Abstract
The selection of emergent plants is of primary importance during the construction of floating treatment wetlands (FTWs). Focusing on the comparison of the nitrogen removal, pot-culture experiments were carried out in floating treatment wetlands constructed with Phragmites australis and Acorus calamus in northeast China, a cold temperate zone. Nitrogen removal and transformation processes were investigated to explore the pathways and factors that influence the nitrogen removal. FTWs showed a high capacity for nitrogen removal. In water with TN concentrations of 9.63 and 4.58 mg L−1, the average TN removal efficiencies of the FTWs constructed with P. australis were 91.5 and 84.2%, respectively, and those of FTWs constructed with A. calamus were 84.2 and 82.8%, respectively. Plant uptake accounted for 36.4 to 77.1% of total N removal. The average TN removal rates of P. australis systems in the first 2 days were 4.20 and 1.77 mg L−1 day−1 for treatments with TN concentrations of 9.36 and 4.58 mg L−1, respectively, significantly higher than those of the A. calamus system, which were 1.75 and 1.04 mg L−1 day−1, respectively. Our results suggested that plant uptake was the main pathway for nitrogen removal in FTWs, and P. australis was a suitable emergent plant species for use in FTW construction in a cold temperate zone.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The eutrophication of surface water has been a serious environmental issue for several decades (Zhao et al. 2012). In China, more than 66 and 22% of the lakes are eutrophic or hypereutrophic, respectively. The large quantities of inorganic nutrients in water bodies, particularly nitrogen (N) and phosphors (P), established the material basis of the eutrophication of surface water (Li et al. 2015). Hence, the removal of N and P has been an urgent task in the remediation of surface water faced with eutrophication (Groh et al. 2015; Headley and Tanner 2012; Li et al. 2015; Lu et al. 2015; Lynch et al. 2015).
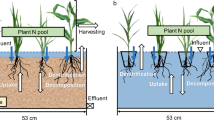
Floating treatment wetlands (FTW) function as a hydroponic system that provides a growing medium for emergent vegetation and microbes, which remove nutrients and/or pollutants from the water (Winston et al. 2013; Zhou et al. 2012). It has been proposed as an in situ remediation technology with high adaptability and low cost on both foundation and land use (Ding et al. 2012; Weragoda et al. 2012). Emergent plants are considered the most important part of the FTW (Pavan et al. 2015; Weragoda et al. 2012). These plants might adsorb N from the water directly, and they are also involved in the indirect removal of N by providing and/or improving the habitat for microbes (Gao et al. 2012; Wang et al. 2014; Zhang et al. 2014). Plenty of studies have been done on the screening of emergent vegetation for constructed wetlands; however, most of this research focused on constructed wetlands rather than FTW (Li et al. 2015; Pavan et al. 2015). Moreover, in consideration of the regional dependence of the emergent plants and the prevention of biological invasion, indigenous plants should be the priority in the construction of FTW. Phragmites australis and Acorus calamus are widely distributed in northeast China. Furthermore, they are of high biomass and widely used in constructed wetlands, which indicated that they might perform well in FTW. Hence, the present study aimed to (1) comparing N removal capacity of FTWs constructed with P. australis and A. calamus in northeastern China and (2) analyzing the relationship between macrophyte and the removal of N.
2 Materials and Methods
2.1 Experimental Design
Pot-cultivar experiments were carried out at the Khanka Lake Wetland Station, Chinese Academy of Sciences. The water temperature was between 15 and 26 °C during the experimental period. Fifteen liters of water was poured into a plastic drum (φ × H = 33 cm × 34 cm), and a plastic foam board (W × L × H = 25 cm × 25 cm × 4 cm), on which five holes (φ = 3 cm) were dug, was set in the pot. P. australis and A. calamus were used in the construction of the FTW. All of the P. australis and A. calamus were wild and collected near Khanka Lake. Uniform seedlings were picked out and fixed with spongia in the holes. One seedling was planted in each hole. The fresh and dry weight of the roots and shoots of the seedling were measured, and the concentration of N in the plant samples was also determined.
Two treatments with different N concentrations (marked with T1 and T2) were arranged in the experiment. A certain amount of NH4NO3 was introduced into the water collected from Khanka Lake to obtain the concentration of N necessary for the experiment. The initial concentrations of N are listed in Table 1. The experiment was arranged in mid-May. After the seedling recovered for 15 days, seven 10-day cycles were implemented. All of the experiments were carried out in triplicate, and a treatment without plants (marked with CK) was also arranged.
2.2 Sample Preparation and Analysis
At the first and last day of each cycle, water samples were collected and analyzed for the pH values and the concentrations of total nitrogen (TN), ammonia nitrogen (NH4 +-N), and nitrate (NO3 −-N). To further explore the nitrogen removal process, more water samples were collected on days 2, 4, and 7 during the third, fifth, and seventh cycles. After the experiment, all of the plants were harvested and separated into shoots and roots. The fresh and dry weights and the N concentrations of the plant samples were measured.
For the water samples, the concentrations of TN, NH4 +-N, and NO3 −-N were detected using alkaline potassium persulfate digestion UV spectrophotometric method (HJ 636-2012), Nessler’s reagent colorimetric method (GB7479-87), and the UV spectrophotometric method (HJ 346–07), respectively. The plant samples were digested by H2SO4-H2O2 and used the salicylic acid spectrophotometric method.
2.3 Data Processing
Data processing was carried out using Excel 2007 (Microsoft, USA) and SPSS18.0 (SPSS Corporation, USA). The Kolmogorov-Smirnov test was applied to verify whether the distribution of the variables was normal. A significance analysis was performed using a one-way ANOVA (LSD test and Student-Newman-Keuls test).
3 Results
3.1 Plant Growth
Plant heights and the relative growth rate of plant heights were used to describe the growth condition of P. australis and A. calamus. The relative growth rate (RGR) is defined in Eq. (1):
in which H 1 and H 2 are the average plant heights at t 1 and t 2.
The changes in plant heights during the experimental period and the RGR values are shown in Fig. 1. The height of A. calamus grew much faster in the initial cycles (with RGR >0.020 day−1) and then decreased sharply. In the last two cycles, the RGR values approach to zero. For P. australis, the RGR values were 0.008–0.025 day−1 in the T1 treatment, except for the last cycle, and 0.004–0.011 day−1 in the T2 treatment, suggesting that the growth of P. australis was much steadier than A. calamus during the whole experimental period. The heights of plants in the treatment T1, which had higher N concentrations, increased more quickly than in the treatment T2. This might be caused by the greater nutrient supplement in the T1 treatment. At the end of the experiment, the height of P. australis and A. calamus were 66.2 and 57.3 cm in the T1 treatment and 53.7 and 41.1 cm in the T2 treatment, respectively. Significant differences (p < 0.05, LSD test) were found among both treatments and plant species.
The biomass of P. australis and A. calamus are depicted in Table 2. The total fresh biomass of P. australis and A. calamus were 130.12 ± 10.91 and 136.24 ± 18.01 g/pot in fresh weight (g FW/pot) under the T1 treatment, and 82.85 ± 5.84 and 63.86 ± 13.66 g FW/pot under the T2 treatment, respectively. Significant decreases (p < 0.05, LSD test) in both the total fresh biomass and the total dry biomass were found in the T2 treatments when compared with the T1 treatment. Moreover, the biomass of A. calamus decreased more sharply than that of P. australis. The biomass of P. australis in the T2 treatment was 63.67% in fresh weight and 67.82% in dry weight of those in the T1 treatment, and those values were only 46.87 and 47.57% for A. calamus. In the same treatment, there were no significant differences (p > 0.05, LSD test) in the total fresh biomass between P. australis and A. calamus.
For shoot biomass and root biomass, the situation was slightly different. The shoot biomass of P. australis was 59.53 ± 5.68 g FW/pot and 27.83 ± 3.89 g DW/pot under the T1 treatment, and 32.09 ± 5.20 g FW/pot and 18.23 ± 2.78 g DW/pot under the T2 treatment. For A. calamus, these data were 46.88 ± 10.68 g FW/pot and 12.45 ± 4.39 g DW/pot under the T1 treatment and 18.90 ± 3.75 g FW/pot and 5.56 ± 0.68 g DW/pot under the T2 treatment. The shoot biomass of P. australis were significantly higher (p < 0.05, LSD test) than that of A. calamus, and the shoot biomass in the T1 treatment was also higher than that in the T2 treatment for both macrophytes. The differences in root biomass of P. australis and A. calamus were smaller than those of the shoot biomass. For P. australis, the root biomass was 70.58 ± 5.95 g FW/pot and 27.95 ± 1.87 g DW/pot and 50.76 ± 5.26 g FW/pot and 19.61 ± 3.03 g DW/pot under the T1 and T2 treatments, respectively. All of these data illustrated that both P. australis and A. calamus grew better in treatments with higher N concentrations. Moreover, P. australis showed a better adaptability to the supply of N than A. calamus.
3.2 N Removal from Water and Accumulation in Plants
The pH values in systems with emergent plants fluctuated from 6.5 to 7.5, while these values increased to 9.0 in systems without plants (CK treatment). Meantime, emergent plants accelerated the removal process of nitrogen. As shown in Fig. 2, the TN concentrations were 0.83 ± 0.24 and 1.03 ± 0.30 mg/L at the end of the cycles for floating wetlands constructed with P. australis and A. calamus in the T1 treatment, with TN removal efficiencies of 91.5 ± 2.4% and 89.9 ± 3.0%, respectively. Floating wetlands constructed with P. australis showed a greater TN removal capacity; however, no significant differences (p > 0.05, LSD test) were observed in either the TN concentration or the TN removal efficiency between plant species. There were significant differences in both the TN concentration (p < 0.01 LSD test) and the TN removal efficiency (p < 0.05 LSD test) between treatments with emergent plants and the CK treatment, of which the average TN concentration was 4.99 ± 0.74 mg/L and the average TN removal efficiency 48.7 ± 8.8%. Similar results were found in the T2 treatment. At the end of cycles, the TN concentrations were 0.75 ± 0.24, 0.80 ± 0.22 and 1.78 ± 0.65 mg/L for the P. australis floating wetland, the A. calamus floating wetland, and CK, respectively. The corresponding TN removal efficiencies were 83.0 ± 4.9, 82.6 ± 4.2, and 61.2 ± 16.9%, respectively.
Plant nitrogen uptake is one of the most important pathways of N removal in floating treatment wetlands. As depicted in Table 3, the contributions of plant uptake to nitrogen removal were 72.2 ± 4.9 and 77.1 ± 2.3% under the T1 treatment and 66.4 ± 11.5 and 35.4 ± 6.7% under the T2 treatment for P. australis and A. calamus, respectively. The N concentrations of the shoots were 17.12 ± 1.80 and 11.34 ± 0.60 mg/g in P. australis and 24.23 ± 6.82 and 19.89 ± 1.92 mg/g for A. calamus under the T1 and T2 treatments, respectively. The corresponding data in the roots were 10.38 ± 0.32 and 9.14 ± 0.59 mg/g and 21.51 ± 2.37 and 15.31 ± 0.48 mg/g. The N concentrations in shoots and roots under the T1 treatment were higher than those under the T2 treatment for both P. australis and A. calamus. However, a significant difference (p < 0.05, LSD test) was only found in the roots of A. calamus under the T2 treatment. Meanwhile, there were no significant differences among the N concentrations in shoots or roots for the same species under the same treatment, although the shoot N concentrations were higher than the root N concentrations under both the T1 and T2 treatments.
N concentrations in P. australis were significantly lower (p < 0.05, LSD test) than those in A. calamus (except for in shoot of the T1 treatment), but a significant difference was only found under the T2 treatment for the total net N uptake. As listed in Table 3, the total net N uptake was 675.43 ± 45.88 and 662.23 ± 211.85 mg/pot for P. australis and A. calamus under the T1 treatment, and 263.92 ± 44.16 and 146.88 ± 28.44 mg/pot for P. australis and A. calamus under the T2 treatment, respectively. The nitrogen distribution followed a different pattern in P. australis and A. calamus. In P. australis, approximately 60% of the total net uptake of nitrogen was in the shoots. Under the T1 and T2 treatments, the net uptake of nitrogen was 427.48 ± 29.70 and 162.90 ± 41.66 mg/pot in the shoots, and 247.96 ± 23.92 and 101.02 ± 24.07 mg/pot in the roots, respectively. The distribution of nitrogen in A. calamus was different. Approximately 60% of the total net uptake of nitrogen was in the roots. The amounts of net nitrogen uptake was only 234.54 ± 64.45 and 59.77 ± 12.81 mg/pot in shoots under the T1 and T2 treatment, respectively, while the net nitrogen uptake in the roots was 427.70 ± 169.65 and 87.11 ± 17.20 mg/pot.
3.3 N Removal Process in the Floating Treatment Wetlands
Floating wetlands constructed with P. australis and A. calamus showed similar nitrogen removal capacities in a 10-day cycle, displaying similar nitrogen concentrations at the end of each cycle (shown in Fig. 2). However, these results could not provide enough information on the removal process of nitrogen. For more detailed information, more water samples were collected on days 2, 4, and 7 of the third, fifth, and seventh cycles (representing the early growth stage, the rapid growth stage, and the late growth stage, respectively), and the results are shown in Fig. 3. Ten days were enough for the degradation of nitrogen. In the floating wetlands constructed with P. australis, the concentration of TN, NH4 +-N, and NO3 −-N degraded to a stable level within 4–6 days, while a similar stable concentration was reached within 6–8 days in the A. calamus floating treatment wetlands. The TN degradation rates in the first 2 days in the floating treatment wetlands were 2.95, 3.85, and 4.20 mg L−1 day−1 in the P. australis floating treatment wetlands, and 1.53, 1.57, and 1.77 mg L−1 day−1 in the A. calamus floating treatment wetlands for the early growth stage, the rapid growth stage, and the late growth stage, respectively. Moreover, the transformation of nitrogen was also slightly different. In the A. calamus floating treatment wetlands, the decrease rate of NO3 −-N sped up after the NH4 +-N reached a stable concentration, indicating that nitrification processes occurred in the system.
4 Discussion
Comparing with other studies carried out in warm areas of China (Luo et al. 2009; Jiang et al. 2011), the growth of macrophytes was a bit slower in the cold temperate zone. This might have negative influence on the performance of FTW. The growth of P. australis was much steadier than that of A. calamus (Fig. 1). Furthermore, P. australis was also more adaptable to nutrient concentrations than A. calamus (Table 2), although a decrease in biomass was also observed when the concentration of nitrogen decreased. Because plant uptake is one of the main pathways of nitrogen removal (Headley and Tanner 2012; Marimon et al. 2013; Wang and Sample 2013), a stable growth rate is conducive to maintaining a stable nitrogen removal performance. Moreover, the shoot biomass was higher than the root biomass in P. australis. Because the shoots could be easily harvested, this fact favored of the management and reuse of FTW.
The processes of nitrogen removal include plant uptake, nitrification/denitrification, microbial uptake, precipitation, and volatilization in FTW (Lynch et al. 2015; Wang et al. 2014; Zhang et al. 2014). The pH value in FTW was maintained approximately neutral (pH = 6.5–7.5), which was suitable for both nitrification and denitrification. FTW showed great capacity for N removal. The N concentration decreased to approximately or less than 1.0 mg/L, with N removal efficiencies higher than 80%, in a 10-day cycle. Due to the absence of a substrate, plant uptake was a more important pathway for nitrogen removal than in constructed wetland because the adsorption of N on a substrate was also a very important removal pathway for N (Li et al. 2015). The plant uptake of nitrogen accounted for more than 65% of the total nitrogen removal except in the T2 treatment of the A. calamus FTW, in which the plant was in a poor condition because of the lack of nutrients. Our data showed that the contribution of plant uptake was higher than in other studies (Reddy et al. 1989; Sekiranda and Kiwanuka 1997; Song et al. 2011; Tanner et al. 1995), which might be caused by the powerful capacity for nitrogen absorption of the emergent plants tested in the present study and the inadequate nutrients supplement in our experiment, suggested by the rapid degradation of nitrogen in the beginning of each cycle (Fig. 3). In treatments without plants (CK), the degradation of nitrogen was also significant, indicating other processes, such as precipitation, microbial uptake, and nitrification/denitrification, could not be neglected (Marimon et al. 2013). However, in floating treatment wetlands, these processes were inhibited because the plants absorbed most of the nitrogen rapidly.
Although the N concentrations in P. australis were slightly lower than those in A. calamus, the total net N uptake of P. australis was higher, as the dry biomass of P. australis was much greater. Moreover, the shoots of P. australis accumulated more than 60% of the nitrogen, while this figure was only 40% in A. calamus, which was convenient for the management and reuse of the P. australis FTW. Furthermore, the large shoot biomass and high nitrogen accumulation made it possible to enhance the performance of the FTW through reaping the shoots of the emergent vegetation periodically (Weragoda et al. 2012; Zhu et al. 2011). All of these results suggested that the amount and distribution of biomass and of the emergent plant should receive attention in the selection of FTW vegetation.
Although similar N removal efficiencies were achieved in a 10-day cycle, the degradation processes of nitrogen were quite different between the FTW with P. australis and A. calamus. The degradation rates of TN in the FTW with P. australis were more than twice as high as those in the FTW with A. calamus. This might be caused by the differences in root systems between P. australis and A. calamus. P. australis has a fibrous root system while A. calamus has rhizomes. Although P. australis has similar or even smaller root biomass than A. calamus, the root surface area of P. australis is much larger. The larger surface area made the absorption and sorption rate of nutrients higher. Thus, the removal rate of nitrogen and phosphorus was higher in the P. australis FTW. Meanwhile, a higher nutrient absorption and sorption rate also resulted in different removal processes of nitrogen. The concentration of NO3 −-N accumulated remarkably in the A. calamus FTW systems, while the accumulation of NO3 −-N could be neglected in the P. australis FTW. This can be explained by the different removal rates of nitrogen in the two systems. In the A. calamus FTW, the lower nutrient absorption rate led to the concentration of NH4 +-N maintaining a high level.
5 Conclusions
-
(1)
Floating treatment wetlands have great capacity to remove nitrogen. Between 83 and 92% of the nitrogen was removed in a 10-day period.
-
(2)
Compared with A. calamus, P. australis was a more suitable species of emergent vegetation for the construction of floating wetlands in northeast China.
References
Ding, Z., Golangoldhirsh, A., Rafiq, M. K., Li, T., Zhao, F., & Yang, X. (2012). Purification of eutrophic water by ryegrass. Water Science & Technology, 66(10), 2138–2145.
Gao, Y., Yi, N., Zhang, Z., Liu, H., & Yan, S. (2012). Fate of (NO3 −)-N-15 and (NH4 +)-N-15 in the treatment of eutrophic water using the floating macrophyte, Eichhornia crassipes. J Environ Qual, 5, 1653–1660.
Groh, T. A., Gentry, L. E., & David, M. B. (2015). Nitrogen removal and greenhouse gas emissions from constructed wetlands receiving tile drainage water. J Environ Qual, 44(3).
Headley, T. R., & Tanner, C. C. (2012). Constructed wetlands with floating emergent macrophytes: an innovative stormwater treatment technology. Critical Reviews in Environmental Science & Technology, volume, 42(21), 2261–2310.
Jiang, Y., Tong, Y., You, W., & Wu, J. (2011). Comparison of the growth characteristics and the optimize configuration modes of the nitrogen and phosphorus uptake capacity of three kinds of plant cultivated on the floating-bed. China Environmental Science, 31(5), 774–780 (in Chinese).
Li, J., Yang, X., Wang, Z., Shan, Y., & Zheng, Z. (2015). Comparison of four aquatic plant treatment systems for nutrient removal from eutrophied water. Bioresour Technol, 179, 1–7.
Lu, H. L., Ku, C. R., & Chang, Y. H. (2015). Water quality improvement with artificial floating islands. Ecol Eng, 74, 371–375.
Luo, G. Y., Zheng, J. F., Xu, X. Y., Cao, J., & Shu, W. Q. (2009). Comparison of the growth characteristics and nutrient uptake of four kinds of plants cultivated on a floating-bed. Acta Scientiae Circumstantiae, 29(2), 285–290 (in Chinese).
Lynch, J., Fox, L. J., Owen, J. S., & Sample, D. J. (2015). Evaluation of commercial floating treatment wetland technologies for nutrient remediation of stormwater. Ecol Eng, 75, 61–69.
Marimon, Z. A., Xuan, Z., & Chang, N. B. (2013). System dynamics modeling with sensitivity analysis for floating treatment wetlands in a stormwater wet pond. Ecol Model, 267(1769), 66–79.
Pavan, F., Breschigliaro, S., & Borin, M. (2015). Screening of 18 species for digestate phytodepuration. Environmental Science & Pollution Research International, 22(4), 2455–2466.
Reddy, K. R., D’Angelo, E. M., & Debusk, T. A. (1989). Oxygen transport through aquatic macrophytes: the role in wastewater treatment. J Environ Qual, 19(2), 261–267.
Sekiranda, S. B. K., & Kiwanuka, S. (1997). A study of nutrient removal efficiency of Phragmites mauritianus in experimental reactors in Uganda. Hydrobiologia, 364(364), 83–91.
Song, H. L., Li, X. N., Wang, X. J., & Lu, X. W. (2011). Enhancing nitrogen removal performance of vegetated floating-bed by adding Hyriopsis cumingii Lea and an artificial medium. Fresen Environ Bull, 20(9), 2435–2441.
Tanner, C. C., Clayton, J. S., & Upsdell, M. P. (1995). Effect of loading rate and planting on treatment of dairy farm wastewaters in constructed wetlands—II. Removal of nitrogen and phosphorus. Water Res, 29(1), 27–34.
Wang, C. Y., & Sample, D. J. (2013). Assessing floating treatment wetlands nutrient removal performance through a first order kinetics model and statistical inference. Ecol Eng, 61(19), 292–302.
Wang, C. Y., Sample, D. J., & Bell, C. (2014). Vegetation effects on floating treatment wetland nutrient removal and harvesting strategies in urban stormwater ponds. Sci Total Environ, 499(1), 384–393.
Weragoda, S. K., Jinadasa, K. B. S. N., Zhang, D. Q., Gersberg, R. M., Tan, S. K., Tanaka, N., et al. (2012). Tropical application of floating treatment wetlands. Wetlands, 32(5), 955–961.
Winston, R. J., Hunt, W. F., Kennedy, S. G., Merriman, L. S., Chandler, J., & Brown, D. (2013). Evaluation of floating treatment wetlands as retrofits to existing stormwater retention ponds. Ecol Eng, 54(5), 254–265.
Zhang, C. B., Liu, W. L., Pan, X. C., Guan, M., Liu, S. Y., Ge, Y., et al. (2014). Comparison of effects of plant and biofilm bacterial community parameters on removal performances of pollutants in floating island systems. Ecol Eng, 73, 58–63.
Zhao, F., Xi, S., Yang, X., Yang, W., Li, J., Gu, B., et al. (2012). Purifying eutrophic river waters with integrated floating island systems. Ecol Eng, 40(3), 53–60.
Zhou, X. H., Wang, G. X., & Yang, F. (2012). Nitrogen removal from eutrophic river waters by using Rumex acetosa cultivated in ecological floating beds. Fresen Environ Bull, 21(7), 1920–1928.
Zhu, L., Li, Z., & Ketola, T. (2011). Biomass accumulations and nutrient uptake of plants cultivated on artificial floating beds in China’s rural area. Ecol Eng, 37(10), 1460–1466.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (grant Nos. 41430637, 41201520 and 41201494), China Postdoctoral Science Foundation funded project (grant No. 2016 M600577), Program for Innovative Research Team (in Science and Technology) in University of Henan Province (grant No. 16IRTSTHN012), and the Education Department of Henan Province (grant No. 16A170007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, X., Guo, R. Comparison of Nitrogen Removal in Floating Treatment Wetlands Constructed with Phragmites australis and Acorus calamus in a Cold Temperate Zone. Water Air Soil Pollut 228, 132 (2017). https://doi.org/10.1007/s11270-017-3266-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-017-3266-z