Abstract
Soybean was used as a model for studies of chemical induction of gene expression in seeds. A chimeric transcriptional activator, VGE, driven by the soybean seed glycinin G1 promoter, was used to induce the expression of an ER-targeted GFPKDEL reporter protein upon addition of the chemical ligand, methoxyfenozide. The chemical gene switch activated gene expression under in vitro conditions in somatic cotyledonary embryos and zygotic seed embryos cultured from transgenic soybean plants, as well as in seeds in planta under greenhouse conditions. The efficiency of induction of GFP expression under different growth conditions was strongly influenced by the developmental stage of the seed and availability of the inducer. The formation of ER-derived GFP-containing protein bodies in seed storage parenchyma cells was correlated with the level of induced expression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A genetic switch that allows for the spatial and temporal activation or disruption of gene expression is a useful tool for functionally analyzing endogenous and exogenous genes. The ability to conditionally control gene expression in specific cell or tissue types has many applications for fundamental and applied research. Manipulating the expression of a single gene or a gene network can increase our understanding of the complexities underlying primary and secondary metabolism, growth and development. In agricultural biotechnology applications the regulation of transgene expression may be particularly important in cases where the synthesis and accumulation of a specific gene product is required in a temporal or tissue-specific manner. Chemically inducible gene expression systems have been used to achieve marker-free plant transformation (Zuo et al. 2002), activation tagging (Sun et al. 2003), conditional genetic complementation and restoration of biological functions (Unger et al. 2002) and induction of RNAi (Wielopolska et al. 2005; Dietrich et al. 2008). Inducible expression systems employing different activator molecules have been developed and used in both dicotyledonous and monocotyledonous plants (reviewed in Padidam 2003; Moore et al. 2006). Future gene switch applications may involve the production of plant-based biopharmacueticals, as well as the commercial production of phytochemicals, industrial oils, lipids, carbohydrates and proteins (Hughes et al. 2004; Sudarshana et al. 2006; Kourtz et al. 2007).
The underlying mechanism behind a genetic switch involves the intracellular formation of a sensitive, stable ligand/receptor complex. In this study, a highly regulated gene switch utilizing the chimeric synthetic transcription factor VGE (Padidam et al. 2003; Koo et al. 2004) was adapted for use in soybean seed. The modular transcriptional activator consists of the Herpes simplex VP16 activation domain (V), the yeast Gal4 binding domain (G, residues 1–147) and the ligand binding domain of the spruce budworm, Chloristoneura fumiferana, ecdysone receptor (E, residues 206–539). The tripartite protein VGE is functionally inactive as a regulator of gene transcription in the absence of ligand. Interaction of the chemical inducer, the nonsteroidal ecdysone analog methoxyfenozide, with VGE leads to its binding to a specific synthetic promoter upstream of the gene of interest resulting in the expression of that gene (Martinez et al. 1999; Unger et al. 2002; Padidam et al. 2003; Koo et al. 2004; Tavva et al. 2006; Kourtz et al. 2007; Dietrich et al. 2008). Methoxyfenozide is the active ingredient in Intrepid (Dow Agrochemicals), an approved insecticide on certain crops.
The effectiveness of the chimeric transcription factor was described by Koo et al. (2004). In this study VGE was expressed in Arabidopsis thaliana from the Cassava mosaic virus promoter on the same T-DNA containing five copies of UASGal4 placed upstream of a tobamovirus coat protein gene (CgCP). In transgenic plants, a 700-fold induction of the viral coat protein synthesis was achieved, resulting in coat-protein mediated disease resistance.
Soybean has many industrial and nutritional applications, and a number of studies have demonstrated that the composition of the seed can be changed by genetic engineering techniques (reviewed in Wang et al. 2003; Schmidt and Herman 2008). Future uses of soybeans for production of modified oils, proteins or secondary metabolites may require development of new approaches to ensure precise gene regulation. We have recently examined the use of soybean for inducible seed-specific gene expression employing the VGE transcriptional activator and methoxyfenozide. The VGE gene switch module was placed under the control of the soybean glycinin promoter and the gene switch system was tested using a green fluorescent protein (GFP) reporter. Qualitative and quantitative analyses of inducible gene expression were conducted with transgenic soybean somatic embryos and seeds from transgenic plants.
Materials and methods
Cassette construction
The coding region of VGE was amplified from pVGE-1300 (Koo et al. 2004) using Pfu ultra polymerase (Stratagene, CA, USA) and primers VGE-S (5′-AAGGGCCCAAAAATGGCCCCCCCGACCGATGTCAG-3′) and VGE-AS (5′-ATTGGGCCCTTACTAGAGATTCGTGGGGGACTCGA-3′), which also introduced Bsp120 flanking sites. The resulting PCR product was T/A tailed using Taq polymerase (New England Biolabs, MA, USA) and cloned into pGEM-T Easy vector (Promega, WI, USA). After sequence verification, the coding region was excised with Bsp120 and subcloned downstream of the glycinin G1 subunit promoter (pGly) into the NotI-digested, dephosphorylated pGly vector (Nielsen et al. 1989; Schmidt and Herman 2008) containing a hygromycin resistance gene controlled by the ubiquitin promoter (Garbarino and Belknap 1994), giving rise to pGly-VGE. Five copies of UASGal4 DNA-response elements fused to the –45 CaMV35S minimal promoter and polylinker region were amplified using the primer pair: (5′-AAAAAGGTACCGCTCGGAGGACAGTACTCC-3′ and 5′-AAAAAGGTACCGGTCACTGGATTTTGGTTTTA-3′) and the CaMV35S terminator region was amplified using the primer pair: (5′-GGTTATAATTAGCGGCCGCTCAGGTCGGTGTCCTCTCCA-3′ and 5′-TCACCTAGGCCCGGGTTATAATTAGCGGCC-3′) from pVGE-1300, introducing a unique NotI site into the polylinker region as well as two flanking Acc65I sites. After subcloning into pGEM-T Easy and verification by sequence analysis, the resulting cassette was released using Acc65I and subcloned into BsiWI-digested, dephosphorylated pGly-VGE, upstream of the pGly promoter giving rise to pGly-VGE-5XM. The DNA fragment coding for an ER-targeted eGFP, termed ER-GFPKDEL (Schmidt and Herman 2008) was then subcloned into NotI-digested, dephosphorylated pGly-VGE-5XM, downstream of the target 5XUASCaMV35S promoter. The final plasmid pGly-VGE-5XM-ER-GFPKDEL was subsequently used for soybean embryo transformation.
Soybean transformation, screening and methoxyfenozide treatment of cotyledonary embryos
Glycine max [L.] Merrill cv. Jack was subjected to biolistic transformation and somatic embryogenesis as described by Schardl et al. (1987) and Trick et al. (1997), and regeneration of fertile plants was done according to Schmidt et al. (2005).
Inducible GFP expression in transformed soybean lines was examined by placing 3–4 week-old cotyledonary embryos in 6-well tissue culture plates (two embryos per well) containing 6 ml of ShaM maturation media per well (Schmidt et al. 2005) with slow shaking at 24°C on a 16 h light (60 μM m−2 s−1)/8 h dark cycle. Embryos were induced with the addition of 1% Intrepid 2F, equivalent to 22.6% (w/v) or ~613 mM (Dow AgroScience LLC, IN, USA), in sterile water, bringing the final concentration of methoxyfenozide (MOF) to 80 μM in the media.
Cultivation and methoxyfenozide treatment of soybean cotyledons in vitro
To study the induction of GFP expression in transgenic soybean seeds in vitro cultures of immature cotyledons were initiated according to Thompson et al. (1977). Briefly, pods were surface sterilized in a 0.5% commercial bleach solution containing 0.1% SDS and then rinsed three times in sterile water. Pods were cut opened under aseptic conditions and immature seeds (7–8 mm or 10–12 mm in length and 60–100 mg in weight) were transferred to Petri dishes. After halving each seed with a scalpel and removing the seed coat, the root and shoot axes were excised from the cotyledon. Cotyledons from one embryo were placed separately in 35 mm tissue culture dishes containing 4 ml of modified MS (Thompson et al. 1977). Cotyledons were submerged into media and shaken at 48 cycles per min at 24°C and light intensity of 110 μM m−2 s−1. After 2 days of incubation, one cotyledon of the pair from a given seed was subjected to 80 μM of MOF in MS.
Transgenic plant growth and methoxyfenozide application
Transgenic as well as control soybean plants were grown in a greenhouse for 120 days at 25°C under 16 h light (1,000 μM m−2 s−1). Approximately 1 week after flowering was initiated, plants were treated with Intrepid 2F by soil drenching. Prior to application the inducer was diluted to the indicated concentrations in Jack’s Professional 15-16-17 peat-lite fertilizing solution (J.R. Peters, Inc, PA, USA) made to the manufacturer’s specifications. Five hundred milliliter of the mixture containing different concentrations of the inducer was added per gallon pot containing one plant. Control plants were drenched with the same volume of fertilizing solution without inducer. The application was repeated three times over a 10 day interval.
Mature dry T1 seeds were harvested and visually selected under a fluorescent dissecting microscope (Nikon SMZ1500, Japan) using 450 nm blue light for excitation. GFP-positive seeds were germinated and the resulting plants were used for experimental studies as well as taken to the T2 generation to obtain homozygous seeds.
The staging and classification of maturing seeds was done according to seed fresh weight and color as described by Miernyk et al. (2009): <10 mg (S1); 30–40 mg (S2); 75–100 mg (S3); 100–150 mg (S4); 200–250 mg (S5); 300–350 mg (S6); dehydrating yellow green (300–350 mg) (S7) and dehydrating yellow (200–250 mg) (S8).
Protein extraction and fluorimetry
Total protein extracts were prepared from seed or cotyledonary embryos ground in 400–1,000 μl of extraction buffer (PBS, pH 7.6, 0.1% Triton) using 3 volumes buffer (w/v) per 100 mg of seed weight, followed by centrifugation at 14,000 rpm for 10 min. Protein concentrations were determined with a CB-Protein Assay Kit (EMD Bioscience, Inc., CA, USA). GFP fluorescence was measured on a fluorescence spectrophotometer (Varian Inc., CA, USA), using excitation and emission wavelengths of 488 and 510 nm, respectively. Recombinant GFP (Vector Laboratories, CA, USA) was used to produce a standard curve.
Western blot analysis
Extracted proteins were separated by standard SDS–PAGE using 12% gels and transferred to nitrocellulose membrane (Protran BA83, Whatman, Germany). The membrane was pre-incubated in PBS containing 2% BSA at room temperature for 1 h. For immunodetection a mouse monoclonal anti-GFP antibody (Clontech, CA, USA), diluted 1:500 was used, followed by an alkaline phosphatase (AP)—conjugated anti-mouse antibody (1:10.000; Sigma, MO, USA). AP activity was determined using the substrate NBT/BCIP (Sigma, USA).
Fluorescence and confocal microscopy
GFP expression in transgenic soybean cotyledons and seeds was observed under a dissecting microscope equipped with a fluorescence module consisting of a 100 W mercury lamp (Nikon, Japan) and GFP filter. Pictures were taken with a Retiga 1300 camera (QImaging, Canada). For observation of single cells, live fluorescent samples were examined under a LSM 510 laser scanning microscope (Carl Zeiss Inc., NY, USA) using an excitation wavelength of 488 nm and detection wavelength between 500 and 520 nm. Confocal images were analyzed using Zeiss LSM image examiner software.
RNA purification and Northern dot-blot hybridization
Total RNA was isolated from seeds after grinding in liquid nitrogen using Trizol reagent (Invitrogen, CA, USA) according to the manufacturer’s instructions. Three seeds of the same developmental stage, taken from the level of the third internode, were used per sample. DNA fragments of VGE (651–1,109 bp; Padidam et al. 2003) and glycinin G1 (1,750–2,308 bp; Sims and Goldberg 1989) genes were radiolabeled with 32P-dCTP by random priming using the Rediprime II labeling system (Amersham Bioscience Inc., USA). For each sample, 10 μg of RNA was denatured and transferred onto Hybond-N membrane (Amersham Bioscience, USA) using a Bio-Dot SF Microfiltration Apparatus (Bio-Rad, USA). RNA blots were hybridized with their respective 32P-labeled probes for 36 h at 42°C in ULTRAhyb buffer (Ambion, TX, USA) and washed according to the manufacturer’s instructions. After washing, blots were exposed to a Phosphor Screen and analyzed using ImageQuant 5.2 software (Molecular Dynamics, Amersham Bioscience, Inc., USA).
Results
Construct design
The soybean gene switch construct pGly-VGE-5XM-ER-GFPKDEL was designed to allow uni-directional expression of the VGE and GFP coding sequences. Expression of VGE was driven by the seed-specific promoter from the Gly1 gene encoding the glycinin G1 subunit (Fischer and Goldberg 1982). Glycinin gene expression appears early in embryogenesis, is highly prevalent during the mid-maturation stage and decays prior to seed dormancy (Nielsen et al. 1989). Use of this promoter allows efficient accumulation of recombinant proteins during seed growth and maturation (Moravec et al. 2007; Schmidt and Herman 2008).
In this study, MOF binding and activation of the chimeric receptor lead to the expression of a GFP gene that was modified to contain the Arabidopsis basic chitinase signal sequence (Samac et al. 1990) and a carboxy-terminal KDEL sequence (Herman et al. 1990) to direct and cause retention of the expressed protein in the endoplasmic reticulum (ER).
Transformation and analysis of somatic embryos
Soybean somatic embryos were transformed with the pGly-VGE-5XM-ER-GFPKDEL construct and hygromycin-resistant lines were isolated after 9 weeks following propagation by repetitive embryogenesis on selective media. Globular embryos were transferred to maturation media (ShaM) and developed to the cotyledonary stage (Schmidt et al. 2005). At about 4 months post-bombardment cell lines were analyzed by PCR for the presence of transgenes using primers to GFP and VGE genes. Inducible GFP expression in transformed lines was first examined in 3–4 week-old cotyledonary embryos at which time the transcription of genes encoding soybean seed storage proteins has been initiated and the glycinin G1 promoter is active (Thibaud-Nissen et al. 2003; Schmidt et al. 2005).
Two transgenic embryonic soybean lines, termed VG1-2 and VG1-4, in which MOF-induction of GFP expression was readily observed, were identified from a total of seventeen PCR-positive lines. Most in vitro studies were performed using cotyledonary embryos and seeds derived from line VG1-2 because fewer embryos were obtained from the transformed VG1-4 line.
Upon close examination, it was ascertained that GFP fluorescence was only observed in the tips of cotyledonary embryos (‘cotyledonary tips’), where the glycinin promoter was active, but not along the embryonic axis (Schmidt et al. 2005). Fluorescence was uniformly dispersed and continuous through all cell layers (Fig. 1a). Up to 30 individual cotyledons per line were tested and found to give the same result. Non-induced transgenic cotyledonary embryos, and induced wild type cotyledonary embryos used as controls, showed no GFP fluorescence. GFP expression was activated in embryos when the first signs of cotyledon morphology appeared. Round-shaped and torpedo-shaped embryos that lacked ‘cotyledonary tips’ did not show fluorescence after MOF treatment. Similarly, GFP expression was not induced in more mature cotyledonary embryos that were aged to 5–6 weeks and appeared yellowish in color. Therefore, the temporal window in which the gene switch was active was set by the stages of cotyledonary development.
Inducible GFP expression in cotyledonary embryos in response to MOF. a Expression of GFP in transformed VG1-2 soybean cotyledonary embryos after 48 h of MOF treatment at 80 μM: Panels (a) and (b) MOF-induced; panel (c) non-induced. Embryos were photographed under white (left panel) and blue light (right panel). b Effect of MOF concentration on accumulation of GFP in VG1-2 cotyledonary embryos. Embryos were induced in ShaM media in tenfold serial dilutions of MOF from 0.08 to 80 μM for 48 h. Protein extracts (500 μg of TSP) prepared from pools of three cotyledonary embryo samples were subjected to quantitative fluorimetry. The averages from two independent experiments are shown with standard deviations
Temporal studies revealed that GFP fluorescence was visible in tips of cultured cotyledons at approximately 5 h after addition of 80 μM MOF to the medium with the intensity of fluorescence increasing over the next 48 h. This kinetic was confirmed by fluorimetry and confocal microscopy. Individual cotyledonary embryos with different morphologies demonstrated relatively uniform fluorescence. A dose–response study revealed that the fluorescence level decreased with dilution of the inducer. Weaker, yet visible fluorescence was observed in cotyledonary embryos treated with 0.8 or 0.08 μM MOF (Fig. 1b).
GFP fluorescence was stable throughout the subsequent maturation stages in individual somatic embryos. Prior treatment of embryos with MOF and subsequent induction of GFP expression had no apparent effect on the germination and regeneration processes in tissue culture, and induced cotyledons could be successfully grown to seed-bearing plants in soil under greenhouse conditions. GFP fluorescence was visible throughout the desiccation and rooting stages of mature cotyledons, but decreased substantially when the first trifoliate leaf appeared on germinating seedlings.
Induction of GFP expression in seeds cultured in vitro
The induction of seed-specific GFP expression was examined in transgenic soybean seeds derived from T0 plants of lines VG1-2 and VG1-4. For these studies, immature cotyledons were cultured in vitro (Thompson et al. 1977). Immature heterozygous seeds were isolated from pods and cotyledons were cultured for 48 h prior to treatment with 80 μM MOF. The twin cotyledon from the same seed was used as the non-induced control. GFP fluorescence appeared only in the MOF-treated half seed and was uniformly dispersed throughout the cotyledon (Fig. 2a). A temporal analysis was conducted using cross-sections from individual cotyledons 0, 6, 12, 24 and 48 h after induction. Based upon fluorimetry and immunoblot analyses GFP was apparent about 12 h after adding MOF and the intensity of the fluorescence increased approximately 10–15 fold during the 48 h induction period in similar manner in all induced seeds (Fig. 2b). The intensity of fluorescence increased more slowly at 72–96 h in the presence of the inducer, but the increase was relatively small compared to that observed during the first 48 h (data not shown).
Inducible GFP expression in response to MOF in VG1-2 in vitro seeds. a Single immature VG1-2 seeds were split into halves with one cotyledon used for induction and the other as the untreated control. Cotyledons were photographed under white (left panel) and blue light (right panel). b Time course study of inducible GFP fluorescence in VG1-2 seeds treated in vitro with 80 μM MOF. Total protein was extracted from cross-sections of tissue taken from one cotyledon at 0, 6, 12, 24 and 48 h after MOF treatment. Each filled bar represents one sample taken from a single induced transgenic cotyledon at the time point indicated. In total, 7 sets of induced samples were tested. White bars, wild type cotyledon; black bars; transgenic non-induced cotyledon; both sampled similarly to induced cotyledon (patterned bars). Each sample for fluorimentry contained 500 μg of TSP. For immunodetection using anti-GFP antibody, 20 μg of TSP was loaded per lane
To determine which stage of seed maturation was the most sensitive to induction, cotyledons from seeds ranging from 30 to 400 mg were placed in culture media and treated with inducer as described. Analysis of GFP fluorescence indicated that seeds with a minimum size of 7–8 mm up to 10–12 mm in length and 60–100 mg (stages S3, S4) were the most sensitive to chemical induction (data not shown). No visible GFP fluorescence was observed in seeds of 30–40 mg or less (stage S2) nor in seeds at late maturation stages (up to 350–400 mg) that were treated with MOF.
Induction of seed-specific GFP expression in planta
Application of the inducer to growing soybean plants was performed by soil drenching. T0 plants grown in one gallon plastic pots were treated at different growth stages to optimize the protocol for GFP induction specifically in soybean seed. Activation of GFP expression in planta was successful using 80 μM MOF, the same concentration used for in vitro studies. However, due to the poor solubility of Intrepid in water, a tenfold higher concentration of MOF was applied to ensure inducer availability to the roots in soil. The final protocol consisted of three 500 ml applications of MOF at 800 μM over a 10 day period. Importantly, the first soil drench was applied at the early flowering stage. It was subsequently shown that a single soil application of the inducer at 800 μM during early flowering was sufficient to initiate and maintain GFP protein accumulation in seeds. No GFP fluorescence was detected in other parts of the plant including the roots, stems or leaves (data not shown). Dry mature T1 seeds from MOF-induced T0 plants were visually examined under a fluorescence microscope to select for seeds that showed the highest levels of GFP fluorescence. Plants derived from the seeds were used to establish homozygous soybean lines. Transgenic plants used in subsequent experiments exhibited a Mendelian 3:1 segregation of induced seeds in the T1 and T2 generations. The presence of a single insertion site of the transgene cassette was confirmed by Southern blot analysis (data not shown).
No obvious effects of MOF were observed on plant growth, flowering or seed formation in T0–T2 plants of VG1 lines. Similarly, transgenic seeds derived from T0, T1 and T2 VG1 plants had normal germination rates compared to wild type seeds. However, we noted that most VG1-4 pods contained a single seed, rather than the typical number of 2–4 seeds per pod observed in VG1-2 and wild type plants. In addition, VG1-4 seeds often exhibited indentations in both induced and non-induced plants.
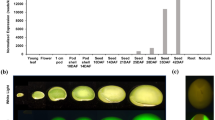
Upon closer examination of induced VG1-2 and VG1-4 seeds, it was revealed that GFP fluorescence was either concentrated in an area near the funiculus opposite the location of the embryo or along the inner side of the seed. In developing as well as mature seeds, fluorescence extended across the seed but a fluorescent gradient remained (Fig. 3). In some seeds a second fluorescent spot appeared near the outer edge of the seed. Interestingly, seeds from lines VG1-2 and VG1-4 exhibited modest differences in the pattern of fluorescence. Extending the time of induction to encompass the entire seed maturation period, or increasing the concentration of inducer, did not result in further quantitative or qualitative changes in the gradient of GFP fluorescence across the seed. In plants treated at the flowering stage, GFP fluorescence first appeared when seeds had matured to the S4 stage and had reached a weight of 120–140 mg (Fig. 4a). In plants induced with MOF after flowering GFP fluorescence was observed about 20 days after MOF application in all seeds that had reached 120–140 mg in weight by that time. At the end of the seed maturation period fluorescent seeds were found in each pod.
GFP expression in VG1-2 and VG1-4 seeds induced in planta by soil drenching with 800 μM MOF. Seeds were split to reveal cotyledonary surfaces. a VG1-2 (stage S4) induced; b VG1-2 (stage S5) induced; c VG1-4 (stage S6) induced; d lateral view of cross-sectioned VG1-2 (stage S5) induced; e VG1-2 (stage S4) non-induced. Seeds were photographed under white (left panel) and blue light (right panel)
a Schematic representation of an induction map determined for a representative homozygous plant, VG1-2-11H. All seeds were analyzed by fluorescence microscopy to observe GFP fluorescence gradient approximately 2 weeks after the first application of MOF. Each circle represents a single pod bearing seeds of the indicated weight. The number of seeds per pod ranged from 2 to 4. The presence or absence of a gradient of fluorescence is indicated by filled and empty circles, respectively. b Changes in VGE and glycinin G1 mRNA levels at different stages during VG1-2-7H soybean seed development. mRNA samples from developing S1-S8 seeds were immobilized on nitrocellulose filters and hybridized with respective P32-labeled probes as described in “Material and Methods”
Semi-quantitative RNA blot analyses performed on seed samples from homozygous plants confirmed that VGE and glycinin G1 mRNAs were expressed coordinately at early stages of seed development and reached their maximum levels by the S5 stage (Fig. 4b). This was expected since expression of the VGE transgene was regulated by the promoter of a glycinin storage protein gene. These results correlate with the glycinin G1 promoter activity and specificity profile described by Nielsen et al. (1989). Expression of both genes continued through seed growth and declined at the S7 stage of maturation when seeds became yellowish in color.
Although there was a slight background of fluorescence in non-induced seeds, the difference between induced and non-induced seeds in planta was significant (Fig. 5). Fluorimetric analysis of protein extracts from T1 seeds showed strong seed-to-seed variability of GFP accumulation in dry mature seeds taken randomly from VG1 heterozygous plants (Fig. 5a). On the other hand, dry mature seeds taken from a single pod of homozygous line VG1-2-7H demonstrated similar levels of GFP fluorescence while seeds from different pods on the same plant showed more variance in fluorescence. In our opinion, this variability can be explained by the differences in physiological maturity of individual seeds (Muasya et al. 2006). Actual differences in quantitative fluorescence among seeds taken from different heights along individual homozygous plants only ranged between the equivalent of 1–3 μg GFP. In these studies there was a tenfold induction of GFP accumulation in homozygous VG1-2-7H seeds during seed maturation, which represents about 0.1% of total seed protein (Fig. 5b). Overall, fluorescence in induced homozygous VG1-2-7H seeds was higher compared to seeds from heterozygous VG1-2 plants.
a Induced GFP fluorescence in dry mature heterozygous T1 seeds of lines VG1-2 and VG1-4. Seeds with visibly detectable expression of GFP were harvested from plants treated with MOF by soil drenching. GFP fluorescence was quantitated by fluorimetry. b Inducible GFP fluorescence in dry mature seeds of homozygous T2 plants VG1-2-7H. Three separate plants were sampled. Each bar represents one seed. A group of three bars represents one pod taken from the following area of the plant: S-side branch; M-the fourth internode; B-base of plant; T-top of plant. 1,2,3-plant number. Each fluorimetry sample contained 500 μg of TSP. NI-seeds from non-induced plants. WT-wild type seeds. Significance of GFP fluorescence in sample groups versus non-induced controls was evaluated using the Mann–Whitney test (* represents P < 0.01)
Intracellular localization of GFP in induced seeds
Confocal microscopy was used to establish the intracellular localization of GFP in cotyledonary embryos, as well as in seeds following MOF induction in vitro and in planta. As shown in Fig. 6b, GFP accumulated in ER-derived protein bodies in induced cotyledonary embryos. During the first 12 h of induction GFP fluorescence was dispersed and did not appear to accumulate in any specific subcellular location (Fig. 6a). Later, as higher levels of expression and accumulation of recombinant protein was achieved, the accumulation of GFP was confined to protein bodies (Fig. 6b). GFP protein remained in this compartment throughout maturation of the cotyledonary embryo. Furthermore, fluorescence was observed in all cell layers of cotyledonary tips as well as immature cotyledons (data not shown). This result suggested that MOF penetration throughout the cotyledonary tissue was sufficient for deep cell induction of GFP expression.
Confocal microscopy of GFP accumulation in VG1-2 seed samples. Blue light images of optical sections taken of cotyledonary embryos at 12 h a and 48 h b after MOF treatment; c in vitro seed, 48 h after MOF treatment; d in planta induced stage S5 seed showing bright zone around the funiculus. GFPKDEL accumulation in subcellular protein bodies indicated by arrows. Bar 5 μm
In seeds induced in vitro, after 48 h of MOF treatment, GFP was localized to protein bodies in cotyledonary cells (Fig. 6c). In contrast, in cotyledons derived from seeds at S3–S4 stages from T1 plants induced by soil drenching, GFP was dispersed throughout the entire fluorescent gradient and not condensed in any particular subcellular location in any of the cells in the cotyledon. However, in more mature seeds GFP-containing protein bodies were detected, being located primarily in the bright fluorescent area along the inner edge of the transgenic seed (Fig. 6d).
Discussion
Chemically-inducible genetic switches have been tested in a number of model plants and plant cell culture systems (Aoyama and Chua 1997; Andersen et al. 2003; Padidam et al. 2003; Hughes et al. 2004; Tang and Newton 2004; Tavva et al. 2006). Although a limited number of studies have been reported on the use of chemically inducible genetic switches in major crop plants, examples include the restoration of male fertility in maize using a chimeric ecdysone receptor-based MOF-inducible system (Unger et al. 2002) and glucocorticoid-inducible gene expression in rice (Ouwerkerk et al. 2001). The approaches used to study inducible gene expression in plants have been mainly based on the constitutive expression of the receptor/activator protein (Aoyama and Chua 1997; Padidam et al. 2003; Koo et al. 2004; Tavva et al. 2006). However, plant tissue/organ-specific promoters have also been used to address the functional complementation of genes (Unger et al. 2002) or to allow the isolation of a specific mutant phenotype (Brand et al. 2006).
In the current study a soybean seed-specific glycinin promoter was used to drive expression of the chimeric receptor VGE in transgenic soybean plants. The expression of glycinin gene family members is highly specific to seeds and most highly elevated during the mid-stages of soybean seed development (Nielsen et al. 1989). Two separately generated inducible transgenic soybean lines were used to demonstrate that a genetic switch based upon the chemical ligand, methoxyfenozide and chimeric EcR receptor, VGE, can be used to induce gene expression with relatively tight control and absolute tissue specificity in soybean seeds.
Soybean somatic cotyledonary embryos cultured on SHaM maturation medium are a good model for the analysis of seed-specific traits prior to plant regeneration. Cotyledonary embryos are similar to seeds in terms of protein and fatty acid/lipid composition (Schmidt et al. 2005). The current study shows that gene switching in this system can be predictive of the results in transgenic plants derived from these tissues. In our study, embryos from both transgenic lines showed a consistent pattern of GFP induction following MOF treatment as well as the highest achievable level of GFP accumulation in the in vitro and in planta protocols tested.
The inducible transgenic soybean lines VG1-2 and VG1-4 were identified among seventeen hygromycin-resistant lines generated after two independent biolistic transformations. Other hygromycin-resistant and PCR-positive lines tested for MOF-inducibility did not respond to addition of the ligand MOF at the cotyledonary embryo stage, or in other cases, after plant regeneration and analyses of T1 seeds followed MOF treatment both in vitro or in planta. Extension of the induction period for up to 5 days did not trigger fluorescence in the PCR-positive, but non-inducible lines. The cause for the lack of inducibility in these lines is as yet unknown, but a few explanations are being considered. PCR analysis revealed full length VGE and gfp sequences in seven of the non-inducible lines. In other lines, it was suspected that tandem insertion and/or fragmentation of the VG1 construct may have occurred during transformation rendering the gene non-functional. It is also well established that DNA delivery by particle bombardment can result in the gene silencing due to integration of fragments or multiple copies of transgene (Dai et al. 2001; Reddy et al. 2003). It was previously shown that the Gal4 DNA-binding domain is intrinsically sensitive to cytosine methylation (Gälweiler et al. 2000).
Isolated immature soybean cotyledons (zygotic embryos) grown aseptically, and herein called in vitro seeds, are commonly used in studies of seed physiology, metabolism and nutrition of seeds (Thompson et al. 1977; Holowach et al. 1984; Saab and Obendorf 1989). It has been reported that growth and storage protein synthesis are more rapid in vitro than in planta (Thompson et al. 1977). In our study in vitro seeds were cultured for 2 days prior to treatment with MOF. The rapid growth of cultivated immature cotyledons (seeds increased in weight ~3 fold during the course of the experiment), combined with direct contact with the inducer, guaranteed by the physical removal of the seed coat, provided optimal conditions to test inducible gene expression. In contrast, the soaking of coated seeds in MOF resulted in only weak induction of GFP fluorescence after 48–72 h.
Time course studies performed by fluorimetry and confocal microscopy showed similar dynamics of fluorescence induction in somatic cotyledonary embryos and in vitro seeds from inducible VG1 lines with the fluorescence signal in somatic cotyledonary embryos being more intense. Extensive formation of GFP-containing ER-derived protein bodies at late stages of induction (after 30–48 h) was observed in both cases. The accumulation of significant levels of recombinant KDEL-tagged GFP protein that in turn induced protein body formation, was similar to previous reports (Moravec et al. 2007; Schmidt and Herman 2008).
Experiments aimed at mapping temporal and spatial patterns of induction suggested that initiation of GFP expression in seeds in planta was determined by the seed growth stage. When inducer was delivered during certain stages of soybean reproductive development, at the time when the glycinin promoter was actively driving VGE expression, significant levels of GFP accumulation occurred. Minor levels of induction were recorded when the inducer was delivered to plants with seeds matured to the S5–S8 stages. No induction occurred in matured, late stage somatic or zygotic embryos in vitro.
Two observations support the notion that uptake and delivery of the inducer to seeds in planta occurred relatively efficiently. First, the induction of GFP fluorescence following MOF application was initiated in a coordinate manner in all seeds that had reached the S3–S4 stage (120–140 mg). Second, a similar induction pattern was seen in planta and in S3–S4 seeds in pods on detached branches that were placed in an 80 μM MOF-solution (data not shown). A similar gradient of GFP fluorescence in the seed was present in both cases. The GFP gradient remained visible throughout all post-induction stages of seed growth and maturation in planta (S4–S8) as well as in dry mature seeds.
Confocal microscopy confirmed that GFP fluorescence and accumulation in ER-derived protein bodies in seeds induced in planta, was most intense in the funicular area and along the inner side of seed cotyledons. Nevertheless, induction of GFP expression and weak fluorescence was spread over the seed cotyledon as confirmed by microscopy, and by analysis using an anti-GFP antibody in a seed printing assay (data not shown). Overall, the smaller numbers of GFP-containing protein bodies that formed in seeds from pod-bearing VG1 plants was correlated with a lower level of GFP accumulation. This is in comparison to GFP florescence levels obtained in vitro and in seeds which carry a pGly:GFP “non-switchable” gene (Schmidt and Herman 2008). The location of the fluorescent gradient and its stability through seed developmental stages suggested that poor physical penetration of the inducer into the seed itself during seed growth and maturation may have occurred.
The accumulation of dry weight in a soybean seed follows a distinct three-stage pattern that consists of a short phase of exponential growth, a period when the growth rate remains relatively constant and a period of decreasing growth rate until the seed reaches its maximum dry weight at physiological maturity (Egli et al. 1981). Cell division in the cotyledons is essentially complete by 2 weeks after flowering, thus the increase in seed size during most of the period when the seed growth rate is constant is primarily associated with cell expansion. Cell expansion during the linear phase of seed growth requires the uptake of water. Seed cotyledons reach their maximum size at about 200–250 mg weight (S5 stage) although the accumulation of dry matter continues (Meinke et al. 1981). During the S3–S6 stages of soybean seed development, glycinin and, therefore, VGE mRNAs are most abundant as the developing seed actively synthesizes storage proteins. The presence of the inducer during that period of seed development resulted in the induction of GFP expression. Our in vitro studies confirmed that the most successful induction of GFP expression occurs during stages of intensive cell growth in the seed.
A large negative osmotic potential develops in seed embryo tissues prior to the onset of maturation, which then declines after the cessation of growth before desiccation. This occurs independent of changes in water relations in maternal tissues (Saab and Obendorf 1989; Wolswinkel 1992). The negative osmotic potential apparently plays a crucial role in maintaining a high rate of assimilate uptake and phloem transport, as seeds are characterized by having a high sink strength, or high capacity to accumulate assimilate. It is speculated that this large negative osmotic potential also helps to maintain the high rate of storage protein biosynthesis that is occurring, and at the same time, suppresses precocious maturation (Wolswinkel 1992). The change of osmotic potential during mid-stages of seed growth/maturation when the glycinin promoter is active could create a physiological barrier for MOF delivery to cotyledonary seed cells that are efficiently expressing VGE in planta. Moreover, the source-sink relationships during seed growth and maturation, perhaps compromised under greenhouse conditions, may have additionally affected MOF penetration into maturing seeds. It has been shown that seed protein mRNAs accumulate within the cotyledons in a wave-like pattern, possibly driven by a gradient of distribution of transcription factors (Perez-Grau and Goldberg 1989). All of these natural phenomena may influence the fluorescent GFP gradient that was observed in seeds induced in planta. Future studies using radioactively-labeled MOF to directly track the movement of the chemical inducer, combined with in situ detection of VGE mRNA in seeds, may shed light on how to explain the transgene induction pattern observed.
In summary, we have successfully developed and tested in soybean an environmentally safe, effective and versatile genetic switch methodology that could be used to alter seed composition, or regulate the expression of genes of interest in soybean seed. The genetic switch functioned successfully under in vitro conditions in somatic cotyledonary and zygotic seed embryos cultured from transgenic soybean lines. Activation of the genetic switch under greenhouse conditions using soil drenching for inducer application to growing plants was also successful with the extent and pattern of transgene expression being strongly influenced by the physiological and developmental state of the seed, as well as the stage of plant maturation. This study on the application of a genetic switch methodology to soybean seed provides a foundation for future research in the development and regulation of new seed traits and products in soybean.
Abbreviations
- TSP:
-
Total soluble protein
- MOF:
-
Methoxyfenozide
- ER:
-
Endoplasmic reticulum
- 35SCaMV promoter:
-
Cauliflower mosaic virus 35S RNA promoter
References
Andersen SU, Cvitanich C, Hougaard BK et al (2003) The glucocorticoid-inducible GVG system causes severe growth defects in both root and shoot of the model legume Lotus japonicus. Mol Plant Microbe Interact 16:1069–1076
Aoyama T, Chua NH (1997) A glucocorticoid-mediated transcriptional induction system in transgenic plants. Plant J 11:605–612
Brand L, Hörler M, Nüesch E et al (2006) A versatile and reliable two-component system for tissue-specific gene induction in Arabidopsis. Plant Physiol 141:1194–1204
Dai S, Zheng P, Marmey P et al (2001) Comparative analysis of transgenic rice plants obtained by Agrobacterium-mediated transformation and particle bombardment. Mol Breed 7:25–33
Dietrich CR, Han G, Chen M et al (2008) Loss-of-function mutations and inducible RNAi suppression of Arabidopsis LCB2 genes reveal the critical role of sphingolipids in gametophytic and sporophytic cell viability. Plant J 54:284–298
Egli DB, Fraser J, Leggett JE et al (1981) Control of Seed Growth in Soya Beans [Glycine max (L.) Merrill]. Ann Bot 48:171–176
Fischer RL, Goldberg RB (1982) Structure and flanking regions of soybean seed protein genes. Cell 29:651–660
Gälweiler L, Conlan RS, Mader P et al (2000) Technical advance: the DNA-binding activity of gal4 is inhibited by methylation of the gal4 binding site in plant chromatin. Plant J 23:143–157
Garbarino JE, Belknap WR (1994) Isolation of a ubiquitin-ribosomal protein gene (ubi3) from potato and expression of its promoter in transgenic plants. Plant Mol Biol 24:119–127
Herman EM, Tague BW, Hoffman LM et al (1990) Retention of phytohemagglutinin with carboxyterminal tetrapeptide KDEL in the nuclear envelope and the endoplasmic reticulum. Planta 182:305–312
Holowach LP, Thompson JF, Madison JT (1984) Storage protein composition of soybean cotyledons grown in vitro in media of various sulfate concentrations in the presence and absence of exogenous L-methionine. Plant Physiol 74:584–589
Hughes EH, Hong S-B, Gibson SI et al (2004) Expression of a feedback-resistant anthranilate synthase in Catharanthus roseus hairy roots provides evidence for tight regulation of terpenoid indole alkaloid levels. Biotech Bioeng 86:18–27
Koo JC, Asurmendi S, Bick J et al (2004) Ecdysone agonist-inducible expression of a coat protein gene from tobacco mosaic virus confers viral resistance in transgenic Arabidopsis. Plant J 37:439–448
Kourtz L, Dillon K, Daughtry S et al (2007) Chemically inducible expression of the PHB biosynthetic pathway in Arabidopsis. Transgenic Res 16:759–769
Martinez A, Sparks C, Hart CA et al (1999) Ecdysone agonist inducible transcription in transgenic tobacco plants. Plant J 19:97–106
Meinke DW, Chen J, Beachy RN (1981) Expression of storage-protein genes during soybean seed development. Planta 153:130–139
Miernyk JA, Johnston ML, Antoine W et al (2009) A directed-proteomics platform for systems analysis of soybean seed development. J Integr Plant Biol (in press)
Moore I, Samalova M, Kurup S (2006) Transactivated and chemically inducible gene expression in plants. Plant J 45:651–683
Moravec T, Schmidt MA, Herman EM et al (2007) Production of Escherichia coli heat labile toxin (LT) B subunit in soybean seed and analysis of its immunogenicity as an oral vaccine. Vaccine 25:1647–1657
Muasya RM, Lommen WJM, Auma EO et al (2006) Relationship between variation in quality of individual seeds and bulk seed quality in common bean (Phaseolus vulgaris L.) seed lots. NJAS 54:5–16
Nielsen NC, Dickinson CD, Cho TJ et al (1989) Characterization of the glycinin gene family in soybean. Plant Cell 1:313–328
Ouwerkerk PB, de Kam RJ, Hoge JH et al (2001) Glucocorticoid-inducible gene expression in rice. Planta 213:370–378
Padidam M (2003) Chemically regulated gene expression in plants. Current Opin Plant Biol 6:169–177
Padidam M, Gore M, Lu DL et al (2003) Chemical-inducible, ecdysone receptor-based gene expression system for plants. Transgenic Res 12:101–109
Perez-Grau L, Goldberg RB (1989) Soybean seed protein genes are regulated spatially during embryogenesis. Plant Cell 1:1095–1109
Reddy MS, Dinkins RD, Collins GB (2003) Gene silencing in transgenic soybean plants transformed via particle bombardment. Plant Cell Rep 21:676–683
Saab IN, Obendorf RL (1989) Soybean seed water relations during in situ and in vitro growth and maturation. Plant Physiol 89:610–616
Samac DA, Hironaka CM, Yallaly PE et al (1990) Isolation and characterization of the genes encoding basic and acidic chitinase in Arabidopsis thaliana. Plant Physiol 93:907–914
Schardl CL, Byrd AD, Benzion G, Altschuler MA et al (1987) Design and construction of a versatile system for the expression of foreign genes in plants. Gene 61:1–11
Schmidt MA, Herman EM (2008) Proteome rebalancing in soybean seeds can be exploited to enhance foreign protein accumulation. Plant Biotechnol J 6:832–842
Schmidt MA, Tucker DM, Cahoon EB et al (2005) Towards normalization of soybean somatic embryo maturation. Plant Cell Rep 24:383–391
Sims TL, Goldberg RB (1989) The glycinin Gy1 gene from soybean. Nucleic Acids Res 17:4386
Sudarshana MR, Plesha MA, Uratsu SL et al (2006) A chemically inducible cucumber mosaic virus amplicon system for expression of heterologous proteins in plant tissues. Plant Biotech J 4:551–559
Sun J, Niu QW, Tarkowski P (2003) The Arabidopsis AtIPT8/PGA22 gene encodes an isopentenyl transferase that is involved in de novo cytokinin biosynthesis. Plant Physiol 131:167–176
Tang W, Newton RJ (2004) Glucocorticoid-inducible transgene expression in loblolly pine (Pinus taeda L.) cell suspension cultures. Plant Sci 166:1351–1358
Tavva VS, Dinkins RD, Palli SR et al (2006) Development of a methoxyfenozide-responsive gene switch for applications in plants. Plant J 45:457–469
Thibaud-Nissen F, Shealy RT, Khanna A et al (2003) Clustering of microarray data reveals transcript patterns associated with somatic embryogenesis in soybean. Plant Physiol 132:118–136
Thompson JF, Madison JT, Muenster A-ME (1977) In vitro culture of immature cotyledons of soya bean (Glycine max L. Merr.). Ann Bot 41:29–39
Trick HN, Dinkins RD, Santarem ER et al (1997) Recent advances in soybean transformation. Plant Tissue Cult Biotech 3:9–26
Unger E, Cigan AM, Trimnell M et al (2002) A chimeric ecdysone receptor facilitates methoxyfenozide-dependent restoration of male fertility in ms45 maize. Transgenic Res 11:455–465
Wang TL, Domoney C, Hedley CL et al (2003) Can we improve the nutritional quality of legume seeds? Plant Physiol 131:886–891
Wielopolska A, Townley H, Moore I et al (2005) A high-throughput inducible RNAi vector for plants. Plant Biotech J 3:583–590
Wolswinkel P (1992) Transport of nutrients into developing seeds: a review of physiological mechanisms. Seed Sci Res 2:59–73
Zuo J, Niu QW, Ikeda Y et al (2002) Marker-free transformation: increasing transformation frequency by the use of regeneration-promoting genes. Curr Opin Biotechnol 13:173–180
Acknowledgments
Dr. Eliot Herman is gratefully acknowledged for providing stimulating discussions and intellectual support. We also thank Dr. Howard Berg in the Integrated Microscopy Facility and Kevin Lutke from Tissue Culture Facility at the Danforth Plant Science Center for their kind assistance throughout this study, and Dr. Mala Padidam (Rohm and Haas Company), Dr. Wayne Parrott (University of Georgia) and Dr. Ed Cahoon (University of Nebraska) for providing certain genetics components for the gene switch and expression systems. This work was supported by grants from the USDA/Illinois-Missouri Alliance for Biotechnology, IMBA (TWT; 2006-5) and from The Consortium for Plant Biotechnology Research, Inc. (CPBR, Inc.) (RNB; GO12026-266).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Semenyuk, E.G., Schmidt, M.A., Beachy, R.N. et al. Adaptation of an ecdysone-based genetic switch for transgene expression in soybean seeds. Transgenic Res 19, 987–999 (2010). https://doi.org/10.1007/s11248-010-9377-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-010-9377-6