Abstract
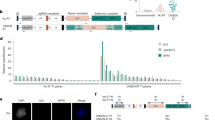
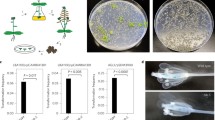
The recognition of the T-DNA left border (LB) repeat is affected by its surrounding sequences. Here, the LB regions were further characterized by molecular analysis of transgenic plants, obtained after Agrobacterium tumefaciens-mediated transformation with T-DNA vectors that had been modified in this LB region. At least the 24-bp LB repeat by itself was insufficient to terminate the T-strand synthesis. Addition of the natural inner and/or outer border regions to at least the LB repeat, even when present at a distance, enhanced the correct recognition of the LB repeat, reducing the number of plants containing vector backbone sequences. In tandem occurrence of both the octopine and nopaline LB regions with their repeats terminated the T-strand synthesis most efficiently at the LB, yielding a reproducibly high number of plants containing only the T-DNA. Furthermore, T-strand synthesis did not terminate efficiently at the right border (RB) repeat, which might indicate that signals in the outer RB region inhibit the termination of T-strand synthesis at the RB repeat.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Afolabi AS, Worland B, Snape JW, Vain P (2004) A large-scale study of rice plants transformed with different T-DNAs provides new insights into locus composition and T-DNA linkage configurations. Theor Appl Genet 109:815–826
Breitler J-C, Meynard D, Van Boxtel J, Royer M, Bonnot D, Cambillau L, Guiderdoni E (2004) A novel two T-DNA binary vector allows efficient generation of marker-free transgeneic plants in three elite cultivars of rice (Oryza sativa L.). Trangenic Res 13:271–187
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Cotsaftis O, Sallaud C, Breitler JC, Meynard D, Greco R, Pereira A, Guiderdoni E (2002) Transposon-mediated generation of T-DNA- and marker-free rice plants expressing a Bt endotoxin gene. Mol Breed 10:165–180
De Buck S, De Wilde C, Van Montagu M, Depicker A (2000) T-DNA vector backbone sequences are frequently integrated into the genome of transgenic plants obtained by Agrobacterium-mediated transformation. Mol Breed 6:459–468
De Buck S, Windels P, De Loose M, Depicker A (2004) The integration position of a single copy â-glucuronidase transgene in Arabidopsis thaliana may have an effect on transgene expression variability but has only a minor effect on the induction of transgene silencing. Cell Mol Life Sci 61:2632–2645
De Loose M, Danthinne X, Van Bockstaele E, Van Montagu M, Depicker A (1995) Different 5′ leader sequences modulate ß-glucuronidase accumulation levels in transgenic Nicotiana tabacum plants. Euphytica 85:209–216
De Neve M, De Buck S, Jacobs A, Van Montagu M, Depicker A (1997) T-DNA integration patterns in co-transformed plant cells suggest that T-DNA repeats originate from ligation of separate T-DNAs. Plant J 11:15–29
Eamens AL, Blanchard CL, Dennis ES, Upadhyaya NM (2004) A bidirectional gene trap construct suitable for T-DNA and Ds-mediated insertional mutagenesis in rice (Oryza sativa L.). Plant Biotechnol J 2:367–380
Fu D, St. Amand PC, Xiao Y, Muthukrishnan S, Liang GH (2006) Characterization of T-DNA integration in creeping bentgrass. Plant Sci. 170:225–237
Gelvin SB (2003) Agrobacterium-mediated plant transformation: the biology behind the “gene-jockeying” tool. Microbiol Mol Biol Rev 67:16–37
Gelvin SB (2000) Agrobacterium and plant genes involved in T-DNA transfer and integration. Annu Rev Plant Physiol Plant Mol Biol 51:223–256
Gheysen G, Angenon G, Van Montagu M (1998) Agrobacterium-mediated plant transformation: a scientifically intriguing story with significant applications. In: Lindsey K (ed) Transgenic plant research. Harwood Academic Publishers, Amsterdam, pp 1–33
Hanson B, Engler D, Moy Y, Newman B, Ralston E, Gutterson N (1999) A simple method to enrich an Agrobacterium-transformed population for plants containing only T-DNA sequences. Plant J 19:727–734
Huang S, Gilbertson LA, Adams TH, Malloy KP, Reisenbigler EK, Birr DH, Snyder MW, Zhang Q, Luethy MH (2004) Generation of marker-free transgenic maize by regular two-border Agrobacterium transformation vectors. Transgenic Res 13:451–461
Jakowitsch J, Papp I, Moscone EA, van der Winden J, Matzke M, Matzke AJM (1999) Molecular and cytogenetic characterization of a transgene locus that induces silencing and methylation of homologous promoters in trans. Plant J 17:131–140
Jen GC, Chilton M-D (1986) The right border region of pTiT37 T-DNA is intrinsically more active than the left border region in promoting T-DNA transformation. Proc. Natl. Acad. Sci. USA 83:3895–3899
Joos H, Inzé D, Caplan A, Sormann M, Van Montagu M, Schell J (1983) Genetic analysis of T-DNA transcripts in nopaline crown galls. Cell 32:1057–1067
Kim S-R, Lee J, Jun S-H, Park S, Kang H-G, Kwon S, An G (2003) Transgene structures in T-DNA-inserted rice plants. Plant Mol Biol 52:761–773
Komari T, Ishida Y, Hiei Y (2004) Plant transformation technology: Agrobacterium-mediated transformation. In: Christou P, Klee H (eds) Handbook of Plant Biotechnology, Vol. 1. John Wiley & Sons, Chichester, pp 233–261
Kononov ME, Bassuner B, Gelvin SB (1997) Integration of T-DNA binary vector ȁ8backboneȁ9 sequences into the tobacco genome: evidence for multiple complex patterns of integration. Plant J 11:945–957
Kuraya Y, Ohta S, Fukuda M, Hiei Y, Murai N, Hamada K, Ueki J, Imaseki H, Komari T (2004) Suppression of transfer of non-T-DNA ȁ8vector backboneȁ9 sequences by multiple left border repeats for transformation of higher plants mediated by Agrobacterium tumefaciens. Mol Breed 14:309–320
Lange M, Vincze E, Møller MG, Holm PB (2006) Molecular analysis of transgene and vector backbone integration into the barley genome following Agrobacterium-mediated transformation. Plant Cell Rep (in press) (DOI 10.1007/s00299-006-0140-7)
Lu H-J, Zhou X-R, Gong Z-X, Upadhyaya NM (2001) Generation of selectable marker-free transgenic rice using double right-border (DRB) binary vectors. Aust J Plant Physiol 28:241–248
Martineau B, Voelker TA, Sanders RA (1994) On defining T-DNA. Plant Cell 6:1032–1033 (Letter to the editor)
McCormac AC, Fowler MR, Chen D-F, Elliott MC (2001) Efficient co-transformation of Nicotiana tabacum by two independent T-DNAs, the effect of T-DNA size and implications for genetic separation. Transgenic Res 10:143–155
Michielse CB, Ram AFJ, Hooykaas PJJ, van den Hondel CAMJJ (2004) Agrobacterium-mediated transformation of Aspergillus awamori in the absence of full-length VirD2, VirC2, or VirE2 leads to insertion of aberrant T-DNA structures. J Bacteriol 186:2038–2045
Miranda A, Janssen G, Hodges L, Peralta EG, Ream W (1992) Agrobacterium tumefaciens transfers extremely long T-DNAs by a unidirectional mechanism. J Bacteriol 174:2288–2297
Peralta EG, Hellmiss R, Ream W (1986) Overdrive, a T-DNA transmission enhancer on the A. tumefaciens tumour-inducing plasmid. EMBO J 5:1137–1142
Ramanathan V, Veluthambi K (1995) Transfer of non-T-DNA portions of the Agrobacterium tumefaciens Ti plasmid pTiA6 from the left terminus of TL-DNA. Plant Mol Biol 28:1149–1154
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning, a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Smith N, Kilpatrick JB, Whitelam GC (2001) Superfluous transgene integration in plants. Crit Rev Plant Sci 20:215–249
Stachel SE, Timmerman B, Zambryski P (1987) Activation of Agrobacterium tumefaciens vir gene expression generates multiple single-stranded T-strand molecules from the pTiA6 T-region: requirements for 5′ virD gene products. EMBO J 6:857–863
Toro N, Datta A, Yanofsky M, Nester E (1988) Role of the overdrive sequence in T-DNA border cleavage in Agrobacterium. Proc Natl Acad Sci USA 85:8558–8562
Toro N, Datta A, Carmi OA, Young C, Prusti RK, Nester EW (1989) The Agrobacterium tumefaciens virC1 gene product binds to overdrive, a T-DNA transfer enhancer. J Bacteriol 171:6845–6849
Travella S, Ross SM, Harden J, Everett C, Snape JW, Harwood WA (2005) A comparison of transgenic barley lines produced by particle bombardment and Agrobacterium-mediated techniques. Plant Cell Rep 23:780–789
Tzfira T, Citovsky V (2002) Partners-in-infection: host proteins involved in the transformation of plant cells by Agrobacterium. Trends Cell Biol 12:121–129
Vain P, Afolabi AS, Worland B, Snape JW (2003) Transgene behaviour in populations of rice plants transformed using a new dual binary vector system: pGreen/pSoup. Theor Appl Genet 107:210–217
Vain P, Harvey A, Worland B, Ross S, Snape JW, Lonsdale D (2004) The effect of additional virulence genes on transformation efficiency, transgene integration and expression in rice plants using the pGreen/pSoup dual binary vector system. Transgenic Res 13:593–603
van der Graaff E, den Dulk-Ras A, Hooykaas PJJ (1996) Deviating T-DNA transfer from Agrobacterium tumefaciens to plants. Plant Mol Biol 31:677–681
Van Haaren MJJ, Pronk JT, Schilperoort RA, Hooykaas PJJ (1987) Functional analysis of the Agrobacterium tumefaciens octopine Ti-plasmid left and right T-region border fragments. Plant Mol Biol 8:95–104
Veluthambi K, Ream W, Gelvin SB (1988) Virulence genes, borders, and overdrive generate single-stranded T-DNA molecules from the A6 Ti plasmid of Agrobacterium tumefaciens. J Bacteriol 170:1523–1532
Wang K, Genetello C, Van Montagu M, Zambryski PC (1987) Sequence context of the T-DNA border repeat element determines its relative activity during T-DNA transfer to plant cells. Mol Gen Genet 210:338–346
Wenck A, Czakó M, Kanevski I, Márton L (1997) Frequent collinear long transfer of DNA inclusive of the whole binary vector during Agrobacterium-mediated transformation. Plant Mol Biol 34:913–922
Wolters A-MA, Trindade LM, Jacobsen E, Visser RGF (1998) Fluorescence in situ hybridization on extended DNA fibres as a tool to analyse complex T-DNA loci in potato. Plant J 13:837–847
Yin Z, Wang G-L (2000) Evidence of multiple complex patterns of T-DNA integration into the rice genome. Theor Appl Genet 100:461–470
Yusibov VM, Steck TR, Gupta V, Gelvin SB (1994) Association of single-stranded transferred DNA from Agrobacterium tumefaciens with tobacco cells. Proc Natl Acad Sci USA 91:2994–2998
Zhu J, Oger PM, Schrammeijer B, Hooykaas PJJ, Farrand SK, Winans SC (2000) The bases of crown gall tumorigenesis. J Bacteriol 182:3885–3895
Zupan J, Muth TR, Draper O, Zambryski P (2000) The transfer of DNA from Agrobacterium tumefaciens into plants: a feast of fundamental insights. Plant J 23:11–28
Acknowledgements
The authors thank Gert Van Der Auwera, Ingrid Peck, and Thierry Aelbrecht for critical reading of the manuscript and many editorial suggestions, Geert Angenon and Willem Broekaert for helpful comments, Vladimir Mironov for generously providing constructs pCDVIB, pCDVIB1, pCDVIB2, and pCDVIB3, Roger Hellens for donating the bar cassette of the pGreen vector, and Martine De Cock for help with the layout. This work was supported by a grant from the European Union BIOTECH program (BIO4-96-0253) and by a STREP project under the 6th framework program of the EU “GENINTEG” (LSHG-CT2003-503303) with additional co-financing from the Flemish Community and a grant from the Special Research Fund of the Ghent University (BOF-01111400). N.P. is indebted to the Institute for the Promotion of Innovation through Science and Technology in Flanders for a predoctoral fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Podevin, N., De Buck, S., De Wilde, C. et al. Insights into recognition of the T-DNA border repeats as termination sites for T-strand synthesis by Agrobacterium tumefaciens . Transgenic Res 15, 557–571 (2006). https://doi.org/10.1007/s11248-006-9003-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-006-9003-9