Abstract
Seven donkeys raised in different locations in Egypt were found to have a unilateral eye showing motile white worms in the aqueous humor. The parasites were surgically removed from the anterior chamber of the eye in five out of the seven donkeys, and were cleared, mounted and identified as Setaria equina based on light microscopic features. The ocular infection with S. equina reported herein may be the first reported aberrant cases in the Egyptian animals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adult Setaria equina is a nematode commonly found floating free within the peritoneal cavity of the equines in all parts of the world (Coleman et al. 1985), and is considered non pathogenic in their natural hosts in most cases. Serious pathogenic effects could occur when such parasite is found in unusual habitats such as ocular glob and central nervous system (Hillyer et al. 2001).
Setaria equina can have erratic locations, mainly in the pleural cavity, the eye, the brain, the spinal medulla and the testicles of horses (Solusby 1982; Vivas et al. 2000; Yadav et al. 2006; Gangwar et al. 2008).
Another filarial worm, Setaria digitata was also reported in the eye of both horses (Jemelka 1976; Yadav et al. 2006) and cattle (Ohtake et al. 1989; Shin et al. 2002). Setaria labiatopapillosa was also reported in subconjunctival eye infection in human in Romania (Panaitescu et al. 1999). In Egypt, the infection with Setaria equina has been recorded by Selim and Fouad (1965), Lotfia (1972), Abu El-Magd and Ahmed (1994) and Abdel- Wahab and Ashour (1999).
The aim of this study was to identify the cause of this condition (the infection of the donkey eyes with Setaria equina) and to set trials for the treatment by surgical interference.
Materials and methods
Between 2005 and 2007, seven donkeys of different sexes and ages from different localities (Kafr El-Sheikh and Algharbia governments) were admitted to the clinic of the Faculty of Veterinary Medicine, Kafr El-Sheikh University, Egypt. All donkeys were found to have one eye opaque. The history supplied by the owner in all cases was a sudden or gradual change in the physical appearance of the eye. Blindness or defective vision had been noted on the affected side. There was no evidence of an ocular pain or discharge.
The naked eye examination of the affected eyes revealed presence of corneal opacities, and deep vascularization and pigmentation. The corneas were cloudy and dirty in color. Corneal opacities appeared as ground glass-like appearance and occupied the entire cornea or have been localized. Pigments were either superficial at the epithelium and the superficial layer of the stroma in the recent cases or deep at the deeper parts of the stroma in the old cases. In contrast to the healthy and the non-affected eyes, the corneas of the affected eyes were leukomatous and therefore the function of the iris could not be determined.
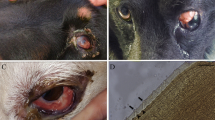
Upon close examination of the recent cases, thread-like white worms were found rapidly moving or swimming within the anterior chamber of the affected eyes (Fig. 1). However, in the old cases the cornea was so severely opaque that the movement of the parasite inside it could be observed only occasionally at the narrow edge of the corneal limbus (Fig. 2). Light stimulation increased the motility of the worm and subsequently, the discomfort of the patient. Except for mild depression, general clinical signs such as body temperature and other physical examinations were all normal.
The worm was removed from the eyes of 5 donkeys as follows. Under the influence of general anesthesia, the affected eye was draped and retractors were positioned to expand the eyelids. A generous amount of oxytetracycline and polymyxine B sulfate ophthalmic ointment (Terramycin, Pfizer) was applied on and around the conjunctiva. A 10 ml disposable syringe with a18-gauge needle was inserted into the anterior chamber of the affected eye at 10 o’clock position of the cornea about 5 mm apart from the limbus. A part of the aqueous humor was to be withdrawn before the parasite could be removed through a small perilimbal corneal incision, approximately the same width as No.15 blade (4 mm). However, the needle got clogged immediately after the piston of the syringe was pulled out to withdraw the aqueous humor. When the needle was pulled out of the eye, the tail or head part of worm plugged the needle hub. As soon as the incision was made and the fluid pressure from the anterior chamber was released, the worm appeared at the incision site and was grasped and withdrawn with alligator forceps inserted through the incision while the cornea was held with tissue forceps. This prevented the damage of the vital parts of the eye, which is always a hazard when the anterior chamber of the eye is surgically invaded. The entire parasite was then successfully removed from the eye. No additional worms were found upon closer examination and then the wound was liberally irrigated with a balanced salt or lactated Ringer’s solution. The defect was sutured with 6–0 polyglactin 910 (Vicryl, Ethicon) and the eye was treated with antibiotics. No additional surgical procedure was done to relieve the blindness of the affected eyes. Postoperatively, topical Terramycin and Atropine sulfate ointments (1%) were applied on the operated eye twice daily for seven days and the eye was protected with a cloth to alleviate atropine- induced photophobia. Atropine was used to prevent anterior synechia.
Blood samples were collected from the infected donkeys for the detection of microfilariae. The collected worms were washed in a physiological saline, and were cleared, mounted and identified according to Solusby (1982) and Abdel Wahab and Ashour (1999).
Results
Seven donkeys were presented in this study of which four were intact males and three were females, ranging in age from 2 to 11 years, with an average age of 7.4 years.
Setaria eqiuna adult female parasites were identified from the eyes of 5 out of the 7 infected donkeys based on light microscopic morphological description. The worms were 10.4 cm in average length. The parasites of the eyes of the other two cases could not be obtained due to the disagreement of the owners.
The mouth opening of the parasite (stoma) is surrounded by a raised cuticular area (cuticular ring) which bears 4 cuticular elevations (two dorsal & two ventral) with separate bases. The amphids (sense organs) are clearly seen on both lateral sides as hilly cuticular elevations (Fig. 3)
The female posterior extremity has a conical shape, with small raised cuticular collar near the tip of the tail. The tail bears several small outgrowths and ends in a simple point (Fig. 4). Upon examination of blood samples from the infected donkeys, no microfilariae were detected.
When the sutures were removed ten days after surgery, all the incisions appeared to have healed. Some scarring was seen in 3 cases. Eight weeks after surgery there was only a slight scar, which would be hidden by the dorsal eyelid. In two operated cases, in which the infection was recent, the iris was functional, the cornea was clear and the vision seemed normal.
Discussion
Adult Setaria spp. are commonly found in the peritoneal cavity of equines and cattle and are generally harmless to the host. Heterotropic parasitism of equines with Setaria spp. is rarely known (Buchoo et al. 2005). The aberrant parasitism of female worms in the eyes of donkeys described herein is rare. Recently, it was reported from equines by Muhammad and Saqib (2007) in Pakistan and by Gangwar et al. (2008) in India. To the best of our knowledge, this is the first report of identification of S. equina in the anterior chamber of the donkey's eye.
In Egypt, previous records of eye infection of donkeys with Setaria equina by Selim and Fouad (1965), Lotfia (1972) and Abu El-Magd and Ahmed (1994) were associated with clinical signs including nasolacrimal duct obstruction, continuous lacrimation, dermatitis and conjunctivitis. These symptoms were never seen in the infected cases of the present investigation, so that previous records of eye infection in the Egyptian donkeys might be attributed to the infection of conjunctival sacs and not of the anterior chamber of the eye with parasites.
The female S.equina that were found in donkey's eyes in this investigation may have been transmitted through mosquitoes or other blood sucking insects, the intermediate host of the parasite, (Sigmund 1973; Wijesundera et al. 1999). However, in the light of earlier reports of prenatal infection with Setaria spp. (Solusby 1982; Fujii et al. 1995) and S. marshalli in Korea (Wee et al. 1996) and Japan (Fujii et al. 1995), the possibility of such a transmission route cannot be disregarded in the case reported herein.
How long the ocular opacity had existed before it was observed is not known. Corneal edema due to dead filarial worm in the anterior chamber with attachments to the endothelium was reported by Basak et al (2007). It is probable that the dead worm liberates toxin into the anterior chamber which is lethal for the endothelium and that leads to corneal edema.
Although medical and surgical therapies have been advocated for the management of equine ocular filariasis (Jemelka 1976; Muhammad and Saqib 2007), surgical removal has been recommended and, as in the present cases, is often curative. Presurgical adulticide therapy is not advised as sever inflammatory reactions to the dead filarial may cause intractable uveitis.
This type of surgery can be performed using a local analgesic. However, we used a general anesthetic to gain a better control of the animal and to prevent unnecessary trauma to vital structures of the eye. The 10 o’clock position was chosen to minimize leakage of aqueous humor due to the flow of fluid under the effect of the gravity during and after surgery. An incision was made as close to the limbus as possible because the cornea is virtually a vascular, with healing depending on the blood supply which originates at the limbus and follows to the site of injury (Jemelka 1976).
Ocular Setaria equina infection in donkeys should send a warning message to breeders of horses especially the highly valuable breeds of Arabian horses to set programs for effective control of filariasis as well as their insect vector. Attention should also be paid to the possibility of prenatal infection.
References
Abdel- Wahab, T.M., and Ashour, A.A., 1999. Scanning electron microscopy of the two filarial nematodes Setaria equina and Onchocerca cervicalis from kafr El-Sheikh area, Egypt. Alexandria Journal of Veterinary Science, 15, 541–547
Abu El-Magd, A., and Ahmed, Z. G., 1994. The occurrence of Setaria equina in donkey’s eyes and their treatment. Assiut Veterinary Medecine Journal, 31, 86–90
Basak,S.K., Hazra,T.K., Bhattacharya,D., 2007. Persistent corneal edema secondary to presumed dead adult filarial worm in the anterior chamber. Indian Journal of Ophthalmology, 55, 67–69
Buchoo, B. A., Pandit, B. A., Shahardar, R. A., Parrah, J. D., Darzl, M. M., 2005. Surgical management and prevalence of ocular filariasis in equines. Indian Veterinary Journal, 82, 81–82
Coleman, S. U., Klei, T. R., Fremch, D. D., 1985. Prevalence of Setaria equina (Nematode: Onchocercidae) in Southeastern Louisiana horses. Journal of Parasitology, 71, 512–513
Fujii, T., Hayashi, T., Ishimoto, A., Takahashi, S., Asan, H., Kato, T., 1995. Prenatal infection with Setaria marshalli (Boulenger 1921) in cattle. Veterinary Parasitology, 56, 303–309
Gangwar, A. K., Devi S., Singh H. N., Singh A., 2008. Ocular filariasis in equines. Indian Veterinary Journal, 85, 547–548
Jemelka, E.D. 1976. Removal of Setaria digitata from the anterior chamber of the equine eye. Veterinary Medicine Small Animal Clinic, 71, 673–675
Hillyer, L., Coles, G., Randle, T., 2001. Setaria equina in the UK. Veterinary Record, 149, 464
Lotfia, S.F., 1972. Some surgical problems caused by filariae in domesticated animals. M.D.Vet. Thesis, Faculty of Veterinary Medicine, Cairo University.
Muhammad, G. and Saqib, M., 2007. Successful treatment of ocular equine microfilariasis (setaria Species) with ivermectin. Veterinary Record, 160, 25–26
Ohtake, O., Sonoda, M., Matsukawa, K., Fukumoto, S., Takashi, K., Kurosawa T., 1989. Clinical studies on bovine autumnal conjunctivitis in Japan. Japanese Journal of Veterinary Science, 51, 618–620
Panaitescu, D., Preda, A., Bain, O., Bugarin, A., 1999. Four cases of human filariasis due to Setaria labiatopapillosa in Bucharest, Romania. Roumania Archieves of Microbiology and Immunology, 58, 203–207
Selim, M.K. and Fouad, K.A., 1965. Incidence of equine filariasis in Egypt. Veterinary Medical Journal of Giza, 10,113–118
Shin, S.S., Cho, K.O., Wee, S.H., 2002. Ocular infection of cattle with Setaria digitata. Journal of Veterinary Medical Science, 64, 7–10
Sigmund, O.E., 1973. The Merck Veterinary Manual 4th. Ed. Merk & Co., Inc., Rahway, N. J., p. 209
Solusby, E.J.L., 1982. Helminths, Arthropods and Protozoa of Domesticated Animals, 7th. Ed. Bailliere Tindall, London, pp. 316–319
Vivas, R.R, Canche, U.D., Lina, E.M., 2000. Filariosis por Setaria equine en un caballo del estado de Yucatán, México. Review Biomedical, 11, 183–185
Wee, S.H., Jang. H., Joo, H.D., Kang, Y.B., Lee, C.G., 1996. Setaria marshalli infection in neonatal calves. Korean Journal of Parasitology, 34, 207–210
Wijesundera, W.S., Chandrasekharan, N.V., Karunangake, E.H., 1999. A sensitive polymerase chain reaction based assay for detection of Setaria digitata: The causative organism of cerebrospinal nematodiasis in goat, sheep and horse. Veterinary Parasitology, 81, 225–233
Yadav, A., Kumar, A., Bhadwal, M.S., Khajuria, J.K., Gupta, A., 2006. Ocular setariosis in horses: A case study. Journal of veterinary parasitology, 20, 2
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marzok, M.A., Desouky, AR.Y. Ocular infection of donkeys (Equus asinus) with Setaria equina . Trop Anim Health Prod 41, 859–863 (2009). https://doi.org/10.1007/s11250-008-9263-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-008-9263-x