Abstract
Purpose
Calcineurin inhibitors (CNI) associated nephrotoxicity remains a risk factor for long-term graft dysfunction after renal transplantation. Everolimus is a mammalian target of rapamycin inhibitor and exhibits synergistic immunosuppressive activity with CNI to permit CNI-reduction. We conducted a systematic review to compare the efficacy of everolimus-based CNI sparing and standard CNI regimens in renal transplantation recipients.
Methods
We searched PubMed and Web of Science databases to identify relevant randomized controlled trials. Glomerular filtration rate (GFR), biopsy-proven acute rejection (BPAR), death or graft loss and incidence of adverse events were the major estimates of renal function, efficacy, and tolerability of the two regimens.
Results
Seven studies providing data for 2,067 patients were included. Six of the seven studies used cyclosporine as the CNI. The patients were divided into two groups: everolimus-based CNI sparing (elimination and minimization) group and standard CNI group. Everolimus-based regimen was associated with increased GFR [P = 0.02; weighted mean difference (WMD) 4.83 mL/min], decreased serum creatinine (P = 0.004; WMD −9.94 μmol/L) and no more death or graft loss [P = 0.72; relative risk (RR) 1.07]. CNI-minimization was not associated with increased BPAR (P = 0.25; RR 0.85) while CNI-elimination was associated with more BPAR Grade 1 (P < 0.00001; RR 4.20). Use of everolimus reduced the risk of CMV infection (P = 0.0002; RR 0.47). There was a higher risk of discontinuation of everolimus (P < 0.00001; RR 1.69) and non-fatal adverse events (P < 0.00001; RR 1.73) in patients on the everolimus based CNI sparing regimens.
Conclusions
Everolimus-based CNI sparing regimen could optimize long-term graft function without leading to more death or graft loss. Although CNI elimination was associated with higher risk of BPAR, everolimus use with CNI minimization did not increase the risk of acute rejections. Use of everolimus was associated with reduction in the incidence of CMV infection, but there was a higher risk of discontinuation of this drug and other non-fatal adverse events.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Calcineurin inhibitors (CNI) are the cornerstone of immunosuppression after solid organ transplantation [1]. In the field of renal transplantation, CNI-based regimens are regarded as most effective and are used worldwide; they have dramatically improved short-term outcomes by reducing acute rejections both in frequency and severity. However, long-term renal graft survival has not been improved to the same extent. The CNI-associated nephrotoxicity contributes to chronic graft dysfunction [2], which is characterized by arteriolar hyalinosis/tubulointerstitial fibrosis [3]. Death with a functioning graft due to cardiovascular disease (CVD) and malignancy were also documented [4]. Thus, current efforts are focused on non-nephrotoxic immunosuppressive regimens that can reduce exposure to CNIs, while maintaining low rates of acute rejections. Everolimus is a member of the mammalian target of rapamycin (mTOR) inhibitor class. The everolimus–receptor complex binds to mTOR, blocking downstream signaling including DNA replication and T cell proliferation. They play an important role in transplant tolerance [5, 6]. Everolimus exhibits synergistic immunosuppressive activity with cyclosporine [7]. The anti-proliferative property of everolimus makes it a potential agent to decrease malignancy, CVD, interstitial fibrosis and tubular atrophy [8, 9] post-transplantation. In fact, significantly improved renal function and comparable immunosuppressive efficacy have been achieved through early conversion from CNIs to everolimus in recipients of heart [10], lung [11], and liver [12] transplantation. Although improved renal function with mammalian target of rapamycin inhibitor (mTORi) in organ transplant recipients has been well-documented [13–22], adverse events that account for the 20–40 % drop-out rate [14] would limit its application.
Several systematic reviews have shown the potency of mTORi (sirolimus and everolimus) for de novo renal transplantation [15, 16], but the clinical use of everolimus is much less documented than sirolimus. They share the same pharmacology, but are different in drug dose and half-life, which may affect the does-dependent side effects. Everolimus was recently approved by FDA for the prevention of rejection in low-to-moderate-risk renal transplant recipients [17, 18]. To figure out the exact role of everolimus on renal transplant recipients, we conducted this systematic review and meta-analysis by pooling relevant studies to compare renal function, treatment failure and adverse events episodes 1–2 year post-transplantation between everolimus-based CNI sparing group and standard CNI exposure group.
Subjects and methods
Search strategy
We searched PubMed and Web of Science for all articles associated with everolimus, reduced-CNIs and renal transplantation. The following key words were used: “everolimus” or “mTORi” or “mammalian target of rapamycin inhibitor”, “calcineurin-inhibitors” or “CNI”, “renal transplantation” or “kidney transplantation”. No other search restrictions were applied. Reference lists of identified papers were searched for relevant studies.
Study selection
The following criteria were to be met by included studies: randomized controlled trials (RCTs) conducted to evaluate the outcomes between everolimus-based CNI-sparing versus traditional CNI-based regiments; recipients aged between 18 and 65 with a single organ transplantation; endpoints designed for measuring renal function, transplantation efficacy or adverse events. Major reasons for exclusion of studies are: review, or editorial, or comment; duplicated studies. The included studies were divided into CNI-elimination group and CNI-minimization group respectively. CNI-elimination refers to total withdrawl of CNI. CNI-minimization refers to reduced exposure of CNI than that applied in standard full-dose CNI immunosuppressive strategy.
Data extraction
Two investigators (Su and Wu) extracted information from all eligible studies independently according to the inclusion criteria listed above. Disagreements were resolved by discussion between the two investigators. The following characteristics were collected from each study: name of the first author, year of publication, study sample size, intervention of each arm, duration of the study, time of CNI switch, CNI doses after minimization or elimination, use of basiliximab. Outcome measures such as glomerular filtration rate (GFR), serum creatinine, biopsy-proven acute rejection (BPAR), death or graft loss, discontinuations of everolimus, and additional information on infections, bone marrow toxicity and metabolic derangements when available were extracted and all the information was entered into RevMan 5 for combination.
Quality assessment
The quality of the trials was independently assessed by Su and Wu based on the Jadad score across five items: randomization, allocation concealment, double blinding, completed data and ITT analysis. A score of three or more is considered good quality. Disagreements on data collection and quality assessment were resolved by consensus.
Statistical analysis
All statistical analyses were performed using Review Manager 5.1 software. Between-study heterogeneity was evaluated using χ 2 test and I 2 statistics. Heterogeneity was considered statistically significant at I 2 > 75 %, calculated as the experimental group (everolimus-based group) versus the control group (CNI-based group). If significant heterogeneity existed, analysis model of random effect was present. When the source of heterogeneity was detected, subgroup analysis was performed accordingly. For binary outcomes (e.g., adverse events), relative risk (RR) was used as a summary statistic, whereas for continuous outcomes (e.g., GFR), weighted mean difference (WMD) was used. These values were combined to provide the overall RR or WMD, with a 95 % confidence interval (CI) for this pooled estimate of the difference. P < 0.05 was considered to be statistically significant.
Results
Included studies
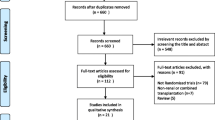
The results of the literature search are illustrated in Fig. 1. Seven randomized clinical trials [19–25] providing data for 2,067 renal transplant recipients were identified. Four studies had a follow-up of period of 12 months and the remaining 3 had 24 months of followup. To avoid duplication, one study reporting 3 year results of the ZESUS trial [26] was excluded, since the original report [20] had been included in the analysis. Twenty seven studies were excluded for irrelevant outcomes as the endpoints of these trials were not designed for measuring renal function, transplantation efficacy or adverse events.
Six of the seven studies evaluated in this meta analysis used cyclosporine as the CNI agent, with only the study by Holdaas et al. [22] using tacrolimus as a CNI in 113 of 294 patients enrolled in the trial. The results of this meta analysis predominantly describe results of cyclosporine elimination/minimization. Total CNI elimination and CNI minimization were investigated in 4 (n = 848) and 4 (n = 1,214) trials respectively. The cyclosporine target concentration of C2 in the CNI sparing groups were illustrated in Table 1. One three-arm study (Hodaas) was considered as two separate studies (one CNI-elimination shown as Holdaas 2011, one CNI-minimization as Holdaas’ 2011) without double-counting any of the participants. All of the included studies used steroids, enteric-coated mycophenolate sodium (EC-MPS) or mycophenolic acid (MPA) as the maintenance therapy at the same time, and basiliximab was taken as the induction therapy in five of the seven studies. The characteristics of eligible studies were illustrated in Table 1.
Each study compared the combination therapy (everolimus based CNI sparing therapy) with standard CNI-exposure therapy for renal transplantation recipients. Six studies started drug conversion in the early period post-transplantation (<6 months) except Holdaas (>6 months). In every report, baseline characteristics of the populations in both arms revealed no significant difference.
Methodological quality
Ninety percent of the RCTs were considered to be of good methodological quality according to the Jadad score (≥3). Bertoni 2011 gained 2. All the included trials conducted randomization and provided complete data. Only one study (Bertoni 2011) did not complete the ITT and one study (Holdaas 2011) failed to follow up 11 of 399 patients.
Graft function
Serum creatinine was used as a marker of graft function in 5 of the 7 studies. The everolimus-based regimen was associated with decreased serum creatinine (WMD −9.94 μmol/L; 95 % CI [−16.66, −3.22 μmol/L]; P = 0.004; heterogeneity I 2 = 0 %; Fig. 2).
Additionally, six studies assessed GFR. Studies by Holdaas et al. and Mjornstedt et al. used measured GFR while another three studies used estimated GFR. Studies by Budde et al. and Chadban et al. analysed estimated GFR using Nankivell formula while the study by Bertoni et al. analysed estimated GFR using Cockroft–Gault formula. Significantly increased GFR (WMD 4.83 mL/min; 95 % CI [0.26, 9.92 mL/min]); P = 0.06; Fig. 3) in patients with everolimus-based CNI sparing strategy was observed when compared with those under standard CNI exposure. Random effect model was applied here due to the existence of obvious heterogeneity. The result reflected varying degrees of renal function improvement as all studies demonstrated superior renal function with everolimus-based protocols.
Death or graft loss
No significant difference was defined in death or graft-loss (RR 1.07; 95 % CI [0.73, 1.58]; P = 0.72; heterogeneity, I 2 = 0 %; Fig. 4) between everolimus-based CNI sparing and standard CNI group.
Acute rejections
CNI elimination was associated with more BPAR (RR 2.51; 95 % CI [1.63, 3.87]; P < 0.0001; heterogeneity, I 2 = 0 %; Fig. 5), while CNI minimization resulted in similar outcomes with standard CNI group (RR 0.85; 95 % CI [0.64, 1.13]; P = 0.25; heterogeneity, I 2 = 0 %; Fig. 5). The heterogeneity was resolved by subgroup analysis.
Further analysis revealed CNI elimination led to more BPAR Grade 1 (RR 4.20; 95 % CI [2.23, 7.91]; P < 0.00001; heterogeneity, I 2 = 0 %). No significant difference was detected in episodes of BPAR Grade 2 and 3 between everolimus-based CNI sparing and standard CNI group (RR 0.79; 95 % CI [0.47, 1.32]; P = 0.36; heterogeneity, I 2 = 0 %).
Discontinuation
Seven studies assessed everolimus discontinuation. Patients under everolimus-based CNI sparing regimens suffered from more discontinuations of everolimus (RR 1.69; 95 % CI [1.44, 1.99]; P < 0.00001; heterogeneity, I 2 = 67 %) than standard CNI-exposure regimens.
Infections
Incidence of infections was evaluated in six studies. Everolimus with CNI sparing did not contribute to any more infections with an RR = 1.05 (95 % CI [0.97, 1.13]; P = 0.2; heterogeneity, I 2 = 0 %). Incidence of CMV infection was lower in CNI sparing group (RR 0.47; 95 % CI [0.32, 0.70]; P = 0.0002; heterogeneity, I 2 = 61 %; Fig. 6).
Other adverse events
Cholesterol and triglycerides level were assessed in six and five studies, respectively. Everolimus-based regimen was associated with significantly increased levels of total cholesterol (WMD 0.70 mmol/L; 95 % CI [0.58, 0.82 mmol/L]; P < 0.00001; heterogeneity, I 2 = 0 %), triglycerides (WMD 0.54 mmol/L; 95 % CI [0.39, 0.68 mmol/L]; P < 0.00001; heterogeneity, I 2 = 57 %), and more New Onset Diabetes after Transplantation (NODAT) (RR 1.38; 95 % CI [1.00, 1.90]; P = 0.05; heterogeneity, I 2 = 38 %).
Everolimus-based regimen was associated with more stomatitis and oral ulcers (RR 6.02; 95 % CI [3.69, 9.80]; P < 0.00001; heterogeneity, I 2 = 50 %) and increased proteinuria (RR 1.66; 95 % CI [1.21, 2.28]; P = 0.002; heterogeneity, I 2 = 0 %). More anemia (RR 1.37; 95 % CI [1.16, 1.62]; P = 0.0002; heterogeneity, I 2 = 0 %) and thrombocytopenia (RR 2.35; 95 % CI [1.18, 4.67]; P = 0.02; heterogeneity, I 2 = 0 %) were also detected in everolimus-based CNI sparing group. The results were presented in Fig. 7.
Discussion
In the field of renal transplantation, efforts are now focused on immunosuppressive strategies with CNI minimization or elimination that can optimize long-term graft function without increasing rejection rates. Everolimus exhibits synergistic immunosuppressive activity with CNI and may permit CNI reduction. Nashan et al. [27] has demonstrated that in renal transplantation recipients an everolimus-based CNI sparing regimen was well tolerated with better graft function and similar rate of treatment failure when compared with the standard CNI exposure regimen. However, controlled clinical trials have not been evaluated systematically until now. Thus, we conducted this meta-analysis to define the impact of everolimus-based CNI sparing regimens on renal transplant recipients. Six of the seven included studies evaluated in this meta analysis used cyclosporine as the CNI agent, with only the study by Holdaas et al. [22] used tacrolimus as a CNI in 113 of 294 patients enrolled in the trial. The results of this meta analysis therefore predominantly describe results of cyclosporine elimination/minimization.
Our results have suggested that everolimus based CNI sparing regimens were associated with significantly improved renal function without any detectable differences in patient or graft survivals over a short follow up period of 1–2 years. There was a reduced risk of CMV infections, but a higher risk of biopsy proven acute rejections and higher rate of discontinuation of the drug due to adverse events. In addition, everolimus based regimens were associated with higher serum cholesterol and triglyceride levels and higher risk of NODAT as compared to patients on a full dose CNI based regimen. Finally, there was more proteinuria, stomatitis, oral ulcerations and bone marrow toxicity in patients on everolimus based regimens as compared to those on full dose CNIs. Our study shows a GFR increase of 0.26–9.92 ml/min (P = 0.02) in the everolimus-based group, indicating that conversion from CNI to everolimus improves renal function. As renal function at 12 months post transplantation is a predictor of long term graft outcomes [28–30], this improvement in renal function with everolimus based regimens could result in improvement in long term graft outcomes. Only the study by Holdaas et al. [22] did not observe significant renal function improvement with everolimus. In this study patients were switched to everolimus based regimens after a mean period of 5.6 years post transplant, during which time they had been exposed to a CNI, whereas in all the other studies the conversion occurred within the first 6 months, suggesting early conversion to everolimus based regimens was associated with improvement in renal functions, before irreversible toxicity sets in with prolonged use of CNIs. This was evidenced by the significantly higher serum creatinines at the time of conversion to everolimus in the study by Holdaas et al. [22]. Our meta analysis also found a significantly increased risk of proteinuria with everolimus based regimens. It has been reported that proteinuria occurs in up to 30 % of kidney transplantation recipients who receive an mTORi. Everolimus has complex effect on podocyte structure and function, possibly compounded by effects on tubular function [31].As such, conversion to everolimus is not advised in patients with preexisting proteinuria >800 mg/day [32]. It has been suggested that proteinuria associated with mTORi can be managed by the initiation of an angiotensin-converting enzyme inhibitor and/or angiotensin receptor blocker [33–35].
Our data showed that everolimus-based CNI sparing regimens (both elimination and minimization) did not increase the risk of death or graft loss. Subgroup analysis showed the 2.5 times incidence of BPAR in the CNI elimination group, which may be due to insufficient immunosuppression with complete CNI withdrawal. CNI elimination strategy would not lead to more BPAR Grade 2 and 3 but resulted in over 4 times incidence of BPAR Grade 1. The increased BPAR Grade 1, if adequately treated, would have minimal or no effect on long-term graft survival [36]. The CNI minimization regimens with everolimus were able to achieve the same efficacy as the standard dose CNI regimens. Although CNI elimination avoids CNI toxicity, it was associated with a higher risk of BPARs and this strategy therefore needs to be used with caution in high risk transplant recipients. In theory CNI-elimination avoids the associated nephrotoxicity once and for all, but CNI-free protocols should be used cautiously in clinic, especially in renal transplant recipients. Some trials showed that sirolimus with complete CNI avoidance would lead to a higher rejection rate [37–39], and this is also supported by our analysis. Perhaps more relevant studies and longer follow-up are warranted to identify long-term survival in recipients of the everolimus with CNI elimination regimen. The present data suggests that everolimus-based CNI minimization seems more reasonable than CNI elimination. This strategy both reduces CNI-associated nephrotoxicity and maintains the low rejection rates achieved by standard CNI exposure.
Although varying doses were used in the enrolled trials, no higher risk of infection was noted in the observed arms. More encouragingly, everolimus-based therapy was associated with a significant decrease in CMV infection, perhaps due to the enhanced CMV-related T-cell response in this group [40, 41]. Both CMV infection and CMV disease negatively influence the outcome, and they are also associated with increased risk of acute rejection and chronic allograft dysfunction [42]. Unfortunately, more recipients discontinued in the intervention arm and most of the discontinuation was caused by the adverse events.
The kidney transplantation population is highly vulnerable to premature CVD, which is the major cause of death with a functioning graft [43]. Consistent with previous studies [44], everolimus-based therapy was associated with overall increased levels of total cholesterol and triglycerides. This suggests that early administration of statins to guard against CVD was necessary, which is supported by two of our studies [21, 22]. Although the association of everolimus with NODAT is not well understood, the presence of diabetes increases postoperative complications such as infection, hypertension, and hyperlipidemia [45]. As the major causes of discontinuation, stomatitis and oral ulcers generally respond to dose reductions and can often be treated effectively [46]. Similar to earlier reports where mTORi therapy was reported to be associated with bone marrow suppression in a dose-dependent manner [14, 19, 21, 47], we also found a higher risk of anemia and thrombocytopenia, although without a higher risk of leukopenia, in the everolimus-based treatment groups. All non-lethal adverse events did not lead to treatment failure, but instead resulted in increased discontinuation.
Effects of everolimus-based regimens on renal transplant recipients illustrated by our meta analysis were very similar to those reported with sirolimus-based regimens. Several studies [48–50] have shown that sirolimus-based CNI sparing regimens are also associated with improved renal function, comparable patient survival rates, more discontinuations from adverse events in renal transplant recipients.
One limitation of this meta-analysis is the lack of patient-centered endpoints (such as quality of life), which are of great clinical significance. Since six of the seven studies included in this meta analysis used everolimus as a cyclosporine sparing or minimising agent, the results of this meta-analysis cannot be generalised to indicate the effects of everolimus as a tacrolimus sparing or minimising agent. Another limitation is the inability to clearly distinguish randomized time points between groups, which was limited by sample size. As a result, we cannot define when the conversion from CNI- to everolimus-based therapy reaches the best balance between efficacy, renal function, and adverse event post-transplantation. For some important endpoints (such renal function), analysis model of random effect was applied to deal with the heterogeneity. Subgroup analysis or the categorization of studies into CNI minimization/elimination has not successfully minimized or eliminated inter-study heterogeneity, although they may fulfill some other endpoints. Longer follow-up is warranted to identify the actual influence of everolimus-based regimens on renal function, rejection, and survival, especially the incidence of malignancy.
Our intention was to identify the risks and benefits of everolimus-based CNI sparing strategy, generate hypothesis and help direct further research and clinical development, rather than to find out the single most effective immunosuppressive regimen. We believe this meta-analysis achieves all these aims and addresses one of the most topical and challenging aspects of transplantation.
In conclusion, this meta-analysis suggests that everolimus based CNI sparing regimens were associated with significantly improved renal functions without any differences in patient or graft survivals. There was a reduced risk of CMV infections, but a higher risk of biopsy proven acute rejections and higher rate of discontinuation of the drug due to adverse events. In addition, everolimus based regimens were associated with higher risk of metabolic disorders and several other adverse events as compared to patients on a full dose CNI based regimen. This sets the stage for further studies to assess an optimum immunosuppression strategy, durability of the benefit, and prophylaxis of adverse events.
References
Calne RY, White DJ, Thiru S et al (1978) Cyclosporin A in patients receiving renal allografts from cadaver donors. Lancet 2(8104–8105):1323–1327
Chapman JR, Valantine H, Albanell J et al (2007) Proliferation signal inhibitors in transplantation: questions at the cutting edge of everolimus therapy. Transplant Proc 39(10):2937–2950
Nankivell BJ, Borrows RJ, Fung CL et al (2003) The natural history of chronic allograft nephropathy. N Engl J Med 349(24):2326–2333
Campbell S, McDonald S, Chang S et al (2011) ANZDATA registry. The 30th annual report. Ch8Transplantation. http://www.anzdata.org.au/anzdata/AnzdataReport/30thReport
Watanabe R, Wei L, Huang J (2011) mTOR signaling, function, novel inhibitors, and therapeutic targets. J Nucl Med 52(4):497–500
Holdaas H, Midtvedt K, Asberg A (2012) A drug safety evaluation of everolimus in kidney transplantation. Expert Opin Drug Saf 11(6):1013–1022
Schuurman HJ, Cottens S, Fuchs S et al (1997) SDZ RAD, a new rapamycin derivative: synergism with cyclosporine. Transplantation 64(1):32–35
Lutz J, Zou H, Liu S et al (2003) Apoptosis and treatment of chronic allograft nephropathy with everolimus. Transplantation 76(3):508–515
Koch M, Mengel M, Poehnert D et al (2007) Effects of everolimus on cellular and humoral immune processes leading to chronic allograft nephropathy in a rat model with sensitized recipients. Transplantation 83(4):498–505
Stypmann J, Engelen MA, Eckernkemper S et al (2011) Calcineurin inhibitor-free immunosuppression using everolimus (Certican) after heart transplantation: 2 years’ follow-up from the University Hospital Munster. Transplant Proc 43(5):1847–1852
Gullestad L, Iversen M, Mortensen SA et al (2010) Everolimus with reduced calcineurin inhibitor in thoracic transplant recipients with renal dysfunction: a multicenter, randomized trial. Transplantation 89(7):864–872
Masetti M, Montalti R, Rompianesi G et al (2010) Early withdrawal of calcineurin inhibitors and everolimus monotherapy in de novo liver transplant recipients preserves renal function. Am J Transplant 10(10):2252–2262
Kamar N, Allard J, Ribes D et al (2005) Assessment of glomerular and tubular functions in renal transplant patients receiving cyclosporine A in combination with either sirolimus or everolimus. Clin Nephrol 63(2):80–86
Rostaing L, Kamar N (2010) mTOR inhibitor/proliferation signal inhibitors: entering or leaving the field? J Nephrol 23(2):133–142
Peddi VR, Wiseman A, Chavin K et al (2013) Review of combination therapy with mTOR inhibitors and tacrolimus minimization after transplantation. Transplant Rev (Orlando) 27(4):97–107
Webster AC, Lee VW, Chapman JR et al (2006) Target of rapamycin inhibitors (sirolimus and everolimus) for primary immunosuppression of kidney transplant recipients: a systematic review and meta-analysis of randomized trials. Transplantation 81(9):1234–1248
Novartis Pharmaceuticals (2010) Zortress (everolimus) [package insert]. Novartis Pharmaceuticals, East Hanover, NJ. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=e082a024-7850-400b-a5c2-2a140612562a
Gabardi S, Baroletti SA (2010) Everolimus: a proliferation signal inhibitor with clinical applications in organ transplantation, oncology, and cardiology. Pharmacotherapy 30(10):1044–1056
Bertoni E, Larti A, Rosso G et al (2011) Good outcomes with cyclosporine very low exposure with everolimus high exposure in renal transplant patients. J Nephrol 24(5):613–618
Budde K, Becker T, Arns W et al (2011) Everolimus-based, calcineurin-inhibitor-free regimen in recipients of de-novo kidney transplants: an open-label, randomised, controlled trial. Lancet 377(9768):837–847
Cibrik D, Silva HJ, Vathsala A et al (2013) Randomized trial of everolimus-facilitated calcineurin inhibitor minimization over 24 months in renal transplantation. Transplantation 95(7):933–942
Holdaas H, Rostaing L, Seron D et al (2011) Conversion of long-term kidney transplant recipients from calcineurin inhibitor therapy to everolimus: a randomized, multicenter, 24-month study. Transplantation 92(4):410–418
Mjornstedt L, Sorensen SS, von Zur MB et al (2012) Improved renal function after early conversion from a calcineurin inhibitor to everolimus: a randomized trial in kidney transplantation. Am J Transplant 12(10):2744–2753
Santos SM, Carlos CM, Cabanayan-Casasola CB et al (2012) Everolimus with reduced-dose cyclosporine versus full-dose cyclosporine and mycophenolate in de novo renal transplant patients: a 2-year single-center experience. Transplant Proc 44(1):154–160
Chadban SJ, Eris JM, Kanellis J et al (2013) A randomized, controlled trial of everolimus-based dual immunosuppression versus standard of care in de novo kidney transplant recipients. Transpl Int 27:302–311
Budde K, Lehner F, Sommerer C et al (2012) Conversion from cyclosporine to everolimus at 4.5 months posttransplant: 3-year results from the randomized ZEUS study. Am J Transplant 12(6):1528–1540
Nashan B, Curtis J, Ponticelli C et al (2004) Everolimus and reduced-exposure cyclosporine in de novo renal-transplant recipients: a three-year phase II, randomized, multicenter, open-label study. Transplantation 78(9):1332–1340
Salvadori M, Rosati A, Bock A et al (2006) Estimated one-year glomerular filtration rate is the best predictor of long-term graft function following renal transplant. Transplantation 81(2):202–206
Helal I, Abderrahim E, Ben HF et al (2009) The first year renal function as a predictor of long-term graft survival after kidney transplantation. Transplant Proc 41(2):648–650
Resende L, Guerra J, Santana A et al (2009) First year renal function as a predictor of kidney allograft outcome. Transplant Proc 41(3):846–848
Letavernier E, Legendre C (2008) mToR inhibitors-induced proteinuria: mechanisms, significance, and management. Transplant Rev (Orlando) 22(2):125–130
Diekmann F, Budde K, Oppenheimer F et al (2004) Predictors of success in conversion from calcineurin inhibitor to sirolimus in chronic allograft dysfunction. Am J Transplant 4(11):1869–1875
Oroszlan M, Bieri M, Ligeti N et al (2010) Sirolimus and everolimus reduce albumin endocytosis in proximal tubule cells via an angiotensin II-dependent pathway. Transpl Immunol 23(3):125–132
Stallone G, Infante B, Grandaliano G et al (2009) Management of side effects of sirolimus therapy. Transplantation 87(8 Suppl):S23–S26
Kaplan B, Qazi Y, Wellen JR (2014) Strategies for the management of adverse events associated with mTOR inhibitors. Transplant Rev (Orlando). doi:10.1016/j.trre.2014.03.002
Sellares J, de Freitas DG, Mengel M et al (2011) Inflammation lesions in kidney transplant biopsies: association with survival is due to the underlying diseases. Am J Transplant 11(3):489–499
Ekberg H, Tedesco-Silva H, Demirbas A et al (2007) Reduced exposure to calcineurin inhibitors in renal transplantation. N Engl J Med 357(25):2562–2575
Flechner SM, Glyda M, Cockfield S et al (2011) The ORION study: comparison of two sirolimus-based regimens versus tacrolimus and mycophenolate mofetil in renal allograft recipients. Am J Transplant 11(8):1633–1644
Asberg A, Midtvedt K, Line PD et al (2006) Calcineurin inhibitor avoidance with daclizumab, mycophenolate mofetil, and prednisolone in DR-matched de novo kidney transplant recipients. Transplantation 82(1):62–68
Vitko S, Margreiter R, Weimar W et al (2004) Everolimus (Certican) 12-month safety and efficacy versus mycophenolate mofetil in de novo renal transplant recipients. Transplantation 78(10):1532–1540
Havenith SH, Yong SL, van Donselaar-van DPK et al (2013) Everolimus-treated renal transplant recipients have a more robust CMV-specific CD8+ T-cell response compared with cyclosporine- or mycophenolate-treated patients. Transplantation 95(1):184–191
Sagedal S, Nordal KP, Hartmann A et al (2002) The impact of cytomegalovirus infection and disease on rejection episodes in renal allograft recipients. Am J Transplant 2(9):850–856
Ojo AO (2006) Cardiovascular complications after renal transplantation and their prevention. Transplantation 82(5):603–611
Eisen H (2006) Long-term cardiovascular risk in transplantation–insights from the use of everolimus in heart transplantation. Nephrol Dial Transplant 21(Suppl 3):i9–i13
Lv C, Chen M, Xu M et al (2014) Influencing factors of new-onset diabetes after a renal transplant and their effects on complications and survival rate. PLoS One 9(6):e99406
Mahe E, Morelon E, Lechaton S et al (2005) Cutaneous adverse events in renal transplant recipients receiving sirolimus-based therapy. Transplantation 79(4):476–482
Ponticelli C (2014) The pros and the cons of mTOR inhibitors in kidney transplantation. Expert Rev Clin Immunol 10(2):295–305
Groth CG, Backman L, Morales JM et al (1999) Sirolimus (rapamycin)-based therapy in human renal transplantation: similar efficacy and different toxicity compared with cyclosporine. Sirolimus European Renal Transplant Study Group. Transplantation 67(7):1036–1042
Glotz D, Charpentier B, Abramovicz D et al (2010) Thymoglobulin induction and sirolimus versus tacrolimus in kidney transplant recipients receiving mycophenolate mofetil and steroids. Transplantation 89(12):1511–1517
Lebranchu Y, Snanoudj R, Toupance O et al (2012) Five-year results of a randomized trial comparing de novo sirolimus and cyclosporine in renal transplantation: the SPIESSER study. Am J Transplant 12(7):1801–1810
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 81102245) and Science and Technology Planning Project of Guangdong Province, China (No. 2011B0318000099), Medical Scientific Research Foundation of Guangdong Province, China (No. B2011072) and Youth teachers cultivation project of Sun Yat-Sen University (No. 12ykpy21).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Liya Su and Ngalei Tam have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Su, L., Tam, N., Deng, R. et al. Everolimus-based calcineurin-inhibitor sparing regimens for kidney transplant recipients: a systematic review and meta-analysis. Int Urol Nephrol 46, 2035–2044 (2014). https://doi.org/10.1007/s11255-014-0783-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-014-0783-1