating wetlands to serve as natural reference sites because small, naturally occurring emergent herbaceous wetlands on private land are uncommon in this region. This is likely due to past wetland drainage programs that incentivized wetland conversion, and restoration of these lost wetlands is a principal justification for both WRP and PFWP (Dahl and Allord 1997). Most natural wetlands in this region are much larger than restorations, are dominated by shrub–scrub vegetation [Alder (Alnus sp.) and Willow (Salix spp.)], and are associated with beaver activity. Though restored wetlands differ in structure from the majority of existing natural wetlands, they represent a wetland type that is rare on the landscape. Therefore, it is likely that restorations serve to replace lost functions associated with small wetlands and increase the beta-diversity of the landscape. Our results support this conclusion, as restorations appeared to be the most beneficial, based on detections, for species that live or breed in wet fields or small emergent herbaceous wetlands, including several SGCN [e.g., American Bittern, Sedge Wren, Blue-winged Teal (Anas discors), Boreal Chorus Frog, Eastern Ribbonsnake]. SGCN and other species that are generally restricted to large wetland complexes were rarely detected at both restored and reference wetlands [e.g., Least Bittern (I. exilis), Pied-billed Grebe (P. podiceps), Marsh Wren (C. palustris)]. Additionally, it is worth noting that a majority of the restorations assessed in this study were small and therefore do not qualify for regulatory oversight. For example, the New York regulatory threshold for wetland protection is 5 ha surface area, and 55% of restorations assessed in this study fell under this threshold. So unless these wetlands qualify for protection under the Clean Water Act, they could be converted without permitting and compulsory mitigation.
The greatest limitation of the snapshot survey approach used in our study is that we are uncertain whether the detection of a species indicates a viable population or important meta-population node, versus an undesirable population sink maintained by immigration (Pulliam 1988; Schlaepfer et al. 2002). It is possible that restorations in this region, being imbedded in an agricultural landscape, may be more prone to agrochemical and sediment run-off, vehicle and farm equipment mortality, predation by subsidized predators, reduced food abundance, or poor quality breeding habitat for due to abundant invasive plant cover and increased disturbance (Zedler and Kercher 2005; Verhoeven et al. 2006). Our study was not designed to evaluate population viability, only species presence, and studies on the population viability of representative SGCN species present at restorations would be worthwhile.
There remains considerable uncertainty about how restoration site selection, restoration age, and restoration techniques impact wetland community assembly (Mitsch and Wilson 1996; Brinson and Rheinhardt 1996; Zedler 2000; Rewa 2005; Brinson and Eckles 2011). Although we did not analyze trends in age formally, it is evident that there have been major improvements in restoration techniques since the inception of these programs nearly 30 years ago. Many older restorations we surveyed are pond-like; they are square or circular in shape, of a uniform depth, and dominated by large stands of cattails (Typha spp.). More recently completed wetland restoration projects include features that enhance habitat heterogeneity, including waterfowl nesting islands, greater shoreline complexity, and micro- and macrotopographical elements (e.g., Larkin et al. 2006; Alsfeld et al. 2009; Shulse et al. 2012).
Next steps for improving the success of WRP and PFWP should include identifying best practices in site selection, project design, hydrological management, and other aspects of landowner stewardship of restorations. It is likely that these and other aspects of pre-restoration planning and post-restoration management have a substantial impact on wildlife colonization and persistence at restorations. Additionally, the conservation of wildlife habitat is only one of the ecosystem services that wetland restorations are intended to augment. It would be worthwhile to consider trade-offs and potential positive synergies between wildlife habitat restoration and the restoration of other ecosystem services. Some restorations may be justified as restoring habitat for wildlife, whereas others may be valuable primarily for surface water and ground water management, or sediment, nutrient, and agrochemical sequestration (Zedler 2003; Rey Benayas et al. 2009). Another next step would be to evaluate how wetland restorations change over time in regards to habitat suitability for SGCN and other wetland-associated species. To evaluate trajectories of wetland restoration community assembly, tracking how a cohort of wetlands develops over time is likely to be more informative than a snapshot of restorations of differing ages. Most restorations are young, less than 20 years old, and given that wetlands change over time due to natural self-organizing processes, one might predict that wetland quality from the perspective of biodiversity might improve over time (Mitsch and Wilson 1996; Mitsch et al. 1998). However, in the future landowners and landscapes will also change, and despite the protections offered by conservation easements, it is uncertain whether next-generation landowners will value and manage restorations with the same ardor as those landowners who initially enrolled in these programs. Understanding the dynamics and trajectory of wetland quality over time is critical for evaluating the long-term sustainability of conservation gains from public–private partnership restoration programs (McLaughlin 2005).
Our study focused on wetlands restored voluntarily by landowners in partnership with federal natural resource agencies, and therefore may not be applicable to the many wetlands restorations done as compulsory mitigation under the stipulations of the Clean Water Act. An additional research priority should be to evaluate whether wetland restorations done as compulsory mitigation also contribute to meeting goals of SWAPs, and if not, whether mitigation projects can be modified to make wetlands more valuable from this perspective (Wilkinson et al. 2009).
Conclusions
We conclude that WRP and PFWP wetland restorations in the St. Lawrence Valley of New York provide habitat for SGCN and other wetland-associated wildlife, and that assemblages at restorations are similar to those at natural reference wetlands. Therefore, WRP and PFWP wetland restorations in this region are meeting federal program-level goals related to the restoration of wildlife habitat, and contribute to the recovery goals for SGCN outlined in the New York SWAP. Given the large area of wetlands that have been restored and conserved through these and other public–private partnership conservation programs nationally, the positive impact on SGCN and wetland-associated species is likely to be substantial.
References
Alsfeld AJ, Bowman JL, Deller-Jacobs A (2009) Effects of woody debris, microtopography, and organic matter amendments on the biotic community of constructed depressional wetlands. Biol Conserv 142:247–255. doi:10.1016/j.biocon.2008.10.017
Balcombe CK, Anderson JT, Fortney RH, Kordek WS (2005) Wildlife use of mitigation and reference wetlands in West Virginia. Ecol Eng 25:85–99. doi:10.1016/j.ecoleng.2005.03.003
Beas BJ, Smith LM (2014) Amphibian community responses to playa restoration in the Rainwater Basin. Wetlands 34:1247–1253. doi:10.1007/s13157-014-0584-4
Bell SM, Herman TB, Wassersug RJ (2007) Ecology of Thamnophis sauritus (Eastern Ribbonsnake) at the northern limit of its range. Northeast Nat 14:279–292. doi:10.1656/1092-6194(2007)14[279:EOTSER]2.0.CO;2
Bibby CJ, Burgess ND, Hill DA, Mustoe SH (2000) Bird census techniques, 2nd edn. Academic, Cambridge
Blouin-Demers G, Weatherhead PJ (2001) Habitat use by Black Rat Snakes (Elaphe obsoleta obsoleta) in fragmented forests. Ecology 82(10):2882–2896. doi:10.1890/0012-9658(2001)082[2882:HUBBRS]2.0.CO;2
Bouvier LD, Cottenie K, Doka SE (2009) Aquatic connectivity and fish metacommunities in wetlands of the lower Great Lakes. Can J Fish Aquat Sci 66:933–948. doi:10.1139/f09-050
Brawley AH, Warren RS, Askins RA (1998) Bird use of restoration and reference marshes within the Barn Island Wildlife Management Area, Stonington, Connecticut, USA. Environ Manag 22:625–633. doi:10.1007/s002679900134
Brinson MM, Eckles SD (2011) US Department of Agriculture conservation program and practice effects on wetland ecosystem services: a synthesis. Ecol Appl 21:S116–S127. doi:10.1890/09-0627.1
Brinson MM, Rheinhardt R (1996) The role of reference wetlands in functional assessment and mitigation. Ecol Appl 6:69–76. doi:10.2307/2269553
Brooks RP, Croonquist MJ (1990) Wetland, habitat, and trophic response guilds for wildlife species in Pennsylvania. J Pa Acad Sci 64:93–102
Brown SC (1998) Remnant seed banks and vegetation as predictors of restored marsh vegetation. Can J Bot 76(4):620–629. doi:10.1139/b98-038
Brown SC (1999) Vegetation similarity and avifaunal food value of restored and natural marshes in Northern New York. Restor Ecol 7:56–68. doi:10.1046/j.1526-100X.1999.07107.x
Brown SC, Bedford BL (1997) Restoration of wetland vegetation with transplanted wetland soil. Wetlands 17(3):424–437. doi:10.1007/BF03161432
Brown SC, Smith CR (1998) Breeding season bird use of recently restored versus natural wetlands in New York. J Wildl Manag 62:1480–1491. doi:10.2307/3802014
Brown SC, Smith K, Batzer D (1997) Macroinvertebrate responses to wetland restoration in Northern New York. Environ Entomol 26(5):1016–1024. doi:10.1093/ee/26.5.1016
Brown DJ, Street GM, Nairn RW, Forstner MRJ (2012) A place to call home: amphibian use of created and restored wetlands. Int J Ecol 2012:1–11. doi:10.1057/9781137394354.0013
Buehler DA, Roth AM, Vallender R, Will TC, Confer JL, Canterbury RA, Swarthout SA, Rosenberg KV, Bulluck LP (2007) Status and conservation priorities of golden-winged warbler (Vermivora chrysoptera) in North America. Auk 124(4):1439–1445. doi:10.1642/0004-8038(2007)124[1439:SACPOG]2.0.CO;2
Conway CJ (2009) Standardized North American marshbird monitoring protocols. US Geological Survey and Arizona Cooperative Fish and Wildlife Research Unit Report #2005-04, Arizona, USA
Cornell Lab of Ornithology (2009) Golden-winged Warbler Atlas Project. Cornell Lab of Ornithology. http://www.birds.cornell.edu/gowap. Materials received Jan 2009
Costanza R, de Groot R, Sutton P, van der Ploeg S, Anderson SJ, Kubiszewski I, Farber S, Turner RK (2014) Changes in the global value of ecosystem services. Glob Environ Change 26:152–158. doi:10.1016/j.gloenvcha.2014.04.002
Cowardin LM, Carter V, Golet FC, LaRoe ET (1979) Classification of wetlands and deepwater habitats of the United States. US Fish and Wildlife Service Report No. FWS/OBS/-79/31. Washington, DC
Dahl TE, Allord GJ (1997) History of wetlands in the conterminous United States. United States Geological Survey Water Supply Paper 2425. US Geological Survey. https://water.usgs.gov/nwsum/WSP2425/history.html. Accessed 9 June 2015
Dahl TE, Johnson CE, Frayer WE (1991) Wetlands status and trends in the conterminous United States mid-1970’s to mid-1980’s. US Department of the Interior, Fish and Wildlife Service, Washington, DC. Available via US Fish and Wildlife Service. http://www.fws.gov/wetlands/Documents/Wetlands-Status-and-Trends-in-the-Conterminous-United-States-Mid-1970s-to-Mid-1980s.pdf. Accessed 9 June 2015
Duriancik LF, Bucks D, Dobrowolski JP, Drewes T, Eckles SD, Jolley L, Kellogg RL, Lund DL, Makuch JR, O’Neill MP, Rewa CA, Walbridge MR, Parry R, Weltz MA (2008) The first five years of the Conservation Effects Assessment Project. J Soil Water Conserv 63:185a–297a. doi:10.2489/jswc.63.6.185A
Euliss NH Jr, Smith LM, Shuguang L, Duffy WG, Faulkner SP, Gleason RA, Eckles D (2010) Integrating estimates of ecosystem services from conservation programs and practices into models for decision makers: the vision for CEAP wetlands. Ecol Appl 21:S128–S134. doi:10.1890/09-0285.1
Fairfax SK, Gwin L, King MA, Raymond L, Watt LA (2005) Buying nature: the limits of land acquisition as a conservation strategy, 1780–2004. MIT Press, Cambridge
Ferner JW (2007) A review of marking and individual recognition techniques for amphibians and reptiles. Herpetological Circular No. 35. Society for the Study of Amphibians and Reptiles, Salt Lake City
Figuerola J, Green AJ (2002) Dispersal of aquatic organisms by waterbirds: a review of past research and priorities for future studies. Freshw Biol 47(3):483–494. doi:10.1046/j.1365-2427.2002.00829.x
Filsinger M, Milmoe J (2012) Restore and enhance—the 25th anniversary of the Service’s Partners for Fish and Wildlife Program. US Fish and Wildlife Service. http://www.fws.gov/partners/aboutus.html. Accessed 5 May 2015
Fleming SK, Kaminski RM, Tietjen TE, Schummer ML, Ervin GN, Nelms KD (2015) Vegetative forage quality and moist-soil management on Wetland Reserve Program Lands in Mississippi. Wetlands 32:919–929. doi:10.2007/s13157-012-0325-5
Fry J, Xian G, Jin S, Dewitz J, Homer C, Yang L, Barnes C, Herold N, Wickham J (2011) Completion of the 2006 National Land Cover Database for the conterminous US. Photogramm Eng Remote Sens 77(9):858–864
Ghermandi A, van den Bergh JCJM, Brander LM, de Groot HLF, Nunes PALD (2010) Values of natural and human-made wetlands: a meta-analysis. Water Resour Res 46:w12516. doi:10.1029/2010WR009071
Gibbs JP, Breisch AR, Ducey PK, Johnson G, Behler J, Bothner R (2007) The amphibians and reptiles of New York State: identification, natural history, and conservation. Oxford University Press, Oxford
Gray RL, Teels BM (2006) Wildlife and fish conservation through the Farm Bill. Wildl Soc Bull 34:906–912. doi:10.2193/0091-7648(2006)34[906:WAFCTT]2.0.CO;2
Gray A, Simenstad CA, Bottom DL, Cornwell TJ (2002) Contrasting functional performance of juvenile salmon habitat in recovering wetlands of the Salmon River Estuary, Oregon, USA. Restor Ecol 10:514–526. doi:10.1046/j.1526-100X.2002.01039.x
Heard L (2000, updated 2008) A comprehensive review of Farm Bill contributions to wildlife conservation, 1985–2000. US Department of Agriculture National Resource Conservation Service Technical Report USDA/NRCS/WHMI-2000, Washington, DC
Heyer WR, Donnelly MA, McDiarmid RW, Hayek LC, Foster MS (1994) Measuring and monitoring biological diversity, standard methods for amphibians. Smithsonian Institution Press, Washington, DC
Hoenig JM, Heisey DM (2001) The abuse of power: the pervasive fallacy of power calculations for data analysis. Am Stat 55:1–6
Juni S, Berry CR (2001) A biodiversity assessment of compensatory mitigation wetlands in Eastern South Dakota. Proc S D Acad Sci 80:185–200
Kahara SN, Duffy WG, DiGaudio R, Records R (2012) Climate, management and habitat associations of avian fauna in restored wetlands of California’s Central Valley, USA. Diversity 4:396–418. doi:10.3390/d4040396
Kaminski MR, Baldassarre GA, Pearse AT (2006) Waterbird responses to hydrologic management of Wetlands Reserve Program habitats in New York. Wildl Soc Bull 24:291–296. doi:10.2193/0091-7648(2006)34[921:WRTHMO]2.0.CO;2
Kihslinger RI (2008) Success of wetland mitigation projects. Nat Wetl News 30:14–16
Larkin D, Vivian-Smith G, Zedler JB (2006) Topographic heterogeneity theory and ecological restoration. In: Falk DA, Palmer MA, Zedler JB (eds) Foundations of restoration ecology. Island Press, Washington, DC
Larkin DJ, Madon SP, West JM, Zedler JB (2008) Topographic heterogeneity influences fish use of an experimentally restored tidal marsh. Ecol Appl 18(2):483–496. doi:10.1890/06-1984.1View
Maresch W, Walbridge MR, Kugler D (2008) Enhancing conservation on agricultural landscapes: a new direction for the Conservation Effects Assessment Project. J Soil Water Conserv 63:198a–203a. doi:10.2489/jswc.63.6.198A
McDairmid RW, Foster MS, Guyer C, Gibbons JW, Chernoff N (2012) Reptile biodiversity: standard methods for inventory and monitoring. University of California Press, Berkeley
McGowan KJ, Corwin K (2008) The second atlas of the breeding birds in New York State. Cornell University Press, Ithaca
McLaughlin NA (2005) Rethinking the perpetual natural of conservation easements. Harv Environ Law Rev 29:422–521
Millennium Ecosystem Assessment (2005) Ecosystems and human well-being: synthesis. Island Press, Washington, DC
Mitsch WJ, Gosselink JC (2015) Wetlands, 5th edn. Wiley, San Francisco
Mitsch WJ, Wilson RF (1996) Improving the success of wetland creation and restoration with know-how, time, and self-design. Ecol Appl 6:77–83. doi:10.2307/2269554
Mitsch WJ, Wu X, Nairn RW, Weihe PE, Wang N, Deal R, Boucher CE (1998) Creating and restoring wetlands: a whole-ecosystem experiment in self-design. Bioscience 48:1019–1030. doi:10.2307/1313458
Monello RJ, Wright GR (1999) Amphibian habitat preferences among artificial ponds in the Palouse Region of Northern Idaho. J Herpetol 33(2):298–303. doi:10.2307/1565727
Mushet DM, Euliss NH Jr, Stockwell CA (2012) Mapping anuran habitat suitability to estimate the effects of grassland and wetland conservation programs. Copeia 2:321–330. doi:10.1643/CH-11-119
National Oceanic Atmospheric Administration National Climatic Data Center, NOAA NCDC (2010) Annual/seasonal normals for Canton, NY, subset used: 1981–2010. https://www.ncdc.noaa.gov/cdo-web/datatools/normals. Accessed 2 July 2017
National Research Council, NRC (2001) Compensating for wetland losses under the Clean Water Act. National Academy Press, Washington, DC
Nedland TS, Wolf A, Reed T (2007) A reexamination of restored wetlands in Manitowoc County, Wisconsin. Wetlands 27(4):99–1015. doi:10.1672/0277-5212(2007)27[999:ARORWI]2.0.CO;2
New York State Department of Environmental Conservation, NYSDEC (2005) New York Comprehensive Wildlife Conservation Strategy. http://www.dec.ny.gov/animals/30483.html. Accessed 5 May 2015
NYSDEC (2015) New York State Wildlife Action Plan. http://www.dec.ny.gov/animals/7179.html. Accessed 28 May 2016
O’Neal BJ, Heske EJ, Stafford JD (2008) Waterbird response to wetlands restored through the Conservation Reserve Enhancement Program. J Wildl Manag 72(3):654–664. doi:10.2193/2007-165
Obbard ME, Brooks RJ (1981) A radio-telemetry and mark-recapture study of activity in the common snapping turtle, Chelydra serpentina. Copeia 3:630–637. doi:10.2307/1444568
Palis JG (2007) If you build it, they will come: herpetofaunal colonization of constructed wetlands and adjacent terrestrial habitat in the Cache River drainage of southern Illinois. Trans Ill State Acad Sci 100:177–189
Paterson JE, Steinberg BD, Litzgus JD (2012) Generally specialized or especially general? Habitat selection by Snapping Turtles (Chelydra serpentina) in central Ontario. Can J Zool 90(2):139–149. doi:10.1139/z11-118
Petranka JW, Holbrook CT (2006) Wetland restoration for amphibians: should local sites be designed to support metapopulations or patchy populations? Restor Ecol 14(3):404–411. doi:10.1111/j.1526-100X.2006.00148.x
Pulliam HR (1988) Sources, sinks, and population regulation. Am Nat 132:652–661. doi:10.1086/284880
R Core Team (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org. Accessed 9 June 2015
Ralph CJ, Droege S, Sauer J (1995) Managing and monitoring birds using point counts: standards and applications. US Department of Agriculture Publication PSW/GTR-149-1995. Washington, DC
Ratti JT, Rocklage AM, Giudice JH, Garton EO, Golner DP (2001) Comparison of avian communities on restored and natural wetlands in North and South Dakota. J Wildl Manag 65(4):676–684. doi:10.2307/3803019
Rehm EM, Baldassarre GA (2007) Temporal variation in detection of marsh birds during broadcast of conspecific calls. J Field Ornithol 78:56–63. doi:10.1111/j.1557-9263.2006.00085.x
Reschke C (1990) Ecological communities of New York State. Available via the NY Natural Heritage Program and NY Department of Environmental Conservation. http://www.dec.ny.gov/animals/29389.html. Accessed 5 May 2015
Rewa C (2005) Wildlife benefits of the Wetlands Reserve Program. In: Haufler JB (ed) Fish and wildlife benefits of the Farm Bill conservation programs, 2002–2005 update. The Wildlife Society Technical Review 05-2, Bethesda
Rey Benayas JM, Newton AC, Diaz A, Bullock JM (2009) Enhancement of biodiversity and ecosystem services by ecological restoration: a meta-analysis. Science 325:1121–1124. doi:10.1126/science.1172460
Robinson JA (2000) Follow-up vegetation and avifauna surveys on wetlands restored through USFWS Partners for Wildlife Program. MS Thesis, State University of New York at Brockport
Ryan TJ, Philippi T, Leiden YA, Dorcas ME, Wigley TB, Gibbons JW (2002) Monitoring herpetofauna in a managed forest landscape: effects of habitat types and census techniques. For Ecol Manag 167:83–90. doi:10.1016/S0378-1127(01)00692-2
Schlaepfer MA, Runge MC, Sherman PW (2002) Ecological and evolutionary traps. Trends Ecol Evol 17:474–480. doi:10.1016/s0169-5347(02)02580-6
Scodari PF (1997) Measuring the benefits of federal wetland programs. Island Press, Washington, DC
Semlitsch RD (2000a) Critical elements for biologically based recovery plans of aquatic-breeding amphibians. Conserv Biol 16:619–629. doi:10.1046/j.1523-1739.2002.00512.x
Semlitsch RD (2000b) Principles for management of aquatic breeding amphibians. J Wildl Manag 60(3):615–631. doi:10.2307/3802732
Semlitsch RD (2008) Differentiating migration and dispersal processes for pond-breeding amphibians. J Wildl Manag 72(1):260–267. doi:10.2193/2007-082
Semlitsch RD, Bodie JR (2003) Biological criteria for buffer zones around wetlands and riparian habitats for amphibians and reptiles. Conserv Biol 17(5):1219–1228. doi:10.1046/j.1523-1739.2003.02177.x
Shulse CD, Semlitsch RD, Trauth KM, Gardener JE (2012) Testing wetland restore to increase amphibian reproductive success and species richness for mitigation and restoration. Ecol Appl 22:1675–1688. doi:10.1890/1051-0761-22.5.1675
Snodgrass JW, Bryan AL, Lide RF, Smith GM (1996) Factors affecting the occurrence and structure of fish assemblages in isolated wetlands of the Upper Coastal Plain, USA. Can J Fish Aquat Sci 53:443–454. doi:10.1139/cjfas-53-2-443
Steen DA, Gibbs JP (2005) Effects of roads on the structure of freshwater turtle populations. Conserv Biol 18:1143–1148. doi:10.1111/j.1523-1739.2004.00240.x
Stryszowska KM, Johnson G, Rivera Mendoza L, Langen TA (2016) Species distribution modeling of the threatened Blanding’s Turtle’s (Emydoidea blandingii) range edge as a tool for conservation planning. J Herpetol 50(3):366–373. doi:10.1670/15-089
Stuber RJ (1982) Habitat suitability index models: Black Bullhead. Western Energy and Land-use Team, Office of Biological Services, and US Fish and Wildlife Service Report #FWS/OBS-82/10.14. Available via the US Army Corps of Engineers. http://cdm16021.contentdm.oclc.org/cdm/ref/collection/p16021coll3/id/252. Accessed 9 June 2015
Talley DM (2000) Ichthyofaunal utilization of newly-created versus natural salt marsh creeks in Mission Bay, California. Wetl Ecol Manag 8:117–132. doi:10.1023/A:1008436301041
US Department of Agriculture Natural Resource Conservation Service, USDA NRCS (2014a) Wetlands Reserve Program. http://www.nrcs.usda.gov/wps/portal/nrcs/main/national/programs/easements/wetlands/. Accessed 5 May 2015
US Fish and Wildlife Service, USFWS (1990) National Wetland Inventory. http://www.fws.gov/wetlands/Data/Data-Download.html. Accessed 5 May 2015
USDA NRCS (2014b) Farm Bill 2014 Agricultural Conservation Easement Program. http://www.nrcs.usda.gov/wps/portal/nrcs/detail/national/programs/easements/acep/?cid=stelprdb1242695. Accessed 5 May 2015
USFWS (2006) St. Lawrence Wetland and Grassland Management District. http://www.fws.gov/northeast/facts/StLawrence06.pdf. Accessed 9 July 2015
USFWS (2015a) Partners in Fish and Wildlife Program. http://www.fws.gov/partners/. Accessed 5 May 2015
USFWS (2015b) State Wildlife Grant Program Overview. http://wsfrprograms.fws.gov/Subpages/GrantPrograms/SWG/SWG.htm. Accessed 9 July 2015
van Leeuwen CHA, van der Velde G, van Lith B, Klaassen M (2012) Experimental quantification of long distance dispersal potential of aquatic snails in the gut of migratory birds. PLoS ONE 7(3):e32292. doi:10.1371/journal.pone.0032292
Verhoeven JTA, Arheimer B, Chengqing Y, Hefting MM (2006) Regional and global concerns over wetlands and water quality. Trends Ecol Evol 21:96–103. doi:10.1016/j.tree.2005.11.015
Walls SC, Waddle JH, Faulkner SP (2014) Wetland Reserve Program enhances site occupancy and species richness in assemblages of anuran amphibians in the Mississippi Alluvial Valley, USA. Wetlands 34:197–207. doi:10.1007/s13157-013-0498-6
Weatherhead PJ, Charland MB (1985) Habitat selection in an Ontario population of the snake, Elaphe obsoleta. J Herpetol 19(1):12–19. doi:10.2307/1564415
Weir LA, Mossman MJ (2005) North American Amphibian Monitoring Program (NAAMP). In: Lannoo MJ (ed) Amphibian declines: conservation status of United States species. University of California Press, Berkeley
Werner R (2004) Freshwater fishes of the northeastern United States: a field guide. Syracuse University Press, Syracuse
Wilkinson JB, McElfish Jr JM, Kihslinger R, Bendick R, McKenney A (2009) The next generation of mitigation: linking current and future mitigation programs with State Wildlife Action Plans and other state and regional plans. The Nature Conservancy and Environmental Law Institute Report. Available via the Environmental Law Institute. http://www.eli.org/sites/default/files/eli-pubs/d19.08executive_summary.pdf. Accessed 9 June 2015
Williams GD, Zedler JB (1999) Fish assemblage composition in constructed and natural tidal marshes of San Diego Bay: relative influence of channel morphology and restoration history. Estuaries 22:702–716. doi:10.2307/1353057
Zedler JB (2000) Progress in wetland restoration ecology. Trends Ecol Evol 15:402–407. doi:10.1016/S0169-5347(00)01959-5
Zedler JB (2003) Wetlands at your service: reducing impacts of agriculture at the watershed scale. Front Ecol Environ 1:65–72. doi:10.1890/1540-9295(2003)001[0065:WAYSRI]2.0.CO;2
Zedler JB, Kercher S (2005) Wetland resources: status, trends, ecosystem services, and restorability. Annu Rev Environ Resour 30:39–74. doi:10.1146/annurev.energy.30.050504.144248
Acknowledgements
Funding in support of this Project was provided by New York State Wildlife Grant #T-9-2, Value of Wetland Restoration Incentive Programs on Privately Owned Land for Species of Greatest Conservation Need, and University of Michigan Water Center Grant Environmental and Socioeconomic Factors Associated with Public–Private Partnership Wetland Restoration Projects Benefiting Wildlife in the Great Lakes Watershed. First, we would like to thank all of the landowners that participated in this study. Without your cooperation, patience, and dedication to the stewardship of our natural resources, this work would not have been possible. We would also like to thank our partners including: Angelena Ross (New York Department of Environmental Conservation); Jim Pullano and Kimberley Farrell (USDA NRCS); Gian Dodici, Carl Schwartz, and Tom Jasikoff (USFWS); Glenn Johnson (SUNY Potsdam); and all of the folks that have helped along the way: Kinga Stryszowska, Cody Merrill, Jayson Hajek, Maria Hargis, Robyn Andrusyszyn, Eric Marcy, Kallen Frey, Brittany Guarna, Laura Barlow, Jeremy Ozolins, Nychele Carley, John Sherry, Felix Grimberg, and Matthew Valente. Survey methods were approved by the Clarkson University IACUC and conducted under permit from the New York State Department of Environmental Conservation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Benson, C.E., Carberry, B. & Langen, T.A. Public–private partnership wetland restoration programs benefit Species of Greatest Conservation Need and other wetland-associated wildlife. Wetlands Ecol Manage 26, 195–211 (2018). https://doi.org/10.1007/s11273-017-9565-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-017-9565-8