Abstract
Dominant canopy tree species have strong effects on the composition and function of understory species, particularly bryophytes. In boreal forests, bryophytes and their associated microbes are a primary source of ecosystem nitrogen (N) inputs, and an important process regulating ecosystem productivity. We investigated how feather moss-associated N2-fixation rates and contribution to N budgets vary in time and space among coniferous and broadleaf deciduous forests. We measured N2-fixation rates using stable isotope (15N2) labeling in two moss species (Pleurozium schreberi and Hylocomium splendens) in broadleaf deciduous (Alaska paper birch—Betula neoalaskana) and coniferous (black spruce—Picea mariana) stands near Fairbanks, interior Alaska, from 2013 to 2015. N2-fixation rates showed substantial inter-annual variation among the 3 years. High N2-fixation was more strongly associated with high precipitation than air temperature or light availability. Overall, contribution of N2-fixation to N budgets was greater in spruce than in birch stands. Our results enhance the knowledge of the processes that drive N2-fixation in boreal forests, which is important for predicting ecosystem consequences of changing forest composition.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Boreal forests of the world are important carbon (C) sinks (DeLuca and Boisvenue 2012), making the productivity of these ecosystems of global importance in regulating atmospheric carbon dioxide (CO2) concentrations (Bonan et al. 1992). Productivity in boreal ecosystems is often limited by nitrogen (N) availability (Tamm 1991; Vitousek and Howarth 1991). Biological N2-fixation (BNF) by various bacteria, such as free-living and symbiotic cyanobacteria, is a major source of N in many ecosystems (Cleveland et al. 1999). Associations between mosses and N2-fixing cyanobacteria have been identified as a source of ecosystem N inputs in mature Scandinavian boreal ecosystems (DeLuca et al. 2002). There, they contribute up to 2–4 kg N ha−1 year−1 (DeLuca et al. 2007; Lagerström et al. 2007), a source of N that is comparable to atmospheric N deposition (Sponseller et al. 2016). Most of this newly fixed N is thought to remain within the moss layer (Hyodo and Wardle 2009; Gavazov et al. 2010) but may become available to vascular plants through mycorrhizae (Parke and Linderman 1980; Carleton and Read 1991).
In boreal and Arctic ecosystems, feather mosses such as Pleurozium schreberi (Brid.) Mitt. and Hylocomium splendens (Hedw.) Schimp. host N2-fixing cyanobacteria on their leaves and stems (DeLuca et al. 2002; Ininbergs et al. 2011). Abiotic conditions are important drivers of BNF by moss-cyanobacteria associations (Rousk and Michelsen 2017). BNF depends on favorable temperatures, moisture, and light conditions (Chapin et al. 1991; Zielke et al. 2005; Rousk et al. 2014; Gundale et al. 2012a, b). BNF is an energetically demanding process, so it is low at sites with high N availability (DeLuca et al. 2008; Gundale et al. 2009). The contribution of P. schreberi’s cyanobacteria communities to N fluxes and pools in Scandinavian boreal forests is well established and the contribution of cyanobacteria on H. splendens is increasingly recognized as substantial (DeLuca et al. 2002; Zackrisson et al. 2004, 2009). Feather mosses are prevalent in the understory of mature boreal forests around the world (Oechel and Van Cleve 1986) and they perform important ecosystem processes because of their high net primary productivity, production of recalcitrant litter, moisture retention, and soil insulation (Turetsky 2003; Turetsky et al. 2012; Lindo et al. 2013).
Most of the work on moss-associated BNF in boreal forests has focused on Fennoscandia’s coniferous boreal forests with an extensive moss cover (e.g., DeLuca et al. 2002; Zackrisson et al. 2004, 2009). However, boreal forests are composed of both coniferous and broadleaf deciduous stands. In the Alaskan boreal forest, coniferous black spruce (Picea mariana Mill. B.S.P.) forests occupy about 40% of the forested landscape and pure deciduous forests occupy about 20% (Calef et al. 2005). Canopy composition is a major driver of moss abundance (Hart and Chen 2006) and has the potential to affect BNF. In interior Alaska, mature black spruce forests (> 40 years old) have a moss cover of over 80%, mostly dominated by feather mosses, while moss cover in deciduous forests in only about 5% (Jean et al. 2017). Broadleaf deciduous stands tend to have warmer and drier soils in the summer and higher N availability than black spruce forests (Melvin et al. 2015). In addition, larger aboveground N pools are found in deciduous stands, while black spruce stands have larger belowground N pools (Melvin et al. 2015; Alexander and Mack 2016). Forests in interior Alaska have a colder and drier climate than boreal Fennoscandia, are regularly affected by large stand-replacing disturbances such as fires (ACIA 2005), and have comparably low N deposition rates (Gundale et al. 2011); these factors may also alter the patterns and ecosystem contributions of moss-associated BNF. Strong spatial (among sites and canopy types), and temporal (among years or months) variability in BNF is to be expected, but how much this variability may affect the overall contribution of moss-cyanobacteria to stand-level N pools is unclear.
Our objective was to characterize the stand-level N contribution of BNF associated with P. schreberi and H. splendens (hereafter referred to by their generic names) in mature Alaska paper birch (Betula neoalaskana Sarg.) and black spruce stands in interior Alaska. Measurements across multiple years and replicate stands provided estimates of how moss-associated BNF rates varied over space and time. Additionally, we related environmental and climatic variables, such as temperature, moisture, light, and N availability, to BNF to understand the mechanisms driving the observed patterns. If rates of BNF per unit moss biomass are primarily limited by light, moisture, or high nutrient availability, then rates should be higher in spruce versus birch stands, as spruce stands have less canopy shading, stable and high moisture levels, and lower available N. In contrast, if temperature limits BNF rates, then the highest rates should be observed in birch stands, where surface temperatures are warmer. At the stand level, N2 fixed per unit area should be higher in spruce than in birch stands, as mosses are more abundant. If yearly or seasonal variation in weather (mostly temperature and precipitation) are driving fixation rates, then variation in those variables should override canopy or moss species effects. Earlier estimates of moss-associated BNF in black spruce-feather moss stands of Alaska (Billington and Alexander 1978, 1983) used the acetylene reduction method (Hardy et al. 1973), and we expect to obtain more sensitive and robust measures using stable isotope labeling to measure BNF rates. Results of this study will give updated estimates of how feather moss-associated BNF contributes to N cycling among deciduous and coniferous boreal forests of interior Alaska.
Methods
Study sites
Our study area was in the Tanana Valley State Forest near Fairbanks, Alaska (64°53N, 148°23W). We established three sites or blocks (A, B, and C) of paired, adjacent stands of mature black spruce and Alaska paper birch (hereafter called spruce and birch) located on a northeast-facing slope that burned in a 1958 fire. Sites A and B were typical of well-drained upland forests, while site C was close to a riparian area with higher soil moisture. This region experiences a cold, continental climate, with a mean annual air temperature of − 2.5 °C, mean annual precipitation of 286 mm (65% as rain), and a growing season of about 135 days (Hinzman et al. 2005).
We installed one weather station (Onset Hobo H21-002) in each stand (n = 6). Weather stations measured soil temperature (S-TMB-M002) and moisture (S-SMD-M005) at 10 cm below the soil surface, and air temperature and relative humidity (S-THB-M002), and photosynthetic active radiation (PAR; S-LIA-M003) at 1.75 m above ground level. Canopy cover was determined by analyzing hemispherical pictures taken 30 cm above the moss in 2014 with Gap Light Analyzer (Frazer et al. 1999; n = 144). Five cores were extracted per stand in 2012 to assess soil characteristics, as well as pools and fluxes of C and nutrients (Melvin et al. 2015).
Moss cover averaged 5% in birch and nearly 100% in spruce stands (visual estimates conducted June 27, 2014 in six 4 m2 plots equally placed along a 60-m transect in each stand). Hylocomium and Pleurozium were the most common bryophytes (Online Resource Table S1) and examination of a few samples revealed the presence of the cyanobacteria genera Nostoc and Anabaena, both with heterocysts. Other taxa known to host N2-fixing bacteria (Van Cleve et al. 1971; Basilier 1980) were present in low amount in some of the stands: Sphagnum spp. and alder (Alnus spp.). Deciduous leaf litter (primarily birch leaves) dominated the forest floor in birch stands (94%) and was present in spruce stands (26%). Additional site description can be found in Melvin et al. (2015), Jean (2017), and Online Resource Table S1.
15N2 incubation assays
The sampling design differed slightly every year from 2013 to 2015 (Table 1). Different moss patches (30 cm diameter) were sampled each year, but the same patches were used within the seasonal 2014 sampling. In 2013, five monospecific Pleurozium or Hylocomium moss patches were selected in the spruce and birch stands from sites A and B. BNF rates were measured in late July on the top 5–8 cm of 20 Pleurozium shoots or 10 Hylocomium shoots per patch (Table 1). In 2014, 12 monospecific moss patches (n = 12 patches per species per stand) were selected along a 60-m transect in each stand of sites A, B, and C. BNF rates were measured in site A in June, July, August, and September 2014 (Table 1), and in sites B and C in August (Table 1). In August 2015, BNF rates were measured in five Hylocomium-dominated patches that also contained Pleurozium in sites A and B (n = 5 patches per stand, Table 1); therefore, both moss species were sampled within the same patch (Jean 2017). In 2014 and 2015, shoots were collected at a length of 5 cm from the apical meristem and BNF rates were measured on 10 Pleurozium or Hylocomium shoots per patch. Incubated samples included both green and brown moss parts.
During incubation trials, we watered moss patches with rainwater 24 h and immediately before the start of the incubation to ensure minimum water availability. Control mosses were collected to measure natural 15N abundance from 2 to 10 shoots per moss species from each patch in each sampling period (5–8 cm in 2013 and 5 cm in 2014–2015). Enriched moss samples were incubated 24 h on their patch of origin in 60-ml translucent polycarbonate syringes, each filled with 10 ml of air and 10 ml of 15N2 (98% enriched, Cambridge Isotope Laboratories Inc., U.S.A.). After incubation, enriched and control samples were weighed wet, dried for 72 h at 60 °C, reweighed, and finely ground. Nitrogen and carbon concentrations and 15N and 13C at% values were determined using an Elemental Analyser coupled to a Continuous-flow Isotope Ratio Mass Spectrometer (University of Florida in 2013 and Northern Arizona University in 2014–2015). BNF was calculated by comparing the δ15N values from enriched and control samples (detailed calculations in Online Resource Table S2). In July 2014, we tested if watering biased estimates of BNF rates by incubating both dry and wet mosses (pooled data from both moss species and forest types in site A; n = 24 for each). Average moss moisture was 40.6% ± 5.4 (mean ± SE) in the non-watered mosses and 76.9% ± 3.8 in the watered treatment but did not affect BNF rates (paired t test: F1,23 = − 1.736, P = 0.096). To extrapolate measurements of BNF to potential N fixed per unit area, we used moss percent cover and excavated five 10-cm-diameter samples dominated by each moss species in each stand to calculate shoots density and dry weight. Depth of the green moss was about 2.2 cm ± 0.2 in spruce and 1.9 cm ± 0.2 in birch stands, but individual moss shoots could be green up to 5–8 cm in length since both moss species have a pleurocarpous growth form. For shoots of similar length, Pleurozium was half to a third the weight and 85% of the volume of Hylocomium (Online Resource Table S1, Table S2).
Data analysis
We investigated inter-annual variation (2013, 2014, and 2015; Table 1) in BNF rates among forest types and moss species using a three-way mixed-model analysis of variance (ANOVA) including all interactions between forest type, moss species, and year of sampling and sites (A and B) as a random effect. Intra-annual BNF rates were compared within the 2014 growing season (June, July, August, and September in site A; Table 1) using a mixed-effect model to account for repeated measures on each moss patch (library lmerTest; Kuznetsova et al. 2015). We used a three-way ANOVA (forest type, moss species, and site) with data from August 2014 to assess spatial variation between sites in BNF rates. In this case, sites are not used as replicates, but as a fixed effect in order to assess how site selection may affect measured BNF rates. We believe that presenting site-to-site variability in BNF rates may stimulate further hypotheses, even if it diverges from our initial sampling design. We interpret those results with caution. We used Tukey honest significant difference (HSD) tests to compare among groups when significant effects were found (P < 0.05). In all analyses, BNF rates were log transformed to meet assumptions of normality and homoscedasticity, and R software was used for statistical calculations (R Development Core Team 2016).
We used a structural equation model (SEM; Grace 2006) to estimate direct and indirect effects of environmental variables on BNF patterns (lavaan package; Rosseel 2011). Data from all years, months, and sites were pooled in a single SEM (n = 363), and we used environmental covariates to account for shared variance associated with spatial and temporal pseudoreplication. We used the natural logarithm of BNF to meet the SEM assumption of linear bivariate relationships, while other relationships between variables were largely linear. Numerical variables were standardized, and we used bootstrapping with 1000 iterations. Additional paths were added individually to our a priori model (Fig. 1) based on the highest modification indices if they made ecological sense (Grace et al. 2012). We determined if the SEM was a good fit by using a Chi-square test (P > 0.05), the root mean square error of approximation (RMSEA with lower 90% confidence intervals close to zero), and the comparative fit index (CFI > 0.90; Grace et al. 2012).
Covariates used in the SEM included three climate variables (average air temperature, average light availability and total precipitation 30 days prior to incubations), a variable representing variation among sites in nutrient availability (N mineralization rate; Melvin et al. 2015), and categorical variables of forest type and moss species. Air temperature was selected over soil temperature, as it was more similar to temperatures at the moss layer surface. Air temperature and light availability were obtained from our six weather stations (one value per stand per sampling period), while total precipitation was acquired from the Fairbanks airport database (ACRC 2016). Monthly averages were used because recovery of moss-associated BNF rates from drought stress can be on the order of hours (Rousk et al. 2014) to weeks (Gundale et al. 2012b). Our a priori model included all direct effects on BNF as well as indirect effects of forest type on temperature, light and N availability, and correlations between light, temperature, and precipitation (Fig. 1).
Stand-level fixation rates and N pools
We used published estimates of ecosystem N sources and requirements from our sites or similar forest stands in interior Alaska (Table 2, Online Resource Table S3). Total N requirements were calculated from the literature by adding N required for annual net primary productivity (ANPP) of trees (foliage, wood, bark, and fine roots) and feather mosses and subtracting N losses in plant litter. N sources were calculated by adding net N mineralization and nitrification, BNF by vascular plants, and atmospheric wet and dry N deposition. Boreal plants can absorb organic N forms (Näsholm et al. 1998), but this was not included in our calculations. N sources accounted for 81 and 133% of N requirements in spruce and birch stands, respectively (Table 2). Estimates of total N losses summed to 0.27 kg ha−1 year−1 in spruce and 0.18 kg ha−1 year−1 in birch, including N leaching (Van Cleve et al. 1983), denitrification (Sponseller et al. 2016), and riverine exports of N (Jones et al. 2005). This approach is similar to what has been done in Stuiver et al. (2015) and, while we did not include ANPP from shrubs, forbs, and grasses, we obtained similar rates of N requirements for coniferous forests of 3.26 kg N ha−1 year−1 in our spruce stands vs. 4.4 kg N ha−1 year−1 in managed Swedish coniferous forests.
We calculated seasonal (June–September) stand-level N2-fixation to assess contributions of moss-associated BNF to N cycling in boreal Alaska using median BNF rates (µg N g dry moss−1 day−1), a well as mean moss cover (%), moss shoot density (stems cm−2), and moss weight per shoot (g dry moss shoot−1). Median BNF was used instead of mean since outliers skewed the distribution. We estimated daily BNF for June to September using measurements made in each month in 2014 and ratios of monthly rates for 2013 and 2015. We estimated N2-fixation for all years in sites A and B and only 2014 in site C. Since it was not logistically possible to measure BNF at all sites and all points in time, we made the assumption that ratios of monthly BNF rates remained the same across years and sites. This assumption relies on the facts that seasonal trends in N2-fixation remain very similar across years and study areas in boreal Scandinavia (e.g., DeLuca et al. 2002, Zackrisson et al. 2004, 2009) and that no seasonal trends are available for interior Alaska other than the data we collected in 2014.
Results
Environmental conditions
The summer of 2014 was unusually wet (370.6 mm rain), while 2013 and 2015 were closer to average rainfall (143.2 mm and 207.5 mm, respectively). Over 5 million acres burned in the summer 2015, resulting in the second-worst fire year on record in Alaska (AICC 2015). Growing season air temperature was similar in both forest types but warmer in 2013 (14.1 °C) and 2015 (14.1 °C) than in 2014 (11.9 °C) (Online Resource Table S1). Soil temperatures were lower in spruce (4.1 °C) than in birch stands (7.7 °C) and remained relatively stable among years. Summer PAR was higher in spruce than in birch stands (384.0 vs. 145.1 µmol m−2 s−1), as canopy cover was lower (69% vs. 85% in birch stands). Soil organic layers were shallower and pH, N mineralization and nitrification rates, and phosphorus (P) availability were higher in birch than in spruce stands, and higher in site C than in site A or B (Online Resource Table S1, Melvin et al. 2015).
Isotopic composition of samples
Natural variation in the range of δ15N was relatively small across years, forest types, moss species and sites (− 2.05‰ ± 0.13 (mean ± SE, n = 71), range − 3.81 to 2.26‰) when compared to values observed in 15N-enriched samples (δ15N = 1043.30‰ ± 51.51 (n = 362), range − 4.15 to 7,561.17‰). Overall, natural δ15N was similar among moss species, averaging − 2.09‰ ± 0.15 (n = 43) for Hylocomium and − 2.30‰ ± 0.12 (n = 44) for Pleurozium, but higher in birch (− 2.01‰ ± 0.14 (n = 42)) than in spruce (− 2.38‰ ± 0.13 (n = 45)) stands, especially in 2015 (Online Resource Table S4).
Inter-annual variation (2013–2015)
Rates of BNF per unit moss biomass varied among years, stand types, and moss species (Fig. 2, Table 3). Sampling year explained about 84% of total variation in BNF rates (P < 0.0001). BNF rates were highest in the rainy summer of 2014 (79.9 ± 4.5) and similar in 2013 (5.2 ± 1.6) and 2015 (1.7 ± 0.5 μg N fixed g dry moss−1 day−1). The impact of forest type and moss species on BNF rates varied according to the year of sampling. BNF rates were higher in spruce than in birch stands in 2013 and 2014, but not in 2015 (P < 0.0001; Table 3). BNF rates were higher in Hylocomium in 2013, higher in Pleurozium in 2014, and, while rates were slightly higher for Hylocomium, the difference among species was not significant in 2015 (P = 0.0011; Table 3; Fig. 2).
N2-fixation rates (μg N g dry moss−1 day−1) of Hylocomium splendens and Pleurozium schreberi in birch and spruce stands from sites A and B (pooled) in late July-early August 2013 (n = 10, 5 per species per forest type per site), 2014 (n = 24, 12 per species per forest type per site), and 2015 (n = 10, 5 per species per forest type per site). While pooled data are presented here, statistical analyses included a random site effect. Medians are indicated by the black horizontal lines, boxes include 25–75% quantiles, and whiskers include 5–95% of the data. Outliers are shown as circles. Lowercase letters indicate forest-type differences across years and uppercase letters indicate moss species differences across years. Different letters indicate that the treatments were significantly different (α = 0.05) following a Tukey HSD posthoc test. Note the log scale of the y-axis
Intra-annual variation (June–September)
Rates of BNF per unit moss biomass changed significantly throughout the growing season (P < 0.0001, Fig. 3, Table 4) and followed weather trends. Air temperatures and precipitation were highest in July (16.1 °C, 147 mm), lowest in September (8.0 °C, 73 mm), and similar between June (14.7 °C, 90 mm) and August (15.0 °C, 58 mm; ACRC 2016). Overall, BNF rates increased from June to August, then declined in September, except for Pleurozium in birch stands, which peaked in July. BNF rates remained higher for Pleurozium than Hylocomium throughout the growing season (P < 0.0001, Table 4), and higher in spruce than in birch stands (P < 0.0001). BNF rates remained low in birch stands throughout the growing season, with higher rates in July and August than June or September (Fig. 3), while in spruce stands BNF rates were highest in August, lowest in June, and intermediate in July and September. BNF rates averaged across 2014 were lowest for Hylocomium in birch stands, similar between Pleurozium in birch stands and Hylocomium in spruce stands, and highest for Pleurozium in spruce stands (P = 0.0007, Fig. 3, Table 4, Tukey HSD P < 0.05).
N2-fixation rates (μg N g dry moss−1 day−1) for Hylocomium splendens and Pleurozium schreberi in spruce and birch stand in June, July, August, and September 2014 (site A only, n = 12). Each point represents one sample. Curves and shadings represent a LOESS (locally weighted scatterplot smoothing) curves with a smoothing parameter of 1 for each series of points and 95% confidence interval. Different letters indicate that the forest type and month groups were significantly different following a Tukey HSD posthoc test (α = 0.05). Letters above the points are associated with spruce and letters below are associated with birch stands. Note the log scale of the y-axis
Between-site variation in N2-fixation
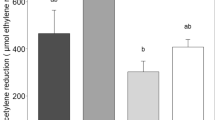
BNF rates differed among moss species, with rates in Pleurozium higher than those in Hylocomium across all sites and forest types (P < 0.0001, Fig. 4, Table 5). Fixation rates were overall higher in spruce stands than in birch stands (P < 0.0001); however, this difference was driven by sites A and B. In birch stands, fixation rates were lowest in site A and highest in site C, while in spruce stands, fixation rates were highest in site A and lowest in site C (P < 0.0001, Fig. 4, Table 5). This pattern was similar to that of N availability, which was higher in birch than in spruce stands, and higher in site C than in sites A and B (Online Resource Table S1). These results indicate that site selection may affect measured differences in BNF rates among forest types.
N2-fixation rates (μg N g dry moss−1 day−1) for Hylocomium splendens and Pleurozium schreberi measured in August 2014 in birch and spruce stands in the 3 sites (n = 12). Different letters indicate a significant difference following a Tukey HSD posthoc test (α = 0.05). Note the log scale of the y-axis
Relation to environmental variables
In the final SEM model (Fig. 5, Online Resource Table S5), the direct effects of moss species and forest types showed higher rates of BNF in spruce than in birch stands, and higher rates in Pleurozium than Hylocomium. BNF was positively associated with precipitation, and negatively with air temperature and light. Forest type affected light availability, which was higher in spruce stands, but did not influence air temperature. N mineralization rates were lower in spruce than in birch stands; once this was accounted for, BNF was positively associated with N mineralization rates. The strong direct path between forest type and BNF rates indicates that other stand-associated variables that were either not measured or not measured at the right scale may also be important drivers of BNF (e.g., moss moisture regime, Fig. 5).
Fitted pathways of a structural equation model of N2-fixation rates (log transformed values, μg N g dry moss−1 day−1). The model fits the data, which included all measurements across forest types, moss species, years, months and sites (n = 363, χ2 = 8.151, 7 degrees of freedom, P = 0.319, RMSEA 90% CI 0.00–0.079, CFI = 0.998). Black lines represent significant regression pathways; solid lines are positive pathways, and dashed lines are negative pathways. Path coefficients are the standardized prediction coefficients from the SEM model with the width of the arrow indicating the magnitude. Significant correlations are indicated by gray double-headed arrows and their coefficients are found in Online Resource Table S5. R2 values are included for all significant endogenous variable pathways
BNF contribution to N budgets
Spruce stands exhibited both high moss cover and high rates of BNF, leading to higher total N2 fixed per unit ground area than in birch stands (Table 6). In the drier 2013 and 2015 years, Hylocomium-associated BNF per unit ground area was dominant, while Pleurozium-associated BNF dominated in the wet year of 2014. BNF rates and total amount of N2 fixed in 2014 were surprisingly high and orders of magnitude larger than in the other two years. Spatial variation among sites in 2014 was smaller (2 to 10-fold, Table 7) than that of the inter-annual variation (46 to 317-fold). The highest amount of fixed N was in the site A spruce stand, which had the highest BNF rates, as well as the highest moss cover and biomass per unit area (Table 7).
The proportion of moss N requirements that could be met by moss-associated N2-fixation varied annually, with values ranging from 2 to 200% in birch stands, and 11 to 429% in spruce stands (Table 6). BNF accounted between 0.01 and 1% of the annual stand-level N requirements in birch stands, in contrast to 2 to 92% in spruce stands. There, N inputs from moss-cyanobacteria associations have the potential to be larger than combined BNF from vascular plants and dry and wet N deposition in a ‘typical’ year (e.g., 2013 and 2015), and sometimes larger than mineralization and nitrification (Table 2), as in the spruce stand of site A in 2014. The amount of N2 fixed by both moss species in the spruce and birch stands from sites A and B in 2014 equaled or exceeded the annual N requirements of feather moss (Table 7). The low abundance of mosses in birch stands meant that the contribution of moss BNF to stand-level N budget was negligible in all three sites. In spruce stands, the contribution to stand-level N requirements of both moss species ranged from 13 to 44% (Table 7).
Discussion
BNF by feather moss-cyanobacteria associations has the potential to be a source of N equal or larger than all other N sources combined in the mature black spruce stands of this study. BNF rates were generally higher in stands dominated by black spruce than in stands of Alaska paper birch for both moss species, although they were low in all stand types in 2015. Environmental factors specific to each forest type are likely important drivers of variation in BNF rates by feather moss-cyanobacteria associations. Our results support the idea that N inputs from feather moss-cyanobacteria associations contribute significantly to N pools in interior Alaska, particularly in black spruce stands where feather mosses are abundant, while their contribution is negligible in deciduous birch stands. Additionally, this study highlights the importance of stochastic factors associated with weather conditions, as year and month of sampling can override the effects of canopy, moss species, and micro-environmental variables on BNF rates.
Boreal forests have large stocks of organic N in their soils (Van Cleve and Alexander 1981), yet plant productivity is often limited by N turnover and availability to plants and microbial symbionts (Tamm 1991; Vitousek and Howarth 1991). We found that the amount of N2 fixed by feather mosses in upland spruce stands was extremely variable among years, contributing up to 92.1% of the stand-level (trees and mosses) N requirements in 2014, 10.4% in 2013, and 2.4% in 2015. These 2013 and 2015 values were comparable to the reported 9.1% contribution of feather mosses to N budgets in managed coniferous forests in Sweden (Stuiver et al. 2015). Feather moss contribution to N budget in birch stands was minor, ranging between 0.01 and 0.63%. The average contribution of both feather moss species over the three years was similar, about 5.7 kg N ha−1 year−1, but the contribution of Hylocomium was larger in 2013 and 2015 and that of Pleurozium was larger in 2014. BNF contributions by feather mosses have been reported to range between 0.01 to 7 kg N ha−1 year−1 in boreal, mostly coniferous, stands (Lindo et al. 2013). BNF rates measured during the wet year of 2014 in spruce stands were much higher (averaging 14 to 44 kg N ha−1 year−1) and exceeded feather moss N requirements. It is still unclear what happens to the fixed N2 and how much of it is trapped in recalcitrant moss litter, transferred to other ecosystem components through mycorrhizal fungi (Carleton and Read 1991; Lagerström et al. 2007), or leached following drying and rewetting events (Carleton and Read 1991; Rousk et al. 2014).
Years were the largest source of variation in BNF rates, with 2014 rates being 9-fold higher than in 2013, and 38-fold higher than in 2015. This pattern of inter-annual variation was associated with precipitation; 2014 received twice the amount of rain of 2013 or 2015 and twice the Fairbanks long-term average (Hinzman et al. 2005). BNF rates also varied through the 2014 growing season, with a peak in fixation rates occurring late-July and early-August for both moss species and in both forest types. This pattern is more similar to reports from the Arctic (Alexander and Schell 1973; Chapin et al. 1991) than to the peaks in June and September found in boreal Scandinavia (DeLuca et al. 2002; Zackrisson et al. 2004). Moss moisture was high (70–95%) in July and August 2014 and may not have been limiting. Variation in BNF rates between sites and forest types was smaller than the inter- and intra-annual variation, suggesting that factors that vary annually, such as weather, are more important in controlling BNF rates than factors that vary in space.
Abiotic conditions, such as temperature, light availability, N availability, and precipitation were associated with BNF patterns. Numerous studies have found a positive impact of temperature on BNF by moss-associated cyanobacteria (e.g., Zielke et al. 2002; Gundale et al. 2012a). Conversely, we found that air temperature (12–16 °C) had a negative association with BNF, possibly because of a decrease in ambient moisture with increased temperatures (Rousk et al. 2015). Our data also did not support a strong dependence of BNF on light availability and moss photosynthesis (Sorensen et al. 2012; Gundale et al. 2012b), possibly because light availability was relatively high across all samples. We found the strongest evidence for moisture (e.g., Gundale et al. 2009; Jackson et al. 2011; Gundale et al. 2012b) in controlling rates of BNF in this part of the boreal forest, as inter- and intra-annual patterns of fixation rates were associated with precipitation patterns.
BNF is an energetically expensive process and rates of BNF and colonization by cyanobacteria are known to decrease under high N availability (Zackrisson et al. 2004; DeLuca et al. 2008; Gundale et al. 2011). Moss-associated BNF is predicted to decrease above N input thresholds of 10.25–12.25 kg N ha−1 year−1 (Zackrisson et al. 2004; Ackermann 2013). N inputs in the birch stands of this study (Table 2) well exceeded this threshold and may explain their lower BNF rates. Low BNF rates in the spruce stand of site C also support a negative effect of N availability, as this site had the highest N mineralization rates in our study. Surprisingly, N availability was positively associated with BNF in our SEM model. However, the low power of our sample design to estimate effects of site-to-site variation (with only one year of measurements across all three sites) means these effects must be considered inconclusive. Nevertheless, these suggest that further research is needed to assess how site variability may affect BNF, even in a small study area. Spatial variation in BNF may have also been affected by other unmeasured factors, such as abundance and colonization of mosses by cyanobacteria (Whiteley and Gonzalez 2016), molybdenum or iron availability (van Groenigen et al. 2006; Rousk et al. 2017), and trophic interactions within the bryosphere (Kardol et al. 2016).
Conclusion
BNF by feather moss-cyanobacteria associations appears to be a significant contributor to N cycling in interior Alaska’s black spruce forests, while it plays a negligible role in birch forests. BNF rates were strongly dependent on the year of sampling and varied with annual precipitation. Moss species, canopy type, and seasonal variation all influenced BNF rates. Fixation rates were higher in spruce stands, in late summer. In birch stands, fixation rates remained low through the summer. Differences in BNF rates among moss species depended on the year; across years, both Pleurozium and Hylocomium contributed similarly to BNF. Our results point towards the importance of moisture as a driver of temporal variation in BNF in interior Alaska. Stand type and associated changes in moss abundance strongly influence spatial variations in stand-level BNF. In this context, changes in climate (e.g., change in precipitation regime) or patterns of forest dominance are likely to significantly alter the dynamics of BNF by moss-cyanobacteria associations. For example, increases in summer precipitation are likely to increase BNF rates and contributions to N budgets, while increases in birch forest cover would reduce landscape-scale contribution of BNF to N cycling.
References
ACIA (Arctic Climate Impact Assessment) (2005) Impacts of a warming arctic. Cambridge University Press, Cambridge
Ackermann K (2013) Nitrogen dynamics in boreal forests: a feather moss’ perspective. Dissertation, Bangor University
Alaska Climate Research Center (ACRC) (2016) Fairbanks annual weather review. Alaska Climate Research Center. http://akclimate.org/. Accessed 12 Feb Dec 2016
Alaska Interagency Coordination Center (AICC) (2015). 2015 Alaska wildfire by area and protection level. Alaska Department of Natural Resources. http://forestry.alaska.gov/. Accessed 15 Dec 2015
Alexander HD, Mack MC (2016) A canopy shift in interior Alaskan boreal forests: consequences for above- and belowground carbon and nitrogen pools during post-fire succession. Ecosystems 19:98–114
Alexander V, Schell DM (1973) Seasonal and spatial variation of nitrogen fixation in the Barrow, Alaska, Tundra. Arct Alp Res 5:77–88
Basilier K (1980) Fixation and uptake of nitrogen in Sphagnum blue-green algal associations. Oikos 34:239–242
Billington M, Alexander V (1978) Nitrogen fixation in a black spruce (Picea mariana [Mill] B.S.P.) forest in Alaska. Ecol Bull 26:209–215
Billington MM, Alexander V (1983) Site-to-site variation in nitrogenase activity in a subarctic black spruce forest. Can J For Res 13:782–788
Bonan GB, Pollard D, Thompson SL (1992) Effects of boreal forest vegetation on global climate. Nature 359:716–718
Calef MP, McGuire AD, Epstein HE, Rupp TS, Shugart HH, Chapin FS (2005) Analysis of vegetation distribution in Interior Alaska and sensitivity to climate change using a logistic regression approach. J Biogeogr 32:863–878
Carleton TJ, Read DJ (1991) Ectomycorrhizas and nutrient transfer in conifer-feather moss ecosystems. Can J Bot 69:778–785
Chapin DC, Bliss LC, Bledsoe LJ (1991) Environmental regulation of nitrogen fixation in a high Arctic lowland ecosystem. Can J Bot 69:2477–2755
Cleveland CC, Townsend AR, Schimel DS, Fisher H, Howarth RW, Hedin LO, Perakis SS, Latty EF, Von Fischer JC, Hlseroad A, Wasson MF, Elseroad A, Wasson MF (1999) Global patterns of terrestrial biological nitrogen (N2) fixation in natural ecosystems. Global Biogeochem Cycles 13:623–645
DeLuca TH, Boisvenue C (2012) Boreal forest soil carbon: distribution, function and modelling. Forestry 85:161–184
DeLuca TH, Zackrisson O, Nilsson M-C, Sellstedt A (2002) Quantifying nitrogen-fixation in feather moss carpets of boreal forests. Lett Nat 419:917
DeLuca TH, Zackrisson O, Gentili F, Sellstedt A, Nilsson M-C (2007) Ecosystem controls on nitrogen fixation in boreal feather moss communities. Oecologia 152:121–130
DeLuca TH, Zackrisson O, Gundale MJ, Nilsson M-C (2008) Ecosystem feedbacks and nitrogen fixation in boreal forests. Science 320:1181
Development Core Team R (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Frazer GW, Canham CD, Lertzman KP (1999) Gap Light Analyzer (GLA). Cary Institute of Ecosystem Studies, Millbrook
Gavazov KS, Soudzilovskaia NA, van Logtestijn RSP, Braster M, Cornelissen JHC (2010) Isotopic analysis of cyanobacterial nitrogen fixation associated with subarctic lichen and bryophyte species. Plant Soil 333:507–517
Grace JB (2006) Structural equation modeling and natural systems. Cambridge University Press, Cambridge
Grace JB, Schoolmaster DR, Guntenspergen GR, Little AM, Mitchell BR, Miller KM, Schweiger EW (2012) Guidelines for a graphtheoretic implementation of structural equation modeling. Ecosphere 3:1–44
Gundale MJ, Gustafsson H, Nilsson M-C (2009) The sensitivity of nitrogen fixation by a feathermoss-cyanobacteria association to litter and moisture variability in young and old boreal forests. Can J For Res 39:2542–2549
Gundale MJ, Deluca TH, Nordin A (2011) Bryophytes attenuate anthropogenic nitrogen inputs in boreal forests. Glob Chang Biol 17:2743–2753
Gundale MJ, Nilsson M, Bansal S, Jäderlund A (2012a) The interactive effects of temperature and light on biological nitrogen fixation in boreal forests. New Phytol 194:453–463
Gundale MJ, Wardle DA, Nilsson M-C (2012b) The effect of altered macroclimate on N-fixation by boreal feather mosses. Biol Lett 8:805–808
Hardy RWF, Burns RC, Holsten RP (1973) Applications of the acetylene-ethylene assay for measurement of nitrogen fixation. Soil Biol Biochem 5:47–48
Hart SA, Chen HYH (2006) Understory vegetation dynamics of North American boreal forests. Crit Rev Plant Sci 25:381–397
Hinzman LD, Bettez ND, Bolton WR, Chapin FS, Dyurgerov MB, Fastie CL, Griffith B, Hollister RD, Hope A, Huntington HP, Jensen AM, Jia GJ, Jorgenson T, Kane DL, Klein DR, Kofinas G, Lynch AH, Lloyd AH, McGuire AD, Nelson FE, Oechel WC, Osterkamp TE, Racine CH, Romanovsky VE, Stone RS, Stow DA, Sturm M, Tweedie CE, Vourlitis GL, Walker MD, Walker DA, Webber PJ, Welker JM, Winker KS, Yoshikawa K (2005) Evidence and implications of recent climate change in Northern Alaska and other Arctic regions. Clim Change 72:251–298
Hinzman LD, Bolton RW, Petrone KC, Jones JB, Adams PC (2006) Watershed hydrology and chemistry in the Alaskan boreal forest: the central role of permafrost. In: Chapin FS, Oswood MW, Van Cleve K, Viereck LA, Verbyla DL (eds) Alaska’s changing boreal forest. Oxford University Press, New York, pp 269–284
Hyodo F, Wardle DA (2009) Effect of ecosystem retrogression on stable nitrogen and carbon isotopes of plants, soils and consumer organisms in boreal forest islands. Rapid Commun Mass Spectrom 23:1892–1898
Ininbergs K, Bay G, Rasmussen U, Wardle DA, Nilsson M-C (2011) Composition and diversity of nifH genes of nitrogen-fixing cyanobacteria associated with boreal forest feather mosses. New Phytol 192:507–517
Jackson BG, Martin P, Nilsson M-C, Wardle DA (2011) Response of feather moss associated N2 fixation and litter decomposition to variations in simulated rainfall intensity and frequency. Oikos 120:570–581
Jean M (2017) Effects of leaf litter and environment on bryophytes in boreal forests of Alaska. Dissertation, University of Saskatchewan
Jean M, Alexander HD, Mack MC, Johnstone JF (2017) Patterns of bryophyte succession in a 160-year chronosequence in deciduous and coniferous forests of boreal Alaska. Can J For Res 1032:1021–1032
Jones JB, Petrone KC, Finlay JC, Hinzman LD, Bolton WR (2005) Nitrogen loss from watersheds of interior Alaska underlain with discontinuous permafrost. Geophys Res Lett 32:1–4
Kardol P, Spitzer CM, Gundale MJ, Nilsson M-C, Wardle DA (2016) Trophic cascades in the bryosphere: the impact of global change factors on top-down control of cyanobacterial N2-fixation. Ecol Lett 19:967–976
Kuznetsova A, Brockhoff PB, Christensen RHB (2015) lmerTest: tests in linear mixed effects models. R package version 2.0-20. https://www.cran.r-projectorg/web/packages/lmerTest. Accessed Nov 2016
Lagerström A, Nilsson M-C, Zackrisson O, Wardle DA (2007) Ecosystem input of nitrogen through biological fixation in feather mosses during ecosystem retrogression. Funct Ecol 21:1027–1033
Lindo Z, Nilsson M-C, Gundale MJ (2013) Bryophyte-cyanobacteria associations as regulators of the northern latitude carbon balance in response to global change. Glob Chang Biol 19:2022–2035
Melvin AM, Mack MC, Johnstone JF, McGuire AD, Genet H, Schuur EAG (2015) Differences in ecosystem carbon distribution and nutrient cycling linked to forest tree species composition in a mid-successional boreal forest. Ecosystems 18:1472–1488
Näsholm T, Ekblad A, Nordin A, Giesler R, Hogberg M, Hogberg P (1998) Boreal forest plants take up organic nitrogen. Nature 392:914–916
Oechel WC, Van Cleve K (1986) The role of bryophytes in nutrient cycling in the taiga. In: Van Cleve K, Chapin FS, Flanagan PW, Viereck LA, Dyrness CT (eds) Forest ecosystems in the Alaskan taiga. Springer, New York, pp 121–137
Parke JL, Linderman RG (1980) Association of vesicular-arbuscular mycorrhizal fungi with the moss Funaria hygrometrica. Can J Bot 58:1898–1904
Rosseel Y (2011) lavaan: an R package for structural equation modeling and more. R package version 0.4-9 (BETA). https://cran.r-project.org/web/packages/lavaan/index.html. Accessed Nov 2016
Rousk K, Michelsen A (2017) Ecosystem nitrogen fixation throughout the snow-free period in subarctic tundra: effects of willow and birch litter addition and warming. Glob Chang Biol 23:1552–1563
Rousk K, Jones DL, DeLuca TH (2014) The resilience of nitrogen fixation in feather moss (Pleurozium schreberi)-cyanobacteria associations after a drying and rewetting cycle. Plant Soil 377:159–167
Rousk K, Sorensen PL, Lett S, Michelsen A (2015) Across-habitat comparison of diazotroph activity in the Subarctic. Microb Ecol 69:778–787
Rousk K, Degboe J, Michelsen A, Bradley R, Bellenger JP (2017) Molybdenum and phosphorus limitation of moss-associated nitrogen fixation in boreal ecosystems. New Phytol 214:97–107
Ruess RW, Van Cleve K, Yarie J, Viereck LA (1996) Contribution of fine root production and turnover to the carbon and nitrogen cycling in taiga forests of the Alaskan interior. Can J For Res 26:1326–1336
Sorensen PL, Lett S, Michelsen A (2012) Moss-specific changes in nitrogen fixation following two decades of warming, shading, and fertilizer addition. Plant Ecol 213:695–706
Sponseller RA, Gundale MJ, Futter M, Ring E, Nordin A, Näsholm T, Laudon H (2016) Nitrogen dynamics in managed boreal forests: recent advances and future research directions. Ambio 45:175–187
Stuiver BM, Gundale MJ, Wardle DA, Nilsson M-C (2015) Nitrogen fixation rates associated with the feather mosses Pleurozium schreberi and Hylocomium splendens during forest stand development following clear-cutting. Forest Ecol Manag 347:130–139
Tamm CO (1991) Nitrogen-limited and nitrogen-depleted terrestrial ecosystems: ecological characteristics. In: Tamm CO (ed) Nitrogen in terrestrial ecosystems. Springer, Berlin, pp 34–49
Turetsky MR (2003) The role of bryophytes in carbon and nitrogen cycling. Bryologist 106:395–409
Turetsky MR, Bond-Lamberty BP, Euskirchen ES, Talbot J, Frolking S, McGuire AD, Tuittila E-S (2012) The resilience and functional role of moss in boreal and arctic ecosystems. New Phytol 196:49–67
Van Cleve K, Alexander V (1981) Nitrogen cycling in tundra and boreal ecosystems. In: Rosswall T, Clark FE (eds) Terrestrial nitrogen cycles. Ecological bulletins, vol 33. Swedish Natural Science Research Council, Stockholm, pp 375–404
Van Cleve K, Viereck LA, Schlentner RL (1971) Accumulation of nitrogen in alder (Alnus) ecosystems near Fairbanks, Alaska. Arct Alp Res 3:101–114
Van Cleve K, Oliver L, Schlentner RE, Viereck LA, Dyrness CT (1983) Productivity and nutrient cycling in taiga forest ecosystems. Can J For Res 13:747–766
van Groenigen K-J, Six J, Hungate BA, de Graaff M-A, Van Breemen N, Van Kessel C (2006) Element interactions limit soil carbon storage. Proc Natl Acad Sci 103:6571–6574
Vitousek PM, Howarth RW (1991) Nitrogen limitation on land and in the sea: how can it occur? Biogeochemistry 13:87–115
Whiteley JA, Gonzalez A (2016) Biotic nitrogen fixation in the bryosphere is inhibited more by drought than warming. Oecologia 181:1243–1258
Zackrisson AO, Deluca TH, Nilsson M, Sellstedt A, Berglund LM, Zackrisson O (2004) Nitrogen fixation increases with successional age in boreal forests. Ecology 85:3327–3334
Zackrisson O, Deluca TH, Gentili F, Sellstedt A, Jäderlund A (2009) Nitrogen fixation in mixed Hylocomium splendens moss communities. Oecologia 160:309–319
Zielke M, Ekker AS, Olsen RA, Spjelkavik S, Solheim B (2002) The influence of abiotic factors on biological nitrogen fixation in different types of vegetation in the High Arctic, Svalbard. Arctic Antarct Alp Res 34:293
Zielke M, Solheim B, Spjelkavik S, Olsen RA (2005) Nitrogen fixation in the High Arctic: role of vegetation and environmental conditions. Arctic Antarct Alp Res 37:372–378
Acknowledgments
We thank Alix Conway, Samantha Miller, Patricia Tomchuk, Dominic Olver, Alexandre Truchon-Savard, and Nicolas Boldt for help in the field/lab. Funding came from the Department of Defense’s Strategic Environmental Research and Development Program (SERDP RC-2109), the Natural Science and Engineering Research Council of Canada (NSERC), the Northern Scientific Training Program (NSTP), NASA Ecosystems and Carbon Cycle, the National Science Foundation’s Bonanza Creek Long Term Ecological Research Site program (NSF DEB-1636476), and USDA Forest Service, Pacific Northwest Research Station.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Joy Nystrom Mast.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jean, M., Mack, M.C. & Johnstone, J.F. Spatial and temporal variation in moss-associated dinitrogen fixation in coniferous- and deciduous-dominated Alaskan boreal forests. Plant Ecol 219, 837–851 (2018). https://doi.org/10.1007/s11258-018-0838-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-018-0838-y