Abstract
Based on the fact that streamwater quality reflects landscape conditions, the objectives of this study were: to investigate nitrogen (N), carbon (C), and major ion concentrations in six streams crossing minimally disturbed Atlantic Forest areas, with similar geomorphological characteristics; to determine N and C fluxes in one of these pristine streams (Indaiá); and assess the impact of human activity on the biogeochemistry of two other streams in the same region, crossing urbanized areas. The distribution pattern of carbon and inorganic nitrogen dissolved forms, as well as the major ion and biogenic gas concentrations in the streamwater, was similar in pristine streams, indicating that the C and N dynamics were determined by influence of some factors, such as climate, atmospheric deposition, geology, soil type, and land covering, which were analogous in the forested watersheds. The urban streams were significantly different from the pristine streams, showing low dissolved oxygen concentrations, high respiration rates, and high concentrations of carbon dioxide, dissolved inorganic nitrogen, dissolved inorganic carbon, and major ion. These differences were attributed to anthropogenic impact on water quality, especially domestic sewage discharge. Additionally, in the Indaiá stream, it was possible to observe the importance of rainfall over temporal dynamics of dissolved carbon forms, and also, the obtained specific flux of dissolved inorganic nitrogen was relatively elevated (approximately 11 kg ha−1 year−1). These results reveal the influence of human activity over the biogeochemistry of coastal streams and also indicate the importance N export of Atlantic Forest to the ocean.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The Coastal Atlantic Forest, located along most of the Brazilian Coast, is one of the oldest and more biologically diverse tropical forests of the world (Murray-Smith et al. 2009). In the State of São Paulo coastal region, the Atlantic Forest is a mosaic of ecosystems, where the main formation is a dense tropical rainforest including areas of restinga (sensu Suguio and Tessler 1984) and several distinct physiognomies according to the altitude: lowland, submontane, and montane forests, varying in altitude from sea level to 1,000 m above sea level (Morellato and Haddad 2000; Veloso et al. 1991). Originally, this phytogeographic unit extended from northeastern to the southern Brazilian coast (Salimon and Negrelle 2001; Vieira et al. 2008). Nowadays, after centuries of deforestation, this vegetation is approximately only 7% of the original area with most of the fragmented remnants distributed over areas of difficult access, such as steep hills and mountainous areas (e.g., Salimon and Negrelle 2001; Vieira et al. 2008). These remnants continue to suffer under severe anthropogenic pressure, including degradation of native streams and rivers caused by loss of riparian vegetation, erosion, organic and chemical pollution, sand mining, and damming (e.g., Salimon and Negrelle 2001; Vieira et al. 2008; Villela et al. 2006).
Despite its importance as one of the richest forests in the world in terms of biodiversity and endemism (Murray-Smith et al. 2009; Myers et al. 2000), relatively little information is available on the structure and function of the Atlantic Forest (Villela et al. 2006). Vieira et al. (2008) warned that, in order to support conservation, management and restoration of this ecosystem based on a strong scientific foundation, more research is urgently needed. Such research, according to the same authors, should focus on understanding the mechanisms that regulate the biodiversity and processes that control the structure and function of these forests.
Watersheds play an important role in biogeochemical cycles of ecologically important elements such as carbon (C), nitrogen (N), phosphorus (P), and sulfur (S) since the water chemistry and nutrient export in streams are determined by interactions between hydrological and biogeochemical processes that occur in the watershed and within the stream channels (e.g., Bluth and Kump 1994; Galloway et al. 2003; Harris 2001; Lu et al. 2007; Markich and Brown 1998; Neal et al. 2006; Peterson et al. 2001; Turgeon and Courchesne 2008; Vink et al. 2007). These processes are influenced by climatic conditions, and in-stream processes as well as a set of watershed features, such as geology, geomorphology, soil properties, soil cover, and also by anthropogenic factors such as change in land use (Bluth and Kump 1994; Lu et al. 2007; Markich and Brown 1998; Neal et al. 2006; Turgeon and Courchesne 2008; Vink et al. 2007).
Human activities, such as agricultural practices and several processes associated with urbanization and industrialization, have greatly modified the biogeochemical cycles at the regional and global level, the most prominent of these changes being manifested in carbon and nitrogen cycles (e.g., Asner et al. 1997; Downing et al. 1999; Filoso et al. 2006; Galloway 1998; Grace 2004; Hedin et al. 1995; Mosier et al. 2002; Solomon et al. 2007; Vink et al. 2007; Vitousek 1994; Vitousek et al. 1997).
Much current understanding of nutrient dynamics in terrestrial and aquatic ecosystems has been derived from studies in temperate regions, particularly in biogeochemically altered areas (Hedin et al. 1995; Perakis and Hedin 2002). Therefore, studies in pristine or minimally disturbed watersheds, which are becoming increasingly scarce, are fundamentally important because they can provide baseline information about natural patterns of nutrient cycling, against which disturbed cycles can be compared (Goller et al. 2006; Hedin et al. 1995; Lewis et al. 1999; Perakis and Hedin 2002). Similarly, studies in tropical watersheds are generally important, since the potential magnitude of elemental fluxes from terrestrial ecosystems to streams, oceans, and atmosphere are large but not well-represented in the literature (Downing et al. 1999; Goller et al. 2006; Lewis et al. 1999; McDowell and Asbury 1994).
The main objective of this study is to present a comprehensive evaluation of carbon, nitrogen, and major ion concentrations and fluxes in six streams crossing minimally disturbed Atlantic Forest areas, in the northern coast of São Paulo State (southeastern Brazil). The watersheds of these streams have similar geomorphological characteristics since all streams are formed at an altitude of approximately 1,000 m above sea level and end in the ocean under the same geological substrate (granites of the pre-Cambrian). In addition to this primary objective, we propose to investigate impact of human activity on the biogeochemistry of two streams located in the same region, having the same geomorphological and geological characteristics as the others, but draining an urbanized area in the final portion of their watersheds. This second objective has practical implications on public policy since the northern coast of the State of São Paulo has been urbanized at an unprecedented rate and without proper domestic sewage treatment required to minimize effects of such urbanization on streams and coastal waters.
2 Materials and Methods
2.1 Study Area
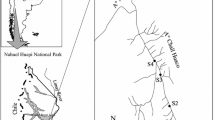
The municipality of Ubatuba (711 km2) is located in the northern coast of São Paulo State—southeastern region of Brazil (Fig. 1). Especially due to its geographic location and environmental characteristics (several coastal beaches and an exuberant vegetation), tourism is the primary economic activity. The majority of the land cover is Atlantic Forest remnants (91% of its area), which are well-preserved, in particular within a State Park's borders (CETESB 2007, 2008; IPT 2000). Urbanization is the second main land use, and agriculture is not significant (CETESB 2006, 2007, 2008; IPT 2000).
Location of Ubatuba municipality (southeastern of Brazil, northern coastal of São Paulo State) and sampling sites: Fazenda (FAZ), Ubatumirim (UBA), Puruba (PUR), Prumirim (PRU), Itamambuca (ITA), Indaiá (IND), Grande (GRA) e Lagoinha (LAG) streams. Note that only Grande and Lagoinha streams were located in urban areas. Details of Indaiá watershed, indicating stage gauge station
The regional climate is predominantly hot and wet, with elevated annual rainfall (on average 2,200 mm year−1). Despite the fact that rain events are usually well-distributed during the year, their intensity are frequently greater in the summer and lower on June, July, and August months (IPT 2000). Similar pattern has been observed for temperature, with monthly average values higher than 24°C in the summer and between 17°C and 20°C in the winter (IPT 2000). The wet deposition of total nitrogen in this area is approximately 2 to 3 kg ha−1 year−1 (P. Camargo, personal communication).
Geologically, the study area is mainly composed of the crystalline basement (pre-Cambrian) of the “Coastal Complex”, with predominance of metamorphic (gneisses and migmatite) and granitic rocks (IPT 2000). Banks of sedimentary rocks are also observed on the “Coastal Plain” (IPT 2000). Soils are acidic, chemically poor, and dystrophic, being characterized by associations between Latosols, Cambisols, and Quartzpsament (Martins 2010).
The hydrographic units are defined by several streams with small stretch and discharge volume that flow from a high altitude in the Serra do Mar mountain range to the Atlantic Ocean (CETESB 2006, 2007; IPT 2000). From this broad hydrological system, we selected eight small watersheds and established one sampling site for each (Fig. 1). The majority of these selected watersheds encompass streams in pristine or minimally disturbed areas of Atlantic Forest, predominantly covered by primary forest. Their local names are: Fazenda, Ubatumirim, Puruba, Prumirim, Itamambuca, and Indaiá streams. In addition, two watersheds that drain urban areas were also selected—Grande and Lagoinha streams. The release of domestic sewage is the main source of pollution of these urban streams, since only 17% of total sewage generation in Ubatuba is treated, the remainder being dumped into water bodies or disposed in septic tanks (IPT 2000). We chose the Indaiá stream as representative of minimally disturbed watersheds and installed a stage gauge station in order to estimate streamwater discharge. The description and location of the sampling sites are summarized in Table 1 and Fig. 1, respectively.
2.2 Sample Design and Analyses
From September 2006 to December 2007, a total of 16 water samples were collected monthly at one site in each watershed. Additionally, intensive collections took place at Indaiá stream, where water samples were taken every 2 days, from December 2006 to March 2008 (totaling 212 samples); and also, every hour during a 48 h rainfall event, in March 2008 (48 samples). To avoid any influence of ocean water dilution, we selected points that were located upstream enough to avoid intrusion of ocean water. This was checked by measuring water conductivity during high tide periods. Additionally, most of time, we sampled our streams during low tide periods of the day.
Water samples taken during monthly collections were analyzed for all studied parameters (described below). Concerning samples intensively collected at Indaiá stream, some parameters were not analyzed, such as total suspended solids (TSS), respiration rates (RR), and stable carbon isotopic composition of DIC (δ13C-DIC).
For all analyses, water samples were collected using a Niskin bottle in midchannel. In the field, during the sampling time, we measured the values of pH (Orion 250A meter), electrical conductivity (EC; Ambar Science 2052 meter), and dissolved oxygen (DO), and water temperature (Yellow Springs 58 meter). Samples were stored in high-density polyethylene flasks, and most were preserved with mercuric chloride (HgCl2, 0.5 µM) to stop microbial activity (except for dissolved inorganic carbon (DIC) and major ion analyses, which were preserved with thymol). In the laboratory, all subsamples were stored at low temperature (4°C) until analyzed. After analysis, the samples preserved with HgCl2 were sent to the Laboratory Waste Management of CENA and submitted to the neutralization process.
For DOC analyses, samples were passed through glass fiber filters (0.7 µm nominal pore diameter) and analyzed in a Shimadzu TOC 5000A. For DIC, samples were filtered through cellulose acetate filters (0.45 μm pore diameter) and also analyzed by Shimadzu model TOC 5000A (with an infrared detector). The speciation of DIC (HCO3, CO3, and CO2) was calculated by equations of the equilibrium thermodynamic, from measurements of pH, temperature, and DIC concentrations (Skirrow 1975). Free dissolved CO2 (pCO2) and dissolved oxygen concentrations are also presented as departure from atmospheric equilibrium, called excess of CO2 (Exc-CO2) and apparent oxygen utilization (AOU), respectively (Butler 1991; Salomão et al. 2008).
Samples for determining anion and cation concentrations were passed through cellulose acetate filters (0.45 µm pore diameter) and analyzed for ionic conductivity with a DIONEX DX500. The analyzed ions were Na+, NH +4 , K+, Mg2+, Ca2+, Cl−, NO −3 , NO −2 , SO 2−4 , and PO 3−4 . Dissolved inorganic nitrogen (DIN) was taken as the sum of ammonium (NH +4 ), nitrate (NO −3 ), and nitrite (NO −2 ) concentrations. The quality control of the analytical data was performed by the charge balance of the individual samples. High significant correlations (r 2FAZ = 0.88; r 2UBA = 0.87; r 2PUR = 0.97; r 2PUR = 0.89; r 2ITA = 0.89; r 2IND = 0.92; r 2GRA = 0.99; r 2LAG = 0.92) and the near-zero intercept between the sum of anions (TZ−) and the sum of cations (TZ+; Table 2) show that the main ions were determined for these samples and that a good ionic balance was achieved.
For δ13C-DIC analyses, water subsamples were acidified (H3PO4) under vacuum, for 5 min. The resulting CO2 was cold-trapped using liquid nitrogen and then analyzed by dual Intel Finnegan Delta Plus mass spectrometer.
For TSS, approximately 20 L of water were sampled and homogenized (US Geological Survey splitter), and 2 L of subsamples were stored. In the laboratory, these subsamples were passed through pre-weighed filters and then the filters were dried to a constant weight. The TSS concentrations (expressed as milligrams per liter) were determined gravimetrically.
For the determination of RR, six 350 mL glass biological oxygen demand bottles were filled in the field. Three bottles (controls) were immediately preserved, and the others were incubated in the dark for 5–7 days, at ambient temperatures. The dissolved oxygen concentrations were measured in the control bottles (initial concentration, DOi) and in the incubated bottles (final concentration, DOf). RR values were calculated as the ratio of DO (DOf–DOi) by the period of incubation and expressed as micromolars per hour.
In addition to water sampling, streamwater discharges were measured in the stage gauge station on the Indaiá stream, from cross-sectional area and flow. Water velocity was measured with a current meter (USGS Model 6205). Water level measurements were taken manually and simultaneously with intensive collections in this station. To obtain the discharge, estimated from the water level data, a stage–discharge curve was developed. The resulting stage–discharge curve is described by equation:
where the water level (H) is in meters and the discharge (Q) in cubic meters per second (r 2 = 0.9706). Using these estimated discharges, the C and N specific fluxes (expressed as kilograms per hectare per year) were calculated as the ratio of annual flow (nutrient concentration × water discharge) by the Indaiá watershed area.
For description of the sampling sites, the instantaneous discharge, water depth, and channel length for each stream were measured (Table 1). These measures were taken from the determination of the cross-sectional area of each stream, and the water velocity was measured with a current meter (USGS Model 6205).
Finally, to characterize the seasonal variation throughout study period, the Indaiá stream discharge values were associated with precipitation values of Ubatuba. The dry period was defined as the period with lower precipitation (May, 2007 to September, 2007). During this period the precipitation values ranged from approximately 50 to 150 mm (August and May months, respectively), with an average value of 100 mm. The precipitation average value for the wet period was 250 mm (ranging from 200 to 380 mm, approximately).
2.3 Data Ordination and Statistical Analyses
In order to differentiate the chemical data with respect to the watershed land use, a principal component analysis (PCA) was conducted using the software PC-ORD, version 3.15 for windows (McCune and Mefford 1997). The PCA was run using quantitative variables (all data collected monthly) and also some categorical variables, as watershed features (e.g., drainage areas and altitude of the sample site). The components used for data interpretation were those in which eigenvalues were higher than “broken-stick” eigenvalues (produced by random hypothesis of this model) (Jackson 1993). To investigate whether the coordinates (scores) produced by PCA ordination presented differences, statistical tests were applied.
All statistical analyses were performed using the software STATISTICA, version 7.1 for Windows (StatSoft 2007). Differences at the 0.05 level were reported as significant. We applied the Shapiro-Wilk and Levene's tests at residual values to test for assumptions of normality and homogeneity of variances, respectively. Due the fact that most of our data did not follow normal distribution, we employed nonparametric statistical analyses. Therefore, median values were used instead of averages. Kruskal–Wallis and Mann–Whitney test were respectively applied to test for statistically significant differences between watersheds and for seasonal differences. Spearman ranking was also used to determinate correlations among data.
3 Results
3.1 Differences among Watersheds
Differences among watersheds were identified by the principal component analysis, explaining 46.8% of total data variance. The first component (PC I), standing for 32.7% of total variation, grouped urban stream results on its positive end, which is characterized by higher values of DIC, NH +4 , EC, and RR, and grouped results of pristine streams on its negative end, characterized by higher values of DO and altitude of the sample site (Fig. 2). The PC I scores of urban streams (Grande and Lagoinha) were statistically different from almost all of pristine stream scores (p < 0.01), with some exception relating to the Puruba stream (p > 0.05). The second component (PC II), standing for 14.1% of total variation, showed some differences among pristine streams, principally regarding watershed features (altitude of sample site and watershed area), and also to C and N concentrations. The PC II grouped the results of the Indaiá stream on its positive end, which is characterized by higher values of pH, NO −3 , and altitude; and grouped Puruba stream results on its negative end, characterized by higher values of TSS, DOC, and area (Fig. 2). Statistical differences for PC II scores were detected between the Indaiá and Puruba streams (p < 0.05). For median values of chemical parameters cited above, see Table 2.
The associations between parameters related to aquatic metabolism (Fig. 3) demonstrated that the DO concentrations were inversely correlated with pCO2 and RR (r s = −0.54 and r s = −0.49, respectively, p < 0.05); and pCO2 values were directly related to RR (r s = 0.43, p < 0.05). Moreover, we found a positive and significant correlation between the median concentrations of AOU and Exc-CO2 (r s = 0.60, p < 0.05; Table 2). In all these associations, urban streams (Grande and Lagoinha) presented the most extreme values. These relationships corroborated the PCA results.
Associations between parameters relating to aquatic metabolism: relationship between median values of respiration rates (RR) and median concentrations of dissolved oxygen saturation (DO); relationship between median concentrations of free dissolved CO2 concentrations (pCO 2 ) and median concentrations of DO; relationship between median values of RR and median concentrations of pCO2. Symbols represent the median values of data collected monthly in each watershed. Bars represent 25% and 75% percentiles
Relating to ion concentrations, compared with the pristine streams, higher values were observed in urban streams (Grande and Lagoinha), expressed as TZ+ and TZ− (p < 0.001; Table 2). However, considering the proportionality of major ions in relation to the sum of charge, as detected from the Piper diagram (Fig. 4), the differences between urban and pristine streams were not considerable. These results are consistent with the watershed geology, reflecting the same geological substrate of then. Moreover, we plotted the concentrations of HCO −3 against the sum of Ca2+ and Mg2+ to assess the importance of rock weathering to the streamwater chemical composition. Only samples collected during the lower water period (May to September) were used to minimize the effects of rain water and surface runoff. The 2:1 ratio for rock weathering was found in the present study (Fig. 5). Higher HCO −3 concentrations than the 2:1 ratio were observed principally in urban streams (Grande and Lagoinha), indicating that this ion also originates from another source than rock weathering.
Relationship among median dissolved bicarbonate (HCO −3 ) and the sum of bivalent basic cations (Ca2++Mg2+) in streamwater samples collected during the low water period in each watershed. Line represents the hypothetical 2:1 ratio. Symbols represent the median values. Bars represent 25% and 75% percentiles
We used the δ13C-DIC as a tool to track the carbon sources. We found that δ13C-DIC values were directly correlated with HCO −3 and free dissolved CO2 concentrations (r s = 0.22 and r s = 0.37, respectively, p < 0.05) and also that Grande and Lagoinha streams presented slightly higher δ13C-DIC values compared with the pristine streams (Table 2).
Concerning seasonal variation, no significant differences were found throughout the period in almost all analyzed parameters in pristine watersheds. However, higher values of EC, DIC, pCO2, DIN, NO −3 , NH +4 , and ion concentrations were obtained in urban watersheds during the lower water period (May to September, 2007) (p < 0.05).
3.2 Distribution Patterns of DOC, DIC and DIN—Indaiá Pristine Watershed
The distribution of dissolved forms of carbon and inorganic nitrogen in the Indaiá stream (chosen as a representative pristine watershed) are shown in Fig. 6. In general, DOC, DIC, and DIN concentrations did not present a significant correlation with streamwater discharge (r s = 0.16, r s = 0.04, and r s = 0.09, respectively, p > 0.05). Inorganic nitrogen was largely dominated by NO −3 . Ammonium (NH +4 ) was regularly detected in low concentration, and NO −2 was not detected in most water sampled (approximately 95%).
Despite this absence of significant relationships between nutrient concentrations and streamwater discharge, a different pattern was found during a 48-h rainfall event at the Indaiá stream for carbon concentrations. The streamwater level and discharge ranged from 0.88 to 1.20 m and from 0.45 to 2.33 m3 s−1, respectively. The carbon dissolved forms reflected this discharge variation in direct proportion to DOC and an inverse proportion to DIC (r s = 0.77 and r s = −0.62, respectively, p < 0.05; Fig. 7). Significant correlations of discharge were not observed for dissolved inorganic nitrogen concentrations during a rainfall event (p > 0.05), as previously noted.
Concerning specific fluxes, the values of DIC flux were higher than DOC (78 and 24 kg ha−1 year−1, respectively). DIN flux was 11 kg ha−1 year−1, and fluxes of NO −3 and NH +4 were 10 and approximately 1 kg ha−1 year−1, respectively.
4 Discussion
4.1 Pristine Versus Urban Watersheds
The correlations between DO and pCO2 among streams demonstrated the importance of the metabolism within the stream on the control of biogenic gas concentrations (Fig. 3). The correlations between DO and RR corroborated this view (Fig. 3), as well as the correlations between Exc-CO2 and AOU. According to these results, we assumed that the aquatic metabolism of studied streams is controlled in part by the same process: internal metabolism.
However, other processes can also control the biogenic gas concentrations in the streams. When the concentrations of biogenic gasses are affected only by the processes of respiration and photosynthesis, a ratio of 1:1 between the concentrations of these gasses is expected (for each mol of CO2 retained in the photosynthetic process 1 mol of O2 is released—the opposite occurring in the respiratory process; Salomão et al. 2008). The Exc-CO2 values, at least two times higher than AOU (Table 2), indicate an excess of respiration over photosynthesis, reflecting the existence of another source of CO2.
Additionally, the metabolism of sampled streams was predominantly heterotrophic, as can be seen by O2 and CO2 concentrations (Table 2), which were below and above the atmospheric equilibrium concentrations, respectively. The aquatic heterotrophic metabolism is sustained principally by allochthonous input of organic matter (Cole et al. 1994; Cole and Caraco 2001; Jarvie et al. 1997). The riverine particulate matter can provide information about such allochthonous input and is interrelated to several factors such as topography, hydrology, and land use (Martinelli et al. 2005). Thus, the observed low values of TSS in forested pristine streams (Table 2) were expected. Low TSS values were also obtained in urban streams, of the same order magnitude. Although, as illustrated by Table 2, these streams exhibited higher DIC and pCO2 values, as well as higher ionic concentrations, and lower DO values—probably as a direct response to anthropogenic inputs of labile organic matter, especially as untreated sewage. These results are in agreement with studies in the southeastern region of Brazil that reported consistent correlation between urban land area and DIC, CO2, and O2 concentrations (Daniel et al. 2002; Martinelli et al. 1999; Salomão et al. 2008).
Another important source of DIC in streamwater is groundwater inputs. Rock weathering contributes a significant portion of HCO −3 to most rivers and thus has a strong influence on their carbon cycle (Barth et al. 2003). Several types of rock weathering reactions released HCO −3 and Ca2+ and Mg2+ in a 2:1 ratio (Barth et al. 2003). This 2:1 balance for rock weathering was found in the present study (Fig. 5), emphasizing the importance of groundwater in the streamwater chemical composition, consistent with results reported by Barth et al. (2003) and Salomão et al. (2008). According to the same authors, the higher HCO −3 concentrations than the 2:1 ratio, which was observed principally in urban streams (Grande and Lagoinha), indicated that this ion originates from other sources than rock weathering.
Within this context, the isotopic composition of DIC is an important tool for tracking carbon sources (e.g., Amiotte-Suchet et al. 1999; Cameron et al. 1995; Salomão et al. 2008; Wachniew 2006). In the present study, δ13C-DIC median values obtained ranged from approximately −10‰ (at Lagoinha stream) to −15‰ (at Puruba stream) (Table 2), and consistent with Amiotte-Suchet et al. (1999) who propose that DIC originated from the silicate weathering and also probably from the water–air exchange, in streams of a small forested watershed that drains a strictly granite basement.
However, the influence of anthropogenic sources at the δ13C-DIC was not well-detected in this study. The labile organic matter inputs and their subsequent aerobic decomposition lead to depletion of the riverine DIC in 13C (Barth et al. 2003). However, the urban streams presented slightly higher δ13C-DIC values in compared with the pristine streams. A possible explanation could be the presence of C4 plants in these urban watersheds, which probably enriched the δ13C-DIC values (e.g., Amiotte-Suchet et al. 1999; Barth et al. 2003; Cameron et al. 1995; Salomão et al. 2008; Wachniew 2006).
In contrast, the influence of anthropogenic inputs in the streamwater chemical composition can be seen in nitrogen concentrations. Among several anthropogenic sources of nitrogen in aquatic ecosystems, the importance of the role of sewage is well-recognized, which is regularly associated with the enrichment of this nutrient in rivers (Filoso et al. 2003; Howarth et al. 1996; Martinelli et al. 1999). Thus, the higher DIN concentrations in Grande and Lagoinha streams probably can be attributed to this source.
Additionally, the results of PCA analyses (Fig. 2) were consistent with this fact. Concerning pristine watersheds, in general, they exhibited similar aquatic functioning. We suggest that C and N dynamics were, overall, determined by influence of factors such as climate, atmospheric deposition, geology, soil type, and land covering, which were analogous in the pristine watersheds. However, differences were detected between Indaiá and Puruba watersheds (p < 0.05), some of them related to environmental characteristics (as sample site altitude and watershed area) and some were associated with C and N concentrations (higher values of TSS and DOC and lower DIN concentrations at Puruba stream; Fig. 2, Table 2). In this case, we assumed that the differences in C and N distribution between Indaiá and Puruba pristine watersheds should be attributed to some watershed's peculiarities, especially those related to the extension and topography of the drainage area and not to anthropogenic influences.
4.2 C and N Temporal Dynamics in a Pristine Watershed of the Atlantic Forest–Indaiá Stream
As shown in Fig. 6, carbon (DOC and DIC) and nitrogen (DIN) concentrations were not related to the streamwater discharge (p > 0.05). However, a different pattern was found during a rainfall event for dissolved carbon forms (Fig. 7)—DOC concentrations were directly proportional to the discharge; and DIC showed an inverse relationship to discharge.
Johnson et al. (2006) evaluating the carbon fluxes by hydrological flowpaths on small watersheds of the Amazon Basin found that terrestrial DOC fluxes sporadically transported by quickflow were nearly the same as DOC transported by base flow on an annual basis. In addition, they showed that deeper hydrological flowpaths were also important C pathways, but for DIC rather than DOC. Despite the fact that, in the present study, we did not analyze C concentrations at such hydrological flowpaths, we observed the runoff responses to rainfall events and subsequent riverine carbon behavior.
Therefore, it was possible to identify the importance of rainfall events on temporal C dynamics at the Indaiá stream. In addition, such patterns probably are reproduced on other minimally perturbed watersheds of the Atlantic Forest with similar hydrological, geomorphologic, and physiographic characteristics.
4.3 Comparisons of C and N Fluxes—Indaiá Versus Other Watersheds
The main role of rivers on the carbon biogeochemical cycle has been the export of such nutrients, and their role as significant sources of releasing CO2 to the atmosphere has also been shown (Cole and Caraco 2001; Richey et al. 1988).
Riverine DOC exports are commonly reported as extremely variable among and within land use and biome types (Aitkenhead and McDowell 2000; Hope et al. 1994). For this reason, the most frequent denominator used to compare river systems of widely varying properties is the annual export of C (Richey et al. 1990).
In order to compare Indaiá DOC export with exports of other regions, we plotted DOC fluxes (kilograms per hectare per year) against the values of specific runoff (millimeters per year; Fig. 8). Data used in this comparison was extracted from several published studies conducted in tropical (McDowell and Asbury 1994; Moreira-Turcq et al. 2003; Richey et al. 1990; Seyler et al. 2005; Waterloo et al. 2006) and temperate regions (Cairns and Lajtha 2005; Campbell et al. 2000; Hope et al. 1994). These selected data were obtained in forested watersheds and principally in regions where human impact was not very significant.
Log–log relationship between annual runoff (millimeters per year) and annual export (kilogram per hectare per year) of DOC in Indaiá stream (present study, IND) and in a set of predominantly forested temperate and tropical watersheds. Data have been extracted from the following published studies: Temperate: Campbell et al. (2000), Cairns and Lajtha (2005) and Hope et al. (1994); Tropical: McDowell and Asbury (1994), Moreira-Turcq et al. (2003), Richey et al. (1990), Seyler et al. (2005) and Waterloo et al. (2006)
The annual specific flux of DOC obtained in the present study was approximately 24 kg ha−1 yr−1. In general, this value is at the lower end of reported data for tropical watersheds (Fig. 8). As shown in Fig. 8, slightly higher values of tropical DOC fluxes refer to backwater rivers of the Negro River Basin in the Amazon region, which are well-recognized as “C-rich” systems (McDowell and Asbury 1994; Moreira-Turcq et al. 2003; Waterloo et al. 2006).
Regarding DIC fluxes, less data are available, which make comparisons among streams more difficult. Hope et al. (1994) reported that the DIC fluxes in several temperate watersheds were similar to total carbon export, ranging from 10 to 100 kg ha−1 year−1. An analogous range was reported by Richey et al. (1990) for a tropical watershed. The DIC flux of the Indaiá stream (almost 80 kg ha−1 year−1) was in the upper reach of the above range.
Finally, studies focused on nitrogen dynamics also frequently employ the annual export as a comparative denominator (Campbell et al. 2004). Since riverine N export is regulated by complex processes that occur within watersheds, it is a useful tool to assess the nutritional status of the terrestrial ecosystems (Campbell et al. 2000, 2004).
In order to compare inorganic nitrogen fluxes between Indaiá and other regions, as previously described, the annual fluxes were related to the specific runoff (Fig. 9). For this comparison, we selected studies conducted in: minimally disturbed forested tropical watersheds, indicated as tropical (Lewis et al. 1999; McDowell and Asbury 1994; Neill et al. 2006); minimally disturbed temperate watersheds predominantly forested and/or with low N atmospheric inputs, indicated as temperate (low) (Cairns and Lajtha 2005; Campbell et al. 2000; Lewis et al. 1999; Lewis 2002; Vanderbilt et al. 2003); temperate watersheds predominantly forested with some anthropogenic impact and/or with high N atmospheric inputs, indicated as temperate (high) (Boyer et al. 2002; Campbell et al. 2004; Castro et al. 2007); and minimally disturbed watersheds dominated by N2-fixing red alder (Alnus rubra), indicated as temperate (red alder) (Compton et al. 2003; Fig.9).
Log–log relationship between annual runoff (millimeters per year) and annual export (kilograms per hectare per year) of DIN, NO −3 , and NH +4 in the Indaiá stream (present study, IND) and in a set of temperate and tropical watersheds. Data extracted from the following published studies: Temperate (low), minimally disturbed watersheds predominantly forested and/or with low N atmospheric inputs: Cairns and Lajtha (2005), Campbell et al. (2000), Lewis (2002), Lewis et al. (1999) and Vanderbilt et al. (2003); Temperate (high), watersheds predominantly forested with some anthropogenic impact and/or with high N atmospheric inputs: Boyer et al. (2002), Campbell et al. (2004) and Castro et al. (2007); Temperate (red alder), minimally disturbed watersheds predominantly composed of N2-fixing red alder (Alnus rubra): Compton et al. (2003); Tropical, minimally disturbed forested watersheds: Lewis et al. (1999), McDowell and Asbury (1994) and Neill et al. (2006)
The annual specific flux obtained in this study was 11 kg ha−1 year−1 for DIN, 10 kg ha−1 year−1 for NO −3 , and approximately 1 kg ha−1 year−1 for NH +4 . In general, these values were slightly higher than most values reported for tropical watersheds and temperate basins with high N atmospheric input and slightly lower than temperate watersheds dominated by N2-fixing red alder (Fig. 9). Elevated atmospheric N deposition may lead to increased availability of inorganic N, which overloaded the biotic demand (condition defined as N saturation; Aber et al. 1998). As described by the same authors, such N saturation results in several effects, such as excess NO −3 in streams. However, there is no evidence of N saturation of the Atlantic Forest. In addition, a higher N exports at the Indaiá stream probably was not due to predominance of N2-fixing plant communities (Campos 2008; Sanchez et al. 1999).
Compared with pristine temperate watersheds (low N atmospheric deposition), we observed higher N export in the Indaiá stream (Fig. 9). The same pattern is frequently reported, given that generally, nitrogen export rates of undisturbed tropical watersheds are significantly greater than those seen in temperate regions with low N atmospheric inputs, given similar rates of runoff (Downing et al. 1999).
References
Aber, J. D., Nadelhoffer, K. J., Steudler, P., & Mellilo, J. M. (1998). Nitrogen saturation in northern forest ecosystems: hypotheses revisited. BioScience, 48, 921–934.
Aitkenhead, J. A., & McDowell, W. H. (2000). Soil C:N ratio as a predictor of annual riverine DOC flux at local and global scales. Global Biogeochemical Cycle, 14, 127–138.
Amiotte-Suchet, P., Aubert, D., Probst, J. L., Gauthier-Lafaye, F., Probst, A., Andreux, F., et al. (1999). δ13C pattern of dissolved inorganic carbon in a small granitic catchment: the Strengbach case study (Vosges Mountains, France). Chemical Geology, 159, 129–145.
Asner, G. P., Seastedt, T. R., & Townsend, A. R. (1997). The decoupling of terrestrial carbon and nitrogen cycles: human influences on land cover and nitrogen supply are altering natural biogeochemical links in the biosphere. BioScience, 47, 226–234.
Barth, J. A. C., Cronin, A. A., Dunlop, J., & Kalin, R. M. (2003). Influence of carbonates on the riverine carbon cycle in an anthropogenically dominated catchment basin: evidence from major elements and stable carbon isotopes in the Lagan River (N. Ireland). Chemical Geology, 200, 203–216.
Bluth, G. J. S., & Kump, L. R. (1994). Lithologic and climatologic controls of river chemistry. Geochimica et Cosmochimica Acta, 58, 2341–2359.
Boyer, E. W., Goodale, C. L., Jaworsk, N. A., & Howarth, R. W. (2002). Anthropogenic nitrogen sources and relationships to riverine export in the northeastern USA. Biogeochemistry, 57, 137–169.
Butler, J. N. (1991). Carbon dioxide equilibria and their applications. Chelsea, MI: Lewis Publishers.
Cairns, M. A., & Lajtha, K. (2005). Effects of succession on nitrogen export in the west-central Cascades, Oregon. Ecosystems, 8, 583–601.
Cameron, E. M., Hall, G. E. M., Veizer, J., & Krouse, H. R. (1995). Isotopic and elemental hydrogeochemistry of major river system: Fraser River, British Columbia, Canada. Chemical Geology, 122, 149–169.
Campbell, J. L., Hornbeck, J. W., McDowell, W. H., Buso, B. C., Shanley, J. B., & Likens, G. E. (2000). Dissolved organic nitrogen budgets for upland, forested ecosystems in New England. Biogeochemistry, 49, 123–142.
Campbell, J. L., Hornbeck, J. W., Mitchell, M. J., Adams, M. B., Castro, M. S., Driscoll, C. T., et al. (2004). Input-output budgets, of inorganic nitrogen for 24 forest watersheds in the northeastern United States: a review. Water Air and Soil Pollution, 151, 373–396.
Campos, M.C.R. (2008). Relação da composição e estrutura do componente arbóreo com variáveis microtopográficas e edáficas da Floresta Ombrófila Densa do Núcleo Picinguaba/PESM, Ubatuba/SP. Master dissertation, Campinas: Universidade Estadual de Campinas.
Castro, M. S., Eshleman, K. N., Pitelka, L. F., Frech, G., Ramsey, M., Thomas, D. M., et al. (2007). Symptoms of nitrogen saturation in a aggrading forested watershed in western Maryland. Biogeochemistry, 84, 333–348.
Cole, J. J., & Caraco, N. F. (2001). Carbon in catchments: connecting terrestrial carbon losses with aquatic metabolism. Marine and Freshwater Research, 52, 101–110.
Cole, J. J., Caraco, N. F., Kling, G. W., & Kratz, T. K. (1994). Carbon dioxide supersaturation in the surface waters of lakes. Science, 265, 1568–1570.
Companhia de Tecnologia de Saneamento Ambiental – CETESB. (2006). Governo do Estado de São Paulo. Relatório da qualidade das águas interiores do Estado de São Paulo, 2005. http://www.cetesb.sp.gov.br/Agua/rios/publicacoes.asp.pdf. Accessed 10 October 2006.
Companhia de Tecnologia de Saneamento Ambiental – CETESB. (2007). Governo do Estado de São Paulo. Relatório da qualidade das águas interiores do Estado de São Paulo, 2006. http://www.cetesb.sp.gov.br/Agua/rios/publicacoes.asp.pdf. Accessed 03 March 2007.
Companhia de Tecnologia de Saneamento Ambiental – CETESB. (2008). Governo do Estado de São Paulo. Relatório da qualidade das águas interiores do Estado de São Paulo, 2007. http://www.cetesb.sp.gov.br/Agua/rios/publicacoes.asp.pdf. Accessed 07 January 2008.
Compton, J. E., Church, M. R., Larned, S. T., & Hogsett, W. E. (2003). Nitrogen export from forested watersheds in the Oregon Coast Range: the role of N2-fixing Red Alder. Ecosystems, 6, 773–785.
Daniel, M. H. B., Montebelo, A. A., Bernardes, M. C., Ometto, J. P. H. B., Camargo, P. B., Krusche, A. V., et al. (2002). Effects of urban sewage on dissolved oxygen, dissolved inorganic carbon, and electrical conductivity of small streams along a gradient of urbanization in the Piracicaba River Basin. Water, Air and Soil Pollution, 136, 189–206.
Downing, J. A., McClain, M., Twilley, R., Melack, J. M., Elser, J., Rabalais, N. N., et al. (1999). The impact of accelerating land use change on the N-cycle of tropical aquatic ecosystems: current conditions and projected changes. Biogeochemistry, 46, 109–148.
Filoso, S., Martinelli, L. A., Williams, M. R., Lara, L. B., Krusche, A., Ballester, V., et al. (2003). Land use and nitrogen export in the Piracicaba River basin, Southeast Brazil. Biogeochemistry, 65, 275–294.
Filoso, S., Martinelli, L. A., Howarth, R. W., Boyer, E. W., & Dentener, F. (2006). Human activities changing the nitrogen cycle in Brazil. Biogeochemistry, 79, 61–89.
Galloway, J. N. (1998). The global nitrogen cycle: changes and consequences. Environmental Pollution, 102, 15–24.
Galloway, J. N., Aber, J. D., Erisman, J. W., Seitzinger, S. P., Howarth, R. W., Cowling, E. B., et al. (2003). The nitrogen cascade. BioScience, 53, 341–356.
Goller, R., Wilcke, W., Fleischbein, K., Valarezo, C., & Zech, W. (2006). Dissolved nitrogen, phosphorus, and sulfur forms in the ecosystem fluxes of a montane forest in Ecuador. Biogeochemistry, 77, 57–89.
Grace, J. (2004). Understanding and managing the global carbon cycle. Journal of Ecology, 92, 189–202.
Harris, G. P. (2001). Biogeochemistry of nitrogen and phosphorus in Australian catchments, rivers and estuaries: effects of land use and flow regulation and comparisons with global patterns. Marine and Freshwater Research, 52, 139–149.
Hedin, L. O., Armesto, J. J., & Johnson, A. H. (1995). Patterns of nutrient loss from unpolluted, old growth temperate forests: evaluation of biogeochemical theory. Ecology, 76, 493–509.
Hope, D., Billet, M. F., & Cresser, M. S. (1994). A review of the export of carbon in river water: fluxes and processes. Environmental Pollution, 84, 301–324.
Howarth, R. W., Billen, G., Swaney, D., Townsend, A., Jaworski, N., Lajtha, K., et al. (1996). Regional nitrogen budgets and riverine N & P fluxes for the drainages to the North Atlantic Ocean: natural and human influences. Biogeochemistry, 35, 75–139.
Instituto de Pesquisa Tecnológica de São Paulo–IPT. (2000). Diagnóstico da situação atual dos recursos hídricos da Unidade de Gerenciamento dos Recursos Hídricos do Litoral Norte–UGRHI-3: Relatório técnico nº 46.172.
Jackson, D. A. (1993). Stopping rules in principal component analyses: a comparison of heuristical and statistical approaches. Ecology, 74, 2204–2214.
Jarvie, H. P., Neal, C., Leach, D. V., Ryland, G. P., House, W. A., & Robson, A. J. (1997). Major ion concentrations and inorganic carbon chemistry of Humber rivers. Science of the Total Environment, 194(195), 285–302.
Johnson, M. S., Lehmann, J., Selva, E. C., Abdo, M., Riha, S., & Couto, E. G. (2006). Organic carbon fluxes within and streamwater exports from headwater catchments in the southern Amazon. Hydrological Processes, 20, 2599–2614.
Lewis, W. M. (2002). Yield of nitrogen from minimally disturbed watersheds of the United States. Biogeochemistry, 57(58), 375–385.
Lewis, W. M., Melack, J. M., McDowell, W. H., McClain, M., & Richey, J. F. (1999). Nitrogen yields from undisturbed watersheds in the Americas. Biogeochemistry, 46, 149–162.
Lu, H., Yin, C., Wang, W., & Shan, B. (2007). A comparison study of nutrient transfer via surface runoff from two small agricultural catchments in north China. Environmental Geology, 52, 1549–1558.
Markich, S. J., & Brown, P. L. (1998). Relative importance of natural and anthropogenic influences on the fresh surface water chemistry of the Hawkesbury-Nepean River, south-eastern Australia. Science of the Total Environment, 217, 201–230.
Martinelli, L. A., Krusche, A. V., Victoria, R. L., Camargo, P. B., Bernardes, M., Ferraz, E. S., et al. (1999). Effects of sewage on the chemical composition of Piracicaba River, Brazil. Water, Air and Soil Pollution, 110, 67–79.
Martinelli, L. A., Camargo, P. B., Bernardes, M. C., & Ometto, J. P. H. B. (2005). Carbon, nitrogen, and stable carbon isotope composition and land-use changes in rivers of Brazil. In E. J. Roose, R. Lal, C. Feller, B. Barthès, & B. A. Stewart (Eds.), Soil erosion and carbon dynamics (pp. 239–254). New York: Taylor & Francis Group.
Martins, S. C. (2010). Caracterização dos solos e serapilheira ao longo do gradiente altitudinal da Mata Atlântica, estado de São Paulo. PhD thesis, Universidade de São Paulo, Piracicaba.
McCune, B., & Mefford, M. J. (1997). PC-ORD: multivariate analysis of ecological data. Version 3.15. Gleneden Beach, Oregon: MjM Software Design.
McDowell, W. H., & Asbury, C. E. (1994). Export of carbon, nitrogen, and major ions from three tropical montane watersheds. Limnology and Oceanography, 39(1), 111–125.
Moreira-Turcq, P., Seyler, P., Guyot, J. L., & Etcheber, H. (2003). Exportation of organic carbon from the Amazon River and its main tributaries. Hydrological Processes, 17, 1329–1344.
Morellato, L. P. C., & Haddad, C. F. B. (2000). Introduction: the Brazilian Atlantic Forest. Biotropica, 32(4b), 786–792.
Mosier, A. R., Bleken, M. A., Chainwanakupt, P., Ellis, E. C., Freney, J. R., Howarth, R. B., et al. (2002). Policy implications of human-accelerated nitrogen cycling. Biogeochemistry, 57, 477–516.
Murray-Smith, C., Brummitt, N. A., Oliveira-Filho, A. T., Bachman, S., Moat, J., Lughadha, E. M. N., et al. (2009). Plant diversity hotspots in the Atlantic Coastal Forests of Brazil. Conservation Biology, 23, 151–163.
Myers, N., Mittermeier, R. A., Mittermeier, C. G., Fonseca, G. A., & Kent, J. (2000). Biodiversity hotspots for conservation priorities. Nature, 403, 853–858.
Neal, C., Jarvie, H. P., Neal, M., Hill, L., & Wickham, H. (2006). Nitrate concentrations in river waters of the upper Thames and its tributaries. Science of the Total Environment, 365, 15–32.
Neill, C., Deegan, L. A., Thomas, S. M., Haupert, C. L., Krusche, A. V., Ballester, V. M., et al. (2006). Deforestation alters the hydraulic and biogeochemical characteristics of small lowland Amazonian streams. Hydrological Processes, 20, 2563–2580.
Perakis, S. S., & Hedin, L. O. (2002). Nitrogen loss from unpolluted South American forests mainly via dissolved organic compounds. Nature, 415, 416–419.
Peterson, B. J., Wollheim, W. M., Mulholland, P. J., Webster, J. R., Meyer, J. L., Tank, J. L., et al. (2001). Control of nitrogen export from watersheds by headwater streams. Science, 292, 86–90.
Richey, J. E., Devol, A. H., Wofsy, S. C., Victoria, R., & Ribeiro, M. N. G. (1988). Biogenic gases and the oxidation and reduction of carbon in Amazon river and floodplain waters. Limnology and Oceanography, 33(4), 551–561.
Richey, J. E., Hedges, J. I., Devol, A. H., Quay, P. D., Victoria, R., Martinelli, L. A., et al. (1990). Biogeochemistry of carbon in the Amazon River. Limnology and Oceanography, 35(2), 352–371.
Salimon, C. I., & Negrelle, R. R. B. (2001). Natural regeneration in quaternary coast plain in southern Brazilian Atlantic Rain Forest. Brazilian Archives of Biology and Technology, 44, 155–163.
Salomão, M. S. M. B., Cole, J. J., Clemente, C. A., Silva, D. M. L., Camargo, P. B., Victoria, R. L., et al. (2008). CO2 and O2 dynamics in human-impacted watersheds in the state of São Paulo, Brazil. Biogeochemistry, 88, 271–283.
Sanchez, M., Pedroni, F., Leitão-Filho, H. F., & Cesar, O. (1999). Composição florística de um trecho de floresta riparia na Mata Atlântica em Picinguaba, Ubatuba, SP. Revista Brasileira de Botânica, 22(1), 31–42.
Seyler, P., Coynel, A., Moreira-Turcq, P., Etcheber, H., Colas, C., Orange, D., et al. (2005). Organic carbon transported by the Equatorial rivers: example of Congo-Zaire and Amazon Basins. In E. J. Roose, R. Lal, C. Feller, B. Barthès, & B. A. Stewart (Eds.), Soil erosion and carbon dynamics (pp. 255–274). New York: Taylor & Francis Group.
Skirrow, G. (1975). The dissolved gases—carbon dioxide. In J. P. Riley & G. Skirrow (Eds.), Chemical oceanography (pp. 1–192). London: Academic Press.
Solomon, D., Lehmann, J., Kinyangi, J., Amelung, W., Lobe, I., Pell, A., et al. (2007). Long-term impacts of anthropogenic perturbations on dynamics and speciation of organic carbon in tropical forest and subtropical grassland ecosystems. Global Change Biology, 13, 511–530.
StatSoft, Inc. (2007). Statistica (data analysis software system). Version 7.1. http://http://www.statsoft.com. Accessed 27 August 2007.
Suguio, K., & Tessler, M. G. (1984). Planícies de cordões litorâneos quaternários do Brasil: Origem e Nomenclatura. In L. D. Lacerda, D. S. Araújo, R. Cerqueira, & B. Turcq (Eds.), Restingas: Origem, Estrutura e Processos (pp. 15–25). Niterói: Centro Educacional Universidade Federal Fluminense (CEUFF).
Turgeon, J. M. L., & Courchesne, F. (2008). Hydrochemical behavior of dissolved nitrogen and carbon in a headwater stream of the Canadian Shield: relevance of antecedent soil moisture conditions. Hydrological Processes, 22, 327–339.
Vanderbilt, K. L., Lajtha, K., & Swanson, F. J. (2003). Biogeochemistry of unpolluted forested watersheds in the Oregon Cascades: temporal patterns of precipitation and stream nitrogen fluxes. Biogeochemistry, 62, 87–117.
Veloso, H. P., Rangel, A. L. R. F., & Lima, J. C. A. (1991). Classificação da vegetação brasileira adaptada a um sistema universal. Rio de Janeiro: Fundação Instituto Brasileiro de Geografia e Estatística.
Vieira, S. A., Alves, L. F., Aidar, M. P. M., Araújo, L. S., Baker, T., Batista, J. L. F., et al. (2008). Estimation of biomass and carbon stocks: the case of the Atlantic Forest. Biota Neotropica, 8, 21–29.
Villela, D. M., Nascimento, M. T., Aragão, L. E. O. C., & Gama, D. M. (2006). Effect of selective logging on forest structure and nutrient cycling in a seasonally dry Brazilian Atlantic forest. Journal of Biogeography, 33, 506–516.
Vink, S., Ford, P. W., Bormans, M., Kelly, C., & Turley, C. (2007). Contrasting nutrient exports from a forested and an agricultural catchment in south-eastern Australia. Biogeochemistry, 84, 247–264.
Vitousek, P. M. (1994). Beyond global warming: ecology and global change. Ecology, 75, 1861–1876.
Vitousek, P. M., Aber, J. D., Howarth, R. W., Likens, G. E., Matson, P. A., Schindler, D. W., et al. (1997). Human alteration of global nitrogen cycle: sources and consequences. Ecological Applications, 7(3), 737–750.
Wachniew, P. (2006). Isotopic composition of dissolved inorganic carbon in a large pollutes river: the Vistula, Poland. Chemical Geology, 233, 293–308.
Waterloo, M. J., Oliveira, S. M., Drucker, D. P., Nobre, A. D., Cuartas, L. A., Hodnett, M. G., et al. (2006). Export of organic carbon in run-off from an Amazonian rainforest blackwater catchment. Hydrological Processes, 20, 2581–2597.
Acknowledgements
This research was supported by the State of São Paulo Research Foundation (FAPESP), process number 05/57812-0, and also as part of the Thematic Project Functional Gradient (process number 03/12595-7), within the BIOTA/FAPESP Program–The Biodiversity Virtual Institute (www.biota.org.br).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Andrade, T.M.B., Camargo, P.B., Silva, D.M.L. et al. Dynamics of Dissolved Forms of Carbon and Inorganic Nitrogen in Small Watersheds of the Coastal Atlantic Forest in Southeast Brazil. Water Air Soil Pollut 214, 393–408 (2011). https://doi.org/10.1007/s11270-010-0431-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-010-0431-z