Abstract
The aims of the present study were to examine the seroprevalence of neosporosis in beef herds from three southern states of Mexico and determine the association with several risk factors. A cross-sectional serological survey for Neospora caninum was carried out by sampling a total of 596 animals from 31 herds in Chiapas, Veracruz, and Yucatan States and tested using an ELISA assay (IDEXX) to detect anti-N. caninum antibodies. The overall prevalence was 11.6%, (95% CI: 0.93–0.14), however, the prevalence for Chiapas was 15% (30/200) (95% CI: 0.11–0.21), in Yucatan 11.3% (21/186) (95% CI: 0.07–0.17) and in Veracruz 8.6% (18/210) (95% CI: 0.05–0.13). Of the 596 serum samples taken, 578 were from females and 18 were bulls., Only one bull sample was found seropositive from one herd in Veracruz State. The overall herd-prevalence was 70.9% (22/31) (95% CI: 0.53–0.84), and by State was: Chiapas 90% (9/10) (95% CI: 0.60–0.98); Yucatan 72.7% (8/12) (95% CI: 0.39–0.86); and Veracruz 50% (5/10) (95% CI: 0.24–0.76), respectively. The highest age-prevalences were found in animals 1 yr old (19.4% [95% CI: 0.01–0.35]) and 4 yrs old (19.6% [95% CI: 0.12–0.29]), and the lowest in animals 3 –yrs of age (6.2%,[95% CI: 0.02–0.20]). No association was found among all the variables tested in this study since most of the ranches have similar management conditions. In conclusion, N. caninum infection is common among beef herds in the Mexican tropics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
N eospora caninum is a heteroxenous cyst-forming apicomplexan parasite which has emerged as an important cause of reproductive failure in cattle worldwide (Dubey et al. 2007). In the Neospora life cycle, dogs (McAllister et al. 1998) and coyotes (Gondim et al. 2004) are both definitive hosts; cattle and other mammals are natural intermediate hosts (Dubey et al. 2007). Cattle are generally infected prenatally by transplacental infection from persistently infected dams (Björkman et al. 1996; Paré et al. 1996; Davison et al. 1999). Epidemiological evidence also suggests horizontal transmission (Davison et al. 1999; Dijkstra et al. 2001). Calves (De-Marez et al. 1999) and pregnant cows (Trees et al. 2002; McCann et al. 2007) can be experimentally infected by Neospora oocyst shedding by dogs when administered orally. In addition, the N. caninum life cycle has been reproduced by cyclical oral transmission between dogs and cattle (Gondim et al. 2002). Neospora caninum DNA was recently reported in fresh and frozen semen from naturally infected bulls (Ortega-Mora et al. 2003; Caetano-da-Silva et al. 2004; Ferre et al. 2005), and the possibility of venereal transmission has been suggested in bovine neosporosis.
In Mexico, bovine neosporosis has been reported in dairy cattle; however, in beef cattle raised in tropical regions there is no information about this parasitic disease.
The aims of the present study were to examine the seroprevalence of neosporosis in beef herds from three southern states of Mexico and determine the association with some risk factors.
Materials and methods
Situation of the herds and animals
A cross-sectional serological survey for Neospora caninum was carried out on cattle beef in three Mexican southern states, Chiapas, Veracruz and Yucatan. A total of 31 herds and 596 animals were sampled to detect anti-N. caninum antibodies, between September to December 2005. A random sample of animals ≥1 year old, was selected for testing from each herd so that the number of cows from each herd was sufficient to estimate the herd prevalence with 5% or less error and 95% confidence interval (CI), conservatively assuming the variance to be 25% and the seroprevalence 50%.
Blood sampling
Blood samples were collected from 578 animals; 71 heifers, 507 cows and 18 bulls in plain vacutainers from the coccygeal or jugular vein and transported to the Parasitologia Veterinaria laboratory in Jiutepec, Morelos, Mexico. After centrifugation at 1000 × g for 15 min, serum was removed and stored at − 20 C until analysis.
Serologic tests
Serum samples were tested using the ELISA test (IDEXX Laboratories) for detection of antibodies against N. caninum. Each sample was tested in replicated tests. Sera with absorbance values above the cut-off level of 0.50 were considered to be positive in accordance with the manufacturer’s instructions. The sensitivity and specificity of this ELISA test was reported 100% and 93%, respectively. The predictive values of the test were 93.75% positive and 100% negative (Wapenaar et al. 2007; Wu et al. 2002)
Data analyses
Seropositive rates for N. caninum antibodies were analyzed in relation to the animal data, such as origin, and age using a two-sided chi-square test or Fisher exact test. P-values, odds ratios (OR), and their 95% confidence intervals (CI) were computed. P-values 0.05 were required to demonstrate statistical significance, (Thurmond and Hietala 1995).
Management practices on farms
A questionnaire was filled out during a personal interview with the farmer or the manager. Animal-level information included age and number of abortions during the last three years. Herd-level information included replacement practices, use of artificial insemination and the presence of dogs on the farm.
Results
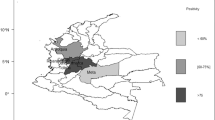
The overall prevalence determined in the study was 11.6% (95% CI: 0.09– 0.14); by state the prevalence was: Chiapas 15% (30/200), Yucatan 11.3% (21/186), and Veracruz 8.6% (18/210). Positive farms were found in 11 of the 15 municipalities studied. Of the 596 serum samples, 578 were from females and 18 from bulls; only one bull sample was found positive in one herd in Veracruz State (Table 1). Chi-square test showed statistical significant differences in the estimated prevalence between animals from Veracruz and Chiapas States (p = .02). The prevalence range by municipality was 0 to 45%.
The highest age-prevalence was found in 1 and 4 yr olds (19.4% and 19.6%, respectively) and the lowest in 3 yr olds (6.2%); there was no significant difference in the animal level prevalence of N. caninum infection among different ages (Table 2).
From the 31 herds tested a herd was considered to be positive when at least one animal was seropositive and the herd-prevalence was 70.9% (22/31) (CI: 0.53–0.84). The herd-prevalence by state was in Chiapas 90% (9/10) (CI: 0.60–0.98), Yucatan 72.72% (8/12) (CI: 0.39–0.86) and Veracruz 50% (5/10) (CI: 0.24–0.76). The range prevalence within herd was 0–50%.
Of the 578 females tested 14 aborted but only two were seropositive. The OR was 1.28 (95% CI 0.28–5.82). The OR between the presence of dogs on the ranches and seropositive results was 0.17 (95% CI.59–.95). From 18 Simmentals tested only one was seropositive in the state of Veracruz.
No association was found among the management variables tested in this study since most of the ranches had similar conditions.
Discussion
The overall estimated animal level seroprevalence of N. caninum infection for beef cattle was 11.6%, (CI: 0.93–0.14), and by comparison the reports in beef cattle from this and other countries are very similar (Dubey et al. 2007). Although the herd exposure to the agent was relatively high 70.9%, (CI: 0.53–0.84), this figure could have underestimated the true prevalence because we only sampled around 20 animals per farm and farms with low within-herd prevalence may have been misclassified as negative. This potential misclassification of herd status could also affect the analysis of potential risk factors.
The parasite appears to be widely distributed geographically in nearly all the municipalities from the three states where cattle were sampled and all had positive herds. The overall prevalence among beef cattle in our study was lower than prevalence found in dairy cattle in other regions in México (Garcia-Vazquez et al. 2005; Morales et al. 2001). These results could be due to a number of factors including an overall higher exposure among animals in dairy herds.
In this study we examined the distribution of N. caninum infection in beef herds in the southern region of Mexico and estimated that 11.1% (CI: 0.09–0.26) and 15.5% (CI: 0.02–0.05) of beef cows and heifers, respectively, had been exposed to this coccidian parasite. From the results of the prevalence in this Mexican region, it is suggested that the major transmission route of N. caninum infection may be transplacental. If horizontal transmission was an important route of N. caninum infection seroprevalence would have been higher than that in heifers because the chance of being exposed to the parasite becomes higher as the animal gets older. Furthermore, the reason why the major route of N. caninum infection in this region may be vertical transmission can be explained by the results showing that there was no clear-cut association between seroprevalence and presence of dogs or coyotes in beef herds. Nonetheless, the possibility of the horizontal transmission occurring among the beef cattle in this region cannot be denied. No decisive conclusion could be drawn on the involvement of canine species in the transmission of the bovine infection in beef cattle in this particular region, since no samples from the dogs in the target farms were available for anti-Neospora antibodies in this study. Furthermore, the putative ways by which dogs may pose an infection risk to cattle have been reported such as defecation by farm dogs on grass, density of the dogs on the farm location, presence and abundance of coyotes (Dubey et al. 2007). More study is required to clarify the transmission of N. caninum infection among beef cattle in Mexico.
The seroprevalence we found in this study has helped us to better understand the association between neosporosis as a cause of abortion in dairy cattle in Mexico (Garcia-Vazquez et al. 2005) and to asses its economic impact and risk factors that could be implicated in N. caninum infection or abortion in the future. Studies in other countries have suggested a significant economic cost to producers due to losses associated with N. caninum (Trees et al. 1999; Häsler et al. 2006; Koiwai et al. 2006). It is unclear if the impacts will be the same under the production conditions in the tropical regions of Mexico.
No reports on beef cattle raised in this region in Mexico have been done in infected beef herds. The primary management goals for infected beef herds include preventing abortions and reducing the risk of both vertical and horizontal transmission of N. caninum so that the prevalence of infection in the herd is reduced in the long term. Reducing the risk of introduction of the parasite into the herd, as discussed above, is also important, so that on-farm transmission control efforts are not offset by the reintroduction of N. caninum from outside the farm. If herd status is important for genetic sales, periodic testing of the herd (every 1 or 2 yrs) to confirm that N. caninum has not been introduced into the herd may be cost-beneficial.
In conclusion, N. caninum infection is common among beef herd in the Mexican tropics. Since beef industry is one of the key industries in this region, the economic effect and risk factors of N. caninum infection among beef cattle in this country should be estimated in the near future.
Abbreviations
- CI:
-
confidence interval
- DNA:
-
deoxyribonucleic acid
- OR:
-
odds ratio
References
Björkman, C, Johansson, O., Stenlund, S., Holmdahl, OJ., Uggla, A., 1996. Neospora species infection in a herd of dairy cattle. Journal of American Veterinary Medical Association, 208,1441–1444.
Caetano-da-Silva, A., Ferre, I., Collantes-Fernandez, E., Navarro, V., Aduriz, G., Ugarte-Garagalza, C., Ortega-Mora, L.M., 2004. Occasional detection of Neospora caninum DNA in frozen extended semen from naturally infected bulls. Theriogenology, 62, 1329–1336 doi:10.1016/j.theriogenology.2004.01.010
Davison, H.C., Otter, A., Trees, A.J., 1999. Estimation of vertical and horizontal transmission parameters of Neospora caninum infections in dairy cattle. International Journal for Parasitology, 29, 1683–1689 doi:10.1016/S0020-7519(99)00129-0
De-Marez, T., Liddell, S., Dubey, J.P., Jenkins, M.C., Gasbarre, L., 1999. Oral infection of calves with Neospora caninum oocysts from dogs: humoral and cellular immune responses. International Journal for Parasitology, 29, 1647–1657 doi:10.1016/S0020-7519(99)00154-X
Dijkstra, T.H., Barkema, H.W., Eysker, M., Wouda, W., 2001. Evidence of post-natal transmission of Neospora caninum in Dutch dairy herds. International Journal for Parasitology, 31, 209–215 doi:10.1016/S0020-7519(00)00160-0
Dubey, J.P., Schares, G., Ortega-Mora, L.M., 2007. Epidemiology and control of neosporosis and Neospora caninum. Clinical Microbiology Reviews, 20, 323–367 doi:10.1128/CMR.00031-06
Ferre, I., Aduriz, G., del-Pozo, I., Regidor-Cerrillo, J., Atxaerandio, R., Collantes-Fernández, E., Hurtado, A., Ugarte-Garagalza, C., Ortega-Mora, L.M., 2005. Detection of Neospora caninum in the semen and blood of naturally infected bulls. Theriogenology, 63, 1504–1518 doi:10.1016/j.theriogenology.2004.07.002
Garcia-Vazquez, Z., Rosario-Cruz, R., Ramos-Aragón, A., Cruz-Vazquez, C., Mapes-Sánchez, G, 2005. Neospora caninum seropositivity and association with abortions in dairy cows in Mexico. Veterinary Parasitology, 134, 61–65 doi:10.1016/j.vetpar.2005.07.007
Gondim, L.F., McAllister, M.M., Pitt, W.C., Zemlicka, D.E., 2004. Coyotes (Canis latrans) are definitive hosts of Neospora caninum. International Journal for Parasitology, 34, 159–161 doi:10.1016/j.ijpara.2004.01.001
Gondim, L.F., Gao, L., McAllister, M.M., 2002. Improved production of Neospora caninum oocysts, cyclical oral transmission between dogs and cattle, and in vitro isolation from oocysts. Journal of Parasitology, 88, 1159–1163.
Häsler, B., Regula, G., Stärk, K.D., Sager, H., Gottstein, B., Reist, M., 2006 Financial analysis of various strategies for the control of Neospora caninum in dairy cattle in Switzerland. Preventive Veterinary Medicine, 773–4:230–53 doi:10.1016/j.prevetmed.2006.07.006
Koiwai, M., Hamaoka, T., Haritani, M., Shimizu, S., Zeniya, Y., Eto, M., Yokoyama, R., Tsutsui, T., Kimura, K., Yamane, I., 2006. Nationwide seroprevalence of Neospora caninum among dairy cattle in Japan. Veterinary Parasitology, 88, 1159–1163.
McAllister, M.M., Dubey, J.P., Lindsay, S.D., Jolley, R.W., Wills, A.R., McAguirre, M.A., 1998. Dogs are definitive host of Neospora caninum. International Journal for Parasitology, 28, 1473–1478 doi:10.1016/S0020-7519(98)00138-6
McCann, M.C., MacAllister, M.M., Gondim, L.F.P., Smith, R.F., Kipar, A., Williams, D.J.L., Tress, A.J., 2007. Neospora caninum in cattle: Experimental infection with oocyst can result in exogenous transplacental infection, but no endogenous transplacental infection in the subsequent pregnancy. International Journal for Parasitology, 37, 1631–1639 doi:10.1016/j.ijpara.2007.05.012
Morales, S.E., Trigo, T.F., Ibarra, F., Puente, C.E., Santacruz, M., 2001. Seroprevalence study of bovine neosporosis in Mexico. Journal of Veterinary Diagnostic Investigation, 13, 413–415.
Ortega-Mora, L.M., Ferre, I., del-Pozo, I., Caetano-da-Silva, A., Collantes-Fernández, E., Regidor-Cerrillo, J., Ugarte-Garagalza, C., Aduriz, G., 2003. Detection of Neospora caninum in semen of bulls. Veterinary Parasitology, 117, 301–308 doi:10.1016/j.vetpar.2003.09.015
Paré, J., Thurmond, M.C., Hietala, S.K., 1996. Congenital Neospora caninum infection in dairy cattle and associated calf hood mortality. Canadian Journal of Veterinary Research, 60, 133–139.
Thurmond, M. C., Hietala, S., 1995. Strategies to control Neospora infection in. cattle. The Bovine Practice, 29, 60–63.
Trees, A.J., McAllister, M.M., Guy, C.S., McGarry, J.W., Smith, R.F., Williams, D.J.L., 2002. Neospora caninum: oocyst challenge of pregnant cows. Veterinary Parasitology, 109, 147–154 doi:10.1016/S0304-4017(02)00234-0
Trees, A.J., Davison, H.C., Innes, E.A., Wasttling, J.M. 1999. Towards evaluating the economic impact of bovine neosporosis. International Journal for Parasitology, 29, 1195–1200 doi:10.1016/S0020-7519(99)00093-4
Wapenaar, W., Barkema, H.W., Vanleeuwen, J.A., McClure. J.T., O'Handley, R.M., Kwok, O.C., Thulliez, P., Dubey, J.P., Jenkins, M.C., 2007. Comparison of serological methods for the diagnosis of Neospora caninum infection in cattle. Veterinary Parasitology, 31;143(2):166–73.
Wu, T.J.J., Dreger, S., Chow, Y.W.E., Bowlby, E.E., 2002. Validation of 2 commercial Neospora caninum antibody linked enzyme linked immunosorbent assays. The Canadian Journal of Veterinary Research, 66, 264–271.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garcia-Vazquez, Z., Rosario-Cruz, R., Mejia-Estrada, F. et al. Seroprevalence of Neospora caninum antibodies in beef cattle in three southern states of Mexico. Trop Anim Health Prod 41, 749–753 (2009). https://doi.org/10.1007/s11250-008-9247-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-008-9247-x