Abstract
The Yangtze River delta is characterized by rapidly accreting sediments that form tidal flats that are quickly colonized by emergent vegetation including Scirpus mariqueter and the invasive species Spartina alterniflora. We measured soil surface elevation, water table depth, soil salinity, water content and compaction in the tidal flat, the Scirpus and Spartina zones and their borders to identify relationships between environmental factors and colonization by Scirpus and Spartina. With increasing elevation from tidal flat to Spartina, inundation frequency and duration, moisture and depth to water table decreased whereas soil salinity, temperature and compaction increased. High soil moisture and groundwater and low salinity were the characteristics of the tidal flat and its border with Scirpus. The Spartina zone and its border with Scirpus were characterized by greater salinity and elevation relative to the other zones. Our findings suggest that soil salinity controls patterns of plant zonation in the newly formed tidal salt marshes whereas elevation is of secondary importance. Our results suggest that patterns of vegetation zonation in tidal marshes of the Yangtze River delta are controlled by environmental factors, especially (low) salinity that favors colonization by Scirpus in the lower elevations of the marsh.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Since the 20th century, ecologists have studied the relationship between environment factors and the distribution of vegetation communities of salt marshes (Simas et al. 2001; Caçador et al. 2007). Salt marshes have attracted extensive attention from plant ecologists because of their low species diversity, simple community structure and striking zonation (Levine et al. 1998).
The distribution of salt marsh vegetation is related to energy and material inputs (i.e. tidal action, soil nutrients), and to the constraints of physical factors (such as flooding, anoxia, salinity, etc.). Tides play an important role in the development of estuarine salt marshes with zonal distribution of plant communities along the gradient of elevation from open water to uplands (Vince and Snow 1984). These zones are determined by differences in elevation that affect inundation and salinity. Most studies to date have focused on the role of abiotic factors in plant zonal pattern, especially salinity and flooding (Pennings et al. 2005; Silvestri et al. 2005).
The mechanisms controlling vegetation zonation in coastal salt marshes may be universal though the importance of individual factors may vary geographically (Pennings et al. 2005; Castillo et al. 2008). One region that is understudied is the tidal marshes of East Asia, including the Yangtze and Yellow River deltas. The eastern headlands of Chongming Island is an important salt marsh of the Yangtze River delta. It contains abundant resources of zoobenthos and vegetation and is a stopover for migrating waterfowls across the Asia–Pacific region (Huang et al. 1993).
In recent years, invasion of salt marsh communities by introduced species has become an important issue of tidal marsh research in the region and elsewhere (Daehler and Strong 1996; Zedler and Kereher 2004). Spartina alterniflora, a salt marsh species native to the east coast of the US, was introduced to China for sediment stabilization in 1979 and spread to Chongming Island by 1995. Because of its adaptability to high salinity and anoxia, Spartina began to colonize the tidal flats and began to displace native species, Scirpus mariqueter and Phragmites australis. It is considered a threat to benthos and birds which use Scirpus as their primary habitat (Yong and Zhang 1992). To date, no studies have focused on the spatial patterns of plant communities, including the invasive species Spartina, and environmental factors in rapidly accreting tidal flats and newly colonizing tidal marshes of the Yangtze River delta.
We measured soil salinity, surface elevation, soil moisture, groundwater, soil compaction, soil temperature and inundation in the mudflat and the stands of Scirpus, Spartina and the transition zones to understand the role of environment factors in Spartina invasion in the Yangtze River delta. Our goal was to identify (1) the environmental factors that structure vegetation zonation in the Yangtze Estuary tidal marshes, and (2) the conditions that favor Spartina invasion into this habitat.
Materials and methods
Study site
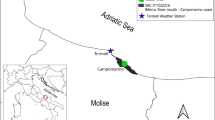
The study site is located at the eastern end of Chongming Island (121°45′E, 31°30′N) in the Yangtze River estuary (Fig. 1). The wetland was established as a national nature reserve in 1992 and was designated as a Ramsar Site in 2001. It has a subtropical monsoon climate, with mean annual sunshine hours of 2138 h, frostless period of 229 days, and average annual temperature of 15.3°C. July and August are the warmest months with monthly average temperature of 26.8°C. The coldest months are January and February with a mean temperature of 3.0°C. Mean annual precipitation ranges from 900 to 1,050 mm, most of which falls from April to September.
The study site has an irregular semidiurnal tide with mean tidal range of 2.43 and 3.08 m (Fig. 2). The soil texture is loamy with finer materials at the higher elevation of the marsh (Salt content of the soil ranges from 0.2 to 0.6%).
The native vegetation consists of Scirpus mariqueter and Phragmites australis. Scirpus is an herbaceous perennial species and reproduces both sexually and asexually though, like many salt marsh species, it spreads mostly by clonal propagation. Since 1995, when Spartina was first reported in marshes of the eastern headlands, Spartina gradually colonized and spread into habitats of the native species. Today, a mosaic pattern of the invasive and native species exists in the marshes with sharp boundaries delineating the different communities.
Experimental design
We established three transects, each perpendicular to the levee and each separated by 200 m. Along each transect, three smaller subtransects were established to transit through the tidal flat, the Scirpus zone and the Spartina zone to sample four vegetated and one unvegetated habitats; tidal flat, tidal flat-Scirpus edge, Scirpus zone, Spartina-Scirpus edge zone and Spartina zone. At each location, three replicate measurements of soil compaction, salinity, temperature and water content, water table depth and elevation were made. Measurements were made four times between May and October, 2008, during period of plant emergence (May), growth (July), flowering (September) and senescence (October). All measurements were made during neap tide phase.
Soil moisture, salinity and temperature were measured 10 cm below the surface using a Hydra soil moisture sensor (including Hydra Data Reader and Hydra Probe II Soil Moisture Sensor (SDI-12/RS485)) (Precision: Moisture, ±0.5% vol; Salinity, ±2%; temperature, ±0.6°C); Stevens Water Monitoring Systems Inc., Australia). Soil compaction was measured using a TJSD-750 soil compaction device (Precision: ± 0.05 kg; Hangzhou Tuopu Ltd. China). Surface elevation was measured using GPS-RTK (Z-Xtreme) (Precision: <1 cm for static GPS, <5 cm for kinematic GPS, cm-dm accuracies for baselines <5 km for kinematic; AshtechTM, USA). The elevations were tied into the nearby Dongtan benchmark site (121°57′, 31°30′), part of the Chinese geodetic level. We augered a hole to depth of 1.5 m to measure water table depth. Once the water table stabilized in the hole, we measured the water table depth from the soil surface.
We use the following formula to calculate the flooding depth of different vegetation zones:
where I represents flooding depth, r represents the changing range of the tide in this region each day (determined from the tide table of the eastern headlands in Chongming island, the Sheshan tide-gauge station (122°14′, 31°25′), adjacent to our study site) and h represents the mean elevation of each vegetation zone.
Statistical analysis
We used analysis of variance (ANOVA) to test for differences in surface elevation and soil properties among the vegetation zones (SPSS 1999). Means were compared using the least significant difference (LSD) test. Principal components analysis (PCA) was used to explore associations between the environmental properties and vegetation zones. We used PC-ORD 5.0 (McCune and Mefford 1999) for PCA using centered and standardized species-environmental variables. According to Jogman et al. (1995), the data were square root-transformed prior to analysis. All tests of significance were conducted at P of 0.05.
Results
Variation of flooding properties among different zones
The tidal cycle of the eastern headlands is characterized by irregular semidiurnal tides (Fig. 2). Combining the tide gauge data with our elevation data, we estimated monthly changes of the flooding duration and depth for the five zones (Fig. 3). One-way ANOVA analysis showed that the depth and duration of flooding in the tidal flat and the tidal flat-Scirpus edge were significantly (P < 0.05) greater than in the other zones and the daily maximum flooding depth was about 60 cm.
Mean monthly flooding depth in the Scirpus zone was greater than in the Spartina zone. Flooding duration in the Scirpus zone also was longer than in the Spartina zone. Both flooding depth and duration were less than the two vegetated zones relative to the un-vegetated mudflat (Fig. 4). The tidal flat was flooded every day and, on most days, it was flooded to a depth of 50 cm or more (Fig. 4). The depth of flooding was shallower in the Scirpus zone than that in the tidal flat, around 50 cm. And the depth of flooding was less than 40 cm in the Spartina zone.
Inundation depth versus frequency in the tidal flat (TF), tidal flat-Scirpus edge (ESM), Scirpus mariqueter (SM), Scirpus-Spartina edge (ESS) and Spartina alterniflora (SA) zones. The density probability curve simulates the trend of frequency (days) versus depth of inundation. The inundation depth and frequency in the Spartina zone are significantly lower than those in Scirpus and tidal flat zone (P < 0.05)
Plots of flooding frequency versus inundation depth (Fig. 4) showed that the Spartina zone experienced more than 100 days with no flooding. In this zone, the maximum flooding depth was 160 cm. In the Scirpus zone, the number of days with no flooding was similar to the Spartina zone though the maximum depth of flooding was greater, about 180 cm (Fig. 4). The tidal flat was inundated longer and to a greater depth than the vegetated zones. In this zone, depth of flooding was 50 cm or more for more than 100 days and the maximum flooding depth was more than 200 cm. The Spartina-Scirpus edge had the lowest duration and depth of inundation of the zones sampled (Fig. 4).
Environmental factors and vegetation zones
Surface elevation and soil properties varied among the different vegetation zones (P < 0.05) as well. Soil surface elevation was greater in the vegetated zones than that in the tidal flat (Fig. 5a). Surface elevation averaged 2.74 m in the tidal flat, 2.95 m in the Scirpus zone and 3.15 m in the Spartina zone. Salinity exhibited a similar trend; it was the lowest in the tidal flat, moderate in the Scirpus zone and the greatest in the Spartina zone (Fig. 5b). Soil water content displayed the opposite trend as it was the greatest in the tidal flat and decreased landward (Fig. 5c). Water table depth, soil compaction and temperature followed the same pattern as elevation, increasing from the tidal flat to the vegetated zones (Fig. 5d, e, f). The Spartina-Scirpus edge had the highest elevation, and greatest depth to water table, soil compaction and temperature.
a Mean surface elevation, b soil salinity, c soil water content, d water table depth, e soil compaction and f temperature (±standard error) in the tidal flat (TF), tidal flat-Scirpus edge (ESM), Scirpus mariqueter (SM), Scirpus-Spartina edge (ESS) and Spartina alterniflora (SA) zones. Means separated by the same letter are not significantly different (P < 0.05) according to the least significant difference (LSD) test
Principle components analysis
Principal components analysis revealed gradients associated with soil salinity, water content and water table depth on the first axis and soil compaction on the second axis (Fig. 6). The first two axes explain 82% of the variation in the data. Tidal flat sites were clustered in areas of high soil water content and water table depth, and low salinity (Fig. 6). Spartina sites were clustered in areas of high soil salinity and compaction. Scirpus sites were intermediate with respect to soil moisture and salinity (Fig. 6) whereas Scirpus-Spartina zone sampling points were clustered in areas of high soil compaction.
Discussion
Expansion of Spartina and the corresponding decline of Scirpus coverage in tidal marshes of the eastern headlands in recent years may be attributed to several factors: (1) Reduced freshwater discharge by the Yangtze River since 2000 (Zhang R et al. 2006) and enhanced saltwater intrusion from the rising sea levels have led to an increase in surface soil salinity in the salt marshes of the region (Day et al. 1995) that made it difficult for Scirpus to colonize newly formed mudflats and (2) Seaward colonization of tidal flats by salt marsh vegetation caused by sediment deposition that increased surface elevation and salinity. Thus, the increase in salinity in recently colonized Spartina zones, in part, may have facilitated the invasion by Spartina. Furthermore, Scirpus reproduction and growth is more intimately tied to freshwater pulsing than Spartina. Li et al. (2010) reported that salinity in the Yangtze estuary is the lowest from May to October which is consistent with the life cycle of Scirpus that is completed during this period. For example, emergence of Scirpus seedling occurs in May, followed by growth in summer, then by plant senescence in October. Spartina, in contrast, continues to grow throughout the year by producing clonal ramets (Livingstone and Patriquin 1981).
Laboratory studies also support Spartina’s greater tolerance to salinity. For example, Scirpus dies at surface water salinities of 1.6% or greater, whereas Spartina tolerates salinities up to 3.2% (Chen et al. 2005). Landin (1991) found that the optimum salinity for the growth of Spartina was 1–2%.
Elevation is also important in structuring patterns of plant zonation in tidal marshes (Sánchez et al. 1996; Boorman et al. 2001; Bockelmann et al. 2002) as it is linked to surface inundation, salinity, soil aeration and other soil properties (Snow and Vince 1984; Adam 1990). In addition to soil salinity, water table depth, compaction and temperature all increased with elevation in our study (Fig. 5). Also, as surface elevation increased, flooding frequency decreased (Fig. 3). Although we observed differences in inundation patterns between Scirpus and Spartina, there was no significant difference in elevation between the two zones (Fig. 5). In addition to larger scale variation in environmental properties along our elevation gradient, there probably are smaller gradients of soil salinity, water content and ground water depth caused by differences in microtopography and other factors (Pennings et al. 2005), including plant species composition.
Principal components analysis also showed that vegetation zonation was strongly linked to soil moisture, salinity, compaction and water table depth (Fig. 6). Longer and deeper inundation and low salinity were characteristics of the tidal flat and tidal flat-Scirpus edge. In contrast, high soil salinity and compaction were characteristics of the Spartina and Spartina-Scirpus edge zone. In addition to salinity and soil moisture, we found that soil temperature differed among Spartina and Scirpus zones, with higher temperature in the Spartina zone. We hypothesize that higher temperature may be attributed to reduced duration and depth of inundation and soil moisture in the Spartina zone.
Differences in plant reproduction strategies also may play an important role in the distribution of tidal marsh vegetation (van Zandt et al. 2003). Clonal reproduction is the main mechanism of reproduction by Spartina in the Yangtze River delta with colonization rates of up to 32 cm in one year (Zhang D et al. 2006). Zhi et al. (2007) also found that, on newly colonized sites, Spartina produces about 53,748 seeds per square meter, of which 70–80% germinate.
In contrast, Scirpus spreads via rhizomes at about one half the rates of Spartina (15 cm one year). Like Spartina, Scirpus also produces considerable numbers of seeds (15–18 per ramet with 2,580 ramets per square meter) that have similar germination rates of 73.8%. Thus, it appears that Spartina’s success may be due more to its rate of clonal spread than its seed production and germination.
In summary, environmental factors, especially salinity, structure plant communities of tidal marshes of the Yangtze River delta. Furthermore, increased salinity appears to favor the spread of Spartina into habitats historically occupied by Scirpus. Other environmental factors, soil moisture and compaction, as well as biological factors, especially rhizome growth also contributes to Spartina’s ability to colonize tidal flat habitat in the delta.
References
Adam P (1990) Saltmarsh ecology. Cambridge University Press, Cambridge, p 461
Bockelmann AC, Bakker JP, Neuhaus R, Lage J (2002) The relation between vegetation zonation, elevation and inundation frequency in a Wadden Sea salt marsh. Aquat Bot 73:211–221
Boorman LA, Hazelden J, Boorman M (2001) The effect of rates of sedimentation and tidal submersion regimes on the growth of salt marsh plants. Cont Shelf Res 21:2155–2165
Caçador I, Tibério S, Cabral HN (2007) Species zonation in Corroios salt marsh in the Tagus estuary (Portugal) and its dynamics in the past fifty years. Hydrobiologia 587:205–211
Castillo JM, Mateos-Naranjo E, Nieva FJ, Figueroa E (2008) Plant zonation at salt marshes of the endangered cordgrass Spartina maritima invaded by Spartina densiflora. Hydrobiologia 614:363–371
Chen ZY, Li B, Chen JK (2005) The effect of salinity and elevation to the growth of Spartina alterniflora. J Yangtze Univ (Nat sci) 2(2):6–9 (in Chinese with English abstract)
Daehler CC, Strong DR (1996) Status, prediction and prevention of introduced cordgrass Spartina spp. invasions in Pacific estuaries, USA. Biol Conserv 78(1):51–58
Day JW, Pont D, Hensel PF, Ibańez C (1995) Impacts of sea-level rise on deltas in the Gulf of Mexico and the Mediterranean: the importance of pulsing events to sustainability. Estuaries 18(4):636–647
Huang ZY, Sun ZH, Yu K (1993) Shanghai birds’ resources and their habitat. Fudan University Press, Shanghai, p 175 (in Chinese)
Jogman RHG, ter Braak CJF, van Tongeren OFR (1995) Data analysis in community and landscape ecology. Pudoc, Wageningen, The Netherlands
Landin M C (1991) Growth habits and other considerations of smooth cordgrass Spartina alterniflora Loisel [A]. In: Mumford TF Jr, Peyton P, Sayce JR et al. (eds) Spartina workshop record [C], Washington Sea Grant Program, University of Washington, Seattle, pp 115–201
Levine JM, Brewer JS, Bertness MD (1998) Nutrients, competition and plant zonation in a New England salt marsh. J Ecol 86:285–292
Li Q, An CG, Ma Q, Xu LL, Zhao YL (2010) Species composition and diversity of zooplankton in tidal creeks of the Chongming Dongtan intertidal flat. Biodiversity Science 18(1):67–75 (in Chinese with English abstract)
Livingstone DC, Patriquin DG (1981) Belowground growth of Spartina alterniflora Loisel, habit, functional biomass and non-structural carbohydrates. Estuar Coast Shelf Sci 12(5):579–587
McCune B, Mefford MJ (1999) PC-ORD. Multivariate analysis of ecological data, Version 4. MjM Software Design, Gleneden Beach, OR, USA, p 237
Pennings SC, Grant MB, Bertness MB (2005) Plant zonation in low-latitude salt marshes: disentangling the roles of flooding, salinity and competition. J Ecol 93:159–167
Sánchez JM, Izco J, Medrano M (1996) Relationships between vegetation zonation and altitude in a salt-marsh system in northwest Spain. J Veg Sci 7:695–702
Silvestri S, Defina A, Marco Marani M (2005) Tidal regime, salinity and salt marsh plant zonation. Estuar Coast Shelf Sci 62:119–130
Simas T, Nunes JP, Ferreira JG (2001) Effects of global climate change on coastal salt marshes. Ecol Model 139:1–15
Snow AA, Vince SW (1984) Plant zonation in an Alaskan salt marsh II: an experimental study of the role of edaphic conditions. J Ecol 72:669–684
SPSS (1999) SPSS 10.0.1 for Windows. SPSS Inc, Chicago, IL
van Zandt PA, Tobler MA, Mouton E, Hasenstein KH, Mopper S (2003) Positive and negative consequences of salinity stress for the growth and reproduction of the clonal plant, Iris hexagona. J Ecol 91:837–846
Vince SW, Snow AA (1984) Plant zonation in an Alaskan salt marsh I: distribution, abundance and environmental factors. J Ecol 72:651–667
Yong XK, Zhang LQ (1992) Studies on reproduction ecology of Scirpus Mariqueter population. J East China Norm Univ (Nat Sci) 4:94–99 (in Chinese with English abstract)
Zedler JB, Kereher S (2004) Causes and consequences of invasive plants in wetlands: opportunities, opportunists, and outcomes. Crit Rev Plant Sci 23:431–452
Zhang D, Yang MM, Li JX, Chen XY (2006) Vegetative dispersal ability of Spartina alterniflora in eastern end of Chongming Island. J East China Norm Univ (Nat Sci) 2:130–135 (in Chinese with English abstract)
Zhang R, Wang YP, Pan SM (2006) Analyses with Wavelet and Hilbert-Huang transform on monthly water discharges at Datong Station, Yangtze River. J Nanjing Univ (Nat Sci) 42(4):423–434 (In Chinese with English abstract)
Zhi YB, Li HL, An SQ (2007) Inter-specific competition: Spartina alterniflora is replacing Spartina anglica in coastal China. Estuar Coast Shelf Sci 74:437–448
Acknowledgments
We thank Wenyong Guo, Zaijun Xin and Yue Jia for their assistance with the collection of field data. Our research was supported by the 111 Project (B08022); Ministry of Education, China; Programme Strategic Scientific Alliances between China and the Netherlands (2008DFB90240); FP7.2009-1 (Contract 244104—THESEUS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
He, Y., Li, X., Craft, C. et al. Relationships between vegetation zonation and environmental factors in newly formed tidal marshes of the Yangtze River estuary. Wetlands Ecol Manage 19, 341–349 (2011). https://doi.org/10.1007/s11273-011-9220-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-011-9220-8