Abstract
Purpose
We aimed to assess the effect of intralesional verapamil on the treatment of Peyronie’s disease.
Materials and methods
This randomized study involved 80 patients. First, they were divided into two groups. The first group (case: 40 patients) received intralesional verapamil and the second group (control: 40 patients) local saline injection. They were followed about 24 weeks and evaluated for the size of plaques, plaque softening, reduction of pain and amelioration of penile deformity and erectile dysfunction (estimated by the International Index of Erectile Function) before and after treatment.
Results
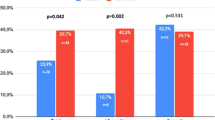
Reduction of plaque size was seen in 17.5% of the case group and 12.8% of the control group (P: 0.755). Pain was reduced in 30% of the case group and 28.2% of the control group (P: 0.99). Curvature was decreased in 17.5% of the case group and 23.1% the control group (P: 0.586). Plaque softening was seen in 30% of the case group compared with 25.6% improvement in the control group (P: 0.803).
Also we found 5% and 2.6% improvement in sexual dysfunction in the case and control groups, respectively. (P: 0.985).
Conclusion
Although in some studies verapamil has been found to be effective in the treatment of Peyronie’s disease, we did not find any improvement in comparison with the control group.
Furthermore, larger scale studies are warranted to assess the effect of this drug on the treatment of Peyronie’s disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Peyronie’s disease (PD) is a localized connective tissue disorder that affects the tunica albugina of the penis. The result is bending and deformity of the erected penis, difficulty in vaginal penetration and change in erectile capacity [1, 2].
This disease was first described in 1743 by the French surgeon, Francois de la Peyroni. According to multivariate logistic regression analysis, the risk factors for PD are: a history of genital and/or perineal injuries, transurethral prostatectomy, cystoscopy, diabetes mellitus, hypertension, lipoma, drugs such as propranolol, Dupuytren’s contracture, smoking and alcohol consumption [3].
While PD was once considered to be relatively uncommon, recent studies suggest that its prevalence is similar to that of diabetes or urolithiasis. A recent epidemiological study reported an overall prevalence of 3.2% [4, 5].
While severe penile deformity and erectile dysfunction are best treated with surgery, numerous oral and intralesional drugs have been used to resolve pain, minimize penile deformity and arrest progression of disease [6–8].
The use of verapamil, a calcium antagonist, is based on its ability to alter fibroblast production of extracellular matrix, macromolecules and collagenase [9–11]. In some studies, verapamil has been shown to be safe and effective in the treatment of Peyronie’s disease [12–14]. Previously, in a pilot study, no improvement was found after verapamil therapy; thus, this study was conducted to ascertain the role of this drug in the treatment of Peyronie’s disease.
Materials and methods
Between March 2002 and October 2005, 80 patients were enrolled in this prospective, single-blind study. The sample size was measured by a statistician considering previous studies. Inclusion criteria were: (1) patients who had Peyronie’s disease as defined by the presence of a palpable plaque upon examination by the same urologist (M.S.); (2) patients who underwent a full course of therapy.
Exclusion criteria were: (1) any history of exposure to oral agents for the treatment of Peyronie’s disease (such as potassium para-aminobenzoate, colchicines and tamoxifen), transdermal verapamil, shock wave therapy or prior intralesional agents within 3 months; (2) patients with calcified plaques; (3) patients who had ventral curvatures; (4) patients who had congenital penile curvature or chordee with hypospedias.
Patients were informed about the nature of disease and the available treatment modalities. Full written consent was obtained before enrollment. We randomized the patients according to a computer-generated random table in two groups: case and control. Patients were blinded to the group to which they belonged. The sonographist was also blinded to the treatment group to which the patients belonged.
Injection technique
At first, dorsal nerve penile block using 1% lidocaine was done. After grasping the plaque between the index finger and thumb, verapamil (10 mg diluted in 10 ml of distilled water) was injected into the plaque with a 10-ml syringe and a 25-gauge needle twice weekly for 12 weeks. Patients had postinjection blood pressure measurements. Precaution was taken not to insert the needle into the corpus cavernosum, dorsal nerves and arteries. The patients were asked to compress the injection site for 5 min to decrease echymosis. The same procedure was performed for patients in the control group with saline (10 ml).
The evaluation was based on the medical history, physical examination, ultrasonography and IIEF questionnaire.
Erectile dysfunction severity is classified into five categories based on the IIEF-5: severe (5–7), moderate (8–11), mild to moderate (12–16), mild (17–21) and no ED (22–25). All of the first four categories were considered as the ED group.
The plaque size was measured by ultrasonography. The pain was subjectively self-evaluated by patients.
Penile deformity was assessed with a protractor at maximum rigidity, and improvement of curvature was defined as an objectively measured curvature of at least 10°. Plaque softening as a subjective datum was recorded by querying the patient about its improvement. The patients were evaluated before and 24 weeks after treatment.
Statistical analysis was done using chi-square and paired test. P values <0.05 were considered significant.
Results
There were no statistically significant differences between the groups in age, onset of the disease, initial plaque size, signs and symptoms (Tables 1 and 2).
The mean curvature at baseline was 49.71° ± 9.3° and 45.57° ± 9.7° in the case and control groups, respectively. The mean plaque area (maximum length × maximum width) was 3.20 ± 0.4 and 3.4 ± 0.9 cm2 in the case and control groups, respectively.
One man dropped out of the case group after the first visit without completing other data, such as curvature and erectile dysfunction assessment. Other patients underwent the full course of treatment.
At the follow-up visit, 24 weeks after treatment, the mean curvature at baseline was 47.57° ± 7.3° and 43.43° ± 8.9° in the case and control groups, respectively. The mean plaque area (maximum length × maximum width) was 2.89 ± 0.39 and 3.02 ± 5.7 cm2 in the case and control groups, respectively. We compared these data before and after treatment in the case and control groups using the paired test. No significant changes were seen (P value = 0.91 and 0.87 for mean curvature and plaque size, respectively).
An at least 5° decrement in curvature, any decrement in plaque size or ED severity based on IIEF-5 was considered as improvement.
Any subjective improvements of pain or plaque softening were considered as positive response (Table 3).
Discussion
Peyronie’s disease frequently develops in response to acute or repeated trauma to an erected penis with microvascular injury and fibrin deposits, leading to recruitment and activation of inflammatory cells, followed by matrix formation and development of fibrotic plaque [15].
A number of oral agents have been investigated for the treatment of PD [16].
Vitamin E was the first oral therapy to be described for this condition [17]. The other agents that have been advised are colchicine (0.6 mg three times per day with meals), potassium aminobenzoate (12 g/day, four to six doses), tamoxifen citrate (20 mg twice a day) and carnitine (1 g twice daily) [18–21].
Direct injection of an agent into the Peyronie’s lesion has an extensive history of investigation [12]. Steroids, collagenase, interferon and verapamil have been used [12].
A number of surgical procedures, such as the Nesbit-type operation plaque excision and dermal grafting, plaque incision and grafting and penile prosthesis have been tried [22–26].
One of the intralesional treatments that has been investigated several times is verapamil [14]. Anecdotal evidence shows some lessening of the scarring process after intralesional injections of a calcium channel blocker. The theory is based on the known action of verapamil in decreasing the production and secretion of extracellular matrix macromolecules by fibroblasts while increasing collagenase production in the same area. Calcium channel blockers significantly decreased both the constitutive and platelet-derived growth factor B-dependent collagen deposition in the ECM formed by human vascular smooth muscle cells and fibroblasts. The drugs inhibited the expression of fibrillar collagens type I and III and of basement membrane type IV collagen. Furthermore, Ca2+ channel blockers specifically increased the proteolytic activity of the 72-kDa type IV collagenase as shown by gelatin zymography and inhibited the transcription of tissue inhibitor of metalloproteinases-2 [11].
The use of verapamil as an intralesional injection was first reported by Levine et al. in 1994. In this non-controlled study, 14 men received biweekly injections of verapamil into the Peyronie’s plaques for 6 months. Subjectively, there was significant improvement in plaque-associated penile narrowing in all patients and curvature in six (42%). Objectively, a decreased plaque volume of >50% was noted in four (30%) of the subjects. Plaque softening was noted in all patients, while 12 (83%) noticed that plaque-related changes in erectile function had improved [14].
In Di Stasi et al.’s prospective study, 96 patients were randomized to be treated with verapamil (5 mg) plus dexamethasone (8 mg) using transdermal electromotive administration. A 43% improvement in plaque size and curvature was noted [27].
Levin et al. published results of another non-placebo-controlled trial involving 156 men treated with intralesional verapamil therapy. Of 128 men who completed the study, 84% of those with pain had complete resolution [28]. But in the present study, the case and control patients did not differ significantly in response to treatment, although curvature change was relatively more compared with the other symptoms.
Sahin et al. evaluated the antifibrotic effect of verapamil and different intracovernosal agents on the corpus cavernosum. It was insufficient to prevent inflammation [29].
Bennett et al. also tried intralesional verapamil injection therapy in men with Peyronie’s disease and found no statistically significant improvement in penile curvature, although the vast majority of patients reported that penile pain resolved after therapy [30].
It is not known why the response rate in our study is so different from that in others. We postulate many possibilities. First, most of the patients were referred to us after complete plaque formation. When we compare this latency with other studies (<18 months), it is significant. Medical and intralesional therapies are clinically and pathologically effective in early stages of the disease, before totally involving the tunica albugina with plaque and scarring. Second, the size of plaques was larger in our study compared to that in other studies (<2 cm in other studies compared to 3 cm in our study) [29], so it is more difficult to achieve complete drug distribution by injection to this large, hardened plaque tissue.
Many limitations warrant mention, such as the single blindness of this study, the small number of the patients and the short-term follow-up.
Conclusion
Based on our study, intralesional verapamil is not effective in the treatment of Peyronie’s disease. No statistically significant improvements were seen in pain, ED, penile curvature, plaque size and softening.
Thus, we recommend other larger, double-blind controlled trials to assess the effect of this drug in the setting of Peyronie’s disease.
References
Smith CJ, Mcmahon C, Shabsigh R (2005) Peyronie’s disease: the epidemiology, aetiology and clinical evaluation of deformity. BJU Int 95:729–732. doi:10.1111/j.1464-410X.2005.05391.x
Gelbard MK, Dorey F, James K (1990) The natural history of Peyronie’s disease. J Urol 144:1376–1379
De la Peyronie F (1743) Sur quelques obstacle qui s opposent a l ejaculation naturelle de la semence. Mem Acad Chir (Paris) 1:318
Sommer F, Schwarzer U, Wassmer G et al (2002) Epidemiology of Peyronie’s disease. Int J Impot Res 14:379–383. doi:10.1038/sj.ijir.3900863
Schwarzer U, Sommer F, Klotz T, Braun M, Reifenrath B, Engelmann U (2001) The prevalence of Peyronie’s disease: results of a large survey. BJU Int 88:727–730. doi:10.1046/j.1464-4096.2001.02436.x
Fitkin J, Ho GT (1999) Peyronie’s disease: current management. Am Fam Physician 60:549–552
Tunuguntlas HS (2001) Management of Peyronie’s disease: a review. World J Urol 19:244–250. doi:10.1007/s003450100209
Anderson MS, Shankey TV, Lubrano T, Mulhall JP (2000) Inhibition of Peyronie’s plaque fibroblast proliferation by biologic agents. Int J Impot Res 12(suppl 3):s25–s31. doi:10.1038/sj.ijir.3900558
Lee RC, Ping J (1990) Calcium antagonists retard extracellular matrix production in connective tissue equivalent. J Surg Res 49:463–466. doi:10.1016/0022-4804(90)90197-A
Doong H, Dissanayaka S, Gowrishankar TR, La Barbera MC, Lee RC (1996) The 1996 Lindberg Award. Calcium antagonists alter cell shape and induce procollagenase synthesis in keloid and normal human dermal fibroblasts. J Burn Care Rehabil 17:497–514. doi:10.1097/00004630-199611000-00005
Roth M, Eickelberg O, Kohler E, Eme P, Block LH (1996) Ca channel blockers modulate metabolism of collagenase within the extracellular matrix. Proc Natl Acad Sci USA 93:5478–5482. doi:10.1073/pnas.93.11.5478
Levine LA (1997) Treatment of Peyronie’s disease with intralesional verapamil injection. J Urol 158(4):1395–1399. doi:10.1016/S0022-5347(01)64224-1
Rehman J, Benet A, Melman A (1998) Use of intralesional verapamil to dissolve Peyronie’s disease plaque: a long-term single blind study. Urology 51(4):620–626. doi:10.1016/S0090-4295(97)00700-0
Levine LA, Merrick PF, Lee RC (1994) Intralesional verapamil injection for the treatment of Peyronie’s disease. J Urol 151(6):1522–1524
Devine CJ Jr, Somers KD, Jordan GH et al (1997) Proposal trauma as the cause of the Peyronie’s lesion. J Urol 157:285–290. doi:10.1016/S0022-5347(01)65361-8
Greenfield JM, Levine LA (2005) Peyronie’s disease: etiology, epidemiology and medical treatment. Urol Clin North Am 32:469–478. doi:10.1016/j.ucl.2005.08.011
Scott WW, Scardino PL (1948) A new concept in the treatment of Peyronie’s disease. South Med J 41:173–177
Akkus E, Carrier S, Rehman J et al (1994) Is colchicine effective in Peyronie’s disease? A pilot study. Urology 44(2):291–295. doi:10.1016/S0090-4295(94)80155-X
Zarafonetis CJ, Horrax TM (1959) Treatment of Peyronie’s disease with potassium para-aminobenzoate (potaba). J Urol 81(6):770–772
Ralph DJ, Brooks MD, Bottazzo GF et al (1992) The treatment of Peyronie’s disease with tamoxifen. Br J Urol 70(6):648–651
Biagiotti G, Cavallini G (2001) Acetyl-L-carnitine vs tamoxifen in the oral therapy of Peyronie’s disease: a preliminary report. BJU Int 88(1):63–67. doi:10.1046/j.1464-410x.2001.02241.x
Ralph DJ, AL-Akraa M, Pryor JP (1995) The Nesbit operation for Peyronie’s disease: 16-year experience. J Urol 154:1362–1363. doi:10.1016/S0022-5347(01)66862-9
Devine CJ, Horton CE (1974) Surgical treatment of Peyronie’s disease with a dermal graft. J Urol 111:44
Lue TF, El-Sakka AI (1998) Venous patch graft for Peyronie’s disease. Part I. technique. J Urol 160:2047–2049. doi:10.1016/S0022-5347(01)62239-0
Wilson SK, Cleves MA, Delk JR (2001) Longterm follow-up of treatment for Peyronie’s disease: modelling the penis over an inflatable penile prosthesis. J Urol 165:825–829. doi:10.1016/S0022-5347(05)66537-8
Carson CC (1998) Penile prosthesis implantation in the treatment of Peyronie’s disease. Int J Impot Res 10:125–128. doi:10.1038/sj.ijir.3900330
Levine LA, Sevier VL (2003) A double blind, placebo-controlled trial of electromotive drug administration (EMDA) using verapamil vs. saline for Peyronie’s disease: preliminary results. J Urol 169(4):274–275
Levine LA, Goldman KE, Greenfield JM (2002) Experience with intraplaque injection of verapamil for Peyronie’s disease. J Urol 168(2):621–625. doi:10.1016/S0022-5347(05)64691-5
Sahin M, Basar MM, Bozdogan O, Atan A (2001) Short-term histopatholoic effects of different intracavernosal agents on corpus cavernosum and antifibrotic activity of intracavernosal verapamil: an experimental study. Urology 58:487–492. doi:10.1016/S0090-4295(01)01211-0
Bennett NE, Guhring P, Mulhall JP (2007) Intralesional verapamil prevents the progression of Peyronie’s disease. Urology 69:1181–1184. doi:10.1016/j.urology.2006.02.048
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shirazi, M., Haghpanah, A.R., Badiee, M. et al. Effect of intralesional verapamil for treatment of Peyronie’s disease: a randomized single-blind, placebo-controlled study. Int Urol Nephrol 41, 467–471 (2009). https://doi.org/10.1007/s11255-009-9522-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-009-9522-4